Back to Journals » International Journal of Women's Health » Volume 7
Cost effectiveness of endometrial ablation with the NovaSure® system versus other global ablation modalities and hysterectomy for treatment of abnormal uterine bleeding: US commercial and Medicaid payer perspectives
Authors Miller J, Lenhart GM, Bonafede M, Basinski CM, Lukes AS, Troeger KA
Received 27 September 2014
Accepted for publication 30 October 2014
Published 6 January 2015 Volume 2015:7 Pages 59—73
DOI https://doi.org/10.2147/IJWH.S75030
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Elie Al-Chaer
Jeffrey D Miller,1 Gregory M Lenhart,1 Machaon M Bonafede,1 Cindy M Basinski,2 Andrea S Lukes,3 Kathleen A Troeger4
1Truven Health Analytics, Cambridge, MA, 2Basinski, LLC, Newburgh, IN, 3Carolina Women’s Research and Wellness Center, Durham, NC, 4Hologic, Inc, Marlborough, MA, USA
Objectives: Abnormal uterine bleeding (AUB) interferes with physical, emotional, and social well-being, impacting the quality of life of more than 10 million women in the USA. Hysterectomy, the most common surgical treatment of AUB, has significant morbidity, low mortality, long recovery, and high associated health care costs. Global endometrial ablation (GEA) provides a surgical alternative with reduced morbidity, cost, and recovery time. The NovaSure® system utilizes unique radiofrequency impedance-based GEA technology. This study evaluated cost effectiveness of AUB treatment with NovaSure ablation versus other GEA modalities and versus hysterectomy from the US commercial and Medicaid payer perspectives.
Methods: A health state transition (semi-Markov) model was developed using epidemiologic, clinical, and economic data from commercial and Medicaid claims database analyses, supplemented by published literature. Three hypothetical cohorts of women receiving AUB interventions were simulated over 1-, 3-, and 5-year horizons to evaluate clinical and economic outcomes for NovaSure, other GEA modalities, and hysterectomy.
Results: Model analyses show lower costs for NovaSure-treated patients than for those treated with other GEA modalities or hysterectomy over all time frames under commercial payer and Medicaid perspectives. By Year 3, cost savings versus other GEA were $930 (commercial) and $3,000 (Medicaid); cost savings versus hysterectomy were $6,500 (commercial) and $8,900 (Medicaid). Coinciding with a 43%–71% reduction in need for re-ablation, there were 69%–88% fewer intervention/reintervention complications for NovaSure-treated patients versus other GEA modalities, and 82%–91% fewer versus hysterectomy. Furthermore, NovaSure-treated patients had fewer days of work absence and short-term disability. Cost-effectiveness metrics showed NovaSure treatment as economically dominant over other GEA modalities in all circumstances. With few exceptions, similar results were shown for NovaSure treatment versus hysterectomy.
Conclusion: Model results demonstrate strong financial favorability for NovaSure ablation versus other GEA modalities and hysterectomy from commercial and Medicaid payer perspectives. Results will interest clinicians, health care payers, and self-insured employers striving for cost-effective AUB treatments.
Keywords: NovaSure, abnormal uterine bleeding, menorrhagia, hysterectomy, global endometrial ablation, cost-effectiveness analysis
Introduction
Abnormal uterine bleeding (AUB) interferes with physical, emotional, and social well-being, impacting the quality of life of more than 10 million women in the USA.1–12 Nearly one-third of all gynecologic office visits are related to AUB.2,8,13 AUB also has substantial cost implications, with estimated annual direct costs in the USA ranging $1–$1.55 billion and indirect costs ranging $12–$36 billion.12 Hysterectomy, a common surgical treatment of AUB, has significant morbidity, long recovery, and high associated health care costs.7,9,14 Global endometrial ablation (GEA) provides a surgical alternative with reduced morbidity, cost, and recovery time.3,7,8,14,15 NovaSure® endometrial ablation (Hologic, Inc, Marlborough, MA, USA) utilizes a radiofrequency impedance-based GEA technology. Since the US Food and Drug Administration approval of NovaSure in 2001,16 substantial data have been generated that support the favorable safety and efficacy profile for use of the procedure in premenopausal women for the treatment of AUB.17–28 Compared to other GEA techniques, the NovaSure procedure has the fastest treatment time, requires no hormonal pretreatment, and can be performed at any time during the menstrual cycle within a hospital or office setting.17–28
Although cost-effective treatments of AUB are particularly appealing in today’s cost-conscious health care environment, which values safe and efficacious treatments that reduce the costs associated with inpatient care,15 costs and cost-effectiveness information about surgical treatment of AUB with GEA and hysterectomy from a US payer perspective have been sparsely published. Cost effectiveness of the NovaSure procedure relative to other GEA modalities and hysterectomy has never been formally evaluated. In this study, we developed a health economic model to evaluate the cost effectiveness of AUB treatment with the NovaSure procedure versus other GEA modalities and versus hysterectomy from the US commercial and Medicaid payer perspectives.
Methods
Model structure and target population
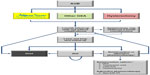
A health state transition (semi-Markov) model was developed using TreeAge Pro 2013 (TreeAge Software, Inc, Williamstown, MA, USA). Three hypothetical cohorts of women receiving AUB interventions were simulated over 1-, 3-, and 5-year horizons in monthly iterations or “cycles” to evaluate clinical and economic outcomes for NovaSure treatment, other GEA modalities, and hysterectomy (Figure 1). Other GEA modalities include second-generation GEA techniques (eg, cryotherapy, microwave endometrial ablation, thermal balloon endometrial ablation, hydrothermal ablation) other than bipolar radiofrequency ablation with the NovaSure system. “Hysterectomy” includes laparoscopic/robotic-assisted hysterectomy, vaginal hysterectomy, and abdominal hysterectomy.
The focus of the modeling analyses was specifically on the stratum of premenopausal women for whom childbearing is complete and who seek a permanent, nonreversible, one-time treatment option for their AUB with the two choices of GEA or hysterectomy. It is implicit that these women may have used medical management (eg, levonorgestrel intrauterine system [LNG-IUS], pharmacological therapy) prior to treatment for their AUB, per American College of Obstetricians and Gynecologists and other treatment guidelines.1,29 In some cases, these other forms of treatment may have failed or, for a variety of reasons, they were not a viable initial option. Regardless, the model analyses initiate on the specific day when each woman in the simulated cohorts underwent her GEA or hysterectomy procedure.
Model simulations begin at the point where AUB treatment is initiated. Site of procedure included office, ambulatory, and inpatient facilities, as naturalistically observed in the underlying claims data used to populate the model parameters. Average starting age in all three cohorts was assumed to be 42 years, consistent with the underlying clinical and economic data30,31 and demographically similar to women cohorts in other economic models of surgical AUB treatment.32–36 Menopause was assumed to occur on or after age 52 (ie, beyond the 1–5-year base case analysis time frame and beyond the maximum 10-year time simulation the model was capable of generating for sensitivity analysis testing) and was not factored into the model calculations, also consistent with other economic models of surgical AUB treatment. In any given model cycle, patients either could remain in or move between 21 discrete health states to replicate the natural course of postsurgical AUB treatment over time. Time-varying transition probabilities, costs, and quality-of-life values were ascribed to each health state and differ according to the characteristics of the particular treatment options being compared. At the end of each simulation, incremental differences in cumulative total costs and outcomes between the treatments were used to generate metrics of cost effectiveness typically used by health care decision makers.
Two versions of the model were created – one containing clinical and economic data oriented from the US commercial health care payer perspective and the other from a US Medicaid perspective. Underlying clinical and cost data differed in the two versions of the model, but the structure and functional operation were identical, with the exception that the commercial payer perspective version of the model was programmed to generate outputs associated with work absence and short-term disability and the associated “indirect costs” from these productivity losses.
The approach used for developing the model is entirely consistent with other economic models of AUB treatment comparing GEA and hysterectomy that have been developed in recent years.32–36
Data inputs and sources
Datatabase analyses
Data inputs for the model and the sources from which they were derived are listed in Tables 1–6. Most clinical and economic data (including treatment patterns, health state transition probabilities, health care resource utilization, direct costs, and productivity costs) were primarily derived from de novo analyses of three large medical claims databases: 1) the Truven Health MarketScan® Commercial Claims and Encounters Database, 2) the Truven Health MarketScan® Medicaid Multi-State Database, and 3) the Truven Health MarketScan® Health Productivity and Management Database.30,31,37 These databases provide information on individuals covered by a variety of employer-sponsored private health insurance plans and employer-paid Medicare supplemental insurance and are considered nationally representative of persons with employer-sponsored health insurance with respect to geography, age, and gender. The database analyses underlying this study focused in a naturalistic, real-world manner on 63,482 women aged 30–55 years with diagnosed AUB who initiated treatment with GEA or hysterectomy (index event) during 2006–2010. Twelve months of continuous enrollment pre- and post-index event were required, and health care utilization and costs assessed in the year following treatment initiation. Probabilities and costs of reintervention were tracked for 3 years following GEA. Workplace absenteeism and short-term disability costs were reported for a subset of patients from the commercial database analyses. More detailed description of the methods and results of these database analyses has been published elsewhere.30,31,37 The small amount of data pertaining to quality of life (ie, utilities) and mortality not obtainable from these database analyses were derived from published literature and various other published and unpublished sources, as documented in Tables 4 and 6.
  | Table 1 Direct costs |
  | Table 2 Monthly probabilities of reintervention and adjunctive/alternative pharmacotherapy† |
  | Table 3 Probabilities of intervention and reintervention complications |
  | Table 5 Work impairment (commercial health care payer perspective) |
  | Table 6 Mortality (both commercial and Medicaid payer perspectives) |
Because of the manner in which GEA insurance claims are coded (ie, no coding differentiation between GEA types/techniques), model parameters pertaining to NovaSure were assumed to be identical to those of GEA, with two exceptions – NovaSure-specific efficacy and safety data (ie, treatment complication rates and reintervention rates) were derived from a literature review and synthesis of 16 published studies (2002–2012) of women utilizing NovaSure treatment for AUB.18–21,23–25,28,38–45 NovaSure treatment reintervention rates (Table 2) and treatment complication rates (Table 3) were found to be relatively low compared to those for other GEA modalities.
Interventions and reinterventions
As mentioned above, different types/techniques of GEA could not be discerned from coding of GEA payment claims within the claims database analyses, so other GEA was modeled as a composite of all GEA types/techniques. First-generation endometrial ablation techniques were excluded from the data analyses. Similarly, hysterectomy was modeled as a composite of laparoscopic/robotic-assisted hysterectomy, vaginal hysterectomy, and abdominal hysterectomy as observed in real-world practice through the claims database analyses. Per the database analyses, a small probability of reintervention after NovaSure and other GEA with LNG-IUS was allowed in the model. Similarly, tranexamic acid also was included in the model as a reintervention option, but limited available claims data for tranexamic acid necessitated substituting some data pertaining to non-intrauterine device hormonal therapies as a proxy. Use of adjunctive/alternative pharmacotherapy with non-intrauterine device hormonal therapy, comprising the large array of available combination oral contraceptives, conjugated estrogens, and progestin-only pills, was accounted for in the model calculations as well. As summarized in Table 2, three sets of reintervention probabilities and reintervention-type distributions are featured in the model pertaining to the 3 sequential years after initial AUB intervention. All values were held constant at the third-year values in the fourth and fifth year of the analyses.
Complications
Probabilities of complications in the first month (30 days) post-intervention or reintervention are listed in Table 3. Complications included are cervical occlusion, cervical trauma (including cervical lacerations and hematometra), uterine perforation, bowel perforation, fluid overload, pregnancy, pyometra, cervicitis, device complications; a subsequent AUB treatment within 30 days, an inpatient stay, or emergency room visit within 2 days of the intervention or reintervention procedure (excluding patients with an inpatient stay for that procedure). All complications data were derived from the MarketScan® database analyses.30,31,37
Costs
As economic inputs to the model, direct costs of intervention and 30-day follow-up for NovaSure, other GEA, and hysterectomy are shown in Table 1, as are the reintervention and 30-day direct costs. Direct costs of NovaSure patients were assumed to be the same as other GEA. Intervention/reintervention costs and associated 30-day follow-up costs encompass “all health care expenditure”, not just gynecological-related health care expenditure. However, chronic monthly costs for patients in a post-intervention/reintervention “well” health state are specifically gynecological-related health care expenditures. All post-intervention costs were assumed to remain constant indefinitely within a simulation, changing only when reintervention is encountered and new post-reintervention costs are ascribed. All cost estimates were derived from the MarketScan® database analyses30,31,37 and expressed as 2013 US dollars (adjusted where necessary using the Medical Care component of the US Consumer Price Index).
Health state utility values
Health state utility scores (between 0= death and 1= perfect health) were used to calculate quality-adjusted life-years (QALYs). As documented in Table 4, utility data were derived from other published economic models of AUB which had obtained estimates from patient-reported outcome studies with quality-of-life assessments.
Work impairment
Work impairment was defined in the model as all-cause absence from work and work loss due to short-term disability. As inputs to the commercial payer version of the model, Table 5 shows the monthly probability of work absence or work loss from short-term disability and the corresponding number of days of lost work. These estimates were derived from the MarketScan® database analyses.30,31,37
Model analysis outcomes
Analytical results generated by the model include an assortment of comparative clinical and economic outcomes, with presentation of incremental cost-effectiveness ratios (ICERs) for NovaSure versus other GEA and NovaSure versus hysterectomy. Clinical outcomes include total and incremental QALYs, total and incremental cases of treatment complications, and total and incremental reinterventions and reintervention hysterectomies (for the NovaSure versus other GEA analyses only). Work productivity outcomes (US commercial payer perspective only) included total and incremental number of work days lost due to worker absence and short-term disability. Economic outcomes include total and incremental direct costs due to worker absence and short-term disability. Cost-effectiveness outcomes include cost per QALY, cost per complication case avoided, cost per reintervention avoided, cost per reintervention hysterectomy avoided, and cost per work day saved.
Analysis time horizon
Although the model was programmed to simulate any time horizon from 1 month up to 5 years (and 10 years as a sensitivity analysis), we selected a few key time points over a 5-year horizon that would be of universal interest, and they are referred to here as Year 1, Year 3, and Year 5.
Sensitivity analyses
A variety of univariate and probabilistic sensitivity analyses (PSAs) were conducted to test the robustness of model parameter values and their impact on the ICERs. Using a second-order Monte-Carlo simulation (1,000 iterations), PSA was conducted on the model’s primary cost-effectiveness outcome and incremental cost per incremental QALY. The parameter values of each probability distribution were calculated from the mean and standard error of the model input parameters. By drawing randomly from those distributions, a large number of estimates of costs, QALYs, and ICERs were generated, thus testing the consequences of varying input parameters. Targeted one-way deterministic sensitivity analyses were performed on the ICERs featuring complications, reinterventions, reintervention hysterectomies, and work productivity. A 10-year analysis scenario was performed to test robustness of the model estimates over long time horizons.
Discounting
Both costs and survival estimates (ie, QALYs) were discounted at an annual discount rate of 3%.
Results
Results of the model analyses show lower total direct costs for NovaSure-treated patients than for patients treated with other GEA modalities or hysterectomy over all time frames under both the commercial payer and Medicaid perspectives (Table 7). As an example, Year 1 direct costs for NovaSure patients were $6,602 and $4,419 in the commercial payer and Medicaid payer perspectives, respectively, which were substantially less than corresponding costs for GEA patients ($7,180 and $6,158, respectively) and less than one-half of the corresponding costs of hysterectomy ($13,221 and $12,716, respectively). The cost differential between the treatments narrowed somewhat over time, but even at Year 3 and Year 5 NovaSure costs were still one-third less than hysterectomy costs. Looking at the incremental differences between NovaSure treatment costs and the comparators, direct cost savings from NovaSure treatment versus other GEA modalities ranged from $578 to $1,309 (commercial payer perspective) and $1,739 to $4,372 (Medicaid perspective) in Year 1 and Year 5, respectively. More dramatic were the estimated potential cost savings for NovaSure treatment versus hysterectomy, which ranged from $6,619 to $6,208 (commercial payer perspective) and $8,297 to $9,259 (Medicaid perspective) in Year 1 and Year 5, respectively.
In the commercial payer perspective analysis, model estimates show that NovaSure-treated patients should experience up to 11% fewer days of work absence and short-term disability when compared with other GEA patients, and up to 47% fewer work days lost compared with hysterectomy patients. For example, in the 1-year analysis, NovaSure patients lost 29.4 days work versus 29.7 and 55.3 work days lost for other GEA and hysterectomy patients, respectively. Corresponding indirect costs were $7,412 for NovaSure patients versus $7,491 and $13,924 for other GEA and hysterectomy patients, respectively.
Quality-of-life outcomes, measured in terms of QALYs, were higher for NovaSure-treated patients than for those treated with other GEA over all time frames under both the commercial payer and Medicaid perspectives (Table 7). For NovaSure versus hysterectomy, QALYs were higher for NovaSure in the Year 1 analysis results, but favorability shifted to hysterectomy in the Year 3 and Year 5 results – a phenomenon attributable to the inherently higher long-term utility weights assigned to hysterectomy, but also due to the cumulative disutility experienced by NovaSure and other GEA patients undergoing repeat ablation and other forms of reintervention naturally avoided by hysterectomy patients. However, the magnitude of the difference in QALYs between NovaSure treatment and hysterectomy is quite small – even by Year 5, QALYs are only about 3% (0.12 QALYs) lower for NovaSure treatment in both the commercial payer and Medicaid scenarios.
Clinical outcomes (ie, reinterventions and occurrence of intervention/reintervention complications) from the modeling analyses bear special mention. From the NovaSure versus other GEA analyses, results show NovaSure-treated patients associated with a 43%–71% reduction in need for reintervention by re-ablation, hysterectomy, or nonsurgical means. Looking at reintervention hysterectomies alone, NovaSure-treated patients had 80% fewer reintervention hysterectomies in the commercial payer analyses, with even larger reductions estimated in the Medicaid analyses. Furthermore, the model predicts 69%–88% fewer intervention/reintervention complications for NovaSure-treated patients versus other GEA modalities, and 82%–91% fewer complications versus hysterectomy. For example, whereas 17.1% of other GEA patients and 35.6% of hysterectomy patients had complications in the first year under the commercial payer perspective, only 4.3% of NovaSure patients had complications. The gap narrows over time as the initial complications of hysterectomy remain constant, while reintervention complications for NovaSure and GEA patients gradually accumulate. However, even by Year 5, NovaSure-treated patients had 69% fewer complications than other GEA patients and 81% fewer complications than hysterectomy patients. Results were similar under the Medicaid perspective.
Cost-effectiveness metrics show NovaSure treatment as economically dominant over other GEA modalities in all circumstances. This represents the potential for improved outcomes among women treated with the NovaSure system (ie, greater QALYs, fewer complications, fewer reinterventions, or fewer work days lost) at lower cost compared with other GEA modalities. Similar results were shown for the NovaSure procedure versus hysterectomy, where NovaSure was typically the dominant strategy (ie, always conferring greater benefit at lower cost). Exceptions were found for the cost-effectiveness metrics involving QALYs in scenarios for Years 3 and 5, where NovaSure treatment conferred cost savings compared with hysterectomy but with reduced benefits (ie, fewer QALYs), resulting in decremental cost-effectiveness ratios ranging from $51,123 to $118,846 saved per QALY lost (commercial perspective) and from $80,902 to $187,884 saved per QALY lost (Medicaid perspective).
Results were not highly sensitive to univariate and probabilistic variation in the parameter values – that is, the changes did not appreciably alter the ICERs (including economic dominance), nor did they generally affect conclusions about the cost effectiveness of the NovaSure procedure. Focusing specifically on the cost-per-QALY ICERs in the 5-year scenario, where the analysis results were somewhat anomalous by showing decremental cost effectiveness for the NovaSure procedure versus hysterectomy, none of the plotted ICER points fell into the upper quadrants of the cost-effectiveness plane – ie, NovaSure treatment consistently always costs less than other GEA modalities and hysterectomy, even under the extremes of probabilistic sampling. For NovaSure procedures versus other GEA, about 50% of the ICERs plotted into the lower left and right quadrants of the cost-effectiveness plane, indicating an even split between decremental and positive cost effectiveness for NovaSure treatment. For NovaSure procedures versus hysterectomy, most (66%) of the ICERs plotted into the lower left quadrant of the plane where decremental cost effectiveness occurs, but over one-third (34%) of the probabilistically generated ICERs plotted into the lower right quadrant of the plane where NovaSure treatment dominates hysterectomy by simultaneously conferring cost savings and more QALYs. As an additional sensitivity analysis, a special 10-year scenario simulation was performed to explore results projected over a long time horizon. Incremental cost differences remained highly favorable for the NovaSure procedure in both the commercial payer and Medicaid perspectives, and indirect costs attributable to work absence and short-term disability leave in the commercial payer perspective also remained advantageous. Cost-effectiveness results seen at Year 5 continued a trend into Year 10, with the NovaSure procedure mostly showing economic dominance over other GEA and hysterectomy in both the commercial and Medicare perspectives.
Discussion
Results of this economic modeling study of AUB show that the direct and indirect costs of NovaSure treatment of AUB are substantially lower than those for other GEA modalities and hysterectomy over short (1-year) and longer (5-year) time horizons, and under both commercial payer and Medicaid perspectives. By 5 years after intervention, direct costs for NovaSure patients are $1,300–$4,400 less compared to those for other GEA patients, and $6,200–$9,300 less compared with hysterectomy patients. Similar results were found for indirect costs, under the commercial payer perspective. Combining these cost savings with clinical and quality-of-life benefits of treatment yielded cost-effectiveness metrics favoring the NovaSure procedure over other GEA modalities and hysterectomy as an AUB treatment strategy. In almost all scenarios analyzed, NovaSure treatment was found to be the economically dominant strategy over other GEA and hysterectomy – that is, NovaSure treatment confers better clinical outcomes and quality-of-life benefits at less cost.
A conservative estimate of the annual indirect economic costs of AUB in the USA is $12 billion,12 which reveals impaired work productivity due to AUB having substantial economic implications, both for individual women and for society.12,46–51 From an employer’s perspective, AUB is an expensive condition, and choice of treatments can profoundly affect the direction of those costs.30,46 Results generated by our model indicate that in the initial year following surgical treatment of AUB, NovaSure-treated patients had almost 26 fewer days of work absence and short-term disability leave compared with hysterectomy patients, leading to an indirect cost (ie, work loss) savings of greater than $6,500. The incremental difference narrows over time, but even by 5 years, NovaSure accounts for more than 30% fewer days of work loss and associated indirect costs. As would be expected, given the technical similarities between the NovaSure procedure and other GEA modalities, comparative results for NovaSure treatment versus other GEA are more modest.
The Medicaid perspective analyses from this study are particularly relevant given recent expansion of the US Medicaid program and the creation of new state-based and federally facilitated competitive marketplaces, or Affordable Insurance Exchanges. Using the average of published AUB prevalence statistics of about 20%,9–12 it follows that a sizeable number of newly eligible women in the expanded Medicaid program will have AUB, many of whom will go on to receive costly pharmacologic and surgical treatments. Hysterectomies will be of particular financial concern, not only because of their high cost but also because they comprise about 27% of the total pharmacologic and surgical AUB interventions that are performed each year in the Medicaid population.31,37 Results of our Medicaid perspective analyses suggest that the NovaSure procedure may save substantial costs – as much as $8,300 per patient in the initial year following intervention and as much as $9,300 by 5 years. Consequently, state-based and federally facilitated exchanges may focus intently on GEA (particularly on less costly, in-office procedures with the NovaSure procedure) as ways to reduce costs.
Limitations
The focus of the analyses was on women who sought and underwent GEA (including NovaSure) or hysterectomy as their chosen AUB treatment option. The analyses did not extend to the greater realm of medical management of AUB and, therefore, we did not evaluate the cost effectiveness of LNG-IUS and pharmacologic therapies in comparison with NovaSure, other GEA, and hysterectomy. There were some data limitations in our study, most of which are inherently tied to using an administrative health care database as the primary data source for the economic model. First, as observed in other retrospective claims database analyses, the most completely recorded data are those that affect reimbursement. Hence, the capture of expenditures is highly accurate, but data on comorbidities and disease severity may not be as carefully recorded. Second, procedure coding in claims data is limited in its ability to differentiate specific types/techniques of GEA or hysterectomy. Analyses of these different procedure types may have revealed differences in clinical and cost outcomes from the model, but it is not possible to speculate about the magnitude and direction of those differences. Third, due to the structure of the underlying claims database analyses, we did not conduct age-stratified subgroup analyses with the model, although patient age may correlate with clinical and economic outcomes. Fourth, the indirect costs included in this analysis are not exhaustive. For example, absenteeism and short-term disability claims do not include transportation costs to and from medical appointments, caregiver time, childcare time, long-term disability, or reduced productivity while at work. Finally, the model’s underlying data pertained to patients with commercial insurance or Medicaid supplemental insurance; therefore, the analyses results may not represent all patients with AUB, especially those uninsured or covered under other Medicaid programs. We also note that although the literature provided health state utility values sufficient for use in the economic model, they are mostly from older studies, some of which predate the advent of second-generation endometrial ablation technologies (notably, the extensively referenced 1998 cost-utility analysis by Sculpher).52 QALY metrics in the analysis results were fairly sensitive to changes in utility values, but it is difficult to say how the results would have changed if more current, robust utility data were available.
Conclusion
The role of GEA in the surgical treatment of AUB has grown in the USA and the NovaSure procedure has secured a strong position in the AUB treatment market. This high regard for NovaSure treatment is reflected by the results of our economic model, which show strong financial favorability for the NovaSure procedure over other GEA modalities and over hysterectomy from both the commercial payer and Medicaid payer perspectives. Results from this study will interest the US commercial health payers and self-insured employers seeking cost-effective treatment for AUB. Similarly, new federal mandates for Medicaid expansion are likely to focus on the attractiveness of outpatient treatments like the NovaSure procedure and other GEA modalities to reduce Medicaid costs.
Disclosure
Portions of this study were presented as a poster (#PIH19) at the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 19th Annual International Meeting, Montreal, QC, Canada, May 31–June 4, 2014. Funding for this study was provided by Hologic, Inc (Marlborough, MA, USA). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
References
American College of Obstetricians and Gynecologists (ACOG), Committee on Practice Bulletins – Gynecology. Practice bulletin no 136: management of abnormal uterine bleeding associated with ovulatory dysfunction. Obstet Gynecol. 2013;122(1):176–185. | |
American Congress of Obstetricians and Gynecologists (ACOG), Committee on Practice Bulletins – Gynecology. Practice bulletin no 128: diagnosis of abnormal uterine bleeding in reproductive-aged women. Obstet Gynecol. 2012;120(1):197–206. | |
Fox KE. Management of heavy menstrual bleeding in general practice. Curr Med Res Opin. 2012;28(9):1517–1525. | |
Munro MG, Critchley HO, Fraser IS. The FIGO classification of causes of abnormal uterine bleeding: Malcolm G Munro, Hilary OD Crithcley, Ian S Fraser, for the FIGO Working Group on Menstrual Disorders. Int J Gynaecol Obstet. 2011;113(1):3–13. | |
UK National Institute for Clinical Excellence (NICE). Heavy Menstrual Bleeding: Clinical Guideline 44; 2007. Available from: http://www.nice.org.uk/nicemedia/pdf/CG44FullGuideline.pdf. Accessed October 18, 2014. | |
Fraser IS, Critchley HO, Munro MG, Broder M. Can we achieve international agreement on terminologies and definitions used to describe abnormalities of menstrual bleeding? Hum Reprod. 2007;22(3):635–643. | |
Apgar BS, Kaufman AH, George-Nwogu U, Kittendorf A. Treatment of menorrhagia. Am Fam Physician. 2007;75(12):1813–1819. | |
Osei J, Critchley H. Menorrhagia, mechanisms and targeted therapies. Curr Opin Obstet Gynecol. 2005;17(4):411–418. | |
Oehler MK, Rees MC. Menorrhagia: an update. Acta Obstet Gynecol Scand. 2003;82(5):405–422. | |
Sweet MG, Schmidt-Dalton TA, Weiss PM, Madsen KP. Evaluation and management of abnormal uterine bleeding in premenopausal women. Am Fam Physician. 2012;85(1):35–43. | |
Centers for Disease Control and Prevention (CDC). Blood Disorders in Women: Heavy Menstrual Bleeding; 2011. Available from: http://www.cdc.gov/ncbddd/blooddisorders/women/menorrhagia.html. Accessed October 18, 2014. | |
Liu Z, Doan QV, Blumenthal P, Dubois RW. A systematic review evaluating health-related quality of life, work impairment, and health-care costs and utilization in abnormal uterine bleeding. Value Health. 2007;10(3):183–194. | |
Spencer CP, Whitehead MI. Endometrial assessment re-visited. Br J Obstet Gynaecol. 1999;106(7):623–632. | |
Stovall DW. Alternatives to hysterectomy: focus on global endometrial ablation, uterine fibroid embolization, and magnetic resonance-guided focused ultrasound. Menopause. 2011;18(4):437–444. | |
Canali C. Global endometrial ablation for menorrhagia: the office option. OBG Manage. 2012;Suppl:S1–S8. Available from: http://www.obgmanagement.com/uploads/media/SupplOBG_Omnia1.pdf. Accessed October 18, 2014. | |
US Food and Drug Administration (FDA). NovaSure™ Impedance Controlled Endometrial Ablation System – P010013; 2001. Available from: http://www.accessdata.fda.gov/cdrh_docs/pdf/P010013a.pdf. Accessed October 18, 2014. | |
Gimpelson RJ. Ten-year literature review of global endometrial ablation with the NovaSure® device. Int J Womens Health. 2014;6:269–280. | |
Cooper J, Gimpelson R, Laberge P, et al. A randomized, multicenter trial of safety and efficacy of the NovaSure system in the treatment of menorrhagia. J Am Assoc Gynecol Laparosc. 2002;9(4):418–428. | |
Cooper J, Gimpelson RJ. Summary of safety and effectiveness data from FDA: a valuable source of information on the performance of global endometrial ablation devices. J Reprod Med. 2004;49(4):267–273. | |
Clark TJ, Samuel N, Malick S, Middleton LJ, Daniels J, Gupta JK. Bipolar radiofrequency compared with thermal balloon endometrial ablation in the office: a randomized controlled trial. Obstet Gynecol. 2011;117(1):109–118. | |
Penninx JP, Mol BW, Engels R, et al. Bipolar radiofrequency endometrial ablation compared with hydrothermablation for dysfunctional uterine bleeding: a randomized controlled trial. Obstet Gynecol. 2010;116(4):819–826. | |
El-Nashar SA, Hopkins MR, Creedon DJ, et al. Prediction of treatment outcomes after global endometrial ablation. Obstet Gynecol. 2009;113(1):97–106. [Erratum in: Obstet Gynecol. Mar 2010; 115(3):663]. | |
Gallinat A. An impedance-controlled system for endometrial ablation: five-year follow-up of 107 patients. J Reprod Med. 2007;52(6):467–472. | |
Fulop T, Rákóczi I, Barna I. NovaSure impedance controlled endometrial ablation: long-term follow-up results. J Minim Invasive Gynecol. 2007;14(1):85–90. | |
Kleijn JH, Engels R, Bourdrez P, Mol BW, Bongers MY. Five-year follow up of a randomised controlled trial comparing NovaSure and ThermaChoice endometrial ablation. BJOG. 2008;115(2):193–198. | |
Bongers MY. Second-generation endometrial ablation treatment: Novasure. Best Pract Res Clin Obstet Gynaecol. 2007;21(6):989–994. | |
Laberge PY. NovaSure():the bipolar radiofrequency endometrial ablation system for dysfunctional uterine bleeding. Womens Health (Lond Engl). 2006;2(5):687–693. | |
Abbott J, Hawe J, Hunter D, Garry R. A double-blind randomized trial comparing the Cavaterm and the NovaSure endometrial ablation systems for the treatment of dysfunctional uterine bleeding. Fertil Steril. 2003;80(1):203–208. | |
American College of Obstetricians and Gynecologists. ACOG committee opinion no 557: management of acute abnormal uterine bleeding in nonpregnant reproductive-aged women. Obstet Gynecol. 2013;121(4):891–896. | |
Bonafede MM, Miller JD, Meyer NM, Lenhart GM. Comparative direct and indirect costs of menorrhagia treatment with global endometrial ablation or hysterectomy. Poster (#PIH17) presented at: The International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 18th Annual International Meeting, May 18–22; 2013; New Orleans, LA. | |
Bonafede MM, Miller JD, Meyer NM, Lenhart GM. Retrospective database analysis of clinical outcomes and costs for treatment of menorrhagia among women enrolled in US Medicaid programs. Poster (#PIH22) presented at: The International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 18th Annual International Meeting, May 18–22; 2013; New Orleans, LA. | |
Bhattacharya S, Middleton LJ, Tsourapas A, et al; International Heavy Menstrual Bleeding Individual Patient Data Meta-analysis Collaborative Group. Hysterectomy, endometrial ablation and Mirena® for heavy menstrual bleeding: a systematic review of clinical effectiveness and cost-effectiveness analysis. Health Technol Assess. 2011;15(19):iii–xvi. , 1–252. | |
Roberts TE, Tsourapas A, Middleton LJ, et al. Hysterectomy, endometrial ablation, and levonorgestrel releasing intrauterine system (Mirena) for treatment of heavy menstrual bleeding: cost effectiveness analysis. BMJ. 2011;342:d2202. | |
Clegg JP, Guest JF, Hurskainen R. Cost-utility of levonorgestrel intrauterine system compared with hysterectomy and second generation endometrial ablative techniques in managing patients with menorrhagia in the UK. Curr Med Res Opin. 2007;23(7):1637–1648. | |
Garside R, Stein K, Wyatt K, Round A, Price A. The effectiveness and cost-effectiveness of microwave and thermal balloon endometrial ablation for heavy menstrual bleeding: a systematic review and economic modelling. Health Technol Assess. 2004;8(3):iii. , 1–155. | |
Garside R, Stein K, Wyatt K, Round A, Pitt M. A cost-utility analysis of microwave and thermal balloon endometrial ablation techniques for the treatment of heavy menstrual bleeding. BJOG. 2004;111(10):1103–1114. | |
Bonafede MM, Miller JD, Laughlin-Tommaso SK, Lukes AS, Meyer NM, Lenhart GM. Retrospective database analysis of clinical outcomes and costs for treatment of abnormal uterine bleeding among women enrolled in US Medicaid programs. Clinicoecon Outcomes Res. 2014;6:423–429. | |
Baskett TF, Clough H, Scott TA. NovaSure bipolar radiofrequency endometrial ablation: report of 200 cases. J Obstet Gynaecol Can. 2005;27(5):473–476. | |
Busund B, Erno LE, Grønmark A, Istre O. Endometrial ablation with NovaSure GEA, a pilot study. Acta Obstet Gynecol Scand. 2003;82(1):65–68. | |
Campbell P, Monaghan C, Parker M. NovaSure endometrial ablation: a review of 400 cases. Gynecol Surg. 2012;9:73–76. | |
Claas MJ, Wirjosoekarto SAM, Bukman A, Lenters E. NovaSure® endometrial ablation: effectiveness and patient satisfaction. J Gynecol Surg. 2012;28(2):89–93. | |
Elmardi A, Furara S, Khan F, Hamza M. NovaSure impedance controlled system for endometrial ablation: the experience of the first UK reference centre. J Obstet Gynaecol. 2009;29(5):419–422. | |
El-Nashar SA, Hopkins MR, Creedon DJ, Cliby WA, Famuyide AO. Efficacy of bipolar radiofrequency endometrial ablation vs thermal balloon ablation for management of menorrhagia: a population-based cohort. J Minim Invasive Gynecol. 2009;16(6):692–699. | |
Ghazizadeh S, Panahi Z, Ghanbari Z, Menshadi AT, Farahmandian T, Javadian P. Comparative efficacy of NovaSure, the levonorgestrel-releasing intrauterine system, and hysteroscopic endometrial resection in the treatment of menorrhagia: randomized clinical trial. J Gynecol Surg. 2014;30(4):215–218. | |
Kalkat RK, Cartmill RS. NovaSure endometrial ablation under local anaesthesia in an outpatient setting: an observational study. J Obstet Gynaecol. 2011;31(2):152–155. | |
Jensen JT, Lefebvre P, Laliberté F, et al. Cost burden and treatment patterns associated with management of heavy menstrual bleeding. J Womens Health (Larchmt). 2012;21(5):539–547. | |
Pron G, Cohen M, Soucie J, et al; Ontario Uterine Fibroid Embolization Collaboration Group. The Ontario Uterine Fibroid Embolization Trial. Part 1. Baseline patient characteristics, fibroid burden, and impact on life. Fertil Steril. 2003;79(1):112–119. | |
Côté I, Jacobs P, Cumming D. Work loss associated with increased menstrual loss in the United States. Obstet Gynecol. 2002;100(4):683–687. | |
Hurskainen R, Aalto AM, Teperi J, et al. Psychosocial and other characteristics of women complaining of menorrhagia, with and without actual increased menstrual blood loss. BJOG. 2001;108(3):281–285. | |
Wasiak R, Filonenko A, Vanness DJ, et al. Impact of estradiol valerate/dienogest on work productivity and activities of daily living in women with heavy menstrual bleeding. J Womens Health (Larchmt). 2013;22(4):378–384. | |
Wasiak R, Filonenko A, Vanness DJ, et al. Impact of estradiol-valerate/dienogest on work productivity and activities of daily living in European and Australian women with heavy menstrual bleeding. Int J Womens Health. 2012;4:271–278. | |
Sculpher M. A cost-utility analysis of abdominal hysterectomy versus transcervical endometrial resection for the surgical treatment of menorrhagia. Int J Technol Assess Health Care. 1998;14(Spring 2):302–319. | |
Hurskainen R, Teperi J, Rissanen P, et al. Clinical outcomes and costs with the levonorgestrel-releasing intrauterine system or hysterectomy for treatment of menorrhagia: randomized trial 5-year follow-up. JAMA. 2004;291(12):1456–1463. | |
Maresh MJ, Metcalfe MA, McPherson K, et al. The VALUE national hysterectomy study: description of the patients and their surgery. BJOG. 2002;109(3):302–312. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.