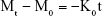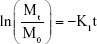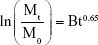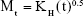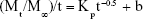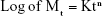Back to Journals » International Journal of Nanomedicine » Volume 13
Controlled release of organic–inorganic nanohybrid:cefadroxil intercalated Zn–Al-layered double hydroxide
Authors Khan SB, Alamry KA, Alyahyawi NA, Asiri AM
Received 4 April 2017
Accepted for publication 28 December 2017
Published 29 May 2018 Volume 2018:13 Pages 3203—3222
DOI https://doi.org/10.2147/IJN.S138840
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster

Sher Bahadar Khan,1,2 Khalid A Alamry,2 Nedaa A Alyahyawi,3 Abdullah M Asiri1,2
1Center of Excellence for Advanced Materials Research (CEAMR), King Abdulaziz University, Jeddah, Saudi Arabia; 2Chemistry Department, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia; 3Chemistry Department, Faculty of Science, University of Jeddah, Jeddah, Saudi Arabia
Background: The intercalation of an antibiotic drug, cefadroxil (CD), into the inter-gallery of Zn, Al nitrate-layered double hydroxide (LDH) was accomplished using a co-precipitation method. This formed a nanostructured organic–inorganic hybrid material that can be exploited for the preparation of a controlled release formulation.
Materials and methods: The drug–LDH nanohybrid was characterized by using field emission scanning electron microscopy (FE-SEM), energy dispersive X-ray spectroscopy (EDS), Fourier transform infrared spectroscopy (FTIR) thermogravimetric (TG) analysis, X-ray powder diffraction (XRD) and UV–visible (UV–vis) absorption spectroscopy, which confirmed the intercalation process. Release tests of nanohybrid in the presence or absence of NaCl or polyacrylamide (PAM) were performed in vitro in gastric (pH 1.2), lysosomal (pH 4.0), intestinal (pH 6.8) and blood (pH 7.4) simulated fluid using UV–vis spectroscopy.
Results: At pH 1.2, LDH was dissolved and intercalated antibiotic released from ZnAl-CD in a molecular form, which led to a significant increase in the antibiotic’s solubility. Results showed that the release of drug from nanohybrid at pH 4.0, 6.8 and 7.4 was a sustained process.
Conclusion: This material might reduce side effects by the release of the drug in a controlled manner. However, it was found that the presence of Cl or PAM species in the release media has a negative impact on the release behavior. The weathering mechanism is responsible for the release of CD from the nanocomposite at pH 1.2, while the mechanism of anion exchange may be responsible for the release behavior at pH 4.0, 6.8 and 7.4. A number of kinetic models were chosen to gain more insights into the mechanisms of drug release. At pH 1.2, the zero-order model most satisfactorily explained the release kinetics of CD, while the release data of CD at pH 4.0, 6.8 and 7.4 were governed by Bhaskar kinetics.
Keywords: drug delivery system, ZnAl-CD LDH nanohybrid, hydrotalcite, antibiotic, drug release, kinetics
Introduction
In recent years, drug administration techniques have received extensive attention from pharmaceutical researchers; in particular, controlled release drug delivery systems release the bioactive agent at a specific rate and at the right place to perform its task with efficiency.1–3 These systems aim to maintain drug concentration in the blood or in target tissues at the desired levels for a longer period of time, to decrease the administration frequency and also minimize possible side effects.4–6
Recently, new materials, namely, nanohybrids or heterostructured nanocomposites, have been widely used in the drug delivery field. Of the heterostructured nanomaterials, layered nanohybrids can be easily manipulated. These materials are typically prepared via intercalation reaction, which can be defined as the incorporation of an organic guest into a two-dimensional (2-D) layered inorganic matrix, yielding interesting hybrid organic/inorganic nanocomposite materials that can used as carriers of drugs and biological active species (Figure 1).7,8
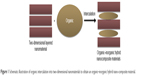  | Figure 1 Schematic illustration of organic intercalation into two-dimensional nanomaterials to obtain an organic–inorganic hybrid nano-composite material. |
As an important class of inorganic layered nanomaterials, clays have been widely studied as active molecular ion delivery vehicles which can be either cationic with negatively charged aluminosilicate layers or anionic with positively charged hydroxide layers.8,9 Layered double hydroxide (LDH) is a synthetic analog of naturally occurring mineral hydrotalcite10 and has contributed to the development of drug delivery applications due to its advantages such as anion exchange properties,11 low cytotoxicity, simple preparation,12 biocompatibility and biodegradability properties,13 basicity of the matrix,14 and low cost.15 It also leads to the achievement of the following: (i) stabilization of the drugs,16 (ii) improving both their solubility and cellular uptake,15,17 and (iii) controllably releasing them.18
The chemical composition of LDH can be described by the formula [MII1-xMIIIx(OH)2]x+(An-)x/n·mH2O, where MII, MIII, and An- represent divalent, trivalent metal ions and an interlayer anion of valence n, respectively. The x value is the molar ratio MIII/(MII + MIII) and m is the amount of water in moles.19 The structure is similar to that of brucite (Mg(OH)2), where a portion of the divalent cations are substituted by trivalent ones, which leads to positively charged sheets.20 These charges are balanced by the interlayer anions.21 Thus, LDH structure is composed of MII and MIII, which can be accommodated in the octahedral sites in the brucite-like layers and the hydroxyl groups located on the vertices of the octahedral.15,19
Due to the effectiveness and importance of biocompatible Zn, Al LDH, we have chosen this material as nanocapsules for cefadroxil (CD). In its ionic form, it is found that zinc plays a vital role in the human body, where it has the ability to prevent DNA damage, as well as being useful in cancer therapy and controlling the activities of many enzymes.22
CD is one of the first-generation cephalosporin β-lactam antibiotics and acts against both Gram-positive and Gram-negative bacteria. Delivery in a nanocapsule may overcome its negative effects and low solubility that lead to poor absorption of drug by living organisms.23
A series of pharmaceutically active compounds including diclofenac, methotrexate, chloramphenicol, salicylate, cefazolin, ketoprofen, lawsone and chlorogenic acid were intercalated into Zn, Al LDHs, and the feasibility of LDH-based drug delivery systems was demonstrated.8,22,24–28 CD has been intercalated into gastroretentive delivery systems (GRDS) in order to analyze its release in acidic pH and to examine the solubility and stability of CD. It was found that the release of CD formulation was sustained for around 14 hours.29 A similar study used hydoxy propyl methyl cellulose as a delivery system for CD to measure the in vitro drug release rate at pH 1.2 and found that the release period was prolonged.30 Conversely, the current study demonstrates that LDH is dissolved rapidly and, consequently, CD is fully released at pH 1.2.
The objectives of this work were to evaluate the ability of LDH to intercalate an antibiotic, and to study the influence of release media pH, and the effect of the presence of exchangeable anions (chloride ions, NaCl) or a macromolecule (polyacrylamide; PAM) in the release medium, on the release rate and the amount of drug released, as well as the effectiveness of the LDH matrix in increasing solubility of the antibiotic and reducing side effects. In order to investigate these objectives, we have intercalated CD in Zn–Al-LDH, containing nitrate as the interlayer anion, by a co-precipitation method. Then, the prepared CD delivery system was characterized by using different physicochemical techniques. After that, we quantitatively monitored the controlled release process of the drug. Finally, the release profile data were fitted by mathematical models in order to evaluate the CD-release mechanisms and kinetics.
Materials and methods
Materials
The metal salts used in the synthesis of the drug–LDH nanohybrid were zinc nitrate hexahydrate (Zn(NO3)2·6H2O, EMD Millipore, Billerica, MA, USA) and aluminum nitrate nonahydrate (Al(NO3)3·9H2O, Fluka). CD (C16H17N3O5S, MW 363.392 g/mol)and sodium hydroxide pellets (98%) were obtained from Sigma-Aldrich Co. (St Louis, MO, USA) and BDH Chemical Ltd, respectively. Deionized water (ELGA, specific resistivity 18 MΩ) was used for the preparation of the sample and washing the obtained product. All chemicals in this synthesis were used as received without further purification.
Preparation and characterization
Synthesis of LDH-intercalated drugs
Intercalation of the drug into LDH was carried using the co-precipitation method at constant pH.18,31 A 200 mL solution containing the metal ions (3.751 g of Al(NO3)3·9H2O (0.01 mol) and 5.949 g of Zn(NO3)2·6H2O (0.02 mol)), was slowly added to the CD solution, which was prepared by dissolving 0.363 g (0.001 mol) of CD in 100 mL of deionized water, Zn2+/Al3+/guest molar ratio 0.2/0.1/0.01. Then the freshly prepared NaOH solution (0.5 M) was added drop by drop to yield pH = 9; when addition was completed the suspension was vigorously stirred at 60°C for 24 h in order to reach equilibrium. Following this, the product was centrifuged at 2,900 rpm for 10 minutes and washed with deionized water three times to remove the unreacted salt ions, impurities and excess drug. The precipitate was dried at room temperature and the sample was named ZnAl-LDH–CD.
Characterization
X-ray diffraction (XRD)
The samples’ crystal structure was recorded by using X-ray diffraction (XRD) with a diffractometer (Thermo Scientific™ ARL™ X’TRA Powder Diffractometer; Thermo Fisher Scientific, Waltham, MA, USA) having λ = 0.154 nm of Cu Kα radiations. The operating current and voltage of the instrument was 50 mA and 40 KV, respectively.
Fourier transform infrared (FTIR)
FTIR spectroscopy was used to obtain direct evidence on the intercalation process as well as the interaction between the drug anions, and LDH and the interlayer water. FTIR spectra of the pristine LDH, and drug–LDH were measured with a Nicolet™ iS™10 FTIR spectrometer (Thermo Fisher Scientific) at 25°C. The spectrum of each sample was collected by cumulating 50 scans, with 32 cm−1 resolution in the mid-infrared (4,000–400 cm−1).
Thermogravimetric (TG) analysis
The TG technique is used to determine the decomposition temperature of the interlayer drug anion, and its content in the ZnAl-CD LDH nanoparticle. Additionally, it can provide information about the thermal stability of drug–LDH. Thermogravimetric (TG) analysis was performed on a Thermal Analysis Q500 (TA Instruments, New Castle, DE, USA) under a nitrogen atmosphere between 25 and 1,000°C at 5°C/minute heating rate.
Ultraviolet–visible (UV–vis) absorption spectroscopy
The potential interactions between host and guest as well as those between guest and guest, stability of the interlayer drug and release from the nanocomposite were studied by UV–vis absorption spectroscopy (GENESYS 10S UV–vis, Thermo Fisher Scientific) using quartz cuvettes.
Field emission scanning electron microscopy (FE-SEM)
The FE-SEM (JSM-7600F, JEOL, Tokyo, Japan) was used to examine the surface morphology of the organic–inorganic nanohybrid.
Scanning electron microscopy/energy-dispersive X-ray spectroscopy (SEM-EDS)
The elemental composition of the drug-LDH sample was examined using EDS (Oxford) operated at 20 kV attached to the SEM (JSM-7600F, JEOL).
Controlled release study of the anions from the nanohybrids into various media
The release experiments were carried out as follows. The release media included buffer A as a simulated gastric fluid (pH 1.2), buffer B as a simulated lysosomal fluid (pH 4.0), buffer C as a simulated intestinal fluid (pH 6.8), and buffer D as a simulated blood fluid (pH 7.4).
A known amount of nanohybrid (10 mg of drug–LDH) was dispersed in release medium solution (100 mL). The mixture was continuously stirred at a slow speed and kept at 37°C. At specified time intervals, a certain volume of solution (2.0 mL) was withdrawn, centrifuged at 1,300 rpm for 100 s and the resulting supernatant was filtered to remove the insoluble particles, then the absorbance of the CD was determined at 230 nm and further the amount of drug was estimated from absorbance using the calibration curve, through which the percentage of drug liberated was calculated.
Release studies also were performed in the presence of NaCl, by dissolving 10 mg of sodium chloride in the flask containing the nanohybrid sample, and analyzing as previously mentioned.
Additionally, the release processes were studied after adding 1.7 mL of PAM ((C3H5NO)n, Sigma-Aldrich Co.) to 100 mL of release medium that containing 8 mg of bionanocomposite sample (drug–LDH).
Mathematical modeling of drug release
The release kinetics of the drug can be determined through mathematical models.
Zero-order kinetics
The zero-order model that describes the dissolution process related to surface reaction controlled kinetics can be given by Equation 1.32,33
| (1) |
where Mt and M0 are the drug quantity released at release time t and the initial quantity of drug in the nanohybrid, respectively, K0 is a proportionality constant expressed in concentration per unit time, and t is the time expressed in minutes.34
First-order kinetics
The first-order model (Equation 2) describes the absorption of some drugs, as well as their elimination.35 Unlike the zero-order model, the rate of the release depends on the drug content existing in the LDH matrix.36
| (2) |
where K1 is the first-order constant (5).
Bhaskar model
The Bhaskar model is expressed by Equation 3:
| (3) |
where B the corresponding kinetic constant. The Bhaskar equation describes the diffusion of drugs from the resins and insoluble inorganic matrixes.37,38
Higuchi model
The Higuchi model explains that drug diffusion from semi-solid or solid matrixes is a rate-determining step of the release process.33
| (4) |
where KH is the diffusion rate constant. The rate of release of the drug is proportional to the reciprocal of the square root of the measurement time.39
Parabolic diffusion model
This model is extensively used to elucidate whether the release process occurs through a surface/edge diffusion process. Equation 5 describes the parabolic model.
| (5) |
where Mt and M∞ are the drug quantities released in time t and infinite time, respectively, Kp is the rate constant for parabolic diffusion model and b is a constant whose chemical significance is not clearly resolved.38,40,41
Korsmeyer–Peppas (K-P) model
In 1984, Korsmeyer and Peppas developed an empirical mathematical equation to confirm the drug mechanism which is represented as:42,43
| (6) |
where K is the rate constant for releasing characteristics of the drug/matrix system, while n is a release exponent that distinguish between release mechanisms of drugs; it was estimated from the slopes of a plot of the log fractional drug release vs log time.44–46
For the K-P model, an n value < 0.45 corresponds to quasi-Fickian diffusion. If the value of n is in the range 0.45–0.89, the release of the drug occurs through two processes, dissolution of LDHs and the diffusion of the drug, and the release process is called non-Fickian transport or anomalous diffusion mechanism. When n > 0.89, drug release follows dissolution of LDHs and the process is called super case transport II.47,48
Results and discussion
Physicochemical characterization of CD-LDH nanohybrid
XRD
The intercalation of CD into LDH was supported by the powder XRD pattern (Figure 2). All characteristic crystalline peaks of pristine LDH were clearly observed in the XRD patterns. However, the nanohybrid did not show any kind of drug crystalline peaks. Generally, LDH exhibited a (003) reflection equivalent to the basal spacing of 8.8–8.0 Å, which reflects the formation of NO3− or CO3− or Cl− or SO4− intercalated LDH. However, the CD-LDH nanohybrid exhibited a (003) peak at 3.4°, which gives the basal spacing from 8.0 Å to 25.1 Å for CD-LDH, indicating the intercalation of drugs into LDH. Subtracting the LDH layer thickness (8.0 Å) from the observed basal spacing, the gallery height can be estimated to be 17.1 Å, which is equal to the lateral molecular length of the CD drug. It suggested that intercalated drug molecules are arranged in a lateral manner in the interlayer space.
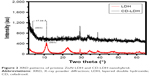  | Figure 2 XRD patterns of pristine ZnAl LDH and CD-LDH nanohybrid. |
FTIR
Figure 3 presents the FTIR analysis of the parent Zn-Al LDH-NO3 and the hybrid of CD-LDH. The pristine LDH spectrum (Figure 3A) shows a broad absorption band at 3,402 cm−1, which can be considered as a common feature for all LDH-like materials, attributable to the −OH stretching vibrations in the Zn/Al-NO3-LDH layers and physically adsorbed and co-intercalated water.49,50 The cause of low-frequency vibration of (OH) group in ZnAl-LDH compared to that of OH-stretching in free water, around 3,600 cm–1, is the hydrogen bonds formed between the interlamellar water and hydroxide groups of the Zn/Al inorganic layers.50,51 Also, the ZnAl–NO3 sample shows a strong peak at 1,338.4 cm–1 (due to ν of NO3−),52 and a weak band at 1,635.4 cm−1 (δH2O).53 Regarding the low wavenumber region of the spectrum (400–800 cm−1), it is expected that bands correspond to metal–oxygen and metal–hydroxyl vibration modes in the LDH sheets.54–56 These bands confirm the Zn–Al LDH structure.57
  | Figure 3 FTIR spectra of (a) pristine ZnAl-NO3 LDH and (b) nanohybrid CD-LDH. |
In the spectrum of the ZnAl-CD LDH nanohybrid (Figure 3B), the characteristic peak associated with the parent LDH-like hydroxide group, which also arises from the phenolic hydroxides, appears at 3,390.4 cm−1, which, together with the emergence of bands below 1,000 cm−1, indicates that the layers structure of LDH has not changed during the intercalation process.58–60 In addition, a pair of absorption bands appears at 1,631.6 and 1,350 cm−1 related to carboxylate asymmetric and symmetric stretching, respectively. Compared to −COO stretching vibration (1,650 and 1,400 cm−1) of pure CD,61 the shift of the two characteristic peaks to lower frequencies was due to ionization of CD (carboxylic groups)62–64 and the strong electrostatic interaction of terminal oxygens of organic anions in interlayer with the electropositive cations in layer as well as the formation of bonding hydrogen between oxygen from carboxylate and the hydroxyl groups in layers or with the interlayer water molecules.65–68 Depending on the above results, the CD is intercalated in its anionic form and is stable in the interlayer space of LDH.1,69 Of particular note is that the stretching frequency peak of nitrate groups may be overlapped with −COO− symmetric-stretching peak, which may not be completely removed from the interlayers during the intercalation process.70 This result is in agreement with the results of Meher71 and Ghotbi and Bin Hussein.58
TG analysis
By analyzing the TG curve of material CD-LDH, it is found that there are seven distinguishable stages of weight loss, which is in agreement with the differential thermogravimetric (DTG) analysis result (Figure 4A and B). In the first and second steps, from 25°C to 160°C, there are about 7.5% and 3.5% losses of the total weight, ascribed to the loss of the physically adsorbed water and water existing within the interlayer galleries, which are attached to the layer of the hydroxide and/or interlayer anion by hydrogen bonds, respectively.72,73 Correspondingly, two peaks at ca. 50°C and 125°C are observed in the DTG analysis.72 The third mass loss (12% weight loss) occurs in the region of 160°C–250°C and corresponds to a peak in the DTG curve at 225°C, possibly due to the loss of hydroxyl group from the Zn–Al layer, the condensation of which leads to formation of water vapor.62,70,74
  | Figure 4 Thermogravimetric (TG) analysis curves: (A) TG and (B) differential thermogravimetric (DTG) of nanohybrid CD-LDH. |
The DTG diagram shows a sharp peak at 260°C and two weak peaks at 340°C and 390°C associated with combustion of the intercalated CD. According to the TG curve, weight loss should take place in three consecutive steps (250°C–440°C), which leads eventually to the producing CO2, H2O and NOx.70,75
The organic CD ions in CD-LDH have a higher decomposition temperature than pure CD; the DTG curve of pure CD shows three weight losses at 191°C, 240°C and 350°C.76 This indicates the improved thermal stability of CD after its intercalation into LDH gallery,77,78 due to the host–guest interactions, which involves electrostatic attraction within the LDH layered structure and hydrogen bonding.70,72 These results are similar to the results of Lonkar et al79 and Wang et al80 who studied the intercalation of antioxidant and glutathione into LDH, respectively.
The final stage at 560°C is due to the formation of ZnO and ZnAl2O4, where a temperature treatment above 440°C causes a collapse of the layered structure of the sample and leads to full calcification of the solid as well as complete burn of drug component, thus giving rise to oxides of the cations.62,81,82
UV–vis spectroscopy
The UV–vis absorption spectrum of pristine CD (Figure 5A) shows two characteristic absorption bands at 230 and 263 nm, which are ascribed to the phenolic structure (π-π* transition) and electronic transitions of the enone form (n-π* transition), respectively.83–85
  | Figure 5 UV–visible absorption of (a) pure cefadroxil and (b) nanohybrid CD-LDH. |
After intercalation of CD in the ZnAl-LDH host (Figure 5B), it is obvious that the absorption band at 230 nm cannot be identified. This may be attributed to the effect of scattering of LDH particles. Meanwhile, the band at 263 nm shows an apparent red shift with Δλ ≈111 nm compared to CD.78,86 These results proved that the drug anions are well intercalated between the layers of the LDH.12 The cause of the occurrence of this shift in the spectrum is the interaction between the host and the guest as indicated by FTIR and TG. It is worth mentioning that electrostatic interaction with the host layer led to the stability of drug anions.78,86 Furthermore, the red shift may occur due to guest–guest interaction through π-π* conjugation of the phenol of interlayer CD ions.87,88 Considering the drug–LDH spectrum, the band became broader compared to that in the CD spectrum and this may be due to π-electron delocalization.89
FE-SEM
The surface morphology of the ZnAl-LDH–CD nanohybrid is shown in Figure 6. The micrographs were obtained with FE-SEM at 430× (Figure 6A), 800× (Figure 6B), 5,000× (Figure 6C), 9,500× (Figure 6D), 10,000× (Figure 6E) and 14,000× (Figure 6F) magnifications.
According to the literature,90 FE-SEM micrographs of the synthesized NO3–LDH show uniform and regular particles with a hexagonal plate-like shape. In contrast, the photographs of the nanohybrid reveal non-uniform irregular agglomerates of compact.91,92 Additionally, the FE-SEM analysis of nanohybrid suggests that the CD-LDH particles have a microblock-like structure.93
SEM-EDS
As illustrated in Figure 7, the EDS pattern of ZnAl-CD confirms the organic and inorganic composition of the CD-LDH nanohybrid, where it shows common energy lines corresponding to C, O, Zn and Al.94 The elemental composition as weight percentages and atomic percentages based on the EDS is shown in Table 1.
  | Table 1 EDS Analysis of CD-LDH |
In vitro drug release testing
The main part of this work is based on the compared release processes of the drug from the organic–inorganic nanohybrid, Zn-Al CD, in the different solution media, in the presence or absence of chloride anions (sodium chloride salt) or macromolecule (PAM). Since the final quantification of the antibiotic is based on a standard curve, relating the absorbance of the solution with the drug concentration, it must build the calibration curve from measurements at 230 nm in all pH medium in the expected concentration range (Figure 8). It is clear that the dependence of the absorbance on the concentration is linear in the investigated range with regression coefficients larger than 0.9 (Table 2).4,26,95 According to the literature, there are two main release mechanisms responsible for the rate of release and the amount released: anion-exchange (coupled with diffusion) of the drug molecules through the LDH material in alkaline or neutral media; and weathering/degradation of the LDH inorganic layers by protonation in strong acidic media.96–98 In mild acidic media, any of the two mechanisms would determine the drug release rate.33
  | Figure 8 Calibration curve of cefadroxil at the different pH values. |
  | Table 2 Linearity data of CD by UV spectroscopy |
CD-Zn/Al-LDH
The drug release profiles, ie, the percentage of CD released into the buffer solution vs time, have been plotted in Figure 9A (strong acidic), 9B (mild acidic), 9C (neutral) and 9D (slightly basic).
In simulated gastric fluid (Figure 9A), under strong acidic conditions, the LDH is dissolved rapidly and completely and the intercalated CD is fully released, in its molecular form, but not fully dissolved: 71% of CD is dissolved after the end of the experiment from drug-intercalated hydrotalcite.33,99,100
Bin Hussein et al92 pointed out that the layered structure of Zn–Al LDH quickly collapses, releasing α-naphthalene acetate (NAA) in strong acidic conditions. The release behavior for the ZnAl–CD nanohybrid does not demonstrate the possibility of the occurrence of ionic exchange between the interlayer CD and the chloride anions of the buffer solution, where the chloride ion affinity to the LDH layers is too low to cause drug replacement. As a result, there is only one mechanism that controls the release of the drug in this media: solid weathering.33,96 The intercalation process, as well as the rapid release of CD in the gastric medium, leads to an increase in the solubility of CD and its dissolution rate at the stomach.33,101 Ambrogi et al102 and Del Arco et al103 have reported that the presence of inorganic LDH matrix improves the solubility of hardly soluble non-steroidal anti-inflammatory drugs (NSAIDs).
In lysosomal, intestinal and blood simulated fluid, a similar behavior is observed for CD-intercalated LDH in all release media at early stages; about 64%, 49% and 58% of CD is released immediately at pH 4.0, 6.8 and 7.4, respectively.
Subsequently, however, the release of CD-LDH is slow and sustained: after 360 min there is a release amount of 84% at pH 4.0 (Figure 9B); ca. 69% at pH 6.8 (Figure 9C) and ca. 71% at pH 7.4 (Figure 9D).33,63,104,105 These findings are consistent with previous research, as reported by Yasin and Ismail27 and Zhang et al106 for samples containing lawsone and methotrexate intercalated in LDHs, where they studied the release of these drugs at different pH values.
The first stage of the release process can be attributed to the release of organic drug that was weakly adsorbed on the surface of the nanocomposite and that in the external part of the lamellar structure (LDH edges). The slow and sustained release of CD in the later stage may depend on the slow anion exchange between interlayered CD and the anions in the fluid.33,63,69 Unlike CD at the interlayer, the CD at the outside layers will be exposed directly to the medium and will be dissolved immediately.99
It is worth mentioning that partial destruction of the lamellar matrix did not happen in simulated lysosomal fluid. This means that the release process is subject to ion exchange mechanism only due to the high crystallinity of the LDH matrix and the great affinity of the intercalated anions for the layers.96 With regard to stability of LDH at and above pH 7, this matrix is able to maintain its lamellar structure because it is more stable at those pH values and, consequently, LDH acts like a drug release modulator and the release process was governed by ion exchange mechanism.107,108
From the release profiles in lysosomal, intestinal and blood simulated fluid, it is clear that the percentage of CD liberated does not reach 100%; this may be because of the characteristics of the ion-exchange reaction. This means that the interlayer anions cannot be exchanged completely.59,109,110 Nejati and Rezvani72 and Xia et al111 have studied the release of olsalazine at pH 7.4 and antihypertensive drugs at pH 4.25; these authors have highlighted that the release of drugs from nanohybrids (drug–LDH) is not complete, for the same reason as above.
The two-step release process can play an active role in therapeutic treatments, where the rapid release in the first stage allows the establishment of a therapeutic dose in a short period of time, while the slow stage allows the maintenance of this dose for long periods, ie, reduces the number of doses that are applied to patients, through the sustained release of drug from the ZnAl-LDH nanohybrid.32,112,113
CD-Zn/Al-LDH/NaCl
As well as the dependence of the CD release rate from the nanohybrid on pH, it is strongly influenced by medium anionic species and their affinity toward the interlayer of the inorganic interlamellar.
Figure 9B–9D show that the release percentages of CD from CD-LDH nanohybrid in the presence of chloride ions are less than those obtained in the absence of exchangeable anion. The release profiles indicated that about 57%, 43% and 50% CD was released immediately at pH 4.0, 6.8 and 7.4, respectively. About 76% of CD is found to be released after 360 min, in lysosomal medium, while a lower release of CD is found in intestinal medium with release of 58% after 6 h. In blood simulated medium, about 64% of the initial CD amount is released during 360 min.
According to the literature,114,115 the effect of various medium anionic species on the release of herbicides from the interlayer of ZnAl-LDH (ZAN) follows the following order: i) for single system release media, carbonate anion > phosphate anion > chloride anion; ii) for binary system release media, carbonate and phosphate anions > chloride and phosphate anions > chloride and carbonate anions; iii) for all systems release media, carbonate anion > phosphate anion > carbonate and phosphate anions > chloride and carbonate and phosphate anions > chloride and phosphate anions > chloride and carbonate anions > chloride anion.
The reason for the increase of the CD release percentage in single system release media (phosphate ion) compared to the binary anions system of chloride–phosphate is ascribed to the decomposition of potassium phosphate, which leads to the formation of HPO42− and H2PO4−, which strongly compete with other anions in the ion exchange process. Phosphate anions are able to exchange intercalated drugs easily due to their high affinity toward the LDH, thus the release rate of the drug is dominated by phosphate ions.107,114 By contrast, when chloride ions are present in the fluid, the rate of the release will be slower, and the amount of drug released will be lower due to the low ion exchange affinity of chloride anion toward the inorganic interlamellar ZnAl-LDH.116,117 This result is in good agreement with previous reports of Berber et al118 and Nakayama et al.119 This suggests that it is the affinity of the sacrificial anion for LDH which governs the ion exchange process.64
CD-Zn/Al-LDH/PAM
The release studies of CD in the presence of PAM (Figure 9B–9D) show similar release behaviors to those in the presence of chloride anions. It is found that the amount of drug liberated is decreased in the presence of PAM.
The immediate release of CD is 48%, 43% and 48% at pH 4.0, 6.8 and 7.4, respectively. At pH 4.0, CD release from CD-LDH reaches a maximum of about 65% within 360 min. At pH 6.8, the release percentage of CD from the nanohybrid reaches 54% after 360 min. About 66% amount of CD is released from the LDH at pH 7.4 after 360 min.
It is known that PAM is not ionized (it is an uncharged polymer), but when it is added to the buffer solutions, it will hydrolyze partially, ie, a partially hydrolyzed polyacrylamide (HPAM) will be formed. Hydrolysis of PAM in the presence of H+ as catalyst at pH 4.0 yields a copolymer composed of acrylamide and acrylic acid (Figure 10A), while its hydrolysis in the presence of OH− as catalyst at pH 7.4 yields a copolymer composed of acrylamide and sodium acrylate (Figure 10B).120,121 In the case of an intestinal medium, the existence of hydroxide ions in this medium would provide considerable assistance to the hydrolysis.122 Since the acrylic acid or sodium acrylate units contain carboxyl groups, they shift such co-polymers to moderately anionic copolymers.
  | Figure 10 General structure for partially hydrolyzed polyacrylamide resulting from the presence of H+ (A) and OH− (B) catalysts in the release media.123,125 |
When HPAM COOH or −COONa groups dissociate into −COO−, the polymer exchanges the CD anions existing on the LDH external sheets and then the carboxylate groups (−COO−) of the polymer can interact with the positive surface charge, which leads to inhibition of release of the drug from the nanohybrid. On the other hand, the formation of the negative layer surrounding the LDH particle inhibits the diffusion of CD anions that have the same charge of the diffusion layer.122,124
Release kinetics of drug from CD-LDH nanohybrid
The release can be described as the rate of mass transport from a solid face into the buffer medium at standard conditions.126 For drug delivery systems, three steps can be distinguished for drug release from the matrix: the initial step is liquid penetration to the matrix, followed by interaction between the nanohybrid and the buffer solution at the solid–liquid interface, and then diffusion of the drug into the exterior liquid (Figure 11).127
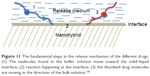  | Figure 11 The fundamental steps in the release mechanism of the different drugs. (1) The molecules found in the buffer solution move toward the solid–liquid interface; (2) reaction happening at the interface; (3) the dissolved drug molecules are moving in the direction of the bulk solution.126 |
In the present work, we used the dissolution-diffusion kinetic models (zero order, first order, Bhaskar, Higuchi, parabolic diffusion and Korsmeyer–Peppas models), in order to describe the overall release process from the drug delivery system, to facilitate quantitative analysis of the data obtained in release study.128
Through the values of linear correlation coefficients (r2), which are shown in Table 3, it is clear that the release mechanisms of the CD from the nanohybrid are the same in all release profiles (II = buffer pH 4.0, III = buffer pH 4.0/NaCl, IV = buffer pH4.0/PAM, V = buffer pH 6.8, VI = buffer pH 6.8/NaCl, VII = buffer pH 6.8/PAM, VIII = buffer pH 7.4, IX = buffer pH 7.4/NaCl and X = buffer pH 7.4/PAM), with the exception of profile I = buffer pH 1.2.
In general, if we look at the values of coefficients related (r2) obtained from release data fitting analyses based on first-order model (Figure 12B and Table 3); Bhaskar model (Figure 12C and Table 3); Higuchi model (Figure 12D and Table 3); parabolic model (Figure 12E and Table 3); and Korsmeyer–Peppas (Figure 12F and Table 3), it can clearly be seen that CD release at pH 1.2 does not depend on (1) the drug concentration in the matrix, (2) the ion-exchange mechanism, (3) the diffusion through the particle, and (4) an external surface/edge diffusion process.33
At pH 4.0, 6.8 and 7.4, since the release of the drug occurs by ion exchange with anions existing in the release medium, the following mechanism may be proposed for release of CD from CD-LDH:
- Reaction in the external parts of the particle: ion exchange reaction happens between the drug anions (large ions) existing in the external parts of the LDH and the anions (small species) of the release media, which leads to the formation of an external phase with smaller interlayer distance (boundary phase) between the external zone in which phosphates have already replaced the drug anions (small spacing) and the internal zone containing intercalated drug (large spacing). As a result, the ion exchange process will become slow.118,129
- Diffusion through the LDH particle: drug anions diffuse from the LDH interlayer region (Figure 13).
- Diffusion of the drug anions from the bulk solution surrounding the LDH particle (Figure 14).15,107
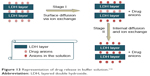  | Figure 13 Representation of drug release in buffer solution.112 |
  | Figure 14 Schematic representation of the diffusion of the drug from the bulk solution surrounding the particle.113 |
The slowest of these processes will determine the rate of exchange of drug anions with the solution medium.107
As an ion-exchange mechanism is involved when using LDH, Bhaskar et al38 created an equation to determine whether the diffusion through the particle is the rate limiting step of the release process. This equation suggests that simply testing for linearity between ln(Mt/M0) and t0.65 is a possible test of the particle diffusion control.38 When the Bhaskar equation was applied to the measured drug release data, the relationship shown above is perfectly linear (Figures 15A, 16A and 17A), which means that the rate limiting step of the release process is the diffusion out of particles.65,124
Since the Bhaskar model provides excellent r2 values equal to or more than 0.95 for all media (from II to X; Table 3), it is possible to elucidate the release processes of CD-LDH nanohybrid on the basis of this model. In other words, through this equation, it is clear that the mechanism of the ion exchange is considered fundamental to determine the overall release rate38,130 and the diffusion of the drug from LDH is subject to strict control by the matrix.8
Another kinetic analysis using an equation suitable for diffusion from the matrix is the Higuchi model. This model fits the release data very well (Figures 15B, 16B and 17B), confirming the significance of the process of the diffusion in controlling the rate of the release of the drug.124
The Korsmeyer–Peppas kinetics mathematical model equation was applied to confirm the diffusion mechanism.131 The correlation coefficients (r2) obtained from the fittings were almost acceptable (Figures 15C, 16C and 17C). Overall, the calculated values of diffusional exponent n are lower than 0.45 (Table 3), which means that the drug release rate is controlled by diffusion from the interlayer of the particle via ion exchange; this process is called quasi-Fickian diffusion mechanism.43,44
By comparing correlation coefficients values obtained by the Bhaskar kinetics model with those obtained by parabolic diffusion equation (Figures 15D, 16D and 17D), it is evident that diffusion of the CD anions from the surface of LDH particles into the medium solution via anion exchange, which takes a short time, cannot be a rate determining step of the release process.63,106
The experimental data show a poor fit with the first order kinetics compared to the Bhaskar kinetic model (Figures 15E, 16E and 17E). This means that the release process cannot be based on the amount of the drug in LDH and drug diffusion from LDH is not passive.8,106
The zero-order model fits the experimental data worse than the Bhaskar model (Figures 15F, 16F and 17F), indicating that the release of the drug is not only considered a single dissolution process.132
Conclusion
CD was successfully intercalated into a Zn-Al nitrate LDH by a co-precipitation technique. The intercalation process was confirmed by FTIR for the nanohybrid, which indicated the presence of functional groups for both the drug and the Zn-Al LDH, confirming the success of the intercalation process. Additionally, TG-DTG analysis proved that thermal stability of CD after intercalation was enhanced compared with its free counterpart, due to interactions between host and guest. Characterization by using UV–vis spectroscopy proved that the interlayer antibiotic anions were stabilized by host–guest and guest–guest interactions. FE-SEM analysis indicated that the CD-LDH nanohybrid has a microblock-like structure. EDS results revealed the nanohybrid contained both organic and inorganic constituents. Release tests of the CD from its interlamellae of the organic–inorganic nanohybrid in a strong acid fluid (pH 1.2) indicated that the LDH was dissolved and intercalated antibiotic released from the sample in a molecular form, while at pH 4.0, 6.8 and 7.4 in the presence or absence of Cl ions or PAM there was immediate release of CD anions that occurred at the beginning of release tests, followed by a slower and sustained release process. So, the ZnAl-CD nanohybrid can be considered as a sustained drug release system allowing increase of the therapeutic efficacy of the antibiotic by maintaining a dose over a long period of time, thus, this system reduces side effects. In general, the release rate of the anion was found to be dependent on the pH of the release medium. The comparison of the release profiles revealed that the percentage of drug released in the absence of the NaCl or PAM in the release media was more than that observed with their presence. The release mechanism had been interpreted on the basis of solid weathering at pH 1.2, while anion-exchange (coupled with diffusion) determined drug release at pH 4.0, 6.8 and 7.4. The release of CD into the gastric simulated fluid can be better expressed by the zero-order model, which means that the release rate of the CD depended on its dissolution. In lysosomal, intestinal, and blood simulated fluid, the Bhaskar model is the best to describe the release behavior kinetic of CD from the CD-LDH nanohybrid. This means that intraparticle diffusion could play a major role in the control of the drug release rate.
Acknowledgment
This project was funded by the Center of Excellence for Advanced Materials Research (CEAMR), King Abdulaziz University, Jeddah, under grant no. (CEAMR-SG-1-438).
Disclosure
The authors report no conflicts of interest in this work.
References
Tammaro L, Costantino U, Nocchetti M, Vittoria V. Incorporation of active nano-hybrids into poly (ε-caprolactone) for local controlled release: antifibrinolytic drug. Appl Clay Sci. 2009;43:350–356. | ||
Parveen S, Misra R, Sahoo SK. Nanoparticles: a boon to drug delivery, therapeutics, diagnostics and imaging. Nanomedicine. 2012;8:147–166. | ||
Mishra B, Patel BB, Tiwari S. Colloidal nanocarriers: a review on formulation technology, types and applications toward targeted drug delivery. Nanomedicine. 2010;6:9–24. | ||
Trikeriotis M, Ghanotakis DF. Intercalation of hydrophilic and hydrophobic antibiotics in layered double hydroxides. Int J Pharm. 2007;332:176–184. | ||
Dash S, Murthy PN, Nath L, Chowdhury P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm. 2010;67:217–223. | ||
Ukmar T, Maver U, Planinšek O, Kaučič V, Gaberšček M, Godec A. Understanding controlled drug release from mesoporous silicates: theory and experiment. J Control Release. 2011;155:409–417. | ||
Park DH, Hwang SJ, Oh JM, Yang JH, Choy JH. Polymer–inorganic supramolecular nanohybrids for red, white, green, and blue applications. Prog Polym Sci. 2013;38:1442–1486. | ||
Perioli L, Posati T, Nocchetti M, Bellezza F, Costantino U, Cipiciani A. Intercalation and release of antiinflammatory drug diclofenac into nanosized ZnAl hydrotalcite-like compound. Appl Clay Sci. 2011;53:374–378. | ||
Sepehrikia S, Arefi D. Determination of Pb and Ni in natural water samples after solid phase extraction with layered double hydroxide (LDH) nano-particles. Am J Nanosci Nanotech. 2013;1:83–86. | ||
Chiang MF, Chen EC, Wu TM. Preparation, mechanical properties and thermal stability of poly (l-lactide)/γ-polyglutamate-modified layered double hydroxide nanocomposites. Polym Degrad Stab. 2012;97:995–1001. | ||
Oh JM, Park DH, Choi SJ, Choy JH. LDH nanocontainers as bio-reservoirs and drug delivery carriers. Recent Pat Nanotechnol. 2012;6:200–217. | ||
Pan D, Zhang H, Zhang T, Duan X. A novel organic–inorganic microhybrids containing anticancer agent doxifluridine and layered double hydroxides: structure and controlled release properties. Chem Eng Sci. 2010;65:3762–3771. | ||
Ribeiro LN, Alcantara AC, Darder M, Aranda P, Araujo-Moreira FM, Ruiz-Hitzky E. Pectin-coated chitosan–LDH bionanocomposite beads as potential systems for colon-targeted drug delivery. Int J Pharm. 2014;463:1–9. | ||
Rives V, Del Arco M, Martín C. Intercalation of drugs in layered double hydroxides and their controlled release: a review. Appl Clay Sci. 2014;88:239–269. | ||
Wei M, Pu M, Guo J, et al. Intercalation of l-Dopa into layered double hydroxides: enhancement of both chemical and stereochemical stabilities of a drug through host-guest interactions. Chem Mater. 2008;20:5169–5180. | ||
Zümreoglu-Karan B, Ay AN. Layered double hydroxides – multifunctional nanomaterials. Chem Pap. 2012;66:1–10. | ||
Oh JM, Biswick TT, Choy JH. Layered nanomaterials for green materials. J Mater Chem. 2009;19:2553–2563. | ||
Kim MH, Park DH, Yang JH, Choy YB, Choy JH. Drug-inorganic-polymer nanohybrid for transdermal delivery. Int J Pharm. 2013;444:120–127. | ||
Shafiei S, Birgani ZT, Darvish A, Azimi MS, Solati-Hashjin M. Layered double hydroxides for diagnostic applications. In: International Congress of Evaluation of Medical Diagnosis Modern Technologies; 2008:1–16; Available from: http://www.academia.edu/19536705/Layered_Double_Hydroxides_for_Diagnostic_Applications. Accessed April 10, 2018. | ||
Basu D, Das A, Stöckelhuber KW, Wagenknecht U, Heinrich G. Advances in layered double hydroxide (LDH)-based elastomer composites. Prog Polym Sci. 2014;39:594–626. | ||
Chakraborti M, Jackson JK, Plackett D, Brunette DM, Burt HM. Drug intercalation in layered double hydroxide clay: application in the development of a nanocomposite film for guided tissue regeneration. Int J Pharm. 2011;416:305–313. | ||
Chakraborty M, Dasgupta S, Soundrapandian C, et al. Methotrexate intercalated ZnAl-layered double hydroxide. J Solid State Chem. 2011;184:2439–2445. | ||
Blazheyevski MY, Labuzova YY. Kinetic spectrophotometric determination of cefadroxil in pure substance and pharmaceutical dosage form. J Chem Pharm Res. 2013;5:115–121. | ||
Frunza M, Lisa G, Popa MI, Miron ND, Nistor DI. Thermogravimetric analysis of layered double hydroxides with chloramphenicol and salicylate in the interlayer space. J Thermal Anal Calor. 2008;93:373–379. | ||
Ryu SJ, Jung H, Oh JM, Lee JK, Choy JH. Layered double hydroxide as novel antibacterial drug delivery system. J Phys Chem Solid. 2010;71:685–688. | ||
San Román MS, Holgado MJ, Salinas B, Rives V. Drug release from layered double hydroxides and from their polylactic acid (PLA) nanocomposites. Appl Clay Sci. 2013;71:1–7. | ||
Yasin Y, Ismail NM. Controlled release of lawsone-intercalated Zn-Al-layered double hydroxide. J Med Sci. 2013;13:453–458. | ||
Barahuie F, Hussein MZ, Arulselvan P, Fakurazi S, Zainal Z. Drug delivery system for an anticancer agent, chlorogenate-Zn/Al-layered double hydroxide nanohybrid synthesised using direct co-precipitation and ion exchange methods. J Solid State Chem. 2014;217:31–41. | ||
Chaudhari SV, Vavia PR. Gastroretentive sustained release floating and swellable cefadroxil formulation. J Bioequiv Bioavail. 2016;8:294–298. | ||
Jatav RK, Bodana M. Formulation and evaluation of floating tablet of cefadroxil. Curr Res Pharm Sci. 2015;5:95–105. | ||
Cavani F, Trifiro F, Vaccari A. Hydrotalcite-type anionic clays: preparation, properties and applications. Catal Today. 1991;11:173–301. | ||
Gu Z, Thomas AC, Xu ZP, Campbell JH, Lu GQ. In vitro sustained release of LMWH from MgAl-layered double hydroxide nanohybrids. Chem Mater. 2008;20:3715–3722. | ||
Rojas R, Palena MC, Jimenez-Kairuz AF, Manzo RH, Giacomelli CE. Modeling drug release from a layered double hydroxide–ibuprofen complex. Appl Clay Sci. 2012;62:15–20. | ||
Xu G, Sunada H. Influence of formulation change on drug release kinetics from hydroxypropylmethylcellulose matrix tablets. Chem Pharm Bull (Tokyo). 1995;43:483–487. | ||
Costa P, Sousa Lobo JM. Modeling and comparison of dissolution profiles. Eur J Pharm Sci. 2001;13:123–133. | ||
Kalam MA, Humayun M, Parvez N, et al. Release kinetics of modified pharmaceutical dosage forms: a review. Cont J Pharm Sci. 2007;1:30–35. | ||
Shoaib MH, Tazeen J, Merchant HA, Yousuf RI. Evaluation of drug release kinetics from ibuprofen matrix tablets using HPMC. Pak J Pharm Sci. 2006;19:119–124. | ||
Bhaskar R, Murthy RSR, Miglani BD, Viswanathan K. Novel method to evaluate diffusion controlled release of drug from resinate. Int J Pharm. 1986;28:59–66. | ||
Higuchi T. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52:1145–1149. | ||
Kosmidis K, Rinaki E, Argyrakis P, Macheras P. Analysis of Case II drug transport with radial and axial release from cylinders. Int J Pharm. 2003;254:183–188. | ||
Serra L, Doménech J, Peppas NA. Drug transport mechanisms and release kinetics from molecularly designed poly (acrylic acid-g-ethylene glycol) hydrogels. Biomaterials. 2006;27:5440–5451. | ||
Korsmeyer RW, Peppas NA. Solute and penetrant diffusion in swellable polymers. III. Drug release from glassy poly (HEMA-co-NVP) copolymers. J Control Release. 1984;1:89–98. | ||
Sahoo S, Chakraborti CK, Behera PK. Development and evaluation of gastroretentive controlled release polymeric suspensions containing ciprofloxacin and carbopol polymers. J Chem Pharm Res. 2012;4:2268–2284. | ||
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15:25–35. | ||
Peppas NA. Analysis of Fickian and non-Fickian drug release from polymers. Pharm Acta Helv. 1985;60:110–111. | ||
Rani GU, Konreddy AK, Mishra S, Sen G. Synthesis and applications of polyacrylamide grafted agar as a matrix for controlled drug release of 5-ASA. Int J Biol Macromol. 2014;65:375–382. | ||
Yadav G, Bansal M, Thakur N, Khare S, Khare P. Multilayer tablets and their drug release kinetic models for oral controlled drug delivery system. ME J Sci Res. 2013;16:782–795. | ||
Siepmann J, Peppas NA. Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv Drug Deliv Rev. 2001;48:139–157. | ||
Wang Z, Liu F, Lu C. Mg–Al–carbonate layered double hydroxides as a novel catalyst of luminol chemiluminescence. Chem Commun. 2011;47:5479–5481. | ||
Nakamoto K. Infrared and Raman Spectra of Inorganic and Coordination Compounds. New York: John Wiley & Sons; 1997. | ||
Bin Hussein MZ, Long CW. Synthesis of organo-mineral nanohybrid material: indole-2-carboxylate in the lamella of Zn–Al-layered double hydroxide. Mater Chem Phys. 2004;85:427–431. | ||
Kura AU, Al Ali SHH, Hussein MZ, Fakurazi S, Arulselvan P. Development of a controlled-release anti-parkinsonian nanodelivery system using levodopa as the active agent. Int J Nanomedicine. 2013;8:1103. | ||
Lambert JB, Shurvell HF, Lightner DA, Cooks RG. Introduction to Organic Spectroscopy. New York: Macmillan Publishing Company; 1987. | ||
Chakraborty M, Dasgupta S, Bose P, et al. Layered double hydroxide: inorganic organic conjugate nanocarrier for methotrexate. J Phys Chem Solid. 2011;72:779–783. | ||
Wei M, Guo J, Shi Z, et al. Preparation and characterization of l-cystine and l-cysteine intercalated layered double hydroxides. J Mater Sci. 2007;42:2684–2689. | ||
Xu ZP, Gu Z, Cheng X, Rasoul F, Whittaker AK, Lu GQM. Controlled release of ketorolac through nanocomposite films of hydrogel and LDH nanoparticles. J Nanopart Res. 2011;13:1253–1264. | ||
DeLeon VH, Nguyen TD, Nar M, D’Souza NA, Golden TD. Polymer nanocomposites for improved drug delivery efficiency. Mater Chem Phys. 2012;132:409–415. | ||
Ghotbi MY, Bin Hussein MZ. Gallate–Zn–Al-layered double hydroxide as an intercalated compound with new controlled release formulation of anticarcinogenic agent. J Phys Chem Solid. 2010;71:1565–1570. | ||
Rives V. Layered Double Hydroxides: Present and Future. New York: Nova Publishers; 2001. | ||
Kloprogge JT, Frost RL. Fourier transform infrared and raman spectroscopic study of the local structure of Mg-, Ni-, and Co-hydrotalcites. J Solid State Chem. 1999;146:506–515. | ||
Rahim N, Naqvi SB, Bibi R, Iffat W, Shakeel S, Muhammad IN. Disintegrants combination: development and optimization of a cefadroxil fast disintegrating tablet. Pak J Pharm Sci. 2014;27:1467–1475. | ||
San Román MS, Holgado MJ, Salinas B, Rives V. Characterisation of diclofenac, ketoprofen or chloramphenicol succinate encapsulated in layered double hydroxides with the hydrotalcite-type structure. Appl Clay Sci. 2012;55:158–163. | ||
Yang JH, Han YS, Park M, Park T, Hwang SJ, Choy JH. New inorganic-based drug delivery system of indole-3-acetic acid-layered metal hydroxide nanohybrids with controlled release rate. Chem Mater. 2007;19:2679–2685. | ||
Bashi AM, Haddawi SM, Al-Yasari AH. Kinetics study and controlled release of synthesized phenoxy herbicides-based nanocomposites from 2,4-dichloro- and 2,4,5-trichlorophenoxy acetates with Zn/Al layered double hydroxide interlamellae. Irq Nat J Chem. 2010;38:279–292. | ||
Zhang H, Zou K, Guo S, Duan X. Nanostructural drug-inorganic clay composites: structure, thermal property and in vitro release of captopril-intercalated Mg–Al-layered double hydroxides. J Solid State Chem. 2006;179:1792–1801. | ||
Santosa SJ, Kunarti ES. Synthesis and utilization of Mg/Al hydrotalcite for removing dissolved humic acid. Appl Surf Sci. 2008;254:7612–7617. | ||
Kameda T, Fubasami Y, Uchiyama N, Yoshioka T. Elimination behavior of nitrogen oxides from a NO3−-intercalated Mg–Al layered double hydroxide during thermal decomposition. Thermochim Acta. 2010;499:106–110. | ||
Morzyk-Ociepa B, Michalska D, Pietraszko A. Structures and vibrational spectra of indole carboxylic acids. Part I. Indole-2-carboxylic acid. J Mol Struct. 2004;688:79–86. | ||
Park MC, Kim H, Park DH, Yang JH, Choy JH. Ketoprofen-LDH nanohybrid for transdermal drug delivery system. Bull Kor Chem Soc. 2012;33:1827–1828. | ||
Al Ali SHH, Al-Qubaisi M, Hussein MZ, Ismail M, Zainal Z, Hakim MN. Controlled release and angiotensin-converting enzyme inhibition properties of an antihypertensive drug based on a perindopril erbumine-layered double hydroxide nanocomposite. Int J Nanomedicine. 2012;7:2129–2141. | ||
Meher RR. Double Hydroxide Nano-carrier for Controlled Delivery of Drug Molecules [PhD thesis]. Rourkela: National Institute of Technology; 2012. | ||
Nejati K, Rezvani Z. Synthesis and characterisation of nanohybrids of olsalazine-intercalated Mg-Al layered double hydroxide. J Exp Nanosci. 2012;7:412–425. | ||
Arizaga GGC, Mangrich AS, da Costa Gardolinski JEF, Wypych F. Chemical modification of zinc hydroxide nitrate and Zn–Al-layered double hydroxide with dicarboxylic acids. J Colloid Interface Sci. 2008;320:168–176. | ||
Zou K, Zhang H, Duan X. Studies on the formation of 5-aminosalicylate intercalated Zn–Al layered double hydroxides as a function of Zn/Al molar ratios and synthesis routes. Chem Eng Sci. 2007;62:2022–2031. | ||
Del Arco M, Fernández A, Martín C, Rives V. Intercalation of mefenamic and meclofenamic acid anions in hydrotalcite-like matrixes. Appl Clay Sci. 2007;36:133–140. | ||
Fulias A, Vlase T, Vlase G, et al. Thermoanalytical study of cefadroxil and its mixtures with different excipients. Rev Chim. 2010;4:11. | ||
Ay AN, Zümreoglu-Karan B, Temel A, Rives V. Bioinorganic magnetic core-shell nanocomposites carrying antiarthritic agents: intercalation of ibuprofen and glucuronic acid into Mg-Al-layered double hydroxides supported on magnesium ferrite. Inorg Chem. 2009;48:8871–8877. | ||
Choy JH, Jung JS, Oh JM, et al. Layered double hydroxide as an efficient drug reservoir for folate derivatives. Biomaterials. 2004;25:3059–3064. | ||
Lonkar SP, Kutlu B, Leuteritz A, Heinrich G. Nanohybrids of phenolic antioxidant intercalated into MgAl-layered double hydroxide clay. Appl Clay Sci. 2013;71:8–14. | ||
Wang Y, Wu P, Li Y, Zhu N, Dang Z. Structural and spectroscopic study of tripeptide/layered double hydroxide hybrids. J Colloid Interface Sci. 2013;394:564–572. | ||
Hibino T, Tsunashima A. Formation of spinel from a hydrotalcite-like compound at low temperature: reaction between edges of crystallites. Clay Clay Mineral. 1997;45:842–853. | ||
Kanezaki E. Thermal behavior of the hydrotalcite-like layered structure of Mg and Al-layered double hydroxides with interlayer carbonate by means of in situ powder HTXRD and DTA/TG. Solid State Ionics. 1998;106:279–284. | ||
Rubino FM. Separation methods for methotrexate, its structural analogues and metabolites. J Chromatogr B Biomed Sci Appl. 2001;764:217–254. | ||
Donald L, Lampman GM, Kriz GS. Introduction to Spectroscopy: A Guide for Students of Organic Chemistry. Fort Worth: Harcourt Brace College Publishers; 1996. | ||
Barreto WJ, Ponzoni S, Sassi P. A raman and UV–vis study of catecholamines oxidized with Mn (III). Spectrochim Acta A Mol Biomol Spectrosc. 1998;55:65–72. | ||
Meng J, Zhang H, Evans DG, Duan X. Study on the supramolecular structure of sorbic acid intercalated Zn-Al layered double hydroxides and its thermal decomposition. Chinese Science Bulletin. 2005;50:2575–2581. | ||
He Q, Yin S, Sato T. Synthesis and photochemical properties of zinc–aluminum layered double hydroxide/organic UV ray absorbing molecule/silica nanocomposites. J Phys Chem Solid. 2004;65:395–402. | ||
Aisawa S, Takahashi S, Ogasawara W, Umetsu Y, Narita E. Direct intercalation of amino acids into layered double hydroxides by coprecipitation. J Solid State Chem. 2001;162:52–62. | ||
Franklin KR, Lee E, Nunn CC. Preparation and characterisation of layered double hydroxides containing monovalent and divalent ions derived from 5-benzoyl-4-hydroxy-2-methoxybenzenesulfonic acid. J Mater Chem. 1995;5:565–569. | ||
Minagawa K, Berber MR, Hafez IH, Mori T, Tanaka M. Target delivery and controlled release of the chemopreventive drug sulindac by using an advanced layered double hydroxide nanomatrix formulation system. J Mater Sci Mater Med. 2012;23:973–981. | ||
Hussein MZ, Jubri ZB, Zainal Z, Yahya AH. Pamoate intercalated Zn-Al layered double hydroxide for the formation of layered organic-inorganic intercalate. Mater Sci Poland. 2004;22:57–67. | ||
Bin Hussein MZ, Zainal Z, Yahaya AH, Foo DWV. Controlled release of a plant growth regulator, α-naphthaleneacetate from the lamella of Zn–Al-layered double hydroxide nanocomposite. J Control Release. 2002;82:417–427. | ||
Belskaya OB, Gulyaeva TI, Leont’eva NN, et al. Formation of platinum sites on layered double hydroxide type basic supports: II. Effect of the nature of the interlayer anion of the layered aluminum-magnesium hydroxides on platinum binding and Pt/MgAlOx formation. Kinetics Catal. 2011;52:876–885. | ||
Carja G, Lehutu G, Dartu L, Mertens M, Cool P. Layered double hydroxides reconstructed in calcium glutamate aqueous solution as a complex delivery system. Appl Clay Sci. 2012;65:37–42. | ||
Tammaro L, Costantino U, Bolognese A, et al. Nanohybrids for controlled antibiotic release in topical applications. Int J Antimicrob Agents. 2007;29:417–423. | ||
Tronto J, dos Reis MJ, Silvério F, Balbo VR, Marchetti JM, Valim JB. In vitro release of citrate anions intercalated in magnesium aluminium layered double hydroxides. J Phys Chem Solid. 2004;65:475–480. | ||
Ambrogi V, Fardella G, Grandolini G, Perioli L. Intercalation compounds of hydrotalcite-like anionic clays with antiinflammatory agents – I. Intercalation and in vitro release of ibuprofen. Int J Pharm. 2001;220:23–32. | ||
Parello ML, Rojas R, Giacomelli CE. Dissolution kinetics and mechanism of Mg–Al layered double hydroxides: a simple approach to describe drug release in acid media. J Colloid Interface Sci. 2010;351:134–139. | ||
Ha JU, Xanthos M. Drug release characteristics from nanoclay hybrids and their dispersions in organic polymers. Int J Pharm. 2011;414:321–331. | ||
Ambrogi V, Perioli L, Ciarnelli V, Nocchetti M, Rossi C. Effect of gliclazide immobilization into layered double hydroxide on drug release. Eur J Pharm Biopharm. 2009;73:285–291. | ||
Berber MR, Minagawa K, Katoh M, Mori T, Tanaka M. Nanocomposites of 2-arylpropionic acid drugs based on Mg–Al layered double hydroxide for dissolution enhancement. Eur J Pharm Sci. 2008;35:354–360. | ||
Ambrogi V, Fardella G, Grandolini G, Nocchetti M, Perioli L. Effect of hydrotalcite-like compounds on the aqueous solubility of some poorly water-soluble drugs. J Pharm Sci. 2003;92:1407–1418. | ||
Del Arco M, Fernandez A, Martin C, Sayalero ML, Rives V. Solubility and release of fenamates intercalated in layered double hydroxides. Clay Minerals. 2008;43:255–265. | ||
Wang Z, Wang E, Gao L, Xu L. Synthesis and properties of Mg2Al layered double hydroxides containing 5-fluorouracil. J Solid State Chem. 2005;178:736–741. | ||
Huang X, Brazel CS. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J Control Release. 2001;73:121–136. | ||
Zhang XQ, Zeng MG, Li SP, Li XD. Methotrexate intercalated layered double hydroxides with different particle sizes: structural study and controlled release properties. Colloids Surf B Biointerfaces. 2014;117:98–106. | ||
Ambrogi V, Fardella G, Grandolini G, Perioli L, Tiralti MC. Intercalation compounds of hydrotalcite-like anionic clays with anti-inflammatory agents, II: uptake of diclofenac for a controlled release formulation. AAPS PharmSciTech. 2002;3:77–82. | ||
Perioli L, Ambrogi V, Di Nauta L, Nocchetti M, Rossi C. Effects of hydrotalcite-like nanostructured compounds on biopharmaceutical properties and release of BCS class II drugs: the case of flurbiprofen. Appl Clay Sci. 2011;51:407–413. | ||
Fogg AM, Dunn JS, Shyu SG, Cary DR, O’Hare D. Selective ion-exchange intercalation of isomeric dicarboxylate anions into the layered double hydroxide [LiAl2 (OH)6] Cl.H2O. Chem Mater. 1998;10:351–355. | ||
Fogg AM, Dunn JS, O’Hare D. Formation of second-stage intermediates in anion-exchange intercalation reactions of the layered double hydroxide [LiAl2(OH)6] Cl. H2O as observed by time-resolved, in situ x-ray diffraction. Chem Mater. 1998;10:356–360. | ||
Xia S, Ni Z, Xu Q, Hu B, Hu J. Layered double hydroxides as supports for intercalation and sustained release of antihypertensive drugs. J Solid State Chem. 2008;181:2610–2619. | ||
Kong X, Jin L, Wei M, Duan X. Antioxidant drugs intercalated into layered double hydroxide: structure and in vitro release. Appl Clay Sci. 2010;49:324–329. | ||
Gunawan P, Xu R. Direct control of drug release behavior from layered double hydroxides through particle interactions. J Pharm Sci. 2008;97:4367–4378. | ||
Hussein MZ, Jaafar AM, Yahaya AH, Zainal Z. The effect of single, binary and ternary anions of chloride, carbonate and phosphate on the release of 2, 4-dichlorophenoxyacetate intercalated into the Zn–Al-layered double hydroxide nanohybrid. Nanoscale Res Lett. 2009;4:1351–1357. | ||
Bin Hussein MZ, Yahaya AH, Zainal Z, Kian LH. Nanocomposite-based controlled release formulation of an herbicide, 2, 4-dichlorophenoxyacetate incapsulated in zinc–aluminium-layered double hydroxide. Sci Technol Adv Mater. 2005;6:956–962. | ||
Miyata S. Anion-exchange properties of hydrotalcite-like compounds. Clays Clay Miner. 1983;31:305–311. | ||
Parker LM, Milestone NB, Newman RH. The use of hydrotalcite as an anion absorbent. Ind Eng Chem Res. 1995;34:1196–1202. | ||
Berber MR, Hafez IH, Minagawa K, Mori T, Tanaka M. Nanocomposite formulation system of lipid-regulating drugs based on layered double hydroxide: synthesis, characterization and drug release properties. Pharm Res. 2010;27:2394–2401. | ||
Nakayama H, Takeshita K, Tsuhako M. Preparation of 1-hydroxyethylidene-1, 1-diphosphonic acid-intercalated layered double hydroxide and its physicochemical properties. J Pharm Sci. 2003;92:2419–2426. | ||
Kurenkov VF, Hartan HG, Lobanov FI. Alkaline hydrolysis of polyacrylamide. Russ J Appl Chem. 2001;74:543–554. | ||
Arinaitwe E, Pawlik M. A method for measuring the degree of anionicity of polyacrylamide-based flocculants. Int J Mineral Process. 2009;91:50–54. | ||
Costantino U, Ambrogi V, Nocchetti M, Perioli L. Hydrotalcite-like compounds: versatile layered hosts of molecular anions with biological activity. Microporous Mesoporous Mater. 2008;107:149–160. | ||
Li B, He J, Evans DG, Duan X. Enteric-coated layered double hydroxides as a controlled release drug delivery system. Int J Pharm. 2004;287:89–95. | ||
Ambrogi V, Perioli L, Ricci M, et al. Eudragit® and hydrotalcite-like anionic clay composite system for diclofenac colonic delivery. Microporous Mesoporous Mater. 2008;115:405–415. | ||
Zeynali ME, Rabii A, Baharvand H. Synthesis of partially hydrolyzed polyacrylamide and investigation of solution properties (viscosity behaviour). Iran Polym J. 2004;13:479–484. | ||
Singhvi G, Singh M. Review: in-vitro drug release characterization models. Int J Pharm Stud Res. 2011;2:77–84. | ||
Kovanda F, Maryšková Z, Kovář P. Intercalation of paracetamol into the hydrotalcite-like host. J Solid State Chem. 2011;184:3329–3335. | ||
Lokhandwala H, Deshpande A, Deshpande S. Kinetic modeling and dissolution profiles comparison: an overview. Int J Pharm Biol Sci. 2013;4:728–737. | ||
Atwood JL, Lehn JM, Alberti G, Bein T. Solid-State Supramolecular Chemistry: Two-and Three-Dimensional Inorganic Networks: Volume 7 (Comprehensive Supramolecular Chemistry). Oxford: Pergamon Press; 1996. | ||
Ngawhirunpat T, Goegebakan E, Duangjit S, Akkaramongkolporn P, Kumpugdee-Vollrath M. Controlled release of chlorpheniramine from resonates through surface coating with Eudragit®RS 100. Int J Pharm Pharm Sci. 2010;2:107–112. | ||
Kabir AKL, Halder S, Shuma ML, Rouf ASS. Formulation development and in vitro evaluation of drug release kinetics from sustained release aceclofenac matrix tablets using hydroxypropyl methyl cellulose. Dhaka Univ J Pharm Sci. 2012;11:37–43. | ||
Zhang H, Pan D, Duan X. Synthesis, characterization, and magnetically controlled release behavior of novel core-shell structural magnetic ibuprofen-intercalated LDH nanohybrids. J Phys Chem C. 2009;113:12140–12148. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.