Back to Journals » Cancer Management and Research » Volume 10
Comparison of totally laparoscopic and open approach in total gastrectomy with D2 lymphadenectomy – systematic review and meta-analysis
Authors Małczak P, Torbicz G, Rubinkiewicz M , Gajewska N, Sajuk N , Rozmus K, Wysocki M , Major P , Budzyński A, Pędziwiatr M
Received 3 August 2018
Accepted for publication 6 November 2018
Published 6 December 2018 Volume 2018:10 Pages 6705—6714
DOI https://doi.org/10.2147/CMAR.S182557
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Piotr Małczak,1,2 Grzegorz Torbicz,1 Mateusz Rubinkiewicz,1 Natalia Gajewska,1 Nadia Sajuk,1 Kamil Rozmus,1 Michał Wysocki,1,2 Piotr Major,1,2 Andrzej Budzyński,1,2 Michał Pędziwiatr1,2
1Department of General Surgery, Jagiellonian University Medical College, Kraków, Poland; 2Centre for Research, Training and Innovation in Surgery (CERTAIN Surgery), Kraków, Poland
Introduction: Gastric cancer is a worldwide health concern, being one of the five most common malignant neoplasms worldwide. Currently, an open approach is the gold standard for surgical treatment. Incorporation of laparoscopy as a method of choice for gastric resections remains controversial because of limited evidence of eligibility. To date, there are no high-quality randomized quality trials on totally laparoscopic D2 total gastrectomies.
Aim: The aim of this study was to assess currently available literature and provide meta-analysis on acquired data regarding short-term outcomes with a subgroup analysis of western and eastern studies.
Materials and methods: We performed a systematic review and meta-analysis according to the PRISMA guidelines. The primary outcomes of interest were morbidity and short-term complications.
Results: An initial reference search yielded 3,073 articles. Finally, we chose eight studies covering 1,582 patients that we included in the quantitative analysis. We did not find statistical differences regarding operative time, anastomotic leakage, surgical site infection, cardiac complications, pulmonary complications, or number of harvested lymph nodes. We found significant differences regarding length of hospital stay and morbidity in the Asian population.
Conclusion: This systematic review indicates that a laparoscopic approach for D2 total gastrectomy does not increase morbidity. Furthermore, it allows for a shorter hospital stay. However, more randomized controlled trials are required to fully assess this approach because available data are of limited quality.
Keywords: systematic review, meta-analysis, total gastrectomy, laparoscopy
Introduction
Gastric cancer is one of the five most common malignant neoplasms worldwide, making it a worldwide health concern.1 Radical resection with adequate lymphadenectomy is required to provide the best oncological response.2 Currently, an open approach is the gold standard. However, minimally invasive approaches such as laparoscopy, laparoscopic-assisted and robotic procedures have been introduced with success in oncological surgery of the stomach.3 Even though the first laparoscopic surgery for gastric cancer by Kitano was performed in 1993, incorporation of laparoscopy as a method of choice for gastric resections remains controversial because of limited evidence of eligibility. To date, no high-quality randomized quality trials regarding totally laparoscopic D2 total gastrectomies (TLDGs) have been conducted. Moreover, most of the observational studies that are available focus on the Asian population, which is of great importance because there are as yet unexplained differences in many aspects of this disease and its treatment compared with western populations.4–6 Large randomized controlled trials (RCTs) are currently underway, but results are not expected until the next few years and the majority of them are being conducted in Asian countries.7,8 Our study aimed to assess currently available literature in this matter and provide meta-analysis on acquired data regarding short-term outcomes with a subgroup analysis of western and eastern studies.
Methods
Study selection
A systematic review of the literature was performed using the Medline, Embase and Cochrane databases to identify all eligible studies that compared patients undergoing TLDG with patients undergoing open D2 gastrectomy (ODG). The search terms were: “laparoscopy,” “minimally invasive,” “classic,” “open,” and “gastrectomy.” These terms were combined using the Boolean operators “AND” and “OR.” The most recent search was performed on November 6, 2017.
Studies eligible for further analysis had to fulfill the following criteria: 1) comparison of patients undergoing TLDG versus ODG; 2) D2 lymphadenectomy; 3) publication in English. Studies were excluded when there was: 1) lack of comparative data; 2) insufficient data to analyze; 3) type of lymphadenectomy unknown 4) laparoscopy-assisted procedures or robotic procedures.
Outcomes of interest
Primary outcomes of interest involved perioperative parameters such as operative time, morbidity (described as the overall complication rate reported by the authors), anastomotic leakage, surgical site infections, cardiopulmonary complications, and length of hospital stay. Secondary outcomes involved oncological effect: the R0 resection rate and lymph node yield.
Data extraction and quality assessment
All references were reviewed and evaluated by two teams of two researchers. In case of any doubts about eligibility for inclusion, an attempt was made to reach consensus within the group. If no resolution was possible, an arbitrary decision was made by another reviewer. Data from the included studies were extracted independently by all teams. Only full-length articles were eligible for extraction. When available, the following data were extracted: first author, year of publication, number of operated subjects, sex, age, tumor size, tumor stage, and outcomes of interest.
Non-randomized studies were evaluated according to the Newcastle–Ottawa Scale (NOS), which consists of three factors: patient selection, comparability of study groups, and assessment of outcomes. A score of 0–9 was assigned to each study, and studies achieving a score of 6 or higher were considered high quality. This study was performed according to the PRISMA guidelines and the Meta-Analysis of Observational Studies in Epidemiology (MOOSE) consensus statement.9,10
Data analysis
Analysis was performed using RevMan 5.3 (freeware from the Cochrane Collaboration). Statistical heterogeneity and inconsistency were measured using Cochran’s Q tests and I2, respectively. Qualitative outcomes from individual studies were analyzed to assess individual and pooled risk ratios (RR) with pertinent 95% CI favoring patients undergoing revisionary surgery and by means of the Mantel–Haenszel random-effects method. When appropriate, mean and SD were calculated from medians and interquartile ranges using a method proposed by Hozo et al.11 Weighted mean differences (WMDs) with a 95% CI are presented for quantitative variables using the inverse variance random-effects method. Statistical significance was observed with a two-tailed 0.05 level for hypotheses, and with 0.10 for heterogeneity testing, while unadjusted P-values were reported accordingly. Additionally, we introduced subgroups, dividing studies based on the nationality of the analyzed population to eastern and western groups.
Results
The initial reference search yielded 3,073 articles. After removing 1,513 duplicates, 1,250 articles had their titles and abstracts evaluated. This resulted in 140 papers suitable for full-text review. Finally, we chose eight studies that were included in the quantitative analysis, which covered 1,582 patients (755 in the TLDG group and 827 in the ODG group). A flowchart of the analyzed studies is presented in Figure 1. The quality of the analyzed studies is moderate, with the majority scoring at least 7 points according to the NOS. Baseline information about the analyzed studies is presented in Table 1 and Figure 2. There were more patients with advanced tumors in open approach in comparison to the laparoscopic group. According to this division, three studies were based on a western population and five studies were based on an Asian population.
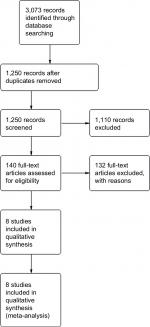  | Figure 1 PRISMA flowchart. |
  | Table 1 Baseline characteristics of included studies Abbreviations: C, cohort; CC, case-control; F/M, female/male; ND, no data; NOS, Newcastle–Ottawa Scale. |
  | Figure 2 Tumor stage distribution among groups. |
The operative time was reported in all studies. There were no differences between analyzed groups, WMD: 13.2; 95% CI [–14.77, –41.18]; P=0.35 (Figure 3). Topal et al,13 Lu et al,15 and Ramagen et al16 reported shorter times for laparoscopy, whereas Wu et al,17 Lu et al,18 and Shu et al19 reported the opposite. The heterogeneity was very high, I2=99%. Sensitivity analysis revealed three studies generating all heterogeneity – Kim et al,14 Lu et al,17 and Ramagen et al.16 After removing those studies, open surgeries were shorter by an average of 50 minutes.
  | Figure 3 Operative time analysis. |
Morbidity was reported in seven studies. Only one study by Kim et al presented data in favor of laparoscopy. After pooling data, meta-analysis revealed significant differences between TLDG (105/708, 14.83%) and ODG (167/763, 21.89%), RR: 0.67; 95% CI [0.51, 0.86]; P=0.002 (Figure 4). Subgroup analysis showed significant differences in the Asian population, RR: 0.63; 95% CI [0.47, 0.85]. A similar analysis of the western population did not show significant differences; however, the sample size was small. The heterogeneity was low, I2=17%.
  | Figure 4 Morbidity analysis. |
Anastomotic leakage was reported in seven studies. There were no statistically significant differences either in total or within subgroups, RR: 0.67; 95% CI [0.36, 1.28]; P=0.23 (Figure 5). The heterogeneity was low.
  | Figure 5 Anastomotic leakage analysis. |
Surgical site infection was reported by four authors. There were no significant differences in the Asian group, RR: 0.44; 95% CI [0.15, 1.3]; P=0.14. There was only one study in the western group and it also did not report a significant difference. The heterogeneity was low and after pooling subgroups, the result remained statistically insignificant (Figure 6).
  | Figure 6 Surgical site infection analysis. |
Cardiac complications were reported only by two authors, whereas pulmonary complications were reported in five studies. There were no differences in cardiac complications between analyzed groups, RR: 0.6; 95% CI [0.13, 2.81]; P=0.52 (Figure 7). Analysis of pulmonary complications resulted in similar conclusions, RR: 0.68; 95% CI [0.41, 1.14]; P=0.14 (Figure 8). In both outcomes, the heterogeneity of included studies was low, I2=0%.
  | Figure 7 Cardiac complications analysis. |
  | Figure 8 Pulmonary complications analysis. |
Length of hospital stay was reported in seven studies. The analysis revealed laparoscopy was associated with a 2.36-day shorter hospital stay (9.59 days in TLDG group versus 12.42 days in ODG group), 95% CI [–3.30, –1.41]; P<0.0001 (Figure 9). All authors, except Kim et al, reported data in favor of laparoscopy. The heterogeneity of the studies was moderate, I2=70%.
  | Figure 9 Length of hospital stay analysis. |
The R0 rate was reported in six studies. There were no differences in analyzed material, RR: 1; 95% CI [0.99, 1.01]; P=0.97 (Figure 10). There was no heterogeneity between the included studies.
  | Figure 10 R0 rate analysis. |
Lymph node yield was reported in seven studies. There were no differences in the number of harvested nodules between analyzed groups, WMD: –0.03; 95% CI [–1.01, 0.95]; P=0.96 (Figure 11). Kim et al and Lu et al reported data in favor of an open approach, whereas Ramagen et al, Wu et al and Lu et al showed results in favor of a laparoscopic approach. The heterogeneity of included studies was high, I2=84%. Sensitivity analysis allowed reduction after removing studies by Lu and Shu to 67%; however, the result remained insignificant.
  | Figure 11 Harvested lymph nodes analysis. |
Discussion
This systematic review, based on 1,582 patients, showed that a laparoscopic approach for total gastrectomy with D2 lymphadenectomy is associated with significantly lower morbidity and shorter length of stay (LOS) with no negative impact on short-term oncological outcomes. All of the included studies were observational; thus, the available data quality is limited. Compared with previous systematic reviews, our study incorporated a subgroup distinction for western and eastern populations.5,20,21 Another major difference in favor of this systematic review was the inclusion of totally laparoscopic total gastrectomies only. It is important because prior meta-analyses incorporated studies involving laparoscopic-assisted procedures or partial gastrectomy.5 This could possibly introduce bias because totally laparoscopic procedures with intracorporeal anastomoses are considered technically more demanding.22,23
Laparoscopy is gradually gaining support in oncological surgery. Its efficacy has been proven best in colorectal surgery in numerous high-quality RCTs and systematic reviews that followed.24–26 As for other neoplasms such as gastric cancer, we can currently rely only on data from observational studies, which in general show that the novel approach has been as good as the classic approach, and in some studies the novel approach provided better results. The only available RCTs regarding gastric cancer were focused on distal gastrectomies.27–29 RCTs regarding laparoscopic versus open gastrectomy for gastric cancer are ongoing in Japan (JCOG1401), China (CLASS02–01), Korea (KLASS03), and the Netherlands (STOMACH).
An important factor determining the difficulty of surgery and, as a result, the potential safety of the technique is the duration of the procedure. In our study, operative time at first did not differ in total between both approaches. However, the heterogeneity was very high. Authors’ reports varied from shorter times for laparoscopy, no difference in duration between the two procedures, or a shorter time in the open approach. After identifying three studies generating the majority of the heterogeneity, (Kim et al, Ramagen, and Lu), we decided to analyze the material without them. This decision resulted in shorter operative time by 50 minutes in favor of open surgery. On one hand, laparoscopy is associated with shorter time spent for proper abdomen closure. On the other, it is more technically demanding. Another possible explanation is the learning curve. Most of the studies did not point out whether surgeons were still on the learning curve or how far beyond it they had gone. The next probable explanation is lack of information about how some authors defined operative time. That is, is it from opening to the closure of the abdomen or beginning of general anesthesia? Although operative time can be considered a reliable benchmark in assessing the operative technique, it would seem that the clinical relevance of this outcome is limited. One could say that it is safer to take a longer time to operate and reduce the chance of overlooking any avoidable complications. In our study, a longer operative time did not increase overall morbidity. On the contrary, morbidity in our study was lower in the laparoscopic group. This is important because the number of complications is another factor of surgery safety. It is interesting that in the subgroup of the western population, the difference was not present. The range of lymphadenectomy may also cause discrepancies in morbidity rate. Another factor underlining this assumption is the lack of significant differences in the rate of anastomotic leakages. That there was no heterogeneity between the western and Asian populations suggests also that, even though laparoscopic esophagoenterostomy is technically demanding and requires meticulous performance, the success rate of proper anastomoses is similar. Another factor that can affect the results is the nutritional status. None of the authors provided this information, which affects both short- and long-term results after gastrectomy.30,31 Finally, the experience of the laparoscopic surgeon is also an important factor to consider in regard to morbidity because gastrectomy is one of the most difficult surgeries; it involves the abdomen and sometimes the chest cavity. All of the above are among the many elements affecting the length of hospital stay. Our meta-analysis showed significantly shorter LOS for the laparoscopic approach, which is in line with the contributions of the laparoscopic approach in different surgical fields.32 However, an unbiased comparison of LOS between countries and hospitals is difficult because it is more associated with local customs than fulfilling clear objective discharge criteria.
The debate on the oncological advantages of various surgical and non-surgical approaches for gastric cancer persists. Currently a preferred surgical method of treatment is gastrectomy with D2 lymphadenectomy. In our study the R0 rate did not differ among the authors with a lack of heterogeneity. Adequate lymphadenectomy during surgery is a crucial part of patient survival. Available meta-analysis of D2 versus D1 lymphadenectomy by Seevaratnam et al and Mocellin et al shows greater mortality and morbidity in D2 resections; however, subgroup analysis showed that this does not occur in more up-to-date studies.33,34 Furthermore, long-term survival reported by El-Sedfy showed better results for patients with T3 cancers.35 While initial experiences with D3 lymphadenectomy showed promise, later RCTs and meta-analysis showed no additional benefit from extensive excision.36,37 In our study, we decided to include studies with D2 lymphadenectomy only. Analysis of lymph node yield was associated with high heterogeneity. Most likely it was caused by different techniques and lack of standardization of lymphadenectomy. De Steur et al, in their study, showed that over 80% of patients did not receive proper lymph station excision.38 No differences in the R0 rate along with indifferent lymph node may show lack of oncological inferiority of laparoscopic approach to open; however, its true oncological value is yet to be determined with long-term survival rates. Chen et al, in their meta-analysis regarding minimally invasive gastrectomies, showed no difference in 5-year overall survival; however extrapolating this to totally laparoscopic gastrectomies may be prone to bias because the subgroup analysis included only two studies.39 This is why the results of ongoing RCTs are so anticipated. None of the studies included in our review provided information on long-term survival, and from an oncological point of view, long-term survival is one of the most important outcomes.
Limitations
The main limitation of this study is the lack of randomized control studies. While most of the studies were of rather high quality, drawing valid conclusions may be prone to bias. The open approach group had more patients with more advanced cancer (stages III and IV), which could be associated with selection, and therefore potentially affect results. Furthermore, none of the analyzed studies provided information on the experience of the surgeons, which could drastically change results. Additionally, none of the papers provided meticulous information on the perioperative care protocol used in every unit. This is important because studies show how novel enhanced recovery after surgery (ERAS) protocols may improve short-term outcomes40–43 The main advantage of our study was the introduction of subgroups for western and Asian populations. It shows that, while we are getting sufficient data regarding the eastern population, the information we have on western countries is scarce. Although we await the results of the STOMACH trial in the Netherlands, it is possible that the number of RCTs in Europe and the US may not increase because patients prefer minimally invasive surgery, which makes them reluctant to enroll in ongoing trials comparing an open approach with laparoscopic surgery.44 Furthermore, the prevalence of gastric cancer in Europe is lower than in Asia, which results in a lower annual number of gastrectomies. For this reason, to achieve sufficient patient numbers, multicenter studies are required. On one hand, it is good because we need to have data on the European population as well. On the other hand, this may cause bias associated with surgical technique. Nevertheless, to provide sufficient answers backed with high-quality data, we need RCTs.
Conclusion
This systematic review indicated that a laparoscopic approach for D2 total gastrectomy does not increase morbidity in comparison to an open approach. Furthermore, a laparoscopic approach allows for a shorter hospital stay. However, more RCTs are required to fully assess this approach because the available data are of limited quality.
Author contributions
Piotr Małczak contributed to concept and design, manuscript preparation, and data collection. Grzegorz Torbicz performed data collection and data analysis. Mateusz Rubinkiewicz involved in manuscript preparation and draft. Natalia Gajewska, Nadia Sajuk, and Kamil Rozmus contributed to data collection. Michał Wysocki performed data analysis. Piotr Major contributed to data interpretation. Andrzej Budzyński and Michał Pędziwiatr provided critical review of the manuscript. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Ferlay J, Partensky C, Bray F. More deaths from pancreatic cancer than breast cancer in the EU by 2017. Acta Oncol. 2016;55(9–10):1158–1160. | ||
Coburn N, Cosby R, Klein L, et al. Staging and surgical approaches in gastric cancer: a clinical practice guideline. Curr Oncol. 2017;24(5):324–331. | ||
Association JGC. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer. 2011;14(2):113–123. | ||
Kitano S, Iso Y, Moriyama M, Sugimachi K. Laparoscopy-assisted Billroth I gastrectomy. Surg Laparosc Endosc. 1994;4(2):146–148. | ||
Xiong JJ, Nunes QM, Huang W, et al. Laparoscopic vs open total gastrectomy for gastric cancer: a meta-analysis. World J Gastroenterol. 2013;19(44):8114–8132. | ||
Strong VE, Song KY, Park CH, et al. Comparison of gastric cancer survival following R0 resection in the United States and Korea using an internationally validated nomogram. Ann Surg. 2010;251(4):640–646. | ||
Hur H, Lee HY, Lee HJ, et al. Efficacy of laparoscopic subtotal gastrectomy with D2 lymphadenectomy for locally advanced gastric cancer: the protocol of the KLASS-02 multicenter randomized controlled clinical trial. BMC Cancer. 2015;15:355. | ||
Haverkamp L, Brenkman HJ, Seesing MF, et al. Laparoscopic versus open gastrectomy for gastric cancer, a multicenter prospectively randomized controlled trial (LOGICA-trial). BMC Cancer. 2015;15:556. | ||
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8(5):336–341. | ||
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–2012. | ||
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. | ||
Dulucq JL, Wintringer P, Stabilini C, Solinas L, Perissat J, Mahajna A. Laparoscopic and open gastric resections for malignant lesions: a prospective comparative study. Surg Endosc. 2005;19(7):933–938. | ||
Topal B, Leys E, Ectors N, Aerts R, Penninckx F. Determinants of complications and adequacy of surgical resection in laparoscopic versus open total gastrectomy for adenocarcinoma. Surg Endosc. 2008;22(4):980–984. | ||
Kim HS, Kim BS, Lee IS, Lee S, Yook JH. Comparison of totally laparoscopic total gastrectomy and open total gastrectomy for gastric cancer. J Laparoendosc Adv Surg Tech A. 2013;23(4):323–331. | ||
Lu J, Huang CM, Zheng CH, et al. Short- and Long-Term Outcomes After Laparoscopic Versus Open Total Gastrectomy for Elderly Gastric Cancer Patients: a Propensity Score-Matched Analysis. J Gastrointest Surg. 2015;19(11):1949–1957. | ||
Ramagem CA, Linhares M, Lacerda CF, Bertulucci PA, Wonrath D, de Oliveira AT. Comparison of laparoscopic total gastrectomy and laparotomic total gastrectomy for gastric cancer. Arq Bras Cir Dig. 2015;28(1):65–69. | ||
Lu Y, Jiang B, Liu T. Laparoscopic versus open total gastrectomy for advanced proximal gastric carcinoma: a matched pair analysis. J BUON. 2016;21(4):903–908. | ||
Shu B, Lei S, Li F, Hua S, Chen Y, Huo Z. Laparoscopic total gastrectomy compared with open resection for gastric carcinoma: a case-matched study with long-term follow-up. J BUON. 2016;21(1):101–107. | ||
Wu H, Li W, Chen G, et al. Outcome of laparoscopic total gastrectomy for gastric carcinoma. J BUON. 2016;21(3):603–608. | ||
Chen K, Pan Y, Cai JQ, Xu XW, Wu D, Mou YP. Totally laparoscopic gastrectomy for gastric cancer: a systematic review and meta-analysis of outcomes compared with open surgery. World J Gastroenterol. 2014;20(42):15867–15878. | ||
Coburn N, Cosby R, Klein L, et al. Staging and surgical approaches in gastric cancer: A systematic review. Cancer Treat Rev. 2018;63:104–115. | ||
Jeong O, Jung MR, Park YK, Ryu SY. Safety and feasibility during the initial learning process of intracorporeal Billroth I (delta-shaped) anastomosis for laparoscopic distal gastrectomy. Surg Endosc. 2015;29(6):1522–1529. | ||
Jeong O, Ryu SY, Choi WY, Piao Z, Park YK. Risk factors and learning curve associated with postoperative morbidity of laparoscopic total gastrectomy for gastric carcinoma. Ann Surg Oncol. 2014;21(9):2994–3001. | ||
van der Pas MH, Haglind E, Cuesta MA, et al. Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol. 2013;14(3):210–218. | ||
Kennedy RH, Francis EA, Wharton R, et al. Multicenter randomized controlled trial of conventional versus laparoscopic surgery for colorectal cancer within an enhanced recovery programme: EnROL. J Clin Oncol. 2014;32(17):1804–1811. | ||
Pedziwiatr M, Malczak P, Mizera M, et al. There is no difference in outcome between laparoscopic and open surgery for rectal cancer: a systematic review and meta-analysis on short- and long-term oncologic outcomes. Tech Coloproctol. 2017;21(8):595–604. | ||
Kim W, Kim HH, Han SU, et al. Decreased Morbidity of Laparoscopic Distal Gastrectomy Compared With Open Distal Gastrectomy for Stage I Gastric Cancer: Short-term Outcomes From a Multicenter Randomized Controlled Trial (KLASS-01). Ann Surg. 2016;263(1):28–35. | ||
Hu Y, Huang C, Sun Y, et al. Morbidity and Mortality of Laparoscopic Versus Open D2 Distal Gastrectomy for Advanced Gastric Cancer: A Randomized Controlled Trial. J Clin Oncol. 2016;34(12):1350–1357. | ||
Katai H, Mizusawa J, Katayama H, et al. Short-term surgical outcomes from a phase III study of laparoscopy-assisted versus open distal gastrectomy with nodal dissection for clinical stage IA/IB gastric cancer: Japan Clinical Oncology Group Study JCOG0912. Gastric Cancer. 2017;20(4):699–708. | ||
Liu X, Zhang D, Lin E, et al. Preoperative controlling nutritional status (CONUT) score as a predictor of long-term outcome after curative resection followed by adjuvant chemotherapy in stage II-III gastric Cancer. BMC Cancer. 2018;18(1):699. | ||
Lee JY, Kim HI, Kim YN, et al. Clinical Significance of the Prognostic Nutritional Index for Predicting Short- and Long-Term Surgical Outcomes After Gastrectomy: A Retrospective Analysis of 7781 Gastric Cancer Patients. Medicine (Baltimore). 2016;95(18):e3539. | ||
Małczak P, Pisarska M, Piotr M, Wysocki M, Budzyński A, Pędziwiatr M. Enhanced Recovery after Bariatric Surgery: Systematic Review and Meta-Analysis. Obes Surg. 2017;27(1):226–235. | ||
Seevaratnam R, Bocicariu A, Cardoso R, et al. A meta-analysis of D1 versus D2 lymph node dissection. Gastric Cancer. 2012;15 Suppl 1:S60–69. | ||
Mocellin S, Nitti D. Lymphadenectomy extent and survival of patients with gastric carcinoma: a systematic review and meta-analysis of time-to-event data from randomized trials. Cancer Treat Rev. 2015;41(5):448–454. | ||
El-Sedfy A, Dixon M, Seevaratnam R, et al. Personalized Surgery for Gastric Adenocarcinoma: A Meta-analysis of D1 versus D2 Lymphadenectomy. Ann Surg Oncol. 2015;22(6):1820–1827. | ||
Tokunaga M, Ohyama S, Hiki N, Fukunaga T, Aikou S, Yamaguchi T. Can superextended lymph node dissection be justified for gastric cancer with pathologically positive para-aortic lymph nodes? Ann Surg Oncol. 2010;17(8):2031–2036. | ||
Sasako M, Sano T, Yamamoto S, et al. D2 lymphadenectomy alone or with para-aortic nodal dissection for gastric cancer. N Engl J Med. 2008;359(5):453–462. | ||
de Steur WO, Hartgrink HH, Dikken JL, Putter H, van de Velde CJ. Quality control of lymph node dissection in the Dutch Gastric Cancer Trial. Br J Surg. 2015;102(11):1388–1393. | ||
Chen XZ, Wen L, Rui YY, et al. Long-term survival outcomes of laparoscopic versus open gastrectomy for gastric cancer: a systematic review and meta-analysis. Medicine (Baltimore). 2015;94(4):e454. | ||
Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, Braga M. Enhanced recovery program in colorectal surgery: a meta-analysis of randomized controlled trials. World J Surg. 2014;38(6):1531–1541. | ||
Matlok M, Pedziwiatr M, Major P, Klek S, Budzynski P, Malczak P. One hundred seventy-nine consecutive bariatric operations after introduction of protocol inspired by the principles of enhanced recovery after surgery (ERAS(R)) in bariatric surgery. Med Sci Monit. 2015;21:791–797. | ||
Pedziwiatr M, Kisialeuski M, Wierdak M, et al. Early implementation of Enhanced Recovery After Surgery (ERAS(R)) protocol - Compliance improves outcomes: A prospective cohort study. Int J Surg. 2015;21:75–81. | ||
Pedziwiatr M, Matlok M, Kisialeuski M, et al. Enhanced recovery (ERAS) protocol in patients undergoing laparoscopic total gastrectomy. Wideochir Inne Tech Maloinwazyjne. 2014;9(2):252–257. | ||
Etoh T, Inomata M, Watanabe M, et al. Success rate of informed consent acquisition and factors influencing participation in a multicenter randomized controlled trial of laparoscopic versus open surgery for stage II/III colon cancer in Japan (JCOG0404). Asian J Endosc Surg. 2015;8(4):419–423. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
