Back to Journals » ClinicoEconomics and Outcomes Research » Volume 12
Cost-Effectiveness of a JAK1/JAK2 Inhibitor vs a Biologic Disease-Modifying Antirheumatic Drug (bDMARD) in a Treat-to-Target Strategy for Rheumatoid Arthritis
Authors Van De Laar CJ , Oude Voshaar MAH, Fakhouri WKH, Zaremba-Pechmann L, De Leonardis F, De La Torre I, Van De Laar MAFJ
Received 18 September 2019
Accepted for publication 25 February 2020
Published 15 April 2020 Volume 2020:12 Pages 213—222
DOI https://doi.org/10.2147/CEOR.S231558
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Giorgio Colombo
Celine J Van De Laar,1 Martijn AH Oude Voshaar,1,2 Walid KH Fakhouri,3 Liliana Zaremba-Pechmann,3 Francesco De Leonardis,3 Inmaculada De La Torre,3 Mart AFJ Van De Laar1,2
1Transparency in Healthcare BV, Hengelo, the Netherlands; 2Department of Psychology and Communication of Health and Risk, Institute for Behavioural Research, University of Twente, Enschede, the Netherlands; 3Eli Lilly & Company, Indianapolis, IN, USA
Correspondence: Celine J Van De Laar
Tel +31 74-43032645
Email [email protected]
Background: Baricitinib is a janus kinase (JAK1/JAK2) inhibitor developed for the treatment of patients suffering from rheumatoid arthritis (RA). Treating RA to the target of remission is current common practice. Cost-effectiveness of different treat-to-target (T2T) strategies, especially ones including new treatments is important for development and preference policy for treatment centers. European League Against Rheumatism (EULAR) and American College of Rheumatology (ACR) guidelines are currently unclear about preference between a JAK1/JAK2 versus a biological disease-modifying antirheumatic drug (bDMARD).
Objective: The main goal of this paper was to evaluate the cost-effectiveness of baricitinib versus first biological for methotrexate inadequate responders in a T2T strategy using a Markov model that incorporates hospital costs as well as societal costs. Costs and utilities over five years were compared between the two strategies.
Methods: A Monte Carlo simulation model was developed to conduct cost–utility analysis from the societal perspective over 5 years. Health states were based on the DAS28-erythrocyte sedimentation rate (ESR) categories. Effectiveness of baricitinib was retrieved from randomized controlled trials. Effectiveness of all other treatments, health state utilities, medical costs, and productivity loss were retrieved from the Dutch RhEumatoid Arthritis Monitoring (DREAM) cohorts. Annual discount rates of 1.5% for utility and 4% for costs were used. Probabilistic sensitivity analysis was employed to incorporate uncertainty and assess robustness of the results.
Results: Probabilistic sensitivity analysis results showed the baricitinib strategy yielded lower costs and higher utility over a 5-year period. Scenario analyses showed the baricitinib strategy to be cost-effective in both the moderate and severe RA populations.
Conclusion: Results suggest that the use of a JAK1/JAK2 inhibitor instead of a bDMARD in a T2T approach is cost-effective in csDMARD refractory RA patients.
Keywords: Markov model, rheumatoid arthritis, baricitinib, cost-effectiveness, health economic model, treat-to-target
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disease with alternating periods of low, moderate and/or high disease activity, usually characterized by inflammation of the synovium. RA can be chronic in which case it may lead to functional impairment and disability if left untreated.1 Accumulating evidence suggests that optimal treatment outcomes may be obtained with timely initiated tight control management of disease activity.
This approach to RA management, also referred to as Treat-to-Target (T2T), involves setting disease activity targets at the onset of treatment. Disease activity is then routinely measured using one of several available composite disease activity indices and medication adjustments are made contingent on the clinical composite score. If the disease activity target is not met, treatment is intensified (eg, by increasing doses, combining treatments, or switching to a different drug). If disease activity is on target during successive measurements, medications may be tapered.2
All major international management guidelines currently endorse a T2T approach to RA management.3,4 Within this general framework, the current recommended approach is to start patients on conventional synthetic disease-modifying antirheumatic drugs (csDMARDs). csDMARDs are a group of traditional RA medications. While csDMARDs have not been developed to target a molecular structure, they have become known for their disease-modifying properties and are recommended first-line agents in light of their favorable cost profile.5,6 If the treatment target cannot be achieved using csDMARDs, international guidelines recommend the initiation of a biological DMARD (bDMARD) or a synthetic drug designed to target particular molecular structures. This class of targeted synthetic DMARDs (tsDMARDs) is relatively new in rheumatology and international recommendations place them at the same level as bDMARDs, with the csDMARD inadequate responders (IR) population as the earliest possible time point for use in the therapeutic algorithm. Few studies have yet directly compared these agents. Of note, the janus kinase (JAK)1/JAK2-inhibitor baricitinib, now approved in more than 50 countries worldwide, was found to be statistically superior for American College of Rheumatology 20 criteria (ACR20) and disease activity score 28 – C-reactive protein (DAS28-CRP) mean change at week 12 against bDMARD adalimumab with background methotrexate (MTX), when administrated in combination with MTX in a population of moderate-to-severe RA patients who are MTX-IR.7 Currently, there is little information on the long-term cost-effectiveness of JAK-inhibitors versus bDMARDs as part of contemporary T2T-based management strategies.
The aim of this study was to simulate long-term patient outcomes and cost-effectiveness of two treatment strategies in which either a JAK1/JAK2-inhibitor (baricitinib) or a bDMARD is initiated in csDMARD-IR by developing a Monte Carlo simulation Markov model.
Materials and Methods
A cost–utility analysis (CUA) was conducted from the Dutch societal perspective using an individual-sampling Monte Carlo simulation Markov model. The Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist was followed in reporting of the analyses. The model was subjected to checks and validation by an external party (IQVIA).
Analytic Framework and Treatment Strategies
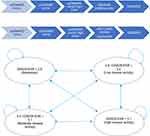
The model was developed using TreeAge Pro 2018, R2 (TreeAge Software, Williamstown, MA) to simulate changes in disease activity, health utility, and costs in patients with early RA (within one year after diagnosis) receiving treatment based on the T2T principle as applied in the Dutch RhEumatoid Arthritis Monitoring registry (DREAM) cohorts from the moment of diagnosis onwards.8,9 In the model, patients transition between disease activity states on a 12-weekly basis. Four commonly distinguished clinically relevant disease activity states based on the Disease Activity Score 28-erythrocyte sedimentation rate (DAS28-ESR) categories, are used in the model: Remission (DAS28-ESR≤2.6), low disease activity (2.6<DAS28-ESR≤3.2), moderate disease activity (3.2<DAS28-ESR≤5.1), and high disease activity (DAS28-ESR>5.1).10 Patients enter the model with an assigned disease activity state. Patient distribution over health states at baseline is based on the observed distribution in the DREAM cohorts. Patients can move from any state to any of the others states every cycle. See Figure 1 for a graphic display of all possible transitions. The transition probabilities depend on the current health state and the treatment that is currently used. This model structure was adapted from a model previously introduced by Welsing et al11,12 and extended by Schipper et al.13 Welsing et al have demonstrated that model-based disease activity trajectories over a period of 5 years closely approximate the disease activity course actually observed in the cohort of patients that transition probabilities were derived from.
In alignment with current management recommendations and the T2T principle, all treatment changes in the model in the present study are governed by the disease activity state of the patient. For all strategies, treatment was targeted at achieving DAS28-ESR remission.8,9 If a patient has been in non-remission for 1 cycle (12 weeks) the next treatment is selected. Patients that are in remission and remain there for 2 cycles (24 weeks) move back to previous medication. This so-called tapering of medication after two cycles is in accordance with the current rheumatology guidelines.3,4 Eventually, in case of long-term sustained remission, patients can reach medication-free remission through tapering. Patients that go through tapering will return to their last effective medication in case of a flare (DAS28-ESR>2.6). Due to reimbursement restrictions in the Netherlands, the first bDMARD will only be selected if DAS28-ESR>3.2, ie, moderate or high disease activity.
Two treatment strategies were evaluated and compared. In the first modelled strategy (DREAM T2T), patients are initially treated with a low-dose csDMARD combination therapy. In case remission is not achieved, higher-dose csDMARD combination is next, before a bDMARD (adalimumab), and ultimately a (second) bDMARD are initiated. Adalimumab was chosen as first bDMARD after inadequate response to TNF-α blockers due to its common use in the Netherlands and internationally. This is also reflected in data availability for effectiveness of TNF-α blockers. In the Baricitinib Strategy, the first bDMARD (adalimumab) is replaced by baricitinib. When patients experience sustained remission (two consecutive cycles spent in remission) their medication is tapered. In case of sustained remission, a patient moves back one treatment step. When the patient is in sustained remission again, he/she can move another step, until arriving at medication-free remission. There is an additional “taper step” between each strategy’s initial treatment and medication-free remission, this is MTX monotherapy in both strategies. For example, a patient in the DREAM T2T strategy on adalimumab who is in sustained remission moves back to MTX Combination therapy, in case of another sustained remission, moves to MTX Monotherapy (the additional taper step before medication-free remission), then in case of another sustained remission will go to medication-free remission. In case a flare happens at any point during the tapering process, the patient returns to the last effective treatment.
The difference between these strategies lies in the initiation of baricitinib vs a bDMARD in csDMARD-IR population. Patients enter the model upon diagnosis and are distributed over the four DAS28 health states as observed in the patient cohorts used as data input. See Figure 1 for an outline of the exact treatment strategies and health states used in the model.
Analyses
Expected values for costs and utility over five years were obtained and uncertainty around these estimates was quantified using probabilistic sensitivity analysis (PSA) with Monte Carlo simulation using 1500x150 samples. For each input parameter, several potentially appropriate distributions according to literature recommendations14 were selected and compared their relative fit using Chi-square tests. Costs and utility were discounted annually at 4% and 1.5%, respectively, according to the Dutch Costing Guidelines for Economic Evaluation.15
Additionally, scenario analyses were performed to explore what the effect would be of lowered drug prices. Specifically, analyses were run where lowered drug prices for adalimumab and baricitinib were put into the model. It can be supposed that due to the introduction of biosimilars, prices of expensive drugs and specifically adalimumab could be lower than the list prices. To take possible discounts or other agreements into consideration the favourability of the two strategies (DREAM T2T strategy and baricitinib 2nd strategy) will be assessed at different levels of lowered medication prices.
Model Inputs
All model inputs can be found in Tables 1, 2, and 3.
 |
Table 1 Model Input Distributions |
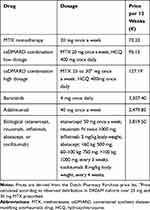 |
Table 2 Medication Costs |
Efficacy
Transition probabilities for each treatment step that does not involve baricitinib were estimated using data from the ongoing “Remission Induction in Early Rheumatoid Arthritis” (Netherlands Trial Register: NTR578) cohorts in the DREAM registry.8,9 Each matrix of observed transitions was filled using data of the first year of follow up (ie a maximum of four (~12 weekly ±~6 weeks) visits) of patients’ first and second therapies with that specific treatment. Transition probabilities were assumed to be independent of time and to occur in evenly spaced time intervals of twelve weeks. The assumption of time independence was tested using Chi-square tests directed at deviations of individual empirical matrices (eg, the matrix of observed transitions from baseline to week 12) from the corresponding input matrix, which was obtained by pooling all observed transitions. The model selects the correct matrix (dependent on current treatment) and then finds the probability of moving to any of the four health states (dependent on current health state) for each sampled patient. The EuroQol-5 dimensions (EQ-5D-3L)16 questionnaire was used to measure the quality of life in all four respective health states and valued using the Dutch tariff.17
Real-world effectiveness data on baricitinib in RA patients have not yet been collected long enough to use in this model, at DREAM or any other registries. Transition probabilities for baricitinib were therefore derived from pivotal trial data.7,18 Transition probabilities for baricitinib were adapted by assuming that for each element in the matrix of estimated baricitinib transitions the relative risk (RR) of observing the transition for baricitinib compared with an available common comparator would be the same in Lilly baricitinib pivotal trials and the DREAM T2T setting.
Medication costs were calculated using the Dutch Pharmacy Purchase prices using the standard Dutch dosage. MTX/Hydroxychloroquine (HCQ) low dosage is daily 20 mg MTX and twice-daily HCQ 200 mg. MTX/HCQ high dosage refers to daily 25 or 30 mg MTX (where 80% is on 30 mg and 20% on 25, as observed in DREAM Remission Induction Cohort) and twice-daily HCQ 200 mg. Adalimumab has a dosage of 40 mg every other week. Baricitinib has a dosage of 4 mg once daily. Next, bDMARD consists of either etanercept (50 mg per week), rituximab (1000 mg every six weeks), infliximab (300 mg every 4 weeks), abatacept (750 mg every 3 weeks), and tocilizumab (800 mg every 4 weeks) with equal probabilities.
For the sensitivity analysis focusing on lowered prices for the expensive medication, adalimumab, and baricitinib, multiple price levels were considered. List price, 75% of list price, 50% of list price and 25% of the list price.
Other costs included in the model are Healthcare Consumption Costs and work productivity loss due to sick leave. Both can be found in Table 3. Both cost sources were collected in RA patients in the Netherlands, being treated in a T2T setting. Healthcare resource usage was assessed using a patient questionnaire and costs were calculated based on the amounts of resource use and unit costs.15 The following information on healthcare resource usage is routinely collected in DREAM by patient questionnaire: number of diagnostic tests; hospital admission days; specialist (rheumatologist and other), nurse and general practitioner visits; visits to psychologists, psychiatrist, and physical therapist, and hours of formal and informal care. These were valued using the Dutch Costing guidelines published by the Dutch Health Care Insurance Board.15 Data were also collected on productivity loss due to sick leave using the Human Capital method (includes hourly salary, social security fees, private car use, administration costs). Sick leave cost was also included in the model, as this means a loss of productivity and costs for the society. Sick leave cost was valued per diem,19 multiplied with the observed percentage of patients in the workforce (in DREAM registry) and added to the total costs per health state.
 |
Table 3 Other Costs per DAS28-ESR Disease Activity State |
Results
Tables 1 and 2 display values and distributions for all parameters. In the PSA, the baricitinib strategy dominated the DREAM T2T strategy, with lower expected costs, and higher expected utility over five years (see Table 4). The Incremental Cost-Effectiveness Ratio (ICER) was estimated to be € −238,418. The negative ICER in this case reflects the dominance of baricitinib strategy, it accumulates lower cost and higher Quality-Adjusted Life Years (QALYs) over 5 years. The estimated difference between the strategies in QALYs accrued over 5 years was small, while the difference in expected costs between the strategies was considerable, leading to a large ICER.
 |
Table 4 Probabilistic Sensitivity Analysis Results |
Figure 2 shows a plot of the distribution of ICER estimates resulting from the Monte Carlo iterations over the cost-effectiveness plane. Almost all (96.07%) estimates of incremental costs were negative (ie, baricitinib strategy incurred less costs); while in 64% of the model replications, the ICER fell in the southeast quadrant (ie, baricitinib incurred less costs and provides better health outcomes). As can be seen in Table 5, baricitinib was likely to be cost-effective at the willingness to pay (WTP) threshold of €60,000 with 81.94% of all iterations below this threshold. Sixty-four percent of iterations were cost saving. Even at more extreme WTP thresholds of €100,000 and €500,000, baricitinib strategy was cost-effective in 75% and 55% of iterations, respectively. Scenario analyses showed the baricitinib strategy to be cost-effective in both the moderate and severe RA populations.
 |
Table 5 ICER Report |
 |
Figure 2 ICER PSA results baricitinib strategy vs DREAM T2T strategy 500 iterations displayed. Abbreviation: QALY, quality-adjusted life year. |
Scenario Analysis
A scenario analysis was run in which lowered prices were considered for adalimumab and baricitinib. Figure 3 shows the accumulated results. For both adalimumab and baricitinib various price levels were considered: list price, 75% of list price, 50% of list price and 25% of list price. Figure 1 shows whether the ICER is below or above the WTP threshold of €60,000, and thus whether the DREAM T2T strategy or the Baricitinib strategy is favorable.
Discussion
In this study, the cost-effectiveness of a T2T strategy in which JAK1/JAK2-inhibitor baricitinib is tried in csDMARD-IR relative to a T2T strategy in which a bDMARD is tried after csDMARD therapy was evaluated. The results suggest that treatment with baricitinib in csDMARD-IR, as part of a tight-control, step up approach is very likely to incur lower costs and likely to be cost-effective at various cost-effectiveness thresholds. However, in case of biological and biosimilar drugs, real-world prices might differ from the commonly used list prices. The results of the scenario analysis suggest that differences in pricing could have an effect on cost-effectiveness profiles of different RA treatment strategies.
The ACR and EULAR management guidelines currently do not distinguish between the relative merit of JAK-inhibitors such as baricitinib compared with bDMARDs as components in contemporary T2T-based management strategies, in light of the limited real-world evidence on their relative effectiveness. However, these recommendations are solely based on direct comparisons of tsDMARDs and bDMARDs in clinical trials. While this provides relevant data on the relative safety and efficacy, integrating health-economics considerations play a crucial factor in the decision-making process for drug prescriptions in RA. In the past, health economic considerations have helped shape T2T sequences. The present study is the first to show the value of JAK1/JAK2-inhibitor baricitinib relative to the current second-line treatment from a societal perspective.
The model used in this study was initially developed and validated by Welsing et al, and later adapted by Schippers et al to model sequential treatment outcomes in RA patients. Unlike many previously published model-based health economic evaluations, the status of patients does not deteriorate over time in this model. This is consistent with studies describing 5-10-year outcomes of early RA T2T cohort studies and was therefore considered appropriate for our current study.
A strength of this work is that, whenever possible, the DREAM daily clinical practice data as inputs for the model were used. Most modeling studies in RA rely on clinical trial data inputs.20 Typically, patients included in clinical trials are younger, have more severe disease, and fewer comorbidities compared with patients seen in daily practice.21,22 The use of clinical trial data in health-economic modelling studies therefore limits the generalizability of the results and might lead to overestimation of health benefits, since these characteristics have previously been found to be associated with achieving treatment response.23 However, unfortunately, real-world effectiveness data on baricitinib in RA patients is not yet available. Therefore, estimates of baricitinib transition probabilities were obtained by assuming that the relative effectiveness of baricitinib and bDMARD would be the same in daily clinical practice and the clinical trial setting. This model thus incorporates data from both the DREAM daily clinical practice data and baricitinib clinical trial program to address the research question. Other limitations of this study are that no individual patient expenses were available in the DREAM cohort study, due to which there were no out-of-pocket costs included in this study. This does not represent a limitation for internal comparison of the two strategies; however, external comparisons should be conducted carefully. Drug discontinuation is not incorporated in the model; however, in the DREAM cohort, it was shown that adherence to the protocol was good.24
Conclusions
In this Monte Carlo simulation, baricitinib strategy was cost-effective compared to DREAM T2T strategy in a T2T setting for RA patients with moderate and severe disease activity. Scenario analysis showed similar results for moderate and severe populations analyzed separately. Future analyses using real-world effectiveness data is important to validate these results.
Abbreviations
ACR20, American College of Rheumatology 20 criteria; bDMARD, biological disease-modifying antirheumatic drug; CHEERS, the Consolidated Health Economic Evaluation Reporting Standards; CI, confidence intervals; combi, combination therapy; csDMARD, conventional synthetic disease-modifying antirheumatic drug, CUA, cost–utility analysis; DAS28 CRP, disease activity score in 28 joints - C-reactive protein; DAS28-ESR, Disease Activity Score 28-erythrocyte sedimentation rate; DREAM, the Dutch RhEumatoid Arthritis Monitoring registry; EQ-5D, the EuroQol-5 dimensions questionnaire; HCQ, hydroxychloroquine; ICER, incremental cost-effectiveness ratio; IR, inadequate responder; JAK1/JAK2-inhibitor, Janus Kinase 1, 2, inhibitor; mono, monotherapy; MTX, Methotrexate; NMB, Net Monetary Benefits; QALYs, Quality-Adjusted Life Years; RR, relative risk; T2T, treat-to-target; tsDMARDs, targeted synthetic DMARDs; WTP, willingness to pay.
Ethics Approval and Informed Consent
Written informed consent was obtained from each patient whose data has been processed and used in the current study. The Medical Ethics Committees of the Medisch Spectrum Twente, Enschede and Isala, Zwolle hospitals determined, in accordance with Dutch Law on medical-scientific research with humans, that no ethical approval was required because all data were collected in the course of regular daily clinical practice. Nonetheless, patients were completely informed and written informed consent was obtained from each patient.
Data Sharing Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request. Requests will be considered by the Dutch RhEumatoid Arthritis Monitoring (DREAM) registry chief science officer.
Acknowledgments
The abstract of this paper was presented at the European League Against Rheumatism (EULAR) Conference annual meeting 2019 in Madrid, Spain, as an oral presentation with interim findings. The abstract was published in “Scientific Abstracts” in Annals of the Rheumatic Diseases; 2019;78:238–239; https://doi.org/10.1136/annrheumdis-2019-eular.906.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was sponsored by Eli Lilly and Company. Publication of the study results was not contingent on the sponsor’s approval or censorship of the manuscript.
Disclosure
C Van De Laar, MAH Oude Voshaar, and MAF Van De Laar received a grant from Eli Lilly & Company to perform the health economic analysis in this study. MAF Van De Laar reports personal fees from Eli Lilly during the conduct of the study. L Zaremba-Pechmann is a contractor at Lilly Deutschland GmbH through HaaPACS GmbH. F De Leonardis, I De La Torre, and WKH Fakhouri are employees at Eli Lilly & Company and stockholders. The authors report no other conflicts of interest in this work.
References
1. Sweeney SE, HarrisJr, ED, Firestein GS, Kelley W, Jeffery RC, Venables PJW. Clinical features of rheumatoid arthritis. In: Firestein GS, Kelley WN, editors. Kelley’s Textbook of Rheumatology. Vol. 38. Philadelphia, PA: Elsevier/Saunders; 2013;1109–1136. doi:10.1016/j.mpmed.2009.12.004
2. Smolen JS. Treat-to-target: rationale and strategies. Clin Exp Rheumatol. 2012;30(4Suppl 73):S2–S6.
3. Singh JA, Saag KG, Bridges SLJ, et al. 2015 American college of rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Care Res (Hoboken). 2016;68:1–25. doi:10.1002/acr.22783
4. Smolen JS, Landewé R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76:960–977. doi:10.1136/annrheumdis-2016-210715
5. Brennan A, Chick SE, Davies R. A taxonomy of model structures for economic evaluation of health technologies. Health Econ. 2006;15(12):1295–1310. doi:10.1002/hec.1148
6. Joensuu JT, Huoponen S, Aaltonen KJ, Konttinen YT, Nordström D, Blom M. The cost-effectiveness of biologics for the treatment of rheumatoid arthritis: A systematic review. PLoS One. 2015;10:e0119683. doi:10.1371/journal.pone.0119683
7. Taylor PC, Keystone EC, van der Heijde D, et al. Baricitinib versus Placebo or adalimumab in rheumatoid arthritis. N Engl J Med. 2017;376(7):652–662. doi:10.1056/NEJMoa1608345
8. Vermeer M, Kuper HH, Hoekstra M, et al. Implementation of a treat-to-target strategy in very early rheumatoid arthritis: results of the Dutch Rheumatoid Arthritis Monitoring remission induction cohort study. Arthritis Rheum. 2011;63(10):2865–2872. doi:10.1002/art.30494
9. Steunebrink LMM, Vonkeman HE, Ten Klooster PM, Hoekstra M, van Riel PLCM, van de Laar MAFJ. Recently diagnosed rheumatoid arthritis patients benefit from a treat-to-target strategy: results from the DREAM registry. Clin Rheumatol. 2016;35(3):609–615. doi:10.1007/s10067-016-3191-3
10. Prevoo ML, van ’T Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995;38(1):44–48. doi:10.1002/art.v38:1
11. Welsing PMJ, Severens JL, Hartman M, PLCM VR, Laan RFJM. Modeling the 5-year cost effectiveness of treatment strategies including tumor necrosis factor-blocking agents and leflunomide for treating rheumatoid arthritis in the Netherlands. Arthritis Care Res. 2004;51:964–973. doi:10.1002/art.20843
12. Welsing PMJ, Severens JL, Hartman M, van Gestel AM, PLCM VR, Laan RFJM. The initial validation of a Markov model for the economic evaluation of (new) treatments for rheumatoid arthritis. Pharmacoeconomics. 2006;24(10):1011–1020. doi:10.2165/00019053-200624100-00008
13. Schipper LG, Kievit W, den Broeder AA, et al. Treatment strategies aiming at remission in early rheumatoid arthritis patients: starting with methotrexate monotherapy is cost-effective. Rheumatology. 2011;50(7):1320–1330. doi:10.1093/rheumatology/ker084
14. Briggs A. Probabilistic analysis of cost-effectiveness models: statistical representation of parameter uncertainty. Value Heal. 2005;8:1–2. doi:10.1111/j.1524-4733.2005.08101.x
15. Hakkaart-van Roijen L, van der Linden N, Bouwmans CAM, Kanters T, Tan SS. Kostenhandleiding. Methodologie van Kostenonderzoek En Referentieprijzen Voor Economische Evaluaties in de Gezondheidszorg [Costing manual: Methodology of costing research and reference prices for economic evaluations in healthcare]. Zorginstituut Nederland. Geactualiseerde versie; 2015. Dutch.
16. Rabin R, De Charro F. EQ-5D: a measure of health status from the EuroQol Group. Ann Med. 2001;33(5):337–343. doi:10.3109/07853890109002087
17. Lamers LM, Stalmeier PF, McDonnell J, Krabbe PF, van Busschbach JJ. Kwaliteit van leven meten in economische evaluaties: het Nederlands EQ-5D-tarief [Measuring the quality of life in economic evaluations: the Dutch EQ-5D tariff]. Ned Tijdschr Geneeskd. 2005;149(28):1574–1578. Dutch.
18. Dougados M, Van Der Heijde D, Chen YC, et al. Baricitinib in patients with inadequate response or intolerance to conventional synthetic DMARDs: results from the RA-BUILD study. Ann Rheum Dis. 2017;76:88–95. doi:10.1136/annrheumdis-2016-210094
19. van der Ploeg CPB, Pal S, Vroome ED, Bossche S. De Kosten van Ziekteverzuim Voor Werkgevers in Nederland [The Cost of Absenteeism for Employers in the Netherlands]. TNO; 2014. Available from: https://www.monitorarbeid.tno.nl/dynamics/modules/SPUB0102/view.php?pub_Id=100294&att_Id=4911. Accessed 20 March, 2020. Dutch.
20. Tosh J, Stevenson M, Akehurst R. Health economic modelling of treatment sequences for rheumatoid arthritis: a systematic review. Curr Rheumatol Rep. 2014;16. doi:10.1007/s11926-014-0447-2.
21. Kievit W, Fransen J, Oerlemans AJM, et al. The efficacy of anti-TNF in rheumatoid arthritis, a comparison between randomised controlled trials and clinical practice. Ann Rheum Dis. 2007;66(11):1473–1478. doi:10.1136/ard.2007.072447
22. Zink A, Strangfeld A, Schneider M, et al. Effectiveness of tumor necrosis factor inhibitors in rheumatoid arthritis in an observational cohort study: comparison of patients according to their eligibility for major randomized clinical trials. Arthritis Rheum. 2006;54:3399–3407. doi:10.1002/art.22193
23. Dmfm VDH, Plcm VR, van Rijswijk MH. van de Putte LBA. Influence of prognostic features on the final outcome in rheumatoid arthritis: a review of the literature. Semin Arthritis Rheum. 1988. doi:10.1016/0049-0172(88)90013-3
24. Vermeer M, Kuper HH, Bernelot Moens HJ, et al. Adherence to a treat-to-target strategy in early rheumatoid arthritis: results of the DREAM remission induction cohort. Arthritis Res Ther. 2012;14(6):R254. doi:10.1186/ar4099
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.