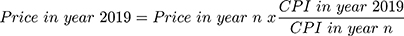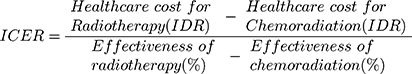Back to Journals » International Journal of Women's Health » Volume 13
Cost-Effectiveness Analysis of Chemoradiation and Radiotherapy Treatment for Stage IIB and IIIB Cervical Cancer Patients
Authors Puspitasari IM , Legianawati D, Sinuraya RK , Suwantika AA
Received 31 October 2020
Accepted for publication 29 January 2021
Published 19 February 2021 Volume 2021:13 Pages 221—229
DOI https://doi.org/10.2147/IJWH.S289781
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Everett Magann
Irma M Puspitasari,1,2 Dewi Legianawati,1 Rano K Sinuraya,1,2 Auliya A Suwantika1,2
1Department of Pharmacology and Clinical Pharmacy, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia; 2Center of Excellence in Higher Education for Pharmaceutical Care Innovation, Universitas Padjadjaran, Sumedang, Indonesia
Correspondence: Irma M Puspitasari
Department of Pharmacology and Clinical Pharmacy, Faculty of Pharmacy, Universitas Padjadjaran, Jalan Bandung, Sumedang KM 21, Jatinangor, Sumedang, 45363, Indonesia
Tel +62 22 84288888 (Ext 3510)
Email [email protected]
Purpose: Cervical cancer is the second most common type of cancer and cause of death from cancer in Indonesia. In 2013, cervical cancer was the most prevalent cancer in Indonesia, with a rate of 0.8 per 1000 women. Based on the National Guidelines for Cervical Cancer Medical Services in Indonesia, the recommended therapy for stages IIB-IIIB cervical cancer is chemoradiation or radiotherapy. This study aimed to evaluate the cost-effectiveness of chemoradiation and radiotherapy for treating stage IIB-IIIB cervical cancer in a national referral hospital in Indonesia.
Patients and Methods: A cross-sectional study from a healthcare perspective using retrospective patient data was conducted. The included patients had stage IIB-IIIB registered cervical cancer, were in the hospital between January 1, 2015 and December 31, 2017, received chemoradiation or radiotherapy, were ≥ 18 years old, and had complete clinical data and detailed cost of therapy data. The incremental cost-effectiveness ratio (ICER) were calculated, and a sensitivity analysis was performed.
Results: The average treatment cost per patient was $2944 and $3231 for radiotherapy and chemoradiation, respectively. Despite the fact that the treatment effectiveness of chemoradiation (69.1%) was considered to be higher than that of radiotherapy (63.2%), chemoradiation had more potential side effects than radiotherapy. In a comparison with radiotherapy, the ICER of chemoradiation was $48.6 per complete response rate. Additionally, the cost of radiotherapy was the most influential parameter impacting the ICER.
Conclusion: Chemoradiation was considered to be more costly than radiotherapy. Additionally, the effectiveness of chemoradiation was higher than that of radiotherapy. A cost utility analysis (CUA) is required for further investigation.
Keywords: Indonesia, cancer treatment, side effects, incremental cost-effectiveness ratio
Introduction
According to 2018 global cancer statistics from the International Agency for Research on Cancer, there were 18.1 million new cases of cancer and 9.6 million cancer deaths worldwide that year. Cervical cancer ranked fourth among women worldwide and is the leading cause of death in women.1 In Indonesia, cervical cancer ranks second for both incidence and cause of death from cancer.1 In 2013, cervical cancer was the most prevalent cancer in Indonesia, with a rate of 0.8 per 1000 women.2
Approximately 76% of new cervical cancer patients are detected at advanced stages (stages IIB and IIIB), and other studies have reported rates as high as 91%.3,4 The choice of therapy for each stage of cancer differs, and it is also determined by other factors that exist in the patient, such as whether the therapy will suit the patient, whether the patient is willing to accept the offered therapy, whether fertility will be maintained, and whether the patient is pregnant.5 However, the most common treatments for cervical cancer are surgery (hysterectomy), radiation (radiotherapy), and chemotherapy.6 Based on the National Guidelines for Cervical Cancer Medical Services in Indonesia, the recommended therapies for stages IIB-IIIB are chemoradiation or radiotherapy.7
Chemoradiation is a combination therapy of chemotherapy and radiation whereas radiotherapy only uses radiation, including external radiation (external beam radiotherapy (EBRT)) and internal radiation (brachytherapy). Several studies have reported that chemoradiation and radiotherapy are effective in healing, increasing survival (overall survival),8 and increasing disease-free survival in cervical cancer patients.9 Another study showed that chemoradiation is more effective than radiotherapy.10 Conversely, a different study reported that radiotherapy is more effective than chemoradiation.11
The cost of cancer therapy varies depending on the stage of cancer at the time of diagnosis; for example, the earlier the stage, the cheaper the cost of therapy.12 Since 2014, the cost of cancer therapy incurred by the Indonesian government has continued to increase. In 2014, total costs of approximately USD 92 million were reported, which increased to USD 164 million in 2016 and USD 214 million in 2017.13 A study in Canada reported that the average cost of cervical cancer therapy per person was approximately CAD 35,519–45,369 or USD 27,783–35,448 per year.14 Considering the large contribution of cervical cancer to mortality and morbidity in Indonesia, the cost of disease therapy is high, and several treatment options are available. Thus, a cost-effectiveness analysis is necessary to determine which therapy has the highest cost-effectiveness. This study aimed to perform a cost-effectiveness analysis of stage IIB-IIIB cervical cancer treated with chemoradiation and radiotherapy in a national referral hospital in Indonesia, which is one of the largest and main referral hospitals in Indonesia, established by the Ministry of Health.
Methods
Study Design
This retrospective cross-sectional study was conducted from a healthcare perspective and used patient data collected from a national referral hospital in Indonesia. Patients who met the following inclusion criteria were eligible for this study: stage IIB-IIIB registered cervical cancer, in the hospital between January 1, 2015 and December 31, 2017, received chemoradiation or radiotherapy, aged ≥18 years, and had complete clinical data and detailed cost of therapy data. Patients who had comorbidities such as kidney failure, heart disease, hypertension, and diabetes; had another cancer diagnosis besides cervical cancer; died before the end of the therapy; and were pregnant were excluded.
Data Collection
The data of patients who met the inclusion criteria were collected. Patients who received chemoradiation were patients treated with chemotherapy and radiation. Patients who received radiotherapy were patients treated with radiation alone. The radiation dose was the actual dose delivered to the patients. Most patients were treated with external beam radiation therapy (EBRT) and brachytherapy with a total average dose 6700–7200 cGy.15 The treatment choice was decided before treatment initiation by the radiation oncologist, based on the patient’s clinical condition and the national guidelines.
Data including medical record number, age, sex, education, occupation, initial symptoms, diagnosis, initial and final tumor size, history of disease, and drug use were obtained from the patients’ medical records. Data on drug costs, radiation costs, consultation fees, costs of consumable medical materials, inpatient care costs, laboratory costs, outpatient service costs, nutritional service costs, and medical doctor fees were obtained from the financial department.
Ethics Approval
This research protocol was approved by the Research Ethics Committee Universitas Padjadjaran, Indonesia, number 1460/UN6.KEP/EC/2018 and was conducted in compliance with the Declaration of Helsinki. Patient consent was not required by Research Ethics Committee since this study used retrospective patient medical records and financial data, also no personal information was collected. All of the data were kept anonymous to ensure confidentiality.
Cost-Effectiveness Analysis
Healthcare Costs
When calculating healthcare costs, which is the actual average cost of treatment delivered to the patients, the prices were adjusted into 2019 prices. The price adjustments were made using the consumer price index (CPI), according to the formula from https://www.inflationtool.com:
The CPI values were obtained from the official website of the Central Statistics Agency of Indonesia.16 The 2019 CPI values were obtained from May 2019 when the price was calculated, whereas the CPI values for 2015, 2016, and 2017 were taken from the CPI values of the first month of therapy for each patient.
Effectiveness of Therapy
Therapy effectiveness was assessed according to the status of tumor remission using the assessment criteria following the standards of the World Health Organization (WHO). Tumor remission status was determined based on the reduction of the final tumor size from the initial tumor size. The criteria for tumor remission status according to the WHO are as follows: (1) complete response (CR): loss of all lesions, assessed ≥4 weeks after therapy; (2) partial response (PR): reduction of lesions by ≥50% of the initial condition, assessed ≥4 weeks after therapy, no metastases, and no progression from other parameters; (3) stable disease (SD): no progressive disease four weeks after therapy, no metastasis, no progression from other parameters; and (4) worsening/no response (progressive disease/no response): ≥25% new growth of lesions or the appearance of new lesions and/or the progression of other tumor parameters.17 The time horizon for this study was four weeks.
The Incremental Cost-Effectiveness Ratio (ICER)
After obtaining healthcare costs and determining the therapy’s effectiveness, the data were analyzed using the ICER to compare the cost-effectiveness between radiotherapy and chemoradiation therapy.18
Sensitivity Analysis
A sensitivity analysis was conducted to measure and evaluate uncertainty,19 particularly that of the dominant parameters influencing the outcome of determining the effectiveness of treatment costs. A sensitivity analysis was performed to determine the factors that have the greatest influence on the ICER value, by adding and subtracting 25% of each cost and effectiveness parameter to the ICER value. The value obtained after addition became the upper limit value and that obtained after subtraction became the lower limit value. The result was presented in a tornado diagram that referred to the summary (stack) of bar graphs representing univariate sensitivity analyses for a wide range of input values, ordered according to the extent (spread) of variation of the resulting model output value (with the widest variation on top). The sensitivity analysis was performed using Microsoft Excel (Microsoft Corp., Washington, USA).
Statistical Analysis
The difference in side effects was tested using the Chi-square test. The differences in the healthcare cost between the two therapies were analyzed with the Mann–Whitney U-test. The difference in treatment effectiveness was analyzed with the Fisher’s exact test. All statistical analyses were performed with SPSS version 22 (IBM Corp., Armonk, NY, USA).
Results
Data Collection Process
Figure 1 presents the data collection process. Of the 844 patients with data, 620 were excluded because of incomplete data, no previous radiotherapy, cancer of a stage other than IIB-IIIB, recurrence, and comorbidities such as hypertension, cardiovascular disease, diabetes, or renal disease. Finally, data from 224 patients who met the inclusion criteria were further analyzed.
 |
Figure 1 Patients’ data collection process. |
Patient Characteristics
Table 1 presents the characteristics of the cervical cancer patients. Most patients (62.1%) were 40–55 years old, 57.6% had an elementary school education, 80.4% were housewives without an occupation, 51.8% had stage IIB disease, and 71% had the histological type of squamous cell carcinoma (SCC). In the chemoradiation and radiotherapy groups, the patients were given radiotherapy with a dose of 6700–7200 cGy consisting of external radiation (EBRT) and internal radiation (brachytherapy), or they received only external radiation. In the chemoradiation group, 79.9% of patients received cisplatin and 11.8% received carboplatin.
 |
Table 1 Data of Patients’ Characteristics |
Side Effects
Table 2 highlights the treatment side effects. More side effects were reported in the patients who received chemoradiation (87.3%) than in those who received radiotherapy (76.3%); this difference was significant (p < 0.01). The most common side effects were nausea and diarrhea. Nausea was more frequently reported in patients who received chemoradiation (49.1%) than in patients who received radiotherapy (41.2%). Conversely, diarrhea was reported more frequently after radiotherapy (34.2%) than after chemoradiation (20.9%).
 |
Table 2 Side Effects of Stage IIB-IIIB Cervical Cancer Patients’ Treatment |
Cost-Effectiveness Analysis
Healthcare Costs
The cost for cervical cancer stage IIB-IIIB treatment from a healthcare perspective is presented in Table 3. Assuming that 1 USD is equivalent to IDR 14,000, the average cost per patient was $3231 with chemoradiation treatment and $2944 with radiotherapy.
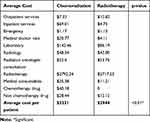 |
Table 3 Healthcare Costs for Stage IIB-IIIB Cervical Cancer Patients’ Treatment |
Treatment Effectiveness
Table 4 compares the treatment effectiveness between chemoradiation and radiotherapy. Chemoradiation therapy resulted in CR in 69.1% of patients; this value was 63.2% with radiotherapy. The effectiveness of chemoradiation therapy was higher than that of radiotherapy, but the difference was not significant (p = 0.61).
 |
Table 4 Effectiveness of Stage IIB-IIIB Cervical Cancer Patients’ Treatment |
ICER
The ICER of chemoradiation to radiotherapy was $48.60 per %CR. Thus, changing from chemoradiation to radiotherapy will save $48.6 for each percent increase in CR.
Sensitivity Analysis
Figure 2 depicts the results of the sensitivity analysis based on the ICER and healthcare cost components after adjustment. Almost all the costs of therapy in Figure 2 were directed to the left, indicating that the lower the costs, the more cost effective the therapy. The opposite result was found for the outpatient service costs. In this case, the higher the cost of outpatient treatment, the more cost effective that treatment. There were four factors that had the greatest influence on the ICER: the cost of radiotherapy had the greatest influence, followed by inpatient services, laboratory examination, and chemotherapy drugs.
 |
Figure 2 Factors influencing the ICER. Abbreviations: ICER, incremental cost-effectiveness ratio, the cost for a 1% gain in complete response. |
Discussion
This present study demonstrated that, from a healthcare perspective, chemoradiation was considered to be more costly than radiotherapy for treating stage IIB-IIIB cervical cancer patients. These results are consistent with Manusirivithaya’s study in Thailand in 2005.11 The average cost per patient was $2944 for radiotherapy and $3231 for chemoradiation. The treatment effectiveness of chemoradiation was higher than that of radiotherapy, but chemoradiation had more side effects. The ICER was $48.60 per %CR, indicating that if chemoradiation is performed instead of radiotherapy, it will cost $48.60 for each percent increase in CR.
The primary factors influencing the ICER in our study were the cost of radiotherapy, the cost of hospitalization, laboratory costs, and the cost of chemotherapy drugs. The most influential factor on the ICER was the cost of radiotherapy, which comprised more than 86% ($2792/$3231) of the total cost of therapy in chemoradiation and more than 92% ($2717/$2944) in patients who received radiotherapy alone. Therefore, if the cost of radiotherapy could be reduced, the therapy would be more cost effective. These results are in line with Kim’s study in 2015, which found that the cost of radiotherapy was the factor with the greatest influence on cost-effectiveness.20 The factor with the second-greatest influence on the ICER was inpatient services because the chemoradiation therapy cost was 14.6 times ($69.01/$4.70) greater than the cost of inpatient services for radiotherapy alone. The cost of laboratory examination for chemoradiation was 1.64 times ($142.46/$86.19) greater than the cost of laboratory examination for radiotherapy alone. The cost of the chemotherapy drugs was the fourth most influential factor contributing to the ICER. This is because based on the national guidelines, the chemotherapy drug in chemoradiation that was delivered to the IIB-IIIB cervical cancer patients was for radiosensitizing so that the treatment requires lower doses of the chemotherapy drug and is relatively cheaper than usual chemotherapy. Nevertheless, the cost of chemotherapy drugs also played a role because patients who received radiotherapy alone had no chemotherapy drug costs.
The sensitivity analysis evaluating outpatient services displayed the opposite results. If the ICER value was increased by 25%, the sensitivity analysis chart pointed to the left, indicating that the higher the cost of outpatient care, the more cost effective the therapy would be. This might be attributed to the fact that most patients who were directly admitted to the emergency department had advanced-stage cancer. They did not undergo outpatient care and cervical screening, even when symptoms such as vaginal discharge or bleeding appeared, albeit a cervical screening programs was available in accordance with the Ministry of Health regulations. If the patients immediately visit the outpatient care when symptoms such as vaginal discharge or bleeding appear, the disease could be diagnosed early and the treatment would be more cost effective.
In this study, most cervical cancer patients were 40–55 years old. This is in line with the study by Kashyap in 2019 in Chandigarh, India, which reported that the most common age range for cervical cancer was 41–60 years.21 Kashyap also identified that a lack of education, not maintaining personal hygiene, using an old cloth repeatedly, place of residence, early age of marriage, not washing genitalia after sexual intercourse, increased number of husband’s sexual partners, a history of sexually transmitted infections and genital warts, and a lack of knowledge about screening for cervical carcinoma as risk factors for cervical cancer.21
Most cervical cancer patients in this study had an elementary school level education. Some studies have reported that education level is a risk factor for cervical cancer,22 potentially because people with low levels of education will also have low knowledge about cervical cancer.23 Additionally, because people with low education levels in Indonesia tend to get married at a younger age, their first sexual intercourse is likely to occur at a young age. Based on a study by Reis et al (2011), first sexual intercourse occurring at a very young age is a risk factor for cervical cancer.24
The most common stage of cervical cancer in this study was stage IIB (51.3%), and the most common histological type was SCC (70.5%). This study was in line with several previous studies in Indonesia, such as Dwipoyono’s study in 2007 at a cancer hospital25 and Watulingas’ study in 2016.26 However, their results differed from a study in Poland, which showed that most cervical cancer patients (48%) were in the early stages (IA-IIA).27 This may be a result of the knowledge on prevention and early detection of cervical cancer in that study.
This study is limited in its use of clinical parameters to assess the tumor remission status based on the WHO criteria to calculate the effectiveness of the treatment, which was determined from the vaginal examination at the time of the last brachytherapy, but was not based on the overall survival. However, by including the tumor remission status in the cost-effectiveness analysis, this study was able to illustrate that chemoradiation was more costly and had a slightly higher level of effectiveness, albeit with more side effects, than radiotherapy.
Conclusion
From a healthcare perspective, chemoradiation was considered to be more costly than radiotherapy for treating stage IIB-IIIB cervical cancer patients. However, the effectiveness of chemoradiation was higher than that of radiotherapy, and the ICER was $48.6 per %CR. The cost of radiotherapy was the most influential parameter impacting the ICER. A cost utility analysis (CUA) is suggested for further investigation.
Funding
This research was supported by Universitas Padjadjaran Indonesia through the Center of Excellence in Higher Education for Pharmaceutical Care Innovation.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
2. Center of Data and Information Ministry of Health Republic of Indonesia. Stop cancer; 2015. Available from: https://www.kemkes.go.id/folder/view/01/structure-publikasi-pusdatin-info-datin.html.
3. Cetina L, Rivera L, Hinojosa J, et al. Routine management of locally advanced cervical cancer with concurrent radiation and cisplatin. Five-year results. BMC Womens Health. 2006;6:3. doi:10.1186/1472-6874-6-3
4. Azad SK, Choudhary V. Treatment results of radical radiotherapy of carcinoma uterine cervix using external beam radiotherapy and high dose rate intracavitary radiotherapy. J Cancer Res Ther. 2010;6(4):482–486. doi:10.4103/0973-1482.77108
5. National Cancer Institute. Cervical cancer treatment (PDQ®)–health professional version. Available from: https://www.cancer.gov/types/cervical/hp/cervical-treatment-pdq.
6. American Cancer Society. Treatment options for cervical cancer, by stage. Available from: https://www.cancer.org/cancer/cervical-cancer/treating/by-stage.html.
7. Ministry of Health Republic of Indonesia. National Guidelines for Cervical Cancer Medical Services; 2017.
8. Green JA, Kirwan JM, Tierney JF, et al. Survival and recurrence after concomitant chemotherapy and radiotherapy for cancer of the uterine cervix: a systematic review and meta-analysis. Lancet (London, England). 2001;358(9284):781–786. doi:10.1016/S0140-6736(01)05965-7
9. Choi IJ, Cha MS, Park ES, et al. The efficacy of concurrent cisplatin and 5-flurouracil chemotherapy and radiation therapy for locally advanced cancer of the uterine cervix. J Gynecol Oncol. 2008;19(2):129–134. doi:10.3802/jgo.2008.19.2.129
10. Li Z, Yang S, Liu L, Han S. A comparison of concurrent chemoradiotherapy and radiotherapy in Chinese patients with locally advanced cervical carcinoma: a multi-center study. Radiat Oncol. 2014;9:212. doi:10.1186/1748-717X-9-212
11. Manusirivithaya S, Sripramote M, Tangjitgamol S, Sanjareonsuttikul N, Pisarnturakit P. Cost effectiveness of concurrent chemoradiation in comparison with radiation alone in locally advanced cervical cancer. J Med Assoc Thai. 2005;88(8):1035–1044.
12. Subramanian S, Trogdon J, Ekwueme DU, Gardner JG, Whitmire JT, Rao C. Cost of cervical cancer treatment: implications for providing coverage to low-income women under the Medicaid expansion for cancer care. Womens Health Issues. 2010;20(6):400–405. doi:10.1016/j.whi.2010.07.002
13. Audriene D Ministry of health: cancer treatment costs in 2017. 2018. Available from: https://www.cnnindonesia.com/gaya-hidup/20180407180941-255-289105/kemenkes-biaya-pengobatan-kanker-di-2017-hampir-rp3-triliun.
14. Pendrith C, Thind A, Zaric GS, Sarma S. Costs of cervical cancer treatment: population-based estimates from Ontario. Curr Oncol. 2016;23(2):e109–e115. doi:10.3747/co.23.2598
15. Legianawati D, Puspitasari IM, Suwantika AA, Kusumadjati A. Management profile of stage IIB–IIIB cervical cancer with radiation therapy and chemoradiation at Dr. Hasan Sadikin General Hospital Bandung in 2015–2017. Indones J Clin Pharm. 2019;8(3):205–216.
16. Central Statistics Agency of Indonesia. Monthly indonesia consumers price indices and inflations. Available from: https://www.bps.go.id/statictable/2009/06/15/907/indeks-harga-konsumen-dan-inflasi-bulanan-indonesia-2006-2020.html.
17. World Health Organization. WHO handbook for reporting results of cancer treatment; 1979. Available from: https://apps.who.int/iris/handle/10665/37200.
18. Ministry of Health Republic of Indonesia. Guideline for Applying Pharmacoeconimics Study; 2013.
19. Jain R, Grabner M, Onukwugha E. Sensitivity analysis in cost-effectiveness studies: from guidelines to practice. Pharmacoeconomics. 2011;29(4):297–314. doi:10.2165/11584630-000000000-00000
20. Kim H, Rajagopalan MS, Beriwal S, Huq MS, Smith KJ. Cost-effectiveness analysis of 3D image-guided brachytherapy compared with 2D brachytherapy in the treatment of locally advanced cervical cancer. Brachytherapy. 2015;14(1):29–36. doi:10.1016/j.brachy.2014.09.002
21. Kashyap N, Krishnan N, Kaur S, Ghai S. Risk factors of cervical cancer: a case-control study. Asia-Pac J Oncol Nurs. 2019;6(3):308–314. doi:10.4103/apjon.apjon_73_18
22. Franceschi S, Plummer M, Clifford G, et al. Differences in the risk of cervical cancer and human papillomavirus infection by education level. Br J Cancer. 2009;101(5):865–870. doi:10.1038/sj.bjc.6605224
23. Segni MT, Demissie HF, Tadesse DM, Amdemichael R, Mamo YT, Worku A. Level of knowledge and associated factor toward cervical cancer among women age (21–64) years visiting health facilities in Gulele Sub-city Addis Ababa Ethiopia. J Pancreas. 2017;18(1):44–48.
24. Reis N. Risk factors for cervical cancer: results from a hospital-based case-control study. Int J Hematol Oncol. 2011;21:153–159. doi:10.4999/uhod.09061
25. Dwipoyono B, Nasdaldy SS, Adisasmita A. Histologic type and age in uteric cervical cancer at Dharmais Cancer Hospital. Indones J Cancer. 2007;1(1):7–11. doi:10.33371/ijoc.v1i1.1
26. Watulingas AM, Loho M, Wagey F. Characteristics of cervical cancer patients at RSUP Prof. Dr. R. D. Kandou Manado for the period of 1 January 2013-31 December 2015. J e-Clinic. 2016;4(2):1–5. doi:10.35790/ecl.4.2.2016.14477
27. Basta T, Knapp P, Blecharz P, et al. Current management of cervical cancer in Poland-Analysis of the questionnaire trial for the years 2002–2014 in relation to ASCO 2016 recommendations. PLoS One. 2019;14(1):e0209901. doi:10.1371/journal.pone.0209901
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.