Back to Journals » Risk Management and Healthcare Policy » Volume 14
Could Adjuvant Chemotherapy Improve Prognosis for Cervical Cancer Patients with Elevated Pretreatment Serum Squamous-Cell Carcinoma Antigen?
Authors Yuan Z, Cao D , Zhang Y, Shen K, Yang J, Yu M , Zhou H
Received 24 July 2020
Accepted for publication 28 September 2020
Published 11 January 2021 Volume 2021:14 Pages 109—116
DOI https://doi.org/10.2147/RMHP.S273848
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Marco Carotenuto
Zhen Yuan, Dongyan Cao, Ying Zhang, Keng Shen, Jiaxin Yang, Mei Yu, Huimei Zhou
Department of Obstetrics and Gynecology, Peking Union Medical College Hospital, Peking Union Medical College, Chinese Academy of Medical Sciences, Beijing, People’s Republic of China
Correspondence: Dongyan Cao; Ying Zhang Email [email protected]; [email protected]
Objective: The aim of this study was to explore whether adjuvant chemotherapy could improve prognosis for cervical cancer patients with elevated pretreatment serum squamous-cell carcinoma antigen (SCC-Ag).
Methods: Propensity-score matching and inverse probability of treatment weighting (IPTW) were used to ensure balanced groups for patients with (arm A) and without adjuvant chemotherapy (arm B). All patients were treated between January 2012 and December 2014 at a single center. Study outcomes were disease-free survival (DFS) and overall survival (OS).
Results: In total, 81 patients were included in this study. By propensity-score matching, 35 patients were included in each group (arm A and arm B). Median follow-up was 60 months in arm A and 66 months in arm B. Overall, 85.7% of patients in arm A and 71.4% of those in arm B received adjuvant radiotherapy. DFS and OS curves were similar between arms A and B (P=0.971 and 0.633, respectively). With IPTW, arm A was not associated with prognosis in terms of DFS (HR 0.946, 95% CI 0.237– 3.784; P=0.938) or OS (HR 1.020, 95%CI 0.357– 2.913; P=0.970).
Conclusion: For patients with elevated pretreatment SCC-Ag, adjuvant chemotherapy was not found to improve prognosis. Also, a considerable proportion of these patients had postoperative indications for adjuvant radiotherapy. For these cervical cancer patients with elevated pretreatment SCC-Ag, the choice of radical hysterectomy and adjuvant chemotherapy should be prudent.
Keywords: adjuvant chemotherapy, cervical cancer, oncologic outcomes, serum squamous-cell carcinoma antigen
Introduction
Histologically, the most common type of uterine cervical cancer is squamous-cell carcinoma, which accounts for approximately 80% of all uterine cervical cancers.1,2 Serum SCCantigen (SCC-Ag) is a kind of tumor-associated antigens related to SCC.
The clinical significance and role of SCC-Ag prognosis in squamous cervical cancer have been explored and reported in many previous studies. Elevated pretreatment and/or posttreatment SCC-Ag levels have been found to be associated with worse survival.2–6
In 2018, a systematic review and meta-analysis was performed to pool the association effects of serum SCC-Ag on recurrence and mortality in patients with squamous-cell cervical cancer, and patients with elevated pretreatment serum SCC-Ag were at higher risk of mortality and recurrence of newly diagnosed cervical cancer.5 Moreover, in 2019 Liu et al reviewed relevant studies published until June 2018, and found that elevated serum SCC-Ag was related to poor prognosis in terms of both disease-free survival (DFS) and overall survival (OS).4 However, serum SCC-Ag is not documented in current guidelines or in routine clinical use for patients with cervical cancer.2
In some studies, adjuvant chemotherapy has been reported to improve prognosis for some patients with cervical cancer.7,8 However, it is unclear whether adjuvant chemotherapy can improve prognosis of these patients with elevated pretreatment SCC-Ag.2
The aim of this preliminary study was to explore whether adjuvant chemotherapy could improve the prognosis of these cervical cancer patients with elevated pretreatment SCC-Ag.
Methods
Inclusion criteria were International Federation of Gynecology and Obstetrics (FIGO) 2009 stage IB-IIA, surgery performed between January 2012 and December 2014, radical hysterectomy with lymphadenectomy, and histological confirmation of SCC with elevated pretreatment serum SCC-Ag. Patients with other histological types of cervical cancer and those with normal SCC-Ag were excluded.
Serum SCC-Ag levels were measured with a chemiluminescent microparticle using an Architect i2000SR (IMX; Abbott Diagnostics, Chicago, IL, USA). As recommended by the manufacturer, the cutoff level was established at 1.5 ng/mL. In our hospital, the measurable range of SCC-Ag is 0–70 ng/mL. The pretreatment SCC-Ag level was determined at the time of cervical cancer diagnosis.
Two experienced gynecological oncologists determined the clinical stage of each case according to the FIGO 2009 stage system. Standard radical hysterectomy (Piver type III hysterectomy) involves resection of parametrial tissue close to the pelvic wall and the upper third or half of the vagina to ensure negative margins and surgical thoroughness.9 Histological diagnosis was confirmed by at least two pathologists. For patients with FIGO stage IB and IIA squamous cervical cancer who chose to undergo primary surgery, the standard treatment was radical hysterectomy plus bilateral pelvic lymph-node dissection with (or without) para-aortic lymph-node dissection and with (or without) adjuvant radiation. Adjuvant radiation was indicated if pathological risk factors were identified. Chemotherapy provided before/after surgery or after radiotherapy, was defined as adjuvant chemotherapy.
Information collected from medical records for analysis was age, clinical stage, serum SCC-Ag, pathological results, treatment modality, and oncological outcomes.
Statistical Analysis
Propensity-score matching was used to select patients, improving the quality of the results reported. Propensity-matched comparison attempts to estimate the effect of a treatment by accounting for possible factors that predict receipt of treatment, thus reducing possible selection biases. Factors used for matching in our study were SCC-Ag, lymph-node metastasis, parametrium invasion, and surgical margin status. Patients undergoing adjuvant chemotherapy were matched 1:1 with a cohort of women not receiving adjuvant chemotherapy using a caliper width ≤0.03 SD of the logit odds of the estimated propensity score. Inverse probability of treatment weighting (IPTW) was also used10 to capture information from patients who otherwise would have been discarded by 1:1 matching.
Categorical variables are summarized in frequency tables and continuous variables presented as medians (range). Frequency distributions were compared using χ2 with Fisher’s exact test and median values using nonparametric tests.
DFS was calculated from the date of surgery to the date of first recurrence or last follow-up in patients, and OS as the date of death or last contact. All follow-up information was censored following March 1, 2019. Survival curves were calculated using the Kaplan–Meier method and compared using the log-rank test. Data were analyzed using SPSS 23 (IBM, Armonk, NY, USA), Prism 7 (GraphPad Software, San Diego, CA), and RStudio 1.1.463. P<0.05 was considered statistically significant using the two-tailed hypothesis.
Results
The patient-selection flowchart is shown in Figure 1. After screening and matching, 35 patients were included in each group: adjuvant therapy (arm A) and no adjuvant therapy (arm B).
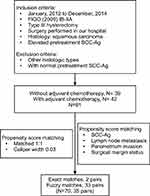 |
Figure 1 Study flowchart.Abbreviations: FIGO, Federation of Gynecology and Obstetrics; SCC-Ag, squamous-cell carcinoma antigen. |
The median ages of patients in arms A and B were 44 and 46 years, respectively, with no significant difference between the two groups (P=0.061). The median pretreatment SCC-Ag level of patients was 3.8 ng/mL in arm A and 2.7 ng/mL in arm B, with no significant difference between two groups (P=0.117). The proportion of patients with FIGO stage IIA in arm A was significantly higher than in arm B (28.6% vs 2.9%, P=0.003). There was no significant difference between the groups in terms of the proportion of patients undergoing the different surgical procedures (transabdominal or laparoscopic radical hysterectomy (P=0.231).
Table 1 shows postoperative pathological high-risk and intermediate-risk factors information for the two groups. Patients with positive pelvic nodes, positive surgical margin, and/or positive parametrium were considered high-risk.6 Owing to the application of a propensity-matching algorithm, baseline characteristics and high risks were similar between the groups. Sedlis criteria consider stromal invasion, lymphatic space involvement, and primary tumor size intermediate risk factors.7 For tumor size, the proportion of patients with tumor diameter >4 cm in arm A was significantly higher than arm B (P=0.001). The proportion of patients with stromal invasion depth of ≥50% in arm A was significantly higher than arm B (P=0.034). Regarding adjuvant radiotherapy, 85.7% and 71.4% of patients in arms A and B received adjuvant radiotherapy, respectively, and proportions in the two groups were not significantly different. Regarding adjuvant chemotherapy in arm A, 14 patients received neoadjuvant chemotherapy before surgery, 14 adjuvant chemotherapy after surgery, and seven chemotherapy both before and after surgery (Figure 2).
 |
Table 1 Clinical and Pathological Characteristics of Patients by Propensity-Score Matching |
 |
Figure 2 Arm A and arm B.Abbreviation: Chemo, chemotherapy |
The median follow-up time of patients was 60 months in arm A and 66 months in arm B. Clinical characteristics of patients with relapse are shown in Table 2. DFS and OS curves (Figure 3), were not significantly different between the groups (P=0.971 and 0.633, respectively). DFS at 3 years was 91% in both arms. OS at 3 years was 91% and 94%, in arms A and B, respectively. Based on IPTW, arm A was not associated with prognosis in terms of DFS (HR 0.946, CI 0.237–3.784; P=0.938) or OS (HR 1.020, HR 0.357–2.913; P=0.970).
 |
Table 2 Clinical and Pathological Characteristics of Patients with Relapse |
 |
Figure 3 Disease-free survival (DFSA) and overall survival (OSB) curves. |
Discussion
Radical hysterectomy with bilateral pelvic lymph-node dissection is the preferred treatment approach for patients with FIGO stage IA2–IIA1 cervical cancers.11 Effective treatments for bulky FIGO IB2 lesions and FIGO stage IIA2, which are associated with poor prognosis, remain controversial, though in the US definitive chemoradiation is typically preferred over radical surgery for select patients with bulky FIGO IB2 lesions and the vast majority of patients with FIGO stage IIA2 or greater cervical cancers.11 Radiotherapy can be associated with severe complications, such as vesicovaginal fistula, and long-term complications, such as vaginal stricture, dyspareunia, and consequent detrimental effects on the patient’s quality of life.12 To reduce long-term complications due to radiotherapy, select FIGO IB2–IIB cases may be treated with radical hysterectomy or neoadjuvant chemotherapy followed by radical hysterectomy,11 especially for younger patients.
For patients with locally advanced cervical cancer, the role of adjuvant chemotherapy in improving prognosis remains controversial. Some studies have reported improved survival,7,13 whereas others have reported no survival benefit.14–17 However, all these studies assessed patients at advanced FIGO stages and not those with elevated pretreatment SCC-Ag levels.
Although the role of adjuvant chemotherapy in improving prognosis is controversial, several studies7,18–20 and meta-analyses have reported that adjuvant chemotherapy can reduce the risk of distant recurrence.13,17,21 Moreover, elevated pretreatment SCC-Ag has been shown to be a risk factors of distant recurrence.20,22,23 Therefore, it may be prudent to explore whether adjuvant chemotherapy could improve prognosis for patients with elevated pretreatment SCC-Ag.
In this study, with propensity score-matching and IPTW, we explored the effect of adjuvant chemotherapy on oncological outcomes of patients with elevated pretreatment SCC-Ag.It did not appear to improve prognosis in terms of distant recurrence, DFS, or OS. However, our findings need to be interpreted with caution, since in arm A there were more patients with more advanced cancer, larger tumors, and deeper stromal invasion, as shown in Table 1. Stromal invasion and primary tumor size were two intermediate risk factors of poor prognosis according to the Sedlis criteria.24 In other words, though after propensity-score matching high-risk factors were equivalent in the two groups, more patients in arm A had intermediate risk factors. Moreover, after neoadjuvant chemotherapy, 10.8%-45% of patients may have pathological complete response rate;14,25,26 therefore, the proportion of patients with high-risk factors in arm A may have been higher than arm B.
For patients with elevated pretreatment SCC-Ag, 85.7% and 71.4% of patients in arms A and B received adjuvant radiotherapy, respectively. This is consistent with one of Reesink-Peters et al’s studies, in which 57%–74% of patients with elevated pretreatment SCC-Ag had postoperative indications for adjuvant radiotherapy.27 In other words, the majority of these patients also received adjuvant radiotherapy and/or chemotherapy, in addition to radical hysterectomy. Therefore, definitive radiotherapy, rather than surgery, should be considered for patients with preoperative elevated SCC-Ag.2
The main limitation of our study was that the number of patients assessed was relatively insufficient. Considering the learning curve required for surgery procedures28 and to ensure adequate follow-up, we selected patients who had undergone surgery after January 2012 and before December 2014. In order to make sure that all surgeries had been performed by experienced oncological gynecological surgeons, we focused our study on a single center. All these factors possibly contributed to the insufficient sample. Moreover, due to the nature of retrospective research, selection bias was inevitable. In this study, the combination of propensity-score matching and IPWT reduced bias as much as possible. To the best of our knowledge, this is the first study to explore whether adjuvant chemotherapy could improve prognosis for cervical cancer patients with elevated pretreatment SCC-Ag using the combination of propensity-score matching and IPTW.
In conclusion, for patients with elevated pretreatment SCC-Ag, adjuvant chemotherapy may not improve survival. Also a considerable proportion of these patientshad postoperative indications for adjuvant radiotherapy. For these patients, the choice of radical hysterectomy and adjuvant chemotherapy should be prudent.
Data-Sharing Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Ethics Approval
This study was approved by the Peking Union Medical College Hospital Ethnics Review Board (S-K1362). All procedures were in accordance with the ethical standards of the responsible committee on human experimentation and with the 1964 Declaration of Helsinki and later versions. Due to the retrospective design, no separate informed consent was necessary in the scope of this study. The data set was kept anonymous to protect patient privacy. All the data were deidentified to protect patient privacy.
Funding
This study was funded by the CAMS Initiative for Innovative Medicine (CAMS-2017-12M-1-002 to Keng Shen).
Disclosure
The authors declare that they have no conflicts of interest and nothing to disclose.
References
1. Cancer ICoESoC Comparison of risk factors for invasive squamous cell carcinoma and adenocarcinoma of the cervix: collaborative reanalysis of individual data on 8097 women with squamous cell carcinoma and 1374 women with adenocarcinoma from 12 epidemiological studies. Int J Cancer. 2007;120(4):885–891. doi:10.1002/ijc.22357
2. Fu J, Wang W, Wang Y, et al. The role of squamous cell carcinoma antigen (SCC Ag) in outcome prediction after concurrent chemoradiotherapy and treatment decisions for patients with cervical cancer. Radiat Oncol. 2019;14(1):146. doi:10.1186/s13014-019-1355-4
3. Guo Q, Zhu J, Wu Y, et al. Predictive value of preoperative serum squamous cell carcinoma antigen (SCC-Ag) level on tumor recurrence in cervical squamous cell carcinoma patients treated with radical surgery: A single-institution study. Eur J Surg Oncol. 2019.
4. Liu Z, Shi H. Prognostic Role of Squamous Cell Carcinoma Antigen in Cervical Cancer: A Meta-analysis. Dis Markers. 2019;2019:6710352. doi:10.1155/2019/6710352
5. Charakorn C, Thadanipon K, Chaijindaratana S, et al. The association between serum squamous cell carcinoma antigen and recurrence and survival of patients with cervical squamous cell carcinoma: A systematic review and meta-analysis. Gynecol Oncol. 2018;150(1):190–200. doi:10.1016/j.ygyno.2018.03.056
6. Wang W, Liu X, Hou X, et al. Posttreatment squamous cell carcinoma antigen predicts treatment failure in patients with cervical squamous cell carcinoma treated with concurrent chemoradiotherapy. Gynecol Oncol. 2019;155(2):224–228. doi:10.1016/j.ygyno.2019.09.003
7. Duenas-Gonzalez A, Zarba JJ, Patel F, et al. Phase III, open-label, randomized study comparing concurrent gemcitabine plus cisplatin and radiation followed by adjuvant gemcitabine and cisplatin versus concurrent cisplatin and radiation in patients with stage IIB to IVA carcinoma of the cervix. J Clin Oncol. 2011;29(13):1678–1685. doi:10.1200/JCO.2009.25.9663
8. Mabuchi S, Isohashi F, Yokoi T, et al. A Phase II study of postoperative concurrent carboplatin and paclitaxel combined with intensity-modulated pelvic radiotherapy followed by consolidation chemotherapy in surgically treated cervical cancer patients with positive pelvic lymph nodes. Gynecol Oncol. 2016;141(2):240–246. doi:10.1016/j.ygyno.2016.02.011
9. Meigs JV. Radical hysterectomy with bilateral dissection of the pelvic lymph nodes for cancer of the cervix (the Wertheim, Reis, Clark, Wertheim-Meigs operation). Surg Clin North Am. 1956;36(4):1083–1116. doi:10.1016/S0039-6109(16)34948-9
10. Austin PC, Stuart EA. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat Med. 2015;34(28):3661–3679. doi:10.1002/sim.6607
11. National Comprehensive Cancer Network. (NCCN) Clinical Practice Guidelines in Oncology. Cervical Cancer (Version 3). 2019. https://www.nccn.org/professionals/physician_gls/f_guidelines.asp.
12. Benedetti Panici P, Bellati F, Plotti F, et al. Neoadjuvant chemotherapy followed by radical surgery in patients affected by vaginal carcinoma. Gynecol Oncol. 2008;111(2):307–311. doi:10.1016/j.ygyno.2008.07.005
13. Collaboration CfCCM-A. Reducing uncertainties about the effects of chemoradiotherapy for cervical cancer: a systematic review and meta-analysis of individual patient data from 18 randomized trials. J Clin Oncol. 2008;26:5802–5812.
14. Zhang J. Neoadjuvant chemotherapy followed by radical hysterectomy for stage IB2-to-IIB cervical cancer: a retrospective cohort study. Int J Clin Oncol. 2019.
15. Yang Z, Chen D, Zhang J, et al. The efficacy and safety of neoadjuvant chemotherapy in the treatment of locally advanced cervical cancer: A randomized multicenter study. Gynecol Oncol. 2016;141(2):231–239. doi:10.1016/j.ygyno.2015.06.027
16. Katsumata N, Yoshikawa H, Kobayashi H, et al. Phase III randomised controlled trial of neoadjuvant chemotherapy plus radical surgery vs radical surgery alone for stages IB2, IIA2, and IIB cervical cancer: a Japan Clinical Oncology Group trial (JCOG 0102). Br J Cancer. 2013;108(10):1957–1963. doi:10.1038/bjc.2013.179
17. Kim HS, Sardi JE, Katsumata N, et al. Efficacy of neoadjuvant chemotherapy in patients with FIGO stage IB1 to IIA cervical cancer: an international collaborative meta-analysis. Eur J Surg Oncol. 2013;39(2):115–124. doi:10.1016/j.ejso.2012.09.003
18. Tang J, Tang Y, Yang J, Huang S. Chemoradiation and adjuvant chemotherapy in advanced cervical adenocarcinoma. Gynecol Oncol. 2012;125:297–302.
19. Lan M, Chen C, Huang Y, et al. Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in nasopharyngeal carcinoma patients with cervical nodal necrosis. Sci Rep. 2017;7(1):42624. doi:10.1038/srep42624
20. Kang S, Nam BH, Park JY, et al. Risk assessment tool for distant recurrence after platinum-based concurrent chemoradiation in patients with locally advanced cervical cancer: a Korean gynecologic oncology group study. J Clin Oncol. 2012;30:2369–2374. doi:10.1200/JCO.2011.37.5923
21. Green JA, Kirwan JM, Tierney JF, et al. Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix: a systematic review and meta-analysis. Lancet. 2001;358(9284):781–786. doi:10.1016/S0140-6736(01)05965-7
22. Huang EY, Huang YJ, Chanchien CC, et al. Pretreatment carcinoembryonic antigen level is a risk factor for para-aortic lymph node recurrence in addition to squamous cell carcinoma antigen following definitive concurrent chemoradiotherapy for squamous cell carcinoma of the uterine cervix. Radiat Oncol. 2012;7:13. doi:10.1186/1748-717X-7-13
23. Hong JH, Tsai CS, Lai CH, et al. Risk stratification of patients with advanced squamous cell carcinoma of cervix treated by radiotherapy alone. Int J Radiat Oncol Biol Phys. 2005;63:492–499. doi:10.1016/j.ijrobp.2005.02.012
24. Sedlis A, Bundy BN, Rotman MZ, et al. A randomized trial of pelvic radiation therapy versus no further therapy in selected patients with stage IB carcinoma of the cervix after radical hysterectomy and pelvic lymphadenectomy: A Gynecologic Oncology Group Study. Gynecol Oncol. 1999;73(2):177–183. doi:10.1006/gyno.1999.5387
25. Ferrandina G, Corrado G, Vitrano G, et al. Dose-dense paclitaxel/carboplatin as neo-adjuvant chemotherapy followed by radical surgery in locally advanced cervical cancer: a prospective phase II study. Cancer Chemother Pharmacol. 2019;83(3):431–438. doi:10.1007/s00280-018-3742-1
26. Ferrandina G, Palluzzi E, Gallotta V, et al. Neo-adjuvant platinum-based chemotherapy followed by chemoradiation and radical surgery in locally advanced cervical cancer (Lacc) patients: A phase II study. Eur J Surg Oncol. 2018;44(7):1062–1068. doi:10.1016/j.ejso.2018.04.019
27. Reesink-Peters N, van der Velden J, Ten HKA, et al. Preoperative serum squamous cell carcinoma antigen levels in clinical decision making for patients with early-stage cervical cancer. J Clin Oncol. 2005;23(7):1455–1462. doi:10.1200/JCO.2005.02.123
28. Tahmasbi Rad M, Wallwiener M, Rom J, et al. Learning curve for laparoscopic staging of early and locally advanced cervical and endometrial cancer. Arch Gynecol Obstet. 2013;288(3):635–642. doi:10.1007/s00404-013-2787-y
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
