Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
COPD-Related Modification to the Airway Epithelium Permits Intracellular Residence of Nontypeable Haemophilus influenzae and May Be Potentiated by Macrolide Arrest of Autophagy
Authors Poh WP , Kicic A , Lester SE, Nguyen PT, Bakaletz LO , Reynolds PN , Hodge S, Roscioli E
Received 13 January 2020
Accepted for publication 30 March 2020
Published 4 June 2020 Volume 2020:15 Pages 1253—1260
DOI https://doi.org/10.2147/COPD.S245819
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Russell
Wee-Peng Poh,1 Anthony Kicic,1– 5 Susan E Lester,6 Phan T Nguyen,7,8 Lauren O Bakaletz,9 Paul N Reynolds,7,8 Sandra Hodge,7,8 Eugene Roscioli7,8
1Telethon Kids Institute, Centre for Health Research, The University of Western Australia, Nedlands 6009, Western Australia, Australia; 2Occupation and Environment, School of Public Health, Curtin University, Perth 6845, Western Australia, Australia; 3School of Biomedical Sciences, The University of Western Australia, Nedlands 6009, Western Australia, Australia; 4Department of Respiratory and Sleep Medicine, Perth Children’s Hospital, Nedlands 6009, Western Australia, Australia; 5Centre for Cell Therapy and Regenerative Medicine, School of Medicine and Pharmacology, The University of Western Australia, Nedlands 6009, Western Australia, Australia; 6Department of Rheumatology, The Queen Elizabeth Hospital, Woodville, SA, Australia; 7Department of Thoracic Medicine, Royal Adelaide Hospital, Adelaide, SA, Australia; 8Department of Medicine, The University of Adelaide, Adelaide, SA, Australia; 9Center for Microbial Pathogenesis, The Research Institute at Nationwide Children’s Hospital and the Ohio State University College of Medicine, Columbus, OH, USA
Correspondence: Eugene Roscioli
Adelaide Health and Medical Science Building, Corner of North Terrace and George St, Adelaide 5005, South Australia, Australia
Tel +61 8 8313 7088
Email [email protected]
Introduction: COPD is an inflammatory airway pathology associated with recurrent infection by nontypeable Haemophilus influenzae (NTHi) that is not effectively managed by macrolide antibiotic therapy. We hypothesised that NTHi is able to reside intracellularly within COPD-derived airway epithelial cells (AEC), and that the factors contained in cigarette smoke when coupled with exposure to erythromycin or azithromycin arrest autophagy, the principle mechanism responsible for clearing intracellular bacteria (called “xenophagy”).
Methods: Cultures of bronchial airway epithelial cells derived from control and COPD participants were differentiated at an air–liquid interface and exposed to macrolide antibiotics, 10% cigarette smoke-extract (CSE) and NTHi. Markers of autophagic flux and intracellular NTHi were assessed using Western blot analysis and transmission electron microscopy.
Results: AEC treated with macrolide antibiotics or 10% CSE exhibited a block in autophagic flux as evidenced by a concomitant increase in LC3-II and Sequestosome abundance (vs control; both P < 0.01). While control AEC showed no clear evidence of intracellular NTHi, COPD-derived cultures exhibited abundant NTHi within the cytoplasm. Further, intracellular NTHi that were encapsulated within vesicles propagated from the apical epithelial layer to the basal cell layer.
Discussion: Taken together, our findings indicate that COPD, cigarette smoke and macrolide antibiotics potentiate the susceptibility to persistent intracellular NTHi. A major mechanism for this is arresting normal autophagic flux in airway epithelial cells. Hence, structural modifications that mitigate this off-target effect of macrolides have significant potential to clear intracellular NTHi and thereby reduce the influence of this pathogen in the airways afflicted by COPD.
Keywords: antibiotic resistance, azithromycin, intracellular bacteria, macrolide, xenophagy
Introduction
COPD is the third leading cause of death worldwide.1 Alongside consumption of cigarettes, infection with NTHi is a primary factor driving declined lung function and mortality, with approximately 50% of COPD exacerbations attributed to NTHi, which is now a WHO classified priority pathogenic bacterium.1,2 Paradoxically, NTHi is also a clinically benign commensal in the nasopharynx of most healthy individuals.2 Hence, disease-related modifications during COPD produce conditions that strongly favour NTHi infection. Growing evidence for an intracellular aspect to NTHi’s pathogenicity,3 aligns with the pathobiology of COPD, whereby protracted activation and injury of airway epithelial cells (AEC) allows the accumulation of intrinsic defects that promote bacterial invasion and an opportunity to exploit the intracellular environment.
Autophagy is a fundamental intracellular process whereby the autophagosome encapsulates potentially harmful cytoplasmic components and is co-digested with the cargo upon fusion with a lysosome, thus releasing biosynthetic intermediates for metabolic processes (the entire process is termed autophagic flux). This is an essential cellular response to cell stress such as starvation, oxidative stress and inflammation.4,5 Defective autophagic flux has been reported in several chronic lung diseases,6,7 although its role in COPD has not been well defined. However, important for COPD, autophagy is the primary mechanism used to clear invading bacteria, a process called xenophagy.
Xenophagy is a selective form of autophagy that rapidly digests intracellular microbes. Autophagy and xenophagy share molecular mechanisms including mTOR (primary inhibitor of initiation), AuTophaGy (ATG) related complexes that are essential for initiation of auto/xenophagic flux, ATG5, −8 and −16L1 for membrane assembly and maturation, the cargo chaperones Sequestosome, Optineurin, and NDP52, and Light Chain 3-II (LC3-II) which is essential throughout auto/xenophagic flux, LAMP2, RAB7, RAB9, and PtdIns3KC3 (many functions including lysosome formation) and IFN-γ, TLR-3/4, ICAM1, and NOD2 (microbe-host cell interactions).8–10 However, clinically important pathogens have evolved mechanisms allowing them to evade, block and usurp the xenophagic apparatus as an essential aspect of their virulence.11 Some bacteria that infect the airways evade or usurp the xenophagic machinery by secreting inhibitory factors (eg Listeria),1 preventing autophagosome-lysosome fusion (eg Legionella), or upregulating and recruiting LC3 to facilitate their survival and replication (eg S. aureus).10,12 One study found NTHi can exist within late endosomes,13 and more recently NTHi was found to promote the generation of de novo autophagic complexes in Hep-2 cells.14
The prevailing consensus is that autophagic flux is blocked during COPD, which is evidenced by an accumulation of autophagosomes and the inhibition of autophagic survival processes that closely corresponds with COPD-related deficits at the epithelium, including nutrient depletion, fragility, and unscheduled senescence.1,15 Further, exogenous exposures associated with COPD, including cigarette smoke and corticosteroids, are known to block autophagic flux,16 and there is a well-described defect in mitophagy (autophagic clearance of defective mitochondria) that aligns with these observations.17 Hence, in a scenario for COPD in which xenophagic flux is similarly inhibited, an increase in defective xenophagic apparatus could serve to enhance NTHi’s entry into fragile AEC and provide a niche for intracellular persistence. This is consistent with the mechanism employed by S aureus which usurps the xenophagic machinery, where it evades intracellular antimicrobial surveillance and remains metabolically active.11,12
Hence, there is converging evidence that intrinsic defects and exogenous exposures synonymous with COPD potentiate host factors which may restrict xenophagic clearance of NTHi, enabling intracellular residence. Yet, there is a surprising paucity of data describing how the usually innocuous NTHi becomes an influential pathogenic vector in the context of disease, let alone COPD. We report our observations for NTHi within COPD-derived AEC unhindered by autophagic activity, and show AEC exposed to macrolide antibiotics and the factors contained in cigarette smoke exhibit a block in autophagic flux far beyond either treatment in isolation.
Methods
COPD and COPD-Related Exposure Modelling of the Human Airway Epithelium
We employed an air–liquid interface (ALI) model to approximate normal AEC-NTHi interactions and responses to exogenous exposures. This contrasts with prior studies examining NTHi infection, which have primarily utilised undifferentiated and submerged cell line systems, without a disease component or exposure, and which have yielded limited insights. Bronchial brushing for ALI culture samples were approved by the Royal Adelaide Hospital Human Research Ethics (Protocol #R20020811), and all participants provided written informed consent. Cultures were derived from controls (n=5 never smokers, one female, 40 years ±22.3 SD, FEV1/FVC 88.6% ±7) and COPD participants (n=3, one female, 63 years ±4.1 SD, FEV1/FVC 53.1% ±5). Basal progenitor AEC stem cells collected from bronchial brushing samples were propagated at an ALI as previously described.15 Briefly, airway stem cells were expanded in T25 culture flasks, and then transferred to transwells and allowed to propagate to confluence (4–6 days). Thereafter, the apical media was removed and the basal chamber media was replaced with differentiation media containing retinoic acid (24–28 days). Cultures were used as experimental models when the mucociliary phenotype was evident (eg Figure 2A–C), via routine light microscopy and when electrical impedance of the epithelial barrier exceeded 500 Ω.cm2. ALI cultures were exposed to a clinical isolate of NTHi (24 h; MOI 50),18 cigarette smoke extract (CSE; as previously described),15 in addition to azithromycin, erythromycin and bafilomycin for 24 h (Merck KGaA Inc, Darmstadt, Germany).
Western Blot Protein Analysis
Protein was isolated from differentiated AEC within the transwells in situ, and Western blot analysis was performed as previously described,19 with the exception of the Rubicon antibody (Cell Signaling Technology Inc., Danvers, MA, USA). Densitometry of histogram analyses was performed using Multi Gauge software (V3.1; Fujifilm, Tokyo, Japan). Density scores were analysed using a random effects gamma (log link) mixed-model regression to allow for correlated treatment responses within each culture. Results were normalised to both β-actin and the biological control and expressed as relative abundance. The statistical analysis was performed using R statistical software (release 3.2.3) for n=4 control cultures.
Electron Microscopy
High-resolution host-microbe and microbe-autophagic apparatus interactions were assessed in situ with previously described methods using transmission electron microscopy (TEM),15 and scanning electron microscopy (SEM).20 For both, the fixative agents were applied directly to cells on the transwell at the end of the exposure period to maintain cell morphology and preserve host–microbe interactions. The transwell membranes were then excised from the transwell frame using a scalpel and embedded/mounted to the TEM mould and SEM peg (respectively), for analysis. TEM and SEM analyses were performed for control (n=3) and COPD (n=3) derived AEC ALI cultures.
Results and Discussion
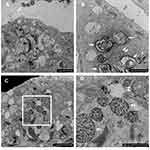
First, we asked whether COPD-derived AEC exhibit protracted (24 h) intracellular survival of NTHi when compared to cultures derived from control participants. Control AEC cultured with NTHi exhibit strong activation of microbial clearance processes as evidenced by abundant vacuolated structures consistent with lysosomes and autophagosomes (Figure 1). The relative efficacy of this response meant we were unable to identify intracellular NTHi with certainty, with the exception of irregularly shaped electron dense structures matching the size of NTHi (approximately 500 nm) within double membrane envelopes (Figure 1B), and digestion products within autolysosomes (Figure 1C and D). However, we cannot rule out that intracellular NTHi was effectively cleared by a related microbial degradative pathway before the 24 h observation interval, and which may be determined by blocking autophagy (eg ATG5 knockout) in control cells using a time course infection protocol. Further, an apparent lack of free NTHi within the cytosol in control AEC may also be explained by effective mucociliary (Figure 2A and B), barrier and defensin activity, and that stem cells from the relatively older COPD participants (albeit non-significant vs controls), may contribute to intracellular infectivity.
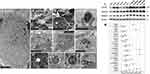
In contrast, COPD-derived AEC were frequently observed interacting with NTHi via villi-like cytoplasmic projections (SEM, Figure 2C, D, and H), that appear to encapsulate and internalise NTHi (as previously described).21 NTHi were observed in COPD-derived AEC, either free in the cytoplasm or encapsulated within membrane-bound vesicles, possibly endosomes, autophagosome and/or LAPosomes (vesicles of the LC3-Associated Phagocytosis (LAP) non-canonical autophagy pathway,22 Figure 2E–G). These observations in COPD-derived AEC support findings for intracellular NTHi identified by Woo et al in the context of otitis media.23 While it is unclear whether an endocytic process is a requisite for NTHi internalisation, we frequently observed an interruption in a NTHi-containing vesicle (NTH-CV), and NTHi in proximity to empty vesicles (Figure 2E–G and I–K), indicating a potential mechanism of vesicle escape. Indeed, in Figure 2F and K, NTHi appears to push out from the NTHi-CV, perhaps as part of a replicative process, as evidenced by its elongated, sigmoid, and lobular shape. Clustering depicted in Figure 2G is also suggestive of cell division, and aligns with the observation of an intracellular bacterial community that was recently applied to NTHi in a clinical model of otitis media.24 Interestingly, their close proximity to the transwell membrane (Figure 2F and G) suggests a mode of propagation from the apical to the basal progenitor cell population, which differentiate to generate the three-dimensional epithelial architecture that defines the ALI culture model. Given basal AEC remain viable for an extended period, the ability to traverse the epithelial compartment may partly explain the clinical persistence of NTHi within the COPD airways, whereby exacerbating factors lead to epithelial fragility and senescence that allows a reservoir of NTHi to express virulence factors after a period of host-cell adaption.11 Importantly, we were unable to definitively resolve NTHi within a double membrane bilayer (the diagnostic feature of an autophagosome; eg Figure 1B and D), and NTHi-CVs were not observed interacting with lysosomes, even when in proximity to autophagic activity (Figure 2F). As autophagy/xenophagy is usually an efficient process,11 these observations suggest that the inhibition of autophagic flux in the context of COPD may be a primary disease-related phenomena that permits intracellular residence, and resistance to clinical interventions.
The COPD airway is exposed to exogenous factors that modulate autophagy, such as cigarette smoke and corticosteroids,15,16 and which may also reprogram progenitor AEC towards chronic disease.25 Although macrolide therapies are relatively efficacious for the clinical signs of NTHi infection, there is a high rate of reinfection in COPD, and there are macrolide-resistant strains. Further, macrolides are known to modulate autophagy. Rapamycin and bafilomycin have well established pro- and anti-autophagic effects (respectively); however, less is known about the macrolides indicated for respiratory infections. Indeed, of the two studies which examine azithromycin in secondary-cell models of airway disease, one reports an increase in autophagic flux in smooth muscle cells for asthma,26 and the other a decrease in flux and elevated bacterial load in macrophages in the context of cystic fibrosis.27
Hence, we asked whether azithromycin or erythromycin, with or without CSE, can elicit a reduction in autophagy in primary differentiated AEC. We observed a restriction in autophagic flux for either macrolide or CSE-exposed AEC as evidenced by a significant increase in the essential autophagy protein LC3-II (all P<0.05), concomitant with an increase in the autophagy receptor, Sequestosome, which is normally co-degraded with the cargo it chaperones to the autophagosome (Figure 2L–M, and see Figure 3). While the macrolide-induced elevation in Sequestosome did not reach significance, a reduction of this autophagy receptor would be expected with a significant increase in LC3-II (which should also be degraded during autophagy) if autophagic activity is uninhibited. As expected, CSE elicited a moderate blockade of autophagic flux, with concomitant and significant increases in both LC3-II and Sequestosome. In contrast, the combination of macrolide/CSE resulted in an appreciable block in autophagy, with a significant increase in both LC3-II and Sequestosome for each exposure (consistent with a log-additive effect). The block in autophagy was most pronounced for 50 mg/mL azithromycin/CSE (LC3-II +38.3-fold, CI95% [18.99–77.06], P<0.001; Sequestosome +3.3-fold, CI95% [1.65–6.68], P=0.001), compared to that of bafilomycin (10 nM; LC3-II +22.3-fold, CI95% [11.09–45.00], P<0.001; Sequestosome +4.9-fold, CI95% [2.43–9.86], P<0.001), which is a potent inhibitor of autophagy and a positive control (Figure 2L–M). Conversely, while Rubicon was also significantly increased following CSE exposure (P<0.05), its abundance was not substantively influenced by either macrolide, suggesting distinct regulation of LAP vs canonical autophagy in the context of these exposures. LAP is known to suppress canonical autophagy and favour internalisation and infectivity of a number of respiratory pathogens,22 which is consistent with our observations.
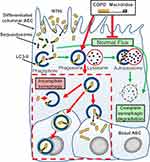
Conclusion
For the most part, current therapeutic options for COPD cannot reverse disease progression after smoking cessation, and it is of concern that some therapeutics may contribute to this phenomenon. Here we show that differentiated cultures of COPD-derive primary AEC permit intracellular residence of NTHi (vs control), and that the macrolide antibiotics erythromycin and azithromycin elicit a potent block in autophagic flux, particularly as a co-exposure with CSE (summarised in Figure 3). Taken together, our findings raise questions regarding the use of macrolide antibiotics to manage NTHi in current smokers, particularly as the epithelium in these individuals are already accumulating defects that potentiate autophagic insufficiency. An essential next question is whether there exists a causative link consistent with COPD, and COPD-related exposures that arrest autophagic flux, as primary factors that effect a block in xenophagic clearance of intracellular NTHi. So while modification of the antibiotic properties of macrolides to curtail the emergence of resistant strains is a high priority, the additional therapeutic challenge is to address the chemical properties of macrolides that produce off-target effects that limit autophagy.
Acknowledgments
We are grateful for the expertise and support of the clinical staff of the Royal Adelaide Hospital Thoracic Unit and the participants enrolled into this study who generously provided their valuable time and tissue samples. This study was funded by the Royal Adelaide Research Committee, Royal Adelaide Hospital Research Fund, The Health Services Charitable Gifts Board, and the Rebecca L Cooper Medical Research Foundation.
Disclosure
Lauren O. Bakaletz is the inventor of IP related to the development of a human vaccine antigen that is licensed to GSK and entitled to royalties. Paul N. Reynolds received travel supports from Boehringer Ingelheim, grants from Royal Adelaide Hospital Research Fund, grants from NHMRC, and salary from South Australia Health, during the conduct of the study; grants from NHMRC, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Barnes PJ, Burney PG, Silverman EK, et al. Chronic obstructive pulmonary disease. Nat Rev Dis Primers. 2015;1:15076. doi:10.1038/nrdp.2015.76
2. Bakaletz LO, Novotny LA. Nontypeable Haemophilus influenzae (NTHi). Trends Microbiol. 2018;26(8):727–728. doi:10.1016/j.tim.2018.05.001
3. King PT, Sharma R. The lung immune response to nontypeable Haemophilus influenzae (lung immunity to NTHi). J Immunol Res. 2015;2015:706376. doi:10.1155/2015/706376
4. Martinet W, Agostinis P, Vanhoecke B, Dewaele M, De Meyeret GRY. Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci. 2009;116(9):697–712. doi:10.1042/CS20080508
5. Yang J, Carra S, Zhu WG, Kampinga HH. The regulation of the autophagic network and its implications for human disease. Int J Biol Sci. 2013;9(10):1121–1133. doi:10.7150/ijbs.6666
6. Mizumura K, Cloonan S, Choi ME, et al. Autophagy: friend or foe in lung disease? Ann Am Thorac Soc. 2016;13(Suppl 1):S40–7. doi:10.1513/AnnalsATS.201507-450MG
7. Nakahira K, Pabon Porras MA, Choi AMK. Autophagy in pulmonary diseases. Am J Respir Crit Care Med. 2016;194(10):1196–1207. doi:10.1164/rccm.201512-2468SO
8. Staples KJ, Taylor S, Thomas S, et al. Relationships between mucosal antibodies, non-typeable Haemophilus influenzae (NTHi) infection and airway inflammation in COPD. PLoS One. 2016;11(11):e0167250. doi:10.1371/journal.pone.0167250
9. Bauckman KA, Owusu-Boaitey N, Mysorekar IU. Selective autophagy: xenophagy. Methods. 2015;75:120–127. doi:10.1016/j.ymeth.2014.12.005
10. Huang J, Brumell JH. Bacteria–autophagy interplay: a battle for survival. Nat Rev Microbiol. 2014;12(2):101–114. doi:10.1038/nrmicro3160
11. Sui X, Liang X, Chen L, et al. Bacterial xenophagy and its possible role in cancer: a potential antimicrobial strategy for cancer prevention and treatment. Autophagy. 2017;13(2):237–247. doi:10.1080/15548627.2016.1252890
12. Chandra P, Kumar D. Selective autophagy gets more selective: uncoupling of autophagy flux and xenophagy flux in Mycobacterium tuberculosis-infected macrophages. Autophagy. 2016;12(3):608–609. doi:10.1080/15548627.2016.1139263
13. Morey P, Cano V, Marti-Lliteras P, et al. Evidence for a non-replicative intracellular stage of nontypable Haemophilus influenzae in epithelial cells. Microbiology. 2011;157(1):234–250. doi:10.1099/mic.0.040451-0
14. Espinoza-Mellado Mdel R, Reyes-Picaso C, Garcés-Pérez MS, et al. NTHi triggers autophagy in HEp-2 cells. Arch Microbiol. 2016;198(2):199–204. doi:10.1007/s00203-015-1167-3
15. Roscioli E, Tran HB, Jersmann H, et al. The uncoupling of autophagy and zinc homeostasis in airway epithelial cells as a fundamental contributor to COPD. Am J Physiol Lung Cell Mol Physiol. 2017;313(3):L453–L65. doi:10.1152/ajplung.00083.2017
16. Wang J, Wang R, Wang H, et al. Glucocorticoids suppress antimicrobial autophagy and nitric oxide production and facilitate mycobacterial survival in macrophages. Sci Rep. 2017;7(1):982. doi:10.1038/s41598-017-01174-9
17. Fujii S, Hara H, Araya J, et al. Insufficient autophagy promotes bronchial epithelial cell senescence in chronic obstructive pulmonary disease. Oncoimmunology. 2012;1(5):
18. Pizzutto SJ, Yerkovich ST, Upham JW, Hales BJ, Thomas WR, Chang AB. Children with chronic suppurative lung disease have a reduced capacity to synthesize interferon-gamma in vitro in response to non-typeable Haemophilus influenzae. PLoS One. 2014;9(8):e104236. doi:10.1371/journal.pone.0104236
19. Hodge G, Roscioli E, Jersmann H, et al. Steroid resistance in COPD is associated with impaired molecular chaperone Hsp90 expression by pro-inflammatory lymphocytes. Respir Res. 2016;17:135. doi:10.1186/s12931-016-0450-4
20. Ha KR, Psaltis AJ, Tan L, Wormald PJ. A sheep model for the study of biofilms in rhinosinusitis. Am J Rhinol. 2007;21(3):339–345. doi:10.2500/ajr.2007.21.3032
21. Ketterer MR, Shao JQ, Hornick DB, Buscher B, Bandi VK, Apicella MA. Infection of primary human bronchial epithelial cells by Haemophilus influenzae: macropinocytosis as a mechanism of airway epithelial cell entry. Infect Immun. 1999;67(8):4161–4170. doi:10.1128/IAI.67.8.4161-4170.1999
22. Mostowy S. Autophagy and bacterial clearance: a not so clear picture. Cell Microbiol. 2013;15(3):395–402. doi:10.1111/cmi.12063
23. Woo JI, Oh S, Webster P, Lee YJ, Lim DJ, Moon SK. NOD2/RICK-dependent β-defensin 2 regulation is protective for nontypeable Haemophilus influenzae-induced middle ear infection. PLoS One. 2014;9(3):e90933. doi:10.1371/journal.pone.0090933
24. Hardison RL, Harrison A, Wallace RM, et al. Microevolution in response to transient heme-iron restriction enhances intracellular bacterial community development and persistence. PLoS Pathog. 2018;14(10):e1007355. doi:10.1371/journal.ppat.1007355
25. Rahman SM, Ji X, Zimmerman LJ, et al. The airway epithelium undergoes metabolic reprogramming in individuals at high risk for lung cancer. JCI Insight. 2016;1(19):e88814. doi:10.1172/jci.insight.88814
26. Stamatiou R, Paraskeva E, Boukas K, Gourgoulianis KI, Molyvdas PA, Hatziefthimiou AA. Azithromycin has an antiproliferative and autophagic effect on airway smooth muscle cells. Eur Respir J. 2009;34(3):721–730. doi:10.1183/09031936.00089407
27. Renna M, Schaffner C, Brown K, et al. Azithromycin blocks autophagy and may predispose cystic fibrosis patients to mycobacterial infection. J Clin Invest. 2011;121(9):3554–3563. doi:10.1172/JCI46095
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.