Back to Journals » Clinical Ophthalmology » Volume 12
Corneal imaging and densitometry measurements in keratoconus patients to monitor disease progression and treatment outcomes after contact lens or Intacs treatment
Authors Alzahrani K , Cristian C , Harper S , Carley F, Brahma A, Morley D, Hillarby MC
Received 10 April 2018
Accepted for publication 29 May 2018
Published 4 September 2018 Volume 2018:12 Pages 1653—1658
DOI https://doi.org/10.2147/OPTH.S168865
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Khaled Alzahrani,1,2 Cristina Cristian,3 Sophie Harper,3 Fiona Carley,3 Arun Brahma,3 Debbie Morley,3 M Chantal Hillarby1
1Division of Pharmacy and Optometry, School of Health Sciences, University of Manchester, Manchester, UK; 2Ophthalmology Department, King Fahad Armed Forces Hospital, Jeddah, Saudi Arabia; 3Manchester Royal Eye Hospital, Manchester, UK
Aim: The aim of this study was to compare the pre- and posttreatment corneal densitometry and corneal thickness value of keratoconus (KCN) patients managed via contact lenses (CLs) or by both intrastromal corneal rings and contact lenses.
Patients and methods: This prospective study was performed at the Manchester Royal Eye Hospital, UK. Patients were recruited before treatment and followed up for 12 months. Data of corneal densitometry and corneal thickness were collected using the Oculus Pentacam at the pretreatment visit and posttreatment visit at 12 months.
Results: Corneal clarity significantly differs between both groups at pre treatment at zone 0–2 mm for the anterior layer (P=0.002). The same diversity is present at zone 2–6 mm for the anterior layer (P=0.003) and posterior layer (P=0.008). The corneal clarity diversity found was not statistically significant at 12 months post treatment (P>0.05). Corneal thickness was found to be statistically significantly different between pre treatment and post treatment for the CL group for central corneal thickness (CCT) and thinnest area (P=0.01 and P=0.02), respectively.
Discussion: This study shows that KCN management with Intacs was found to be effective in maintaining corneal clarity for a longer time than that with CL alone. On the other hand, corneal clarity reduces with disease progression in cases managed with CLs only. Analysis of Oculus Pentacam images provides an objective evaluation to monitor the corneal status after these different pathways of management.
Keywords: keratoconus, contact lenses, densitometry, Intacs, keratocyte, haze
Introduction
Keratoconus (KCN) is noninflammatory, progressive bilateral dystrophy with corneal stromal thinning.1 Spectacles and/or contact lenses (CLs) are the optional managements available at early stage of the disease. KCN is a progressive disease; when the advanced stage is reached, the only treatment option is corneal transplantation.2,3 In some cases when the cornea is considered clear and the only reasons for corneal transplantation are intolerance of CL or decreased visual acuity (VA), using alternative treatment may be in the best interest of both surgeon and patients.4
CLs have been used for long time to improve VA in KCN even at an advanced stage.5–8 Improving VA without causing any damage to the corneal structures is the most important aim.9 Rigid gas permeable (RGP) lenses are the most widely used lens for keratoconic management at the early stage of the disease.10 The three-point-touch fitting method is the most commonly used method for RGP lenses.11
Intrastromal Corneal Rings Intacs (Addition Technology, Fremont, CA, USA) as keratoconus treatment was first done by Colin et al.12,13 Intacs have been proved to improve the topographic regularity and CL intolerance, both leading to better VA.14–16 Intacs work via shortening the arc and flattening the central cornea along with providing a biomechanical strengthening for the corneal ectatic thinning.17,18 However, an Intac may give a good result in early and moderate stages of the disease, but the Intac is considered poor in advanced stages of keratoconus.19
Corneal clarity in KCN is stated to be at a high level when compared with normal, healthy eyes.20 However, most of keratoconics report increased corneal haze to differing levels. Less attention has been given to the effect of CLs and Intacs on formations of corneal haze.
This study aims to use the Oculus Pentacam (OCULUS Optikgerate GmbH, Wetzlar, Germany) to assess the corneal densitometry level changes in a keratoconic eye before and after fitting with a CL; second, compare changes that takes place in corneal densitometry before and after implanting Intacs in the eye for keratoconic management; and finally, assess the corneal clarity changes between both treatment procedures at pre treatment and post treatment.
Patients and methods
This is a prospective, comparative, and nonrandomized cross-sectional study and was conducted at Manchester Royal Eye Hospital (MREH). This study was approved by Central Manchester University Hospitals NHS Foundation Trust, Manchester, UK, and NREC local ethics committee (ref: 15/NE/0363). The research followed the tenets of the Declaration of Helsinki.
Patients who were aged >13 years at the CL fitting or Intacs and CL placements and had a 1 year follow-up post treatment were selected for this study. A written informed consent was obtained from the patients older than 18 years. For those younger than 18 years, a written consent was obtained from parents and legal guardian in addition to an assent form from the patients. These patients were identified and then recruited following a clinic visit. Clinical notes were examined, where relevant information including patient age, gender, and date of procedures were recorded for descriptive data analysis. Posttreatment parameters including best-corrected visual acuity (BCVA) in logMAR, refraction, and relevant clinical details were extracted from the clinical notes for data analysis. Pretreatment and posttreatment data of corneal densitometry and central corneal thickness (CCT) were retrieved from the Oculus Pentacam.
CL fitting
All patients were fitted with RGP CLs by an optometrist at MREH. The fitting included analysis of the topography results and slit-lamp examination. The fitting procedures followed standard methods for keratoconic patients.11,21,22
Intacs procedure
Intacs fitting was performed by one surgeon for all subjects in this study. The procedure was done using local anesthesia. Mean corneal thickness was >425 μm for all patients and >450 μm at the proposed insertion locations. Keratometry reading showed 58 diopter (D) with mean 50 D and standard deviation (SD) 5.27 for Simk1 and mean 52.77 D and SD 8.34 for Simk2 with clear central cornea. Endothelial cell count was >1,000 cells/mm2. Two Intacs segments of 150–350 μm thickness for each eye were used to embrace the steepest keratoconic meridian at a depth between 300 and 400 μm. The aim was to achieve maximum flattening of the cornea.23 All the cases were fitted with RGP CL based on unsatisfactory VA with spectacles or soft CL correction.
Pentacam imaging
Pentacam topography images are routinely used clinically to assess patients mostly as a corneal diagnostic tool. Densitometry software allows the corneal clarity to be measured from the same image. The protocol takes only 5 minutes, and it is noninvasive, so carries no risks to the patient.24 A good single image of the cornea was taken at each visit, which met the quality requirement determined by Pentacam analysis. The measurements from the Pentacam images were compared for pre treatment and post treatment for both groups at 12 months. The same measurements were compared between groups at 1 year post treatment.
Statistical analyses
IBM SPSS Statistics for Mac, Version 23.0 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism version 7.00 for Windows (GraphPad Software, Inc., La Jolla, CA, USA) were used to perform analysis. Normality of data was assessed using the Shapiro–Wilk W-test. Data were represented in each follow-up as the mean (M)±SD at baseline and 12 months. Independent t-test for data was used to compare both groups. A paired t-test analysis was used to analyze follow-ups as compared to the baseline. A P-value of <0.05 was considered as a statistically significant result.
Results
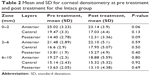
A total of 27 eyes from 16 patients met our criteria at 12 months posttreatment follow-up. These were seven patients (nine eyes) for Intac treatment with a mean±SD age of 32.04±8.38 years and nine patients (18 eyes) for CL treatment with a mean±SD age of 30.22±7.27 years, which is not statistically significant (P>0.05). The two groups showed similar demographic data.
At pre treatment, the mean (SD) of BCVA logMAR was found to be 0.11 (0.14) for the CL group, whereas it was 0.42 (0.25) for the Intacs group. At post treatment, the mean (SD) of BCVA logMAR was found to be 0.16 (0.35) for the CL group and 0.24 (0.35) for the Intacs group. When both groups were compared in terms of BCVA, a significant difference was found in pre treatment (P<0.001). In contrast, at 12 months post treatment, no statistically significant difference was found between both groups (P>0.05).
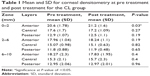
The study found a statistically significant increase for the CL group for mean corneal densitometry only in the anterior layer of zone 0–2 mm between pre treatment and 12 months post treatment (P<0.05; Table 1). However, the Intacs group showed no statistical difference between corneal densitometry at pre treatment and 12 months post treatment (P>0.05; Table 2).
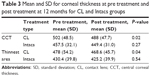
  | Table 1 Mean and SD for corneal densitometry at pre treatment and post treatment for the CL group |
  | Table 2 Mean and SD for corneal densitometry at pre treatment and post treatment for the Intacs group |
Corneal densitometry at pretreatment comparison between both groups showed a difference at zone 0–2 mm for the anterior layer (P=0.0001). A significant difference was also seen in the 2–6 mm zone for the anterior layer (P=0.006) and the posterior layer (P=0.01).
Comparing the mean corneal densitometry value between both groups at 12 months post treatment reveals no difference in any zones or layers (P>0.05; Figure 1).
Corneal thickness was found to decrease statistically significant between pre treatment and post treatment for the CL group in both CCT and the thinnest area (P=0.02 and P=0.04), respectively. This difference was not seen for the Intacs group (P>0.05, Table 3). Additionally there was a significant difference between both groups at pre treatment for CCT (P=0.02) and thinnest area (P=0.02). Similar differences were found at 12 months post treatment in CCT (P=0.3) and thinnest area (P=0.2; Table 3).
Discussion
Corneal clarity change in KCN has been reported in recent studies.25,26 Factors that can alter corneal clarity include age.27–29 Viral corneal disease as well as keratoconus and Fuchs disease (Fuchs endothelial dystrophy [FED]) can have an impact of decrease in corneal clarity. KCN progression shows decreased corneal clarity.20,30 Management of KCN by corneal collagen cross-linking (CXL) was found to have an effect on corneal clarity and increased corneal haze.31,32 However no reports about the effect of CLs or Intacs on corneal clarity using Pentacam have been published yet.
CL management of KCN is a well-documented evidence-based option for patients. This research did not find any statistical differences in corneal clarity between baseline and 12 months post fitting of CL except at the anterior layer of zone 0–2 mm.
KCN management can involve the Intacs procedure for some advanced cases where patients have difficulty using RGP lenses. This study found no significant change in corneal clarity after 12 months after Intacs were fitted compared to the baseline.
The results for both the Intac and CL groups showed higher corneal haze than published data for age-matched healthy controls at both pretreatment and posttreatment imaging.28,33 Both treatment groups (CLs and INTACS) showed significant differences pretreatment clarity with that at 12 months post treatment, however, there was no difference between the two treatment groups. Other studies of KCN management show a difference between pre treatment and post treatment of corneal clarity, for example, collagen cross-linking where corneal clarity is increased (CXL).32,34
Damage of keratocytes has been associated with haze development post CXL in many reports.33,35,36 It has been reported that corneal morphology can be altered due to a long-period wearing of CLs.37 However, keratocyte density does not seem to be affected by wearing daily CLs for a long period.38 Correspondingly, endothelial cell density was found to be unaffected in keratoconus for those patients who were not wearing CLs.39 However, there are conflicting reports on changes in endothelial density, increasing,40 no change,41 or decreasing,42 for long-time wearing of CL. On the other hand, Intacs placed in the cornea lead to a decrease in irregular astigmatism caused by KCN without attacking the center of the cornea and its tissues.4,43 It is commonly believed that lipid deposits will accumulate after Intac insertion.44 Lipids affect the extracellular matrix in the cornea due to change in the biomechanics,45 and this may change corneal clarity. However, we showed that clarity was increased 12 months after Intacs were fitted.
CLs may cause alteration of corneal thickness.46,47 This could be linked to structural alteration in the cornea due to reduced basal epithelial cells.48 Our result shows statistically significant change between pre treatment and post treatment, which increases the CCT.49 These changes in corneal thickness could be due to inflammation caused by CL wear or induced by an increase in proinflammatory cytokines in tears,50 which increased with wearing CLs.51 However, this study did not find any effect of the Intacs procedure on corneal thickness. These differences of the effect on corneal thickness between both treatment forms despite using CL could be due to lens mechanical effect.52,53
This study was limited by a small sample size; therefore, result is preliminary and needs to be replicated in a large study group. Another limitation was the average age of the participants, which may reflect a stable stage of keratoconic disease progression.
Conclusion
Corneal clarity seems not to be affected by management of the disease progression either by CL alone or Intacs and CL. However, treatment with Intacs seems to stabilize the corneal clarity in comparison with that with CLs alone. Intacs slow down the decrease in clarity associated with the CL treatment alone. Maybe there is less irritation and hence less inflammation after the Intacs are fitted. This study represents the first report, to our knowledge, about change in clarity measured using the Pentacam in keratoconic patients under management by CLs alone or Intacs and CLs. Analysis of Oculus Pentacam images provided an objective evaluation to monitor the corneal status after these treatments.
Acknowledgments
The authors thank the MREH (corneal clinic) for supporting patient’s recruitments for this study. This study was supported by Armed Forces Medical Services, Ministry of Defence, Riyadh, Saudi Arabia.
Disclosure
The authors report no conflicts of interest in this work.
References
Krachmer JH, Feder RS, Belin MW. Keratoconus and related noninflammatory corneal thinning disorders. Surv Ophthalmol. 1984;28(4):293–322. | ||
Kymionis GD, Siganos CS, Tsiklis NS, et al. Long-term follow-up of Intacs in keratoconus. Am J Ophthalmol. 2007;143(2):236–244. e231. | ||
Romero-Jiménez M, Santodomingo-Rubido J, Wolffsohn JS. Keratoconus: a review. Cont Lens Anterior Eye. 2010;33(4):157–166. | ||
Colin J. European clinical evaluation: use of Intacs for the treatment of keratoconus. J Cataract Refract Surg. 2006;32(5):747–755. | ||
Mandathara Sudharman P, Rathi V, Dumapati S. Rose K lenses for keratoconus – an Indian experience. Eye Contact Lens. 2010;36(4):220–222. | ||
Kok JH, van Mil C. Piggyback lenses in keratoconus. Cornea. 1993;12(1):60–64. | ||
Barnett M, Mannis MJ. Contact lenses in the management of keratoconus. Cornea. 2011;30(12):1510–1516. | ||
Yeung K, Eghbali F, Weissman BA. Clinical experience with piggyback contact lens systems on keratoconic eyes. J Am Optom Assoc. 1995;66(9):539–543. | ||
Barr JT, Zadnik K, Wilson BS, et al. Factors associated with corneal scarring in the Collaborative Longitudinal Evaluation of Keratoconus (CLEK) Study. Cornea. 2000;19(4):501–507. | ||
Mandell RB. Contemporary management of keratoconus. Int Cont Lens Clin. 1997;24(2):43–58. | ||
Leung KK. RGP fitting philosophies for keratoconus. Clin Exp Optom. 1999;82(6):230–235. | ||
Colin J, Cochener B, Savary G, Malet F. Correcting keratoconus with intracorneal rings. J Cataract Refract Surg. 2000;26(8):1117–1122. | ||
Rabinowitz YS. INTACS for keratoconus. Int Ophthalmol Clin. 2006;46(3):91–103. | ||
Colin J, Cochener B, Savary G, Malet F, Holmes-Higgin D. INTACS inserts for treating keratoconus: one-year results. Ophthalmology. 2001;108(8):1409–1414. | ||
Siganos CS, Kymionis GD, Kartakis N, Theodorakis MA, Astyrakakis N, Pallikaris IG. Management of keratoconus with Intacs. Am J Ophthalmol. 2003;135(1):64–70. | ||
Boxer Wachler BS, Christie JP, Chandra NS, Chou B, Korn T, Nepomuceno R. Intacs for keratoconus. Ophthalmology. 2003;110(5):1031–1040. | ||
Shetty R, Kurian M, Anand D, Mhaske P, Narayana KM, Shetty BK. Intacs in advanced keratoconus. Cornea. 2008;27(9):1022–1029. | ||
Alió JL, Shabayek MH, Artola A. Intracorneal ring segments for keratoconus correction: long-term follow-up. J Cataract Refract Surg. 2006;32(6):978–985. | ||
Alió JL, Shabayek MH, Belda JI, Correas P, Feijoo ED. Analysis of results related to good and bad outcomes of Intacs implantation for keratoconus correction. J Cataract Refract Surg. 2006;32(5):756–761. | ||
Lopes B, Ramos I, Ambrósio R. Corneal densitometry in keratoconus. Cornea. 2014;33(12):1282–1286. | ||
Lee JL, Kim MK. Clinical performance and fitting characteristics with a multicurve lens for keratoconus. Eye Contact Lens. 2004;30(1):20–24. | ||
Das S, John B, Mohan S, Rajan M. Contact lens fitting in keratoconus–a prospective study on visual and functional outcomes. Int J Res Med Sci. 2015;3(8):1851–1857. | ||
Armitage WJ, Tullo AB, Larkin DF. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222–1223. | ||
Cho YK, Chang HS, La TY, Ty L, et al. Anterior segment parameters using Pentacam and prediction of corneal endothelial cell loss after cataract surgery. Korean J Ophthalmol. 2010;24(5):284–290. | ||
Anayol MA, Sekeroglu MA, Ceran BB, Dogan M, Gunaydin S, Yilmazbas P. Quantitative assessment of corneal clarity in keratoconus: a case control study of corneal densitometry. Eur J Ophthalmol. 2016;26(1):18–23. | ||
Alzahrani K, Carley F, Brahma A, et al. Corneal haze in juvenile and adult keratoconus patients after corneal cross-linking. Acta Ophthalmol. 2017;95(S259). | ||
Dhubhghaill SN, Rozema JJ, Jongenelen S, Hidalgo IR, Zakaria N, Tassignon MJ. Normative values for corneal densitometry analysis by Scheimpflug optical assessment. Invest Ophthalmol Vis Sci. 2014;55(1):162–168. | ||
Alzahrani K, Carley F, Brahma A, Morley D, Hillarby MC. Corneal clarity measurements in healthy volunteers across different age groups: observational study. Medicine. 2017;96(46):e8563. | ||
Garzón N, Poyales F, Illarramendi I, et al. Corneal densitometry and its correlation with age, pachymetry, corneal curvature, and refraction. Int Ophthalmol. 2017;37(6):1263–1268. | ||
Mofty H, Alzahrani K, Carley F, et al. Evaluation of corneal symmetry after UV corneal crosslinking for keratoconus. Clin Ophthalmol. 2017;11:11. | ||
Gutiérrez R, Lopez I, Villa-Collar C, González-Méijome JM. Corneal transparency after cross-linking for keratoconus: 1-year follow-up. J Refract Surg. 2012;28(11):781–786. | ||
Greenstein SA, Fry KL, Bhatt J, Hersh PS. Natural history of corneal haze after collagen crosslinking for keratoconus and corneal ectasia: Scheimpflug and biomicroscopic analysis. J Cataract Refract Surg. 2010;36(12):2105–2114. | ||
Wollensak G, Spoerl E, Wilsch M, Seiler T. Keratocyte apoptosis after corneal collagen cross-linking using riboflavin/UVA treatment. Cornea. 2004;23(1):43–49. | ||
Herrmann CIA, Hammer T, Duncker GIW. Haze-Bildung nach Vernetzungstherapie bei Keratokonus [Haze formation after crosslinking therapy in keratoconus]. Der Ophthalmologe. 2008;105(5):485–487. German. | ||
Mazzotta C, Balestrazzi A, Baiocchi S, Traversi C, Caporossi A. Stromal haze after combined riboflavin-UVA corneal collagen cross-linking in keratoconus: in vivo confocal microscopic evaluation. Clin Exp Ophthalmol. 2007;35(6):580–582. | ||
Dhaliwal JS, Kaufman SC. Corneal collagen cross-linking: a confocal, electron, and light microscopy study of eye bank corneas. Cornea. 2009;28(1):62–67. | ||
Carlson KH, Bourne WM. Endothelial morphologic features and function after long-term extended wear of contact lenses. Arch Ophthalmol. 1988;106(12):1677–1679. | ||
Patel SV, Mclaren JW, Hodge DO, Bourne WM. Confocal microscopy in vivo in corneas of long-term contact lens wearers. Invest Ophthalmol Vis Sci. 2002;43(4):995–1003. | ||
Timucin OB, Karadag MF, Cinal A, Asker M, Asker S, Timucin D. Assessment of corneal endothelial cell density in patients with keratoconus not using contact lenses. Cont Lens Anterior Eye. 2013;36(2):80–85. | ||
Hollingsworth JG, Efron N, Tullo AB. In vivo corneal confocal microscopy in keratoconus. Ophthalmic Physiol Opt. 2005;25(3):254–260. | ||
Yeniad B, Yilmaz S, Bilgin LK. Evaluation of the microstructure of cornea by in vivo confocal microscopy in contact lens wearing and non-contact lens wearing keratoconus patients. Cont Lens Anterior Eye. 2010;33(4):167–170. | ||
Niederer RL, Perumal D, Sherwin T, Mcghee CN. Laser scanning in vivo confocal microscopy reveals reduced innervation and reduction in cell density in all layers of the keratoconic cornea. Invest Ophthalmol Vis Sci. 2008;49(7):2964–2970. | ||
Ertan A, Kamburoğlu G. Intacs implantation using a femtosecond laser for management of keratoconus: comparison of 306 cases in different stages. J Cataract Refract Surg. 2008;34(9):1521–1526. | ||
Twa MD, Kash RL, Costello M, Schanzlin DJ. Morphologic characteristics of lamellar channel deposits in the human eye: a case report. Cornea. 2004;23(4):412–420. | ||
Ly LT, Mcculley JP, Verity SM, Cavanagh HD, Bowman RW, Petroll WM. Evaluation of intrastromal lipid deposits after intacs implantation using in vivo confocal microscopy. Eye Contact Lens. 2006;32(4):211–215. | ||
Yeniad B, Yiğit B, Işsever H, Közer Bilgin L, Bilgin LK. Effects of contact lenses on corneal thickness and corneal curvature during usage. Eye Contact Lens. 2003;29(4):223–229. | ||
Liu Z, Pflugfelder SC. The effects of long-term contact lens wear on corneal thickness, curvature, and surface regularity. Ophthalmology. 2000;107(1):105–111. | ||
Bitirgen G, Ozkagnici A, Malik RA, Oltulu R. Evaluation of contact lens-induced changes in keratoconic corneas using in vivo confocal microscopy. Invest Ophthalmol Vis Sci. 2013;54(8):5385–5391. | ||
Romero-Jiménez M, Santodomingo-Rubido J, Flores-Rodríguez P, González-Méijome JM. Short-term corneal changes with gas-permeable contact lens wear in keratoconus subjects: a comparison of two fitting approaches. J Optom. 2015;8(1):48–55. | ||
Lema I, Durán JA. Inflammatory molecules in the tears of patients with keratoconus. Ophthalmology. 2005;112(4):654–659. | ||
Lema I, Durán JA, Ruiz C, Díez-Feijoo E, Acera A, Merayo J. Inflammatory response to contact lenses in patients with keratoconus compared with myopic subjects. Cornea. 2008;27(7):758–763. | ||
Rathi VM, Mandathara PS, Dumpati S. Contact lens in keratoconus. Indian J Ophthalmol. 2013;61(8):410. | ||
Ghosh S, Mutalib HA, Sharanjeet-Kaur, Ghoshal R, Retnasabapathy S. Effects of contact lens wearing on keratoconus: a confocal microscopy observation. Int J Ophthalmol. 2017;10(2):228. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.