Back to Journals » Clinical Ophthalmology » Volume 8
Confocal microscopy findings in deep anterior lamellar keratoplasty performed after Descemet's stripping automated endothelial keratoplasty
Authors Pang A, Mohamed-Noriega K ![]() , Chan A, Mehta J
, Chan A, Mehta J
Received 22 September 2013
Accepted for publication 18 October 2013
Published 16 January 2014 Volume 2014:8 Pages 243—249
DOI https://doi.org/10.2147/OPTH.S54824
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Audrey Pang,1,2 Karim Mohamed-Noriega,1 Anita S Chan,1,3–5 Jodbhir S Mehta1,3
1Singapore National Eye Centre, 2Department of Ophthalmology, Tan Tock Seng Hospital, 3Singapore Eye Research Institute, 4Department of Histopathology, Pathology, Singapore General Hospital, 5Department of Ophthalmology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore
Background: This study describes the in vivo confocal microscopy findings in two patients who had deep anterior lamellar keratoplasty (DALK) following Descemet's stripping automated endothelial keratoplasty (DSAEK).
Methods: The study reviewed the cases of two patients who first underwent DSAEK followed by DALK when their vision failed to improve due to residual stromal scarring. In the first case, a DSAEK was performed for a patient with pseudophakic bullous keratopathy. After surgery, the patient's vision failed to improve satisfactorily due to residual anterior stromal opacity and irregularity. Subsequently, the patient underwent a DALK. The same two consecutive operations were performed for a second patient with keratoconus whose previous penetrating keratoplasty had failed and had secondary graft ectasia. In vivo confocal microscopy was performed 2 months after the DALK surgery in both cases.
Results: At 3 months after DALK, the best-corrected visual acuity was 6/30 in case 1 and 6/24 in case 2. In vivo confocal microscopy in both cases revealed the presence of quiescent keratocytes in the stroma layers of the DSAEK and DALK grafts, which was similar in the central and peripheral cornea. There was no activated keratocytes or haze noted in the interface between the grafts.
Conclusion: Our short-term results show that performing a DALK after a DSAEK is an effective way of restoring cornea clarity in patients with residual anterior stromal opacity. In vivo confocal microscopy showed that there were no activated keratocytes seen in the interface of the grafts, which suggests that optimal visual acuity may be obtained with minimal interface haze.
Keywords: cornea, transplant, keratocytes, histology
Introduction
Over the past 10 years, selective replacement of diseased layers of the cornea in lamellar keratoplasty has been increasingly performed over penetrating keratoplasty (PK). Descemet’s stripping automated endothelial keratoplasty (DSAEK) provides more rapid visual improvement in patients with corneal endothelial disease, while deep anterior lamellar keratoplasty (DALK) eliminates the risk of endothelial rejection in patients with corneal stromal disease.1
Busin et al first reported a technique of performing a DALK after DSAEK surgery in two patients who had residual anterior stromal opacities.2 He showed that a DALK instead of a PK can be performed to preserve the DSAEK graft in patients who have residual cornea opacities. This maintains the structural integrity of the eye and reduces the risk of intraocular complications. In our study, we performed a DALK after DSAEK with a modified technique which preserved the host’s Descemet’s membrane (DM) in the periphery of the grafts. It is possible to leave the DM, as we can directly visualize it on the stromal bed of the DSAEK intraoperatively. We also describe the confocal microscopic features seen postoperatively to assess the presence of activated keratocytes and possible interface haze.
Methods
Case study 1
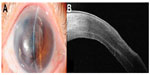
A 73-year-old female with a history of prior cataract surgery in the left eye more than 30 years previously, and a recent secondary anterior chamber intraocular lens implantation 5 years ago, presented to our clinic in 2011 with left blurring of vision due to pseudophakic bullous keratopathy (Figure 1A). At presentation, her best-corrected visual acuity (BCVA) was counting fingers at 50 cm distance. Anterior segment optical coherence tomography (OCT) by Visante (Carl Zeiss, Meditec, Jena, Germany) showed mild diffuse anterior stromal opacity which was inferred to be due to the chronic nature of the bullous edema. An uncomplicated left DSAEK was performed in March 2011 (as described below). However, intraoperatively, it was noted following superficial keratectomy that she had an irregular scar on the anterior stromal surface.
Her BCVA improved to 6/60 (+1.50/−0.50 × 180) at 3 months post-DSAEK. Repeat anterior segment OCT scans using the Optovue system (Optovue Inc, Fremont, CA, USA) revealed anterior stroma scarring and mild irregular corneal thinning overlying the scar (Figure 1B). In addition, posterior segment OCT revealed mild chronic macula edema. Since there was no change in the irregular thinning over a year, and due to the presence of a guarded visual prognosis from the chronic macula edema, after counseling, we elected to perform a DALK 14 months after the DSAEK surgery.
Case study 2
A 38-year-old male with bilateral keratoconus, a history of bilateral PKs, and a right trabeculectomy more than 10 years previously, had been on regular follow-up in our clinic. In September 2012, his BCVA was 6/12 (+2.75/−4.00 × 5) in the right eye and 6/9 (plano/−5.00 × 40) in the left eye. However, he developed left corneal graft failure and was noted to have inferior graft ectasia in January 2013 (Figure 2). His BCVA dropped to counting fingers at 1 meter distance. He also had a left cataract. He underwent a DSAEK and phacoemulsification surgery in January 2013. During the DSAEK procedure, following the air tamponade, his inferior graft ectasia became worse. In view of this, he underwent a left DALK 1 week later.
  | Figure 2 Left penetrating keratoplasty failure due to graft ectasia and recurrent keratoconus in case 2. |
Surgical procedures
The DSAEK was performed under general anesthesia using a previously described technique.3 The DM was stripped under air through a temporal sclera incision in case 1 and no stripping was performed in case 2. An inferior peripheral iridectomy was performed. Four venting incisions were made. The lamellar graft was harvested from the donor cornea using the Moria ALTK (automated lamellar therapeutic keratoplasty) microkeratome (Moria Inc., Doylestown, PA, USA).
The diameter of the lamellar graft was 8.75 mm in case 1 and 9.0 mm in case 2. The lamellar graft was inserted into the anterior chamber using the Endoglide DSAEK insertion device (Network Medical Products, Ripon, UK).3 Once the graft was inserted, an air bubble was injected into the anterior chamber, and a full air-fill was maintained for 10 minutes. Aqueous fluid was expressed through the venting incisions to ensure good graft–host apposition.
The DALK was performed under general anesthesia. The corneal surface was first marked with a Solan trephine (Medtronics, Inc., Jacksonville, FL, USA). The diameter of the trephine was 7.5 mm in case 1 and 8.0 mm in case 2. The size of the graft was chosen to allow at least 0.5 mm rim of the DSAEK around the DALK. A free-hand manual dissection was performed with gentle traction of the recipient anterior stroma until a clear interface could be reached. Once an interface was identified, it was carefully exposed using a marginal lamellar dissector (ASICO, Westmont, IL, USA), and the recipient stroma was cut along the previous trephine mark. Following complete exposure of the interface, we noted that, in fact, the recipient’s DM had been split into anterior and posterior layers. In case 1 the central cornea was devoid of any DM following Descemet’s stripping as part of the DSAEK procedure. The paracentral/peripheral interface had the presence of the recipient’s posterior nonbanded DM. This was visualized intraoperatively as a bright, reflective surface on the interface bed and was confirmed histologically (Figure 3). The anterior banded layer of DM could be visualized on the removed corneal button. In the histological specimen from case 2, the complete posterior nonbanded part of the DM could be seen attached to the DSAEK, and the peripheral interface had remnants of the anterior DM layer, the central anterior layer attached to the recipient’s removed cornea. Intraoperatively, the split between the banded and nonbanded part of the DM is only possible since there is DM present on both the recipient tissue and the DSAEK graft following the removal of the corneal button, hence there must be a split in the DM to achieve this. The donor cornea was trephined with a 7.5 mm and 8.0 mm trephine (Moria Inc.) in case 1 and case 2, respectively. The DM was removed before suturing the graft in place with two 10/0 nylon sutures.
Confocal microscopy
In vivo confocal microscopy was performed at 2 months postoperative using the Heidelberg Retina Tomography 2 Rostock Cornea Module (HRT 2 RCM; Heidelberg Engineering, Germany). After a drop of contact gel (Vidisic Gel; Bausch and Lomb, Rochester, NY, USA) was applied onto the front of the microscope lens, a TomoCap (Heidelberg Engineering) was mounted onto the holder to cover the microscope lens. The HRT 2 RCM uses a 60× immersion objective lens (Olympus Europa GmbH, Hamburg, Germany) and a 670 nm diode laser as the light source. The central cornea was examined in layers using the volumetric scan, advancing from the epithelium to the endothelium. Both the central cornea and peripheral cornea between the two grafts were examined. Four examinations of the eye were performed.
Results
At 3 months postoperative, the case 1 patient’s BCVA improved to 6/30 (−6.00/−1.00 × 110). Slit lamp biomicroscopy revealed clear corneal grafts in the left eye (Figure 4A). Posterior segment OCT revealed the presence of chronic mild cystoid macula edema. Anterior segment OCT showed a regular corneal surface and a smooth interface between the DSAEK and DALK grafts (Figure 4B). The host DM was clearly seen at the periphery, between the two lamellar grafts.
In case 2, the patient’s BCVA improved to 6/24 (+4.50/−7.00 × 60) at 3 months postoperative. His suboptimal visual acuity was due to coexisting advanced glaucoma. Slit lamp biomicroscopy revealed clear cornea grafts in the left eye (Figure 5A). Anterior segment OCT also showed a regular corneal surface and a smooth interface between the DSAEK and DALK grafts (Figure 5B).
In both cases, the histology of the host corneal button showed remnants of the anterior DM of the host cornea (Figure 6). At the periphery, the host DM had split, with the anterior portion attached to the host, leaving the posterior portion adhered to the DSAEK graft.
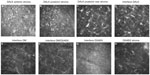
In vivo confocal microscopy of the central cornea in both patients demonstrated normal quiescent keratocytes in the anterior and posterior stromal layers of the DSAEK and DALK, respectively (Figures 7 and 8).
Typically, quiescent keratocytes were seen as regular oval-shaped cells with a bright nucleus and were well separated from each other. No activated keratocytes were seen in the stroma or the interface between the grafts. No inflammatory cells or corneal nerves were seen in the DALK and DSAEK grafts.
The interface between the grafts was identified as an amorphous, acellular zone with variable background extracellular reflectivity. A few hyperreflective birefringent particles, which were likely debris, were observed at the interface. Microfolds within the graft stroma near the interface were seen as dark hyporeflective striae.
At the periphery of the two corneal grafts there was a similar finding of quiescent keratocytes noted in the stroma of the DALK and DSAEK grafts (Figures 9 and 10). There was neither activated keratocytes nor haze noted at the interface. Small hyperreflective particles corresponding to debris were also noted in the interface.
Discussion
It is occasionally difficult to predict the visual significance of stroma opacities in edematous corneas, which affects the choice between lamellar and PK procedures. As previously described by Busin et al, the approach of performing a DALK after DSAEK surgery in patients is an effective treatment in patients who fail to achieve satisfactory visual improvement after DSAEK.2 Previously Busin et al used a Hessburg–Baron trephine to create a partial-thickness trephination aligned with the edge of the DSAEK graft. The incision was deepened manually until the stromal surface of the DSAEK graft was reached. No adhesion was noted between the host and the DSAEK graft, and the donor tissue was punched to size and sutured in place.
In our study, we sized our partial-thickness trephine to 1 mm smaller than the DSAEK graft. A free-hand manual dissection was performed until a clear interface was identified and carefully exposed using a marginal lamellar dissector. After complete exposure of the interface, we noted that the recipient’s DM in the periphery had been split as described in the surgical procedures above. The central cornea was devoid of any DM as expected following Descemet’s stripping during the DSAEK procedure. The peripheral interface had the presence of the recipient’s remnant posterior DM, which was visualized intraoperatively as a bright, reflective surface on the interface bed. Our cases suggest that there is stronger adhesion between the DSAEK graft stromal surface and the posterior nonbanded DM than between the individual DM layers.
Normal corneal stroma consists of a dense network of collagen fibers, keratocytes, and proteoglycans. Keratocytes in the stroma are usually mitotically quiescent and have numerous intercellular contacts. During corneal wound healing, keratocyte apoptosis occurs in the region of injury, and repopulation often occurs by cells that become activated to undergo mitosis and migrate to repopulate the apoptotic area.4 Activated keratocytes involved in cellular repair have been previously identified in corneal transplants by their large, brightly reflective appearance associated with visible cytoplasmic processes.5 Confocal microscopy has shown the presence of active keratocytes in recipient stroma after DSEK.6 These prior studies show that activated keratocytes play a role in the repair of the extracellular matrix; however, the altered composition of the extracellular matrix and haphazard deposition of keratocytes scatter light and is believed to contribute to postoperative haze formation.7
Ultrastructural analysis of the grafts in our cases was performed using in vivo confocal microscopy. Our study showed that there were no activated keratocytes noted in the stroma of both the DALK and DSAEK grafts, especially the interfaces between the grafts. This microscopic finding was similar both in the center and periphery of the grafts. Similar findings of quiescent keratocytes have been described after individual manual deep lamellar keratoplasty,5 DSAEK,8 DALK,9 and laser-assisted in situ keratomileusis10 surgeries. In the periphery of the grafts, we did not expect the presence of postoperative corneal haze, as this is analogous to the interface in a DM baring DALK. Confocal microscopy in our study has also shown that there were no activated keratocytes in the central area as well as the periphery despite having two interfaces in the periphery of case 1 and in both the center and periphery of case 2. The two interfaces are the donor DALK stroma/host DM interface, and the host DM/DSAEK stromal interface. We recognize that the indications for serial DSAEK and DALK are relatively rare, and this study describes only two cases. More cases will need to be described to show the validity of this procedure. Nevertheless, the absence of activated keratocytes in these two cases provides encouraging evidence, although limited, that the risk of developing corneal haze in patients who have undergone a DALK after DSAEK surgery is low.
Conclusion
Our results suggest that a DALK performed after DSAEK surgery can aid in the visual rehabilitation of patients with residual stromal scarring. There was minimal evidence of interface haze or scarring.
Disclosure
The authors report no conflicts of interest in this work.
References
Tan DT, Dart JK, Holland EJ, Kinoshita S. Cornea transplantation. Lancet. 2012;379(9827):1749–1761. | |
Busin M, Beltz J. Deep anterior lamellar keratoplasty after descemet stripping automated endothelial keratoplasty. Cornea. 2011;30(9):1048–1050. | |
Khor WB, Mehta JS, Tan DT. Descemet stripping automated endothelial keratoplasty with a graft insertion device: surgical technique and early clinical results. Am J Ophthalmol. 2011;151(2):223–232. e2. | |
Mohan RR, Hutcheon AE, Choi R, et al. Apoptosis, necrosis, proliferation, and myofibroblast generation in the stroma following LASIK and PRK. Exp Eye Res. 2003;76(1):71–87. | |
Bourne WM. Cellular changes in transplanted human corneas. Cornea. 2001;20(6):560–569. | |
Savastano A, Sbordone S, Piccirillo V, et al. Confocal microscopy after descemet stripping endothelial keratoplasty: a case report. Cornea. 2009;28(5):570–574. | |
Zieske JD. Extracellular matrix and wound healing. Curr Opin Ophthalmol. 2001;12(4):237–241. | |
Kobayashi A, Mawatari Y, Yokogawa H, Sugiyama K. In vivo laser confocal microscopy after descemet stripping with automated endothelial keratoplasty. Am J Ophthalmol. 2008;145(6):977–985. | |
Feizi S, Javadi MA, Kanavi MR. Cellular changes of donor corneal tissue after deep anterior lamellar keratoplasty versus penetrating keratoplasty in eyes with keratoconus: a confocal study. Cornea. 2010;29(8):866–870. | |
Perez-Gomez I, Efron N. Confocal microscopic evaluation of particles at the corneal flap interface after myopic laser in situ keratomileusis. J Cataract Refract Surg. 2003;29(7):1373–1377. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.