Back to Journals » Clinical Interventions in Aging » Volume 17
Cortical Trajectory Fixation Versus Traditional Pedicle-Screw Fixation in the Treatment of Lumbar Degenerative Patients with Osteoporosis: A Prospective Randomized Controlled Trial
Authors Ding H , Hai Y, Liu Y , Guan L, Pan A, Zhang X, Han B , Li Y, Yin P
Received 16 November 2021
Accepted for publication 31 January 2022
Published 23 February 2022 Volume 2022:17 Pages 175—184
DOI https://doi.org/10.2147/CIA.S349533
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Hongtao Ding, Yong Hai, Yuzeng Liu, Li Guan, Aixing Pan, Xinuo Zhang, Bo Han, Yue Li, Peng Yin
Department of Spine Surgery, Beijing Chao-Yang Hospital, Capital Medical University of China, Beijing, People’s Republic of China
Correspondence: Yong Hai; Yuzeng Liu, Department of Spine Surgery, Beijing Chao-Yang Hospital, Capital Medical University of China, Beijing, People’s Republic of China, Tel +86 10-85231229 ; +86 13801221889 ; +86 13811552832, Email [email protected]; [email protected]; [email protected]
Study Design: This was a prospective randomized controlled trial study.
Objective: To elucidate clinical and radiographic outcomes and complications of cortical bone trajectory (CBT)-screw fixation in patients with osteoporosis at 24-month follow-up and to compare the results with those after transforaminal lumbar interbody fusion (TLIF) using traditional pedicle screw (PS) fixation.
Methods: We enrolled 124 patients and randomly assigned them to two groups (each group had 62 participants). The primary outcome was fusion rate. Secondary outcomes were VAS, Oswestry Disability Index (ODI), and Japanese Orthopaedic Association (JOA) scores, operation duration, incision length, estimated blood loss, drainage volume, radiological outcomes, and complications.
Results: At the 6- and 12-month follow-up points, similar fusion rates were observed based on CT scans in both groups (P=0.583 and 0.583). CBT provided significantly better short-term functional status at 3 months postoperation on ODI and JOA scores (P=0.012 and 0) and similar improvements in pain intensity and functional status at other follow-up points. In addition, CBT resulted in significantly better surgical characteristics. Notably, CBT fixation led to lower incidence of screw loosening (P=0.006).
Conclusion: CBT-screw fixation for single-level lumbar fusion in patients with osteoporosis provided improvement in clinical symptoms comparable to that of TLIF using PS fixation. Significantly better lumbar stability was found in the CBT group. We suggest that CBT-screw fixation is a reasonable and superior alternative to PS in TLIF in osteoporosis.
Trial Registration Number: ChiCTR1900022658.
Date of Registration: April 20, 2019.
Keywords: cortical bone-trajectory screw, pedicle screw, lumbar fusion, osteoporosis, randomized controlled trial
Introduction
Posterior lumbar fusion is one of the most used surgical methods for the treatment of lumbar degenerative diseases for its advantages of solid construction and high fusion rate of pedicle-screw internal fixation.1–3 However, some drawbacks have been reported as well, including extensive soft-tissue and paravertebral muscle exposure, lack of purchase strength in osteoporosis patients, and superior facet-joint violation.4–6 The cortical bone trajectory (CBT) technique, first described in 2009 by Santoni et al, was developed as a solution to reduce the incidence of complications associated with conventional pedicle screws (PSs).7–10 While traditional screws engage with cancellous bone, directed convergently along the anatomical axis of the pedicle, CBT screws increase the pullout strength by maximizing thread contact with the cortical bone of the pedicle and vertebrae, which start at the junction of the superior articular process and pars, following a sagittal caudocephalad path and a medial–lateral path in the transverse plane.7,9–11
The superior pullout strength, higher insertional torque, and satisfactory stability of the screw-rod construction compared to traditional fixation have been confirmed by biomechanical studies.7,11–14 Also, the comparable clinical outcomes, less multifidus muscle damage, and fewer complications associated with instrumentation compared to PSs have been observed in some retrospective clinical studies.15–17 However, to our knowledge, even though CBT fixation was introduced to improve internal fixation efficacy in osteoporosis patients, there have been few prospective studies on osteoporotic cohorts based on a large patient population. The aim of our study was to elucidate clinical and radiographic outcomes and complications of CBT-screw fixation in patients with osteoporosis at 18 months postoperation and to compare the results with those after transforaminal lumbar interbody fusion (TLIF) using traditional PS fixation.
Methods
Patients
The study was approved by the institutional ethics committee of the authors’ hospital. Inclusion criteria were: diagnosis of lumbar degenerative disease (LDD) according to radiography, computed tomography, and magnetic resonance imaging, as well as an additional diagnosis of osteoporosis based on World Health Organization criteria (dual-energy X-ray absorptiometry scan, t-score <–2.5), and meeting indications for lumbar surgery. Patients needed to present both clinical symptoms and radiological evidence of severe single-level LDD, and the symptoms and physical examination had to be consistent with radiological evidence. In addition, all patients underwent conservative treatment for 6 months with poor response. Patients were volunteers, provided written consent, and were required to complete at least 2 years’ follow-up. Exclusion criteria were secondary osteoporosis, history of previous lumbar spinal surgery, fractures, infection or tumors in the lumbar spine, inability to accurately complete preoperative and postoperative questionnaires, and lack of suitability as judged by the corresponding author. The inclusion and exclusion criteria were applied to avoid confounding effects on outcome variables.
Sample-Size Estimation
As this was an open-label, clinical superiority trial in patients with LDD and osteoporosis to determine whether CBT screws are stabler in posterior lumbar surgery than PSs based on fusion rate and clinical outcomes, we used noninferiority criteria to calculate the appropriate sample size.18 The primary end point was fusion rate, and the expected fusion rate of the control group using PSs was 93%, which agreed with several studies.3 For noninferiority of CBT versus PS in TLIF with two-sided 1% significance, power of 80%, and noninferiority margin of 15%, a sample of 56 patients for each group was necessary. Given an anticipated dropout rate of 10%, at least 124 patients were required.
Randomization
Patients were randomly assigned to undergo either midline lumbar fusion with CBT screws or TLIF combined with traditional PS fixation (PS group). Randomization was performed using nQuery Advisor PPS 6.01, which assigns numbers in strict chronological sequences and enters regular sequences for each group. Randomization was stratified via age (>60 versus <60 years), smoking status (smokers versus nonsmokers), and BMI (>24 versus <24). Each study participant was allocated a unique randomization number on screening completion. This was a single-blind study for patients, rather than the surgeon and other health providers.
Surgical Procedures and Postoperative Protocols
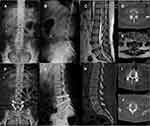
All surgeries were performed using the same operative technique. Transforaminal decompression was performed via a midline posterior incision. One polyether ether ketone interbody cage with the autograft bone locally obtained during decompression was routinely inserted in each patient for interbody fusion. In the CBT group, a bilateral screw-rod system with a cortical screw-fixation system (Medtronic CD Solera 4.75 spinal system) was used under fluoroscopic guidance. In the PS group, a bilateral screw-rod system with traditional PS fixation (CD Horizon Legacy spinal system) was used. Lastly, one drainage tube was placed beneath the paraspinal muscles and pulled out until the drainage volume in 24 hours was <50 mL. All patients were treated with the same postoperative protocols. They were permitted to ambulate after the drainage tube had been removed, and most were discharged on postoperative days 5–7. Patients were encouraged to wear a customized lumbar brace and avoid standing or sitting for long periods during the first month after surgery. They were able to resume normal activities at 3 months postoperation (Figures 1 and 2).
Outcome Measures
Primary End Point
The primary end point of our study was fusion rate. Fusion status was evaluated at 12 months postoperation. Fusion was defined as the presence of a continuous fusion mass inside or outside the cage on CT imaging. Status was regarded as nonunion when the fusion mass on CT was discontinuous. Two independent orthopedic spine surgeons blind to the surgery assessed the fusion status by CT. Radiographic data measurements were carried out using Picture Archiving and Communication Systems. All CT scans were taken using 1 mm intervals (Siemens Somatom Sensation 16).
Secondary End Points
Several secondary end points were measured: intensity of low-back and lower-extremity pain (using a VAS), functional status (using the Oswestry Disability Index [ODI] and Japanese Orthopaedic Association [JOA] scores), surgical morbidity (based on operating duration, incision length, estimated blood loss, and drainage volume), and incidence of intraoperative complications (nerve-root injury, durotomy, and screw misplacement) and postoperative complications (infection, cage malposition, screw loosening, and revision surgery). CBT-screw accuracy was evaluated according to the method described by Ding et al19 and PS-screw accuracy by the method of Abul-Kasim et al20 on postoperative CT scanning. Data were collected prospectively by the corresponding author, both perioperatively and at each follow-up visit.
Statistical Analysis
SPSS 18 was used for statistical analysis. Two-samples independent t-tests were conducted to assess differences in continuous variables of parametric data between the two cohorts. Fisher’s exact and x2 tests were used to analyze differences in categorical outcome variables, and P≤0.05 denoted statistical significance.
Results
Demographic Data
In sum, 124 eligible patients were enrolled and randomly allocated to a CBT group (62 patients) and PS group (62 patients). Eventually, 61 in the CBT group and 60 in the PS group qualified for the study, each of whom fully complied with the inclusion criteria (Figure 3). No significant differences were found between the groups in terms of age, sex, follow-up, smoking status, body-mass index, bone-mineral density, or surgical level distribution (Table 1).
 |
Table 1 Patient demographics and perioperative parameters |
 |
Figure 3 Flowchart of enrollment. |
Primary Outcome Measure
Based on CT scans, at 6 months postoperation 37 patients (60.66%) in the CBT group and 33 (55%) in the PS group showed fusion, with no significant difference between the groups (P=0.583). At 1 year postoperation, fusion had been achieved in 55 patients (90.16%) in the CBT group and 52 (86.67%) in the PS group, with no significant difference between the groups (P=0.583, Table 2).
 |
Table 2 Fusion status and complications |
Secondary Outcome Measures
Both had similar improvements in mean VAS scores postoperatively. Pain levels at last follow-up were significantly lower than preoperation for both groups, with means decreasing from 6.20±0.87 preoperatively to 1.49±0.74 at last follow-up in the CBT group and from 6.22±0.90 to 1.48±0.81 in the PS group, with no significant difference between the groups (Table 3). ODI scores decreased from 66.62±6.83 preoperation to 31.54±4.69 at 3 months postoperation to 15.87±2.15 at last follow-up in the CBT group and from 66.05±6.03 preoperation to 34.08±6.20 at 3 months postoperation to 15.90±2.27 at last follow-up in the PS group, a significant reduction. ODI scores at 3 months postoperation in the CBT group were significantly lower than the PS group, but showed no difference at other follow-up points (Table 3). JOA scores also improved from 12.21±1.92 preoperatively to 16.41±1.70 at 3 months postoperation and 25.05±1.10 at last follow-up in the CBT group and from 12.22±1.42 preoperatively to 15.30±1.45 at 3 months postoperation and 24.98±1.23 at last follow-up in the PS group. These ODI scores were not significantly different between the two groups, except that at 3 months postoperation (Table 3).
 |
Table 3 Clinical outcomes |
Perioperative parameters, ie, average surgery time, intraoperative blood loss, length of incision, and postoperative drainage, are given in Table 1. The CBT group had better outcomes than the PS groupon all of these. Perioperative complications, ie, durotomy, nerve-root injury and surgical site infection, between the groups were similar. Screw misplacement on postoperative CT occurred with two of 244 screws (0.82%) in the CBT group and three of 240 (1.25%) in the PS group, which indicated no significant difference between the groups (P=0.684). During follow-up, three of 244 (1.23%) screws in the CBT group were loose and had migrated on CT scans, the incidence of which was significantly lower than the PS group, with 14 of 240 (5.83%) screws loosening (P=0.006). One of 61 patients in the CBT group had had cage migration at 3 months post operation, while there was none in the PS group. No significant difference was found.
Two patients (3.28%) in the CBT group returned to hospital during follow-up because of screw loosening (one) and cage migration (one), with the latter receiving revision surgery and obtaining relief. Four patients (6.67%) in the PS group returned to hospital during follow-up because of screw loosening and internal fixation failure, of which three received revision surgery and obtained relief. However, there was no significant difference between the groups in terms of revision surgery. Complications occurred in four of 61 patients (6.56%) in the CBT group and 12 of 60 (20.0%) in the PS group during follow-up, with significantly higher incidence in the PS group (P=0.034, Table 2).
Discussion
According to rough estimates, there are approximately 200 million people worldwide with osteoporosis,21 and as the population ages and the expected life span increases, the prevalence of osteoporosis continues to rise. Rates of osteopenia and osteoporosis among patients undergoing spine surgery have been reviewed recent years. It was reported that 46.1% of male patients and 41.4% of female patients had osteopenia and 14% of male patients and 51.3% of female patients had osteoporosis in Chin et al.22 According to Bjerke et al,23 of 140 patients undergoing primary posterior thoracolumbar or lumbar fusion, the prevalence of osteopenia was 58.6% (82 of 140), and osteoporosis 10% (14 of 140). It has been well proven that surgical complications, such as instrument failure (cage migration, screw loosening, and pseudarthrosis) and subsequent vertebral compression fractures, correlate with osteoporosis.24,25 Both PS loosening and cage migration are correlated with an increase in back pain, rate of nonunion, and pseudarthrosis, leading to poor quality of life.26,27 In the treatment of elder patients with osteoporotic LDD, many surgical techniques have hsve shown satisfactory fusion rates and good stability, including cement augmentation of PS, expandable PSs, and CBT-screw fixation.28 CBT-screw fixation is considered a potential option for patients with osteoporosis undergoing lumbar fusion.29,30 However, the utilization of CBT screws in the osteoporotic lumbar spine are supported by limited data. This is the first study to thoroughly evaluate at least 2-year follow-up outcomes in patients with osteoporosis undergoing single-level TLIF using CBT screws and to compare these outcomes with those after single-level TLIF using PSs via prospectively collected data.
In the current study, similar clinical recovery was recorded in the two osteoporotic groups. Both pain intensity and function status in terms of VAS, ODI, and JOA scores in the two groups showed significant improvement compared to preoperation scores. There was no statistically significant difference between the two groups in improvements in VAS, ODI, and JOA scores. However, in the short-term postoperative period, within 3 months of surgery CBT fixation provided significantly better ODI and JOA scores tha PSs as a result of smaller skin incisions and less muscle dissection. In addition, CBT fixation resulted in lower surgical morbidity measured by average surgical time, intraoperative blood loss, length of incision, and postoperative drainage than the PS group. It has been reported that CBT fixation could offer several advantages that may overcome the limitations of PSs.8,15,16,31 The reduction in surgery-related morbidity may be attributed to the medial insertion point of CBT screws, less lateral muscle dissection, and shorter skin incisions. As an indicator of postoperative recovery, less postoperative drainage means better and faster recovery. Purposeful attention paid to soft-tissue handling, protection of muscles, and hemostasis leads to less trauma and damage, which could yield visible short-term and potential further benefits to patients, translating to significantly better ODI and JOA scores than those of PS patients in a 3-month postoperative period. The results from two prospective randomized controlled trials support our findings of CBT fixation’s advantages.32,33
For patients with osteoporosis who undergo PS fixation and interbody fusion, the stability of instrumentation and rate of union are the biggest concerns. As the primary end point of the current study, fusion rates between the two groups were compared. The fusion rate in the osteoporotic population is considered one of the most important factors in evaluation of the safety and efficacy of CBT fixation in lumbar fusion surgery and influences postoperative clinical outcomes and patient satisfaction. The fusion rate at 1-year follow-up was 86.67% (52 of 60) in the PS group and 90.16% (55 of 61) in the CBT group on CT scans (P=0.583). It was even higher in CBT group, while fusion rates at 1-year follow-up showed no significant differences between the groups. A meta-analysis of 14 studies (466 patients in a CBT group and 488 in a PS group) reported fusion rates of 92.6% (452 of 488 levels) in the CBT group and 92.7% (471 of 508 levels) in the PS group based on dynamic radiography and CT images between the 12- and 40-month follow-up, demonstrating no statistically significant difference in fusion rates between the two groups.34 The reported fusion rates in the two groups were similar to those in our current study. In a previous comparative study of CBT- and PS-screw fixation used in the treatment of osteoporotic LDD, CBT fixation achieved a higher fusion rate (24 of 31 vs 18 of 32, P=0.06) at 1 year postoperation on CT scans, with no significant difference. The results reported by Liu et al29 were similar to our study.
Loosening of PSs is known to be the main reason for failure in posterior stabilization for osteoporosis. Stability of an implanted PS depends on bone quality, implantation technique, and screw properties.35 In the current study, the incidence of screw loosening was much higher in the PS group than thef CBT group (P=0.006). The advantage of CBT screws lies in the superior biomechanical foundation in the osteoporotic spine. Cortical screws are minimally affected by the enhanced cancellous properties of osteoporotic lumbar pedicles, because they target the highly dense cortical bone, which is mostly spared by the osteoporotic disease process. According to the human cadaveric study of Weiser et al,36 mean fatigue load at failure was 264.9 N for standard PSs and increased significantly to 324.7 N by the use of cortical screws (P=0.014). This effect was particularly evident in reduced BMD vertebrae, rather than normal BMD.
CBT screws rely on four-point purchase between the dorsal cortex at the site of insertion, the medial and lateral cortices of the pedicle wall, and the curvature of the vertebral body wall to maximize cortical bone contact to improve purchase strength.14 Inaccurate placement may give rise to suboptimal fixation, even nerve injury. In our study, the incidence of misplacement in CBT group was similar to the PS group. The narrow nail trajectory of CBT screws puts higher demands on the technique of the surgeon. In terms of other surgical complications, such as durotomy, nerve-root injury, and surgical site infection, no significant difference was found between our two groups.
Our study has some limitations. It was conducted only on patients undergoing single-level TLIF surgery, which means the conclusions drawn may not be applied to subjects having multilevel fusion surgeries. The study was not blinded to the surgeons because of its nature, which might have produced performance bias. Moreover, this was a single-institution study, which may have led to some bias. Despite these limitations, the present study has some unique strengths. This is the first clinical research study with a prospective randomized design to evaluate CBT-fixation outcomes in an osteoporotic population. In our opinion, this study is meaningful in reporting 2-year follow-up outcomes, because it provides a reference for further studies regarding CBT fixation in osteoporotic patients undergoing lumbar fusion surgery.
Conclusion
CBT-screw fixation for single-level lumbar fusion in patients with osteoporosis provided improvement in clinical symptoms comparable to that of TLIF using PS fixation. In addition, statistically significant lumbar stability was found in the CBT group. We suggest that CBT-screw fixation is a reasonable and superior alternative to PSs in TLIF for osteoporotic populations.
Abbreviations
CBT, cortical bone trajectory; PS, pedicle screw; TLIF, transforaminal lumbar interbody fusion; VAS, visual analogue scale; ODI, Oswestry Disability Index; JOA, Japanese Orthopaedic Association; BMI, body-mass index; BMD, bone-mineral density.
Data Sharing Statement
The research is ongoing, and the authors are not allowed to share detailed data before the project finishes. If readers are interested in the relevant data, they can obtain these from the corresponding author after permission.
Ethics Approval and Informed Consent
The studies were approved by the institutional ethics committee of Beijing Chaoyang Hospital (2019-KE-2). All procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, the 1964 Declaration of Helsinki, and its later amendments, or comparable ethical standards. Informed consent was obtained from all participants.
Author Contributions
All authors made a significant contribution to the work reported, whether in conception, study design, execution, acquisition of data, analysis and interpretation, or all these areas, took part in drafting, revising, or critically reviewing the article, gave final approval to the version to be published, have agreed on the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
Funding
The study received funding support from Beijing Natural Science Funds (Haidian Original Innovation Joint Fund L202006).
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Lee N, Kim KN, Yi S, et al. Comparison of outcomes of anterior, posterior, and transforaminal lumbar interbody fusion surgery at a single lumbar level with degenerative spinal disease. World Neurosurg. 2017;101:216–226. doi:10.1016/j.wneu.2017.01.114
2. Cheng X, Zhang K, Sun X, et al. Clinical and radiographic outcomes of bilateral decompression via a unilateral approach with transforaminal lumbar interbody fusion for degenerative lumbar spondylolisthesis with stenosis. Spine J. 2017;17(8):1127–1133. doi:10.1016/j.spinee.2017.04.011
3. Lidar Z, Beaumont A, Lifshutz J, Maiman DJ. Clinical and radiological relationship between posterior lumbar interbody fusion and posterolateral lumbar fusion. Surg Neurol. 2005;64(4):303–308. doi:10.1016/j.surneu.2005.03.025
4. Babu R, Park JG, Mehta AI, et al. Comparison of superior-level facet joint violations during open and percutaneous pedicle screw placement. Neurosurgery. 2012;71(5):962–970. doi:10.1227/NEU.0b013e31826a88c8
5. Liu J, Deng H, Long X, Chen X, Xu R, Liu Z. A comparative study of perioperative complications between transforaminal versus posterior lumbar interbody fusion in degenerative lumbar spondylolisthesis. Eur Spine J. 2016;25(5):1575–1580. doi:10.1007/s00586-015-4086-8
6. Mehta VA, McGirt MJ, Garces Ambrossi GL, et al. Trans-foraminal versus posterior lumbar interbody fusion: comparison of surgical morbidity. Neurol Res. 2011;33(1):38–42. doi:10.1179/016164110X12681290831289
7. Santoni BG, Hynes RA, McGilvray KC, et al. Cortical bone trajectory for lumbar pedicle screws. Spine J. 2009;9(5):366–373. doi:10.1016/j.spinee.2008.07.008
8. Hung CW, Wu MF, Hong RT, Weng MJ, Yu GF, Kao CH. Comparison of multifidus muscle atrophy after posterior lumbar interbody fusion with conventional and cortical bone trajectory. Clin Neurol Neurosurg. 2016;145:41–45. doi:10.1016/j.clineuro.2016.03.005
9. Matsukawa K, Yato Y, Nemoto O, Imabayashi H, Asazuma T, Nemoto K. Morphometric measurement of cortical bone trajectory for lumbar pedicle screw insertion using computed tomography. J Spinal Disord Tech. 2013;26(6):E248–E253. doi:10.1097/BSD.0b013e318288ac39
10. Matsukawa K, Kato T, Yato Y, et al. Incidence and risk factors of adjacent cranial facet joint violation following pedicle screw insertion using cortical bone trajectory technique. Spine. 2016;41(14):E851–E856. doi:10.1097/BRS.0000000000001459
11. Matsukawa K, Taguchi E, Yato Y, et al. Evaluation of the fixation strength of pedicle screws using cortical bone trajectory: what is the ideal trajectory for optimal fixation? Spine. 2015;40(15):E873–E878. doi:10.1097/BRS.0000000000000983
12. Matsukawa K, Yato Y, Hynes RA, et al. Cortical bone trajectory for thoracic pedicle screws: a technical note. Clin Spine Surg. 2017;30(5):E497–E504. doi:10.1097/BSD.0000000000000130
13. Matsukawa K, Yato Y, Imabayashi H, Hosogane N, Asazuma T, Nemoto K. Biomechanical evaluation of cross trajectory technique for pedicle screw insertion: combined use of traditional trajectory and cortical bone trajectory. Orthop Surg. 2015;7(4):317–323. doi:10.1111/os.12212
14. Matsukawa K, Yato Y, Kato T, Imabayashi H, Asazuma T, Nemoto K. In vivo analysis of insertional torque during pedicle screwing using cortical bone trajectory technique. Spine. 2014;39(4):E240–E245. doi:10.1097/BRS.0000000000000116
15. Sakaura H, Miwa T, Yamashita T, Kuroda Y, Ohwada T. Posterior lumbar interbody fusion with cortical bone trajectory screw fixation versus posterior lumbar interbody fusion using traditional pedicle screw fixation for degenerative lumbar spondylolisthesis: a comparative study. J Neurosurg Spine. 2016;25(5):591–595. doi:10.3171/2016.3.SPINE151525
16. Sakaura H, Miwa T, Yamashita T, Kuroda Y, Ohwada T. Cortical bone trajectory screw fixation versus traditional pedicle screw fixation for 2-level posterior lumbar interbody fusion: comparison of surgical outcomes for 2-level degenerative lumbar spondylolisthesis. J Neurosurg Spine. 2018;28(1):57–62. doi:10.3171/2017.5.SPINE161154
17. Liu YZ, Hai Y, Zhang XN, et al. [Comparison of cortical bone trajectory screw fixation and pedicle screw fixation in posterior lumbar fusion]. Zhonghua Yi Xue Za Zhi. 2019;99(19):1473–1478. Chinese. doi:10.3760/cma.j.issn.0376-2491.2019.19.008
18. Piaggio G, Elbourne DR, Pocock SJ, Evans SJ, Altman DG, Group C. Reporting of noninferiority and equivalence randomized trials: extension of the CONSORT 2010 statement. JAMA. 2012;308(24):2594–2604. doi:10.1001/jama.2012.87802
19. Ding H, Han B, Hai Y, et al. The feasibility of assessing the cortical bone trajectory screw placement accuracy using a traditional pedicle screw insertion evaluation system. Clin Spine Surg. 2020. doi:10.1097/BSD.0000000000001059
20. Abul-Kasim K, Strombeck A, Ohlin A, Maly P, Sundgren PC. Reliability of low-radiation dose CT in the assessment of screw placement after posterior scoliosis surgery, evaluated with a new grading system. Spine. 2009;34(9):941–948. doi:10.1097/BRS.0b013e31819b22a4
21. Carlson BB, Salzmann SN, Shirahata T, et al. Prevalence of osteoporosis and osteopenia diagnosed using quantitative CT in 296 consecutive lumbar fusion patients. Neurosurg Focus. 2020;49(2):E5. doi:10.3171/2020.5.FOCUS20241
22. Chin DK, Park JY, Yoon YS, et al. Prevalence of osteoporosis in patients requiring spine surgery: incidence and significance of osteoporosis in spine disease. Osteoporos Int. 2007;18(9):1219–1224. doi:10.1007/s00198-007-0370-8
23. Bjerke BT, Zarrabian M, Aleem IS, et al. Incidence of osteoporosis-related complications following posterior lumbar fusion. Global Spine J. 2018;8(6):563–569. doi:10.1177/2192568217743727
24. Khalid SI, Nunna RS, Maasarani S, et al. Association of osteopenia and osteoporosis with higher rates of pseudarthrosis and revision surgery in adult patients undergoing single-level lumbar fusion. Neurosurg Focus. 2020;49(2):E6. doi:10.3171/2020.5.FOCUS20289
25. Park MK, Kim KT, Bang WS, et al. Risk factors for cage migration and cage retropulsion following transforaminal lumbar interbody fusion. Spine J. 2019;19(3):437–447. doi:10.1016/j.spinee.2018.08.007
26. Cho JH, Hwang CJ, Kim H, Joo YS, Lee DH, Lee CS. Effect of osteoporosis on the clinical and radiological outcomes following one-level posterior lumbar interbody fusion. J Orthop Sci. 2018;23(6):870–877. doi:10.1016/j.jos.2018.06.009
27. Formby PM, Kang DG, Helgeson MD, Wagner SC. Clinical and radiographic outcomes of transforaminal lumbar interbody fusion in patients with osteoporosis. Global Spine J. 2016;6(7):660–664. doi:10.1055/s-0036-1578804
28. Fischer CR, Hanson G, Eller M, Lehman RA. A systematic review of treatment strategies for degenerative lumbar spine fusion surgery in patients with osteoporosis. Geriatr Orthop Surg Rehabil. 2016;7(4):188–196. doi:10.1177/2151458516669204
29. Liu L, Zhang S, Liu G, Yang B, Wu X. Early clinical outcome of lumbar spinal fixation with cortical bone trajectory pedicle screws in patients with osteoporosis with degenerative disease. Orthopedics. 2019;42(5):e465–e471. doi:10.3928/01477447-20190604-01
30. Chen H, Liu S, Zhang J, et al. [A comparative study on treatment of lumbar degenerative disease with osteoporosis by manual and robot-assisted cortical bone trajectory screws fixation]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2020;34(9):1142–1148. Chinese. doi:10.7507/1002-1892.202001070
31. Takenaka S, Mukai Y, Tateishi K, Hosono N, Fuji T, Kaito T. Clinical outcomes after posterior lumbar interbody fusion: comparison of cortical bone trajectory and conventional pedicle screw insertion. Clin Spine Surg. 2017;30(10):E1411–E1418. doi:10.1097/BSD.0000000000000514
32. Lee GW, Son JH, Ahn MW, Kim HJ, Yeom JS. The comparison of pedicle screw and cortical screw in posterior lumbar interbody fusion: a prospective randomized noninferiority trial. Spine J. 2015;15(7):1519–1526. doi:10.1016/j.spinee.2015.02.038
33. Lee GW, Ahn MW. Comparative study of cortical bone trajectory-pedicle screw (cortical screw) versus conventional pedicle screw in single-level posterior lumbar interbody fusion: a 2-year post hoc analysis from prospectively randomized data. World Neurosurg. 2018;109:e194–e202. doi:10.1016/j.wneu.2017.09.137
34. Wang J, He X, Sun T. Comparative clinical efficacy and safety of cortical bone trajectory screw fixation and traditional pedicle screw fixation in posterior lumbar fusion: a systematic review and meta-analysis. Eur Spine J. 2019;28(7):1678–1689. doi:10.1007/s00586-019-05999-y
35. Cho W, Cho SK, Wu C. The biomechanics of pedicle screw-based instrumentation. J Bone Joint Surg Br. 2010;92(8):1061–1065. doi:10.1302/0301-620X.92B8.24237
36. Weiser L, Sellenschloh K, Puschel K, et al. Cortical threaded pedicle screw improves fatigue strength in decreased bone quality. Eur Spine J. 2020. doi:10.1007/s00586-020-06593-3
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.