Back to Journals » OncoTargets and Therapy » Volume 7
Comparison of short-term effect of thoracoscopic segmentectomy and thoracoscopic lobectomy for the solitary pulmonary nodule and early-stage lung cancer
Authors Ren M, Meng Q, Zhou W, Kong F, Yang B, Yuan J, Wu D, Zhang J, Li Q, Lin Y, Viswanathan V, Song X
Received 22 February 2014
Accepted for publication 13 May 2014
Published 24 July 2014 Volume 2014:7 Pages 1343—1347
DOI https://doi.org/10.2147/OTT.S62880
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Mingming Ren,1,* Qingjun Meng,1,* Wenyan Zhou,1 Fanyi Kong,1 Bo Yang,1 Jun Yuan,1 Dongwen Wu,2,* Jing Zhang,3,* Qiaqia Li,2 Yunshou Lin,3 Vidya Bhavani Viswanathan,4 Xiang Song1,*
1Department of Thoracic Surgery, Cangzhou Central Hospital, Hebei, People’s Republic of China; 2The Xiangya Medical School of Central-South University, Changsha, People’s Republic of China; 3Tianjin Medical University, Tianjin, People’s Republic of China; 4Harvard University, Cambridge, MA, USA
*These authors contributed equally to this work
Purpose: To compare the short-term effect of anatomic video-assisted thoracoscopic surgery (VATS) segmentectomy and VATS lobectomy.
Patients and methods: From January 2011 to December 2012, 21 patients underwent VATS segmentectomy and 61 underwent VATS lobectomy. Intraoperative blood loss, operating time, postoperative drainage time, length of hospital stay, postoperative complications, local recurrence, and survival were compared between the two groups.
Results: The intraoperative blood loss and average hospital stay were less in the segmentectomy group than in the lobectomy group (P<0.05). There was no significant difference in the operating time, number of lymph nodes dissected, postoperative drainage time, or 1-year survival between the two groups (P>0.05). Only one patient died because of heart disease. The two groups had a similar incidence of postoperative complications (P>0.05). There was one (4.8%) local recurrence after segmentectomy and two (3.3%) after lobectomy (P>0.05).
Conclusion: VATS segmentectomy could be performed safely and is a method with favorable 1-year survival. It may be the ideal surgical procedure for patients with solitary pulmonary nodules in early stage lung cancer, especially for those with limited cardiopulmonary reserve or significant comorbidities.
Keywords: thoracoscopes, segmentectomy, lobectomy, solitary pulmonary nodules
Introduction
A recent report from the National Lung Screening trial showed a significant 20% reduction in lung cancer mortality among those screened with low-dose spiral computed tomography (CT) compared to chest radiography.1 Today, with multislice spiral CT widely available, increasing numbers of small pulmonary nodules have been identified in higher-risk surgical patients. In 1993, Video-Assisted Thoracic Surgery (VATS) lobectomy was first described by Kirby et al2 for the treatment of early stage non-small-cell lung cancer and benign nodules. It has been proven safe, minimally invasive, and radical in the decades since. Roviaro et al3 reported the first case of VATS segmentectomy in 1993.
During the past decade, thoracoscopic lobectomy has gained popularity, whereas thoracoscopic segmentectomy has continued to be an area of controversy because of the complexity of the procedure and the fear of increased local recurrence.4 However, numerous published studies suggest the outcomes of VATS lobectomy and segmentectomy procedures for clinical stage I non-small-cell carcinoma (NSCLC) were equivalent for early-stage NSCLC patients.5,6 There is still a lack of published studies examining the short-term effect after VATS segmentectomy relative to that after VATS lobectomy, especially in the People’s Republic of China. We retrospectively analyzed 82 cases under thoracoscope, including 21 patients who underwent VATS segmentectomy and 61 who underwent VATS lobectomy. On follow-up, the efficacy, safety, benefits, and limitations of VATS segmentectomy and VATS lobectomy were compared for all patients.
Material and methods
Participants
For this retrospective study, the data were actively collected from the medical records of patients with small-sized stage IA NSCLC (≤3 cm) treated at the Cangzhou Central Hospital, Third Xiangya Hospital, and Tianjin General Hospital, from Jan 2011 to Dec 2012. Patients had undergone either VATS segmentectomy or VATS lobectomy. Patients’ physical status and comorbidities such as cardiovascular disease, pulmonary disease with low pulmonary function, and tumor diameter were considered when selecting the appropriate surgical procedure.
Before the treatment, patients’ liver and renal function, tumor markers, and blood coagulate functions were tested. Routine examinations such as urinalysis and blood count were completed. Cranial CT, abdominal CT, or ultrasound, emission computed tomography, or positron emission computed tomography were performed in order to confirm the absence of distant metastases. To those patients with pure ground-glass opacity or nodes too small to locate intraoperatively, the position was fixed under CT and marked by hook wire after puncture.
Operative technique
The patients were placed in the lateral decubitus position for thoracotomy. The procedures were performed under general anesthesia with single-lung ventilation. The surgeon was positioned on the anterior side of the patient. All procedures were conducted under video vision without direct vision or rib spreading. Three incisions were used in all patients. A 10 mm, 30-degree thoracoscope through a thoracoscopic port was placed in the anterior axillary line in the eighth intercostal space. Two additional access incisions were placed in the fourth or fifth intercostal spaces in the anterior and posterior axillary lines, according to the position of the tumor. The operative technique of thoracoscopic lobectomy was similar to that of previous reports.7 Segmentectomy was performed with resection of the segmental artery, intersegmental vein, and segmental bronchus, respectively, and the intersegmental lung parenchyma was divided using an automated suturing device or with electrocautery.5 In most VATS segmentectomies, the segmental bronchus was clamped to check reventilation before stapling. The parenchymal surgical margin for all thoracoscopic segmentectomies was at least 2 cm. Systemic lymphadenectomy was performed in both groups.
Outcome measures
Operating time, intraoperative blood loss, number of lymph nodes dissected, postoperative drainage time, hospital stay, complication rate, local recurrence, and death rate were assessed.
Statistical analyses
Statistical analysis was performed using SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). All data are expressed as mean ± standard deviation. Differences between groups with continuous variables were assessed by chi-square test or the Fisher exact test. Statistical significance was accepted as P<0.05 throughout the study.
Results
Eighty-two patients who underwent either VATS segmentectomy or VATS lobectomy for NSCLC were examined in this study. Of the 21 patients from segmentectomy group, eight were male and 13 were female, with an average age of 56 years. Of the 61 patients from the lobectomy group, 23 were male and 38 were female, with an average age of 51 years. In the segmentectomy group, 18 of the tumors were adenocarcinomas and three were squamous cell carcinomas. In the VATS lobectomy group, 55 tumors were adenocarcinomas, five were squamous cell carcinomas, and one was a squamous adenocarcinoma. No down-staged case appeared in this study. Patient characteristics and tumor location details are shown in Tables 1 and 2.
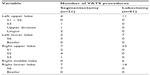  | Table 2 Location of the tumor |
No conversion or reoperation to thoracotomy was performed in any of the 82 patients. No atelectasis, lung infection, air leaks, arrhythmia, or death during the perioperative period occurred. Patients from both groups were discharged from hospital after complete recovery. There was no significant difference between the anatomic segmentectomy group and lobectomy group in average surgery time, postoperative drainage time, and number of lymph nodes dissected (P>0.05). The segmentectomy group showed a faster recovery, including significantly reduced blood loss and length of hospital stay (P<0.05) (Table 3).
  | Table 3 Patient recovery and follow-up |
The postoperative complications in the segmentectomy group included atrial fibrillation (n=1) and respiratory failure (n=1). In patients with lobectomy, atrial fibrillation (n=2), respiratory failure (n=1), pneumonia (n=1), and vocal cord paralysis (n=1) occurred. The two groups showed similar postoperative complication rates, at 9.5% in patients who underwent segmentectomy and 8.2% in patients who underwent lobectomy.
Follow-up was completed for all patients, with a median of 1.5 years (range, 3 months to 2.5 years). Local recurrences were documented: one (4.8%) in the group that underwent segmentectomy and two (3.3%) in the group that underwent lobectomy. After 6 months of follow-up, the one patient with a local recurrence in the segmentectomy group had a marginal recurrence in left S6, but no lymph node recurrence appeared. In the lobectomy group, a patient who underwent right upper-lobe lobectomy had a chest CT scan at a one-year follow-up that showed right hilar lymph node enlargement, which is highly suggestive of lymph node metastasis. Another patient in the lobectomy group underwent left upper-lobe lobectomy, and at a 1.5 year follow-up, a chest CT scan showed two small nodules in the left lower lobe, suggesting metastasis. Local recurrence rates were similar between the two groups (P>0.05). No distant recurrences were reported. Only one patient in the thoracoscopic lobectomy group died because of heart disease during the follow-up period. The overall estimated 1-year survival rate for the entire study population was 98.8%. There was no significant difference between the two groups in 1-year survival (P>0.05).
Discussion
One advantage of segmentectomy over lobectomy is that it spares pulmonary parenchyma. Therefore, in some scenarios in which wedge resection cannot provide an adequate operation, segmentectomy may be the most feasible surgical option for patients with pulmonary carcinoid or malignant tumors. However, as for primary lung cancer, the practical value of sublobar resection remains controversial. In 1995, a randomized trial performed by Ginsberg and Rubinstein8 showed that there was a significant threefold increase in the locoregional recurrence rate for the limited resection group, compared with the lobectomy treatment group. For these reasons, sublobar resections have been used as parenchymal sparing procedures in patients with significantly reduced pulmonary function. However, in the 1995 report a high percentage of patients in the sublobar resection group (32.8%) underwent a wedge resection. None of the data stratified by tumor diameter was further analyzed.8
Due to the dramatic development of radiographic technology, the diagnosis of small-sized peripheral NSCLC has been widely improved. Recently, several studies have revealed that for stage IA disease (≤2.0 cm), there was no significant difference between anatomic segmentectomy and lobectomy groups in long-term effects, demonstrating an 83.0%–96.7% 5-year survival of the former.6,9–12 The National Comprehensive Cancer Network guidelines suggest that anatomic segmentectomy is an option in some cases of early-stage peripheral pulmonary cancer. The criteria are as follows: first, patients with a comorbid disease or decreased cardiopulmonary function that may not tolerate lobectomy; second, tumors ≤2 cm, with one of the following conditions: adenocarcinoma in situ; ground-glass opacity ratio ≥50%; or CT follow-up showed a doubling time ≥400 days. To define whether or not segmental resection can be applied in a broader sense, we are looking forward to the results of two ongoing prospective randomized trials of lobar versus sublobar resection in patients with small peripheral NSCLC (≤2 cm).13,14
Thoracoscopic segmentectomy has produced smaller trauma, less postoperative pain, and better functional results.6 Previous research has shown that, as for stage IA NSCLC, the VATS segmentectomy showed no significant difference compared with lobectomy in terms of the number of groups undergoing lymph node resection, number of harvested lymph nodes, local recurrence, and survival rates.15,16 Compared with wedge resection, segmentectomy produced adequate margins, better lymphadenectomy between pulmonary lobes, lower recurrence rates, and a better 5-year survival rate.17
Our contrastive study comparing segmentectomy and lobectomy (21 patients versus 61 patients) demonstrated that no extensive bleeding occurred and no conversion to thoracotomy was performed. The VATS segmentectomy group experienced reduced blood loss and faster recovery, causing shorter hospitalization and higher bed rotation and utilization rates. The postoperative complication rates, local recurrence rates, and 1-year survival rates were similar between the two groups. Thus, we can conclude that VATS segmentectomy shares the advantages (minimally invasive, safe, radical) of lobectomy in the treatment of solitary pulmonary nodules, with decreased blood loss and shorter hospital stays. It preserves long-term respiratory function in a minimally invasive way.18
The limitations of our study include the retrospective analysis and inherent differences in the tumor size and follow-up period between the two groups. Since there was no randomization in this study, selection bias by surgeons was possible. Another limitation is the relatively short period of follow-up to observe differences in local recurrence and overall survival between the two methods. Longer follow-up and increasing the number of patients in both groups may help us to further confirm these results.
Conclusion
VATS segmentectomy could be performed safely and is a method with a favorable 1-year survival. It may be the ideal surgical procedure for patients with solitary pulmonary nodules in early-stage lung cancer, especially for those with limited cardiopulmonary reserve or significant comorbidities.
Disclosure
The authors report no conflicts of interest in this work.
References
Wahidi MM, Govert JA, Goudar RK, Gould MK, McCrory DC; American College of Chest Physicians. Evidence for the treatment of patients with pulmonary nodules: when is it lung cancer?: ACCP evidence-based clinical practice guidelines (2nd edition). Chest. 2007;132(Suppl 3):94S–107S. | |
Kirby TJ, Rice TW. Thoracoscopic lobectomy. Ann Thorac Surg. 1993;56(3):784–786. | |
Roviaro GC, Rebuffat C, Varoli F, et al. Videoendoscopic thoracic surgery. Int Surg. 1993;78(1):4–9. | |
Zhong C, Fang W, Mao T, Yao F, Chen W, Hu D. Comparison of thoracoscopic segmentectomy and thoracoscopic lobectomy for small-sized stage IA lung cancer. Ann Thorac Surg. 2012;94(2):362–367. | |
Nakamura H, Taniguchi Y, Miwa K, et al. Comparison of the surgical outcomes of thoracoscopic lobectomy, segmentectomy, and wedge resection for clinical stage I non-small cell lung cancer. Thorac Cardiovasc Surg. 2011;59(3):137–141. | |
Shapiro M, Weiser TS, Wisnivesky JP, Chin C, Arustamyan M, Swanson SJ. Thoracoscopic segmentectomy compares favorably with thoracoscopic lobectomy for patients with small stage I lung cancer. J Thorac Cardiovasc Surg. 2009;137(6):1388–1393. | |
Shiraishi T, Shirakusa T, Iwasaki A, Hiratsuka M, Yamamoto S, Kawahara K. Video-assisted thoracoscopic surgery (VATS) segmentectomy for small peripheral lung cancer tumors: Intermediate results. Surg Endosc. 2004;18(11):1657–1662. | |
Ginsberg RJ, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg. 1995;60(3):615–622; discussion 622–623. | |
Henschke CI; I-ELCAP Investigators. CT screening for lung cancer: update 2005. Surg Oncol Clin N Am. 2005;14(4):761–776. | |
Landreneau RJ, Hazelrigg SR, Mack MJ, et al. Postoperative pain-related morbidity: video-assisted thoracic surgery versus thoracotomy. Ann Thorac Surg. 1993;56(6):1285–1289. | |
Pettiford BL, Schuchert MJ, Santos R, Landreneau RJ. Role of sublobar resection (segmentectomy and wedge resection) in the surgical management of non-small cell lung cancer. Thorac Surg Clin. 2007;17(2):175–190. | |
Pennathur A, Abbas G, Christie N, Landreneau R, Luketich JD. Video assisted thoracoscopic surgery and lobectomy, sublobar resection, radiofrequency ablation, and stereotactic radiosurgery: advances and controversies in the management of early stage non-small cell lung cancer. Curr Opin Pulm Med. 2007;13(4):267–270. | |
Tschernko EM, Hofer S, Bieglmayer C, Wisser W, Haider W. Early postoperative stress: video-assisted wedge resection/lobectomy vs conventional axillary thoracotomy. Chest. 1996; 109(6):1636–1642. | |
Demmy TL, Curtis JJ. Minimally invasive lobectomy directed toward frail and high-risk patients: a case-control study. Ann Thorac Surg. 1999;68(1):194–200. | |
El-Sherif A, Gooding WE, Santos R, et al. Outcomes of sublobar resection versus lobectomy for stage I non-small cell lung cancer: a 13-year analysis. Ann Thorac Surg. 2006;82(2):408–415; discussion 415–416. | |
Schuchert MJ, Pettiford BL, Keeley S, et al. Anatomic segmentectomy in the treatment of stage I non-small cell lung cancer. Ann Thorac Surg. 2007;84(3):926–932; discussion 932–933. | |
Okada M, Nishio W, Sakamoto T, et al. Effect of tumor size on prognosis in patients with non-small cell lung cancer: the role of segmentectomy as a type of lesser resection. J Thorac Cardiovasc Surg. 2005;129(1):87–93. | |
Keenan RJ, Landreneau RJ, Maley RH, et al. Segmental resection spares pulmonary function in patients with stage I lung cancer. Ann Thorac Surg. 2004;78(1):228–233; discussion 228–233. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

