Back to Journals » Vascular Health and Risk Management » Volume 15
Comparison of perioperative costs with fast-track vs standard endovascular aneurysm repair
Authors Krajcer Z , Ramaiah VG, Henao EA, Nelson WK, Moursi MM, Rajasinghe HA, Anderson LH , Miller LE
Received 30 March 2019
Accepted for publication 17 August 2019
Published 3 September 2019 Volume 2019:15 Pages 385—393
DOI https://doi.org/10.2147/VHRM.S210593
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Harry Struijker-Boudier
Zvonimir Krajcer1, Venkatesh G Ramaiah2, Esteban A Henao3, Wayne K Nelson4, Mohammed M Moursi5, Hiranya A Rajasinghe6, Louise H Anderson7, Larry E Miller8
1Department of Cardiology, CHI St. Luke’s Health, Houston, TX, USA; 2Department of Vascular Surgery, Honor Health, Scottsdale, AZ, USA; 3Department of Vascular Surgery, Heart Hospital of New Mexico, Albuquerque, NM, USA; 4Department of Vascular Surgery, St. Charles Hospital, Bend, OR, USA; 5Department of Vascular Surgery, Central Arkansas Veterans Healthcare System, Little Rock, AR, USA; 6Department of Vascular Surgery, NCH Healthcare Systems, Naples, FL, USA; 7Technomics Research, Long Lake, MN, USA; 8Miller Scientific Consulting, Inc., Asheville, NC, USA
Correspondence: Zvonimir Krajcer
Department of Cardiology, CHI St. Luke’s Health, 6624 Fannin #2780, Houston, TX 77005, USA
Email [email protected]
Background: Perioperative health care utilization and costs in patients undergoing elective fast-track vs standard endovascular aneurysm repair (EVAR) remain unclear.
Methods: The fast-track EVAR group included patients treated with a 14 Fr stent graft, bilateral percutaneous access, no general anesthesia or intensive care monitoring, and next-day hospital discharge. The standard EVAR group was identified from Medicare administrative claims using a matching algorithm to adjust for imbalances in patient characteristics. Hospital outcomes included operating room time, intensive care monitoring, hospital stay, secondary interventions, and major adverse events (MAEs). Perioperative outcomes occurring from hospital discharge to 30 days postdischarge included MAE, secondary interventions, and unrelated readmissions.
Results: Among 1000 matched patients (250 fast-track; 750 standard), hospital outcomes favored the fast-track EVAR group, including shorter operating room time (2.30 vs 2.83 hrs, P<0.001), shorter hospital stay (1.16 vs 1.69 d, P<0.001), less need for intensive care monitoring (4.4% vs 48.0%, P<0.001), and lower secondary intervention rate (0% vs 2.4%, P=0.01). Postdischarge outcomes also favored fast-track EVAR with a lower rate of MAE (0% vs 7.2%, P<0.001) and all-cause readmission (1.6% vs 6.8%, P=0.001). The total cost to the health care system during the perioperative period was $26,730 with fast-track EVAR vs $30,730 with standard EVAR. Total perioperative health care costs were $4000 (95% CI: $3130–$4830) lower with fast-track EVAR vs standard EVAR, with $2980 in savings to hospitals and $1030 savings to health care payers.
Conclusion: A fast-track EVAR protocol using a 14 Fr stent graft resulted in shorter procedure time, lower intensive care utilization, faster discharge, lower incidence of MAE, lower readmission rates, and lower perioperative costs compared to standard EVAR.
Keywords: abdominal aortic aneurysm, cost, EVAR, fast-track, Medicare, percutaneous, perioperative
Introduction
Endovascular aneurysm repair (EVAR) has become the first-line treatment for abdominal aortic aneurysm (AAA) requiring intervention at most centers. Compared to open surgical AAA repair, EVAR is associated with lower risk for early mortality and morbidity.1 However, EVAR remains costlier than surgical repair2 and results in negative operating margins for hospitals.2,3 In 2015, the Centers for Medicare and Medicaid Services (CMS) increased EVAR reimbursements by 14% for uncomplicated cases and 24% for cases with major comorbidity or complication,4 but these increases are insufficient to make EVAR a profitable procedure at most hospitals. In the current economic environment with bundled payments5 and financial penalties for higher than average readmission rates,6 adoption of treatment pathways that improve operational efficiency without compromising patient safety may potentially improve hospital profit margins.
Enhanced recovery after surgery pathways, also known as fast-track surgery, have been implemented across many medical disciplines, including vascular surgery.7 Over the last decade, several studies have evaluated the feasibility of fast-track EVAR in well-selected patients.8–12 While the elements of these programs differ slightly, the general premise involves regional/local anesthesia, percutaneous vascular access, no intensive care monitoring, and/or early discharge. While cost reductions with fast-track EVAR have been reported, these studies were performed outside the US9 or evaluated outpatient EVAR,11,12 a procedure not currently eligible for reimbursement in the US. Thus, the applicability of these findings to the US health care system is unclear. The purpose of this study was to compare perioperative health care utilization and costs from a hospital and health care payer perspective in patients undergoing fast-track EVAR vs matched patients undergoing standard EVAR.
Methods
Data sources
The fast-track EVAR population included patients enrolled in the prospective multicenter LIFE registry of elective EVAR utilizing bilateral percutaneous access with a 14 Fr stent graft (Ovation Prime, Endologix, Inc., Irvine, CA), no general anesthesia and intensive care monitoring, and next-day hospital discharge. A total of 250 patients were enrolled at 31 centers in the United States from October 2014 to May 2016. The study was prospectively registered at ClinicalTrials.gov (NCT02224794). The study design13 and perioperative outcomes14 from this study have been presented elsewhere. Patients were enrolled in consecutive fashion and were eligible for the study if they had a AAA anatomically suitable for elective EVAR and were deemed by the treating physician to be appropriate for a fast-track EVAR protocol based on pre-defined study eligibility criteria (Table 1).
 |
Table 1 Main study entry criteria for patients treated with fast-track endovascular aneurysm repair |
After obtaining approval from Western Institutional Review Board (Puyallup, WA) with a waiver of consent given the use of deidentified claims data, a standard EVAR population was identified using administrative claims on all Original Medicare beneficiaries available from CMS. Medicare is the primary health care payer in 79% of EVAR procedures;15 thus, the data derived from Medicare claims are representative of nationwide EVAR outcomes. We included all hospital claims submitted to the Inpatient Prospective Payment System and Outpatient Prospective Payment System, which were linked by beneficiary in blinded fashion across the continuum of care.
The standard EVAR population included adult Medicare beneficiaries who underwent elective EVAR between January 2014 and August 2015. The latter date was selected to allow for follow-up through September 2015, which corresponds to the date of transition from International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) to ICD-10-CM codes. Patients were identified using a combination of Medicare Severity-Diagnosis Related Group (MS-DRG) codes and ICD-9-CM codes. We initially included patients with MS-DRG code 237 (Major cardiovascular procedures with major complication or comorbidity or thoracic aortic aneurysm repair) or 238 (Major cardiovascular procedures without major complication or comorbidity), ICD-9-CM diagnosis code of 441.4 (Abdominal aneurysm without mention of rupture), and ICD-9-CM procedure code of 39.71 (Endovascular implantation of other graft in abdominal aorta). We then excluded patients with any of the following ICD-9-CM diagnosis codes: 441.0 (Dissection of aorta), 441.1 (Thoracic aneurysm, ruptured), 441.2 (Thoracic aneurysm without mention of rupture), 441.3 (Abdominal aneurysm, ruptured), 441.5 (Aortic aneurysm of unspecified site, ruptured), 441.6 (Thoracoabdominal aneurysm, ruptured), 441.7 (Thoracoabdominal aneurysm without mention of rupture), 441.9 (Aortic aneurysm of unspecified site without mention of rupture), 421.0 (Acute and subacute bacterial endocarditis), or 759.82 (Marfan syndrome). In order to account for the main exclusion criteria that were applied in the fast-track EVAR study, we also excluded patients with congestive heart failure, renal insufficiency, unstable angina, critical limb ischemia, major surgery, or reintervention procedure 30 days prior to the EVAR procedure, myocardial infarction, or stroke within the last 3 months, and patients with life expectancy less than 1 year estimated by a Charlson comorbidity index of 5 or greater.16 Exclusion conditions in the standard EVAR group were identified from Medicare claims in the 12-month period prior to the procedure.
Patient matching
In order to adjust for remaining imbalances in baseline patient characteristics between fast-track vs standard EVAR groups, we applied a 3-to-1 (standard-to-fast-track) algorithm that matched patients on key characteristics, including age, sex, chronic obstructive pulmonary disease, diabetes mellitus, coronary artery disease, valvular heart disease, cardiomyopathy, myocardial infarction, hypertension, peripheral vascular disease, and carotid artery disease. Matching was performed using the nearest neighbor algorithm that optimized the number of characteristics shared between groups.17
Health care utilization
Hospital outcomes included operating room time, need for intensive care monitoring, length of hospital stay, secondary intervention, and major adverse event (MAE). An MAE was defined as myocardial infarction, stroke, renal failure, respiratory failure, paralysis, or bowel ischemia. Secondary interventions included endovascular or open surgical reinterventions using the coding scheme of Edwards and colleagues.18 Perioperative outcomes occurring over 30 days postdischarge included MAE, secondary interventions, and readmissions for reasons other than secondary intervention.
Health care cost estimation
Health care utilization costs were obtained from Medicare Hospital Standard Analytic Files. Costs were estimated from a hospital perspective, a health care payer perspective, and a total health care utilization perspective. The hospital perspective included hospital utilization and costs. Hospital costs were estimated from hospital charges multiplied by the hospital’s cost-to-charge ratio. The health care payer perspective included perioperative utilization occurring over 30 days postdischarge, with costs adjusted for health care payer mix. Costs were adjusted to represent national averages by assuming a nationwide primary health care payer mix of 83% Medicare and 17% private payers. This was a reasonable approximation since Medicare and private payers account for 95% of the primary health care payers in elective EVAR cases.15 We estimated private payer costs by applying a private payer to Medicare payment ratio for cardiovascular procedures.19 The total health care utilization perspective was calculated as the sum of hospital costs and health care payer costs.
Data analysis
The cost of individual elements contributing to overall hospital cost was estimated by multivariable linear regression. Total hospital costs were regressed on hospital length of stay and indicator variables for intensive care monitoring, MAE, and secondary intervention. Residuals plots were assessed for evidence of non-linearity or bias. Costs were not adjusted for inflation since all cases in both groups were performed over a 20-month period. Probability parameters were modeled using a beta distribution and continuous parameters were modeled with a normal distribution. The private payer to Medicare payment ratio was modeled with a triangular distribution. For outcomes with no observed events, we used a beta distribution with mean of 0.1% to accommodate model requirements. Monte Carlo probabilistic sensitivity analysis with 10,000 iterations examined the effect of combined uncertainty across all model parameters. Additional sensitivity analyses were performed to assess the robustness of the base-case results. Cost models were developed using TreeAge Pro 2017 (TreeAge Software, Williamstown, MA).
Results
Patient characteristics
Before matching, we identified 10,429 patients electively treated with standard EVAR. Compared to the fast-track EVAR group, these patients were older with more comorbidities. Following matching, this study included 1000 patients treated with elective EVAR—250 with fast-track EVAR and 750 with standard EVAR. Baseline patient characteristics were well matched between groups, with exceptions of higher incidence of renal insufficiency (10% vs 0%) and congestive heart failure (4% vs 0%) in the fast-track EVAR group. Since creatinine >2.0 mg/dL, or New York Heart Association class III or IV were exclusion criteria in the fast-track EVAR study, patients with renal insufficiency and creatinine ≤2.0 mg/dL, or CHF classified as NYHA class I or II remained eligible. Therefore, we conservatively excluded patients with renal insufficiency and congestive heart failure from the matched standard EVAR group (Table 2). Mean aortic characteristics in the fast-track EVAR group included AAA diameter of 51±8 mm, proximal neck angle of 24±17 degrees, proximal neck length of 24±14 mm, and external iliac diameter of 7.4±1.7 mm. Aortic characteristics were not available in Medicare claims for the standard EVAR group.
 |
Table 2 Baseline characteristics of patients undergoing standard and fast-track EVARa |
Perioperative clinical outcomes
Hospital outcomes favored the fast-track EVAR group, including shorter operating room time (2.30 vs 2.83 hr, P<0.001), shorter hospital stay (1.16 vs 1.69 d, P<0.001), less need for intensive care monitoring (4.4% vs 48.0%, P<0.001), and lower secondary intervention rate (0% vs 2.4%, P=0.01). Postdischarge outcomes generally favored fast-track EVAR with a lower rate of MAE (0% vs 7.2%, P<0.001) and all-cause readmission (1.6% vs 6.8%, P=0.001). Utilization and cost estimates for all model parameters are reported in Table 3. The all-cause readmission rate in the fast-track EVAR group was 1.6% (4/250); readmissions were attributable to renal insufficiency (0.4% [1/250]), cardiomyopathy (0.4% [1/250]), diverticulosis (0.4% [1/250]), and chronic obstructive pulmonary disease (0.4% [1/250]). In matched patients undergoing standard EVAR, the all-cause readmission rate was 6.8% (51/750). The most common reasons for readmission in this group were classified as respiratory (1.5% [11/750]), rehabilitation (1.2% [9/750]), infection (0.7% [5/750]), gastrointestinal (0.7% [5/750]), cardiovascular (0.5% [4/750]), and urological (0.5% [4/750]). Thirty-day mortality was 0.3% with standard EVAR and 0.4% with fast-track EVAR.
 |
Table 3 Perioperative utilization and cost estimates in matched patients undergoing standard and fast-track EVARa |
Perioperative health care costs
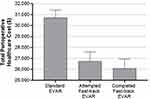
The total cost to the health care system over 30 days from discharge following standard EVAR was $30,730–$29,300 incurred by hospitals and $1430 incurred by health care payers. In the fast-track EVAR group, the total cost was $26,730–$26,320 incurred by hospitals and $410 incurred by health care payers. Ultimately, total health care costs were $4000 lower with fast-track EVAR vs standard EVAR, with $2980 in savings to hospitals and $1030 savings to health care payers. The primary factors contributing to these cost reductions were shorter hospital stay, shorter operating room time, lower rate of intensive care monitoring, and fewer postdischarge MAEs (Table 4). When accounting for the combined uncertainty in all model parameters using Monte Carlo simulations, the mean savings with fast-track EVAR remained $4000, with a 95% confidence interval ranging from $3130 to $4830 (Figure 1).
 |
Table 4 Perioperative cost per patient with standard and fast-track EVARa |
Sensitivity analyses
This study investigated the primary perioperative elements that may substantially influence costs with standard EVAR and fast-track EVAR. However, two elements of the fast-track EVAR program—no general anesthesia and bilateral percutaneous access—were not included in base-case cost models given the absence of these data within Medicare claims. Therefore, we performed sensitivity analyses to assess the potential impact of these exclusions. Regarding the choice of anesthesia, we assumed a $210 cost reduction with regional/local anesthesia vs general anesthesia (Premier Research Services, email communication, November, 2015). and that 81% of the standard EVAR patients underwent general anesthesia.20 Given that general anesthesia was used in 2.4% of the fast-track EVAR patients, inclusion of anesthesia type in the model would have yielded approximately $170 per patient in additional cost savings with fast-track EVAR. Regarding the choice of vascular access, the cost of vascular closure devices for bilateral PEVAR was estimated at $105012 and the cost of a cutdown tray and sutures was estimated at $390.21 Assuming 23% of the standard EVAR patients are treated with bilateral PEVAR22 vs 100% in the fast-track EVAR group, this would yield an additional expense of approximately $510 per patient with fast-track EVAR had vascular closure devices been modeled. Collectively, these data suggest that exclusion of anesthesia and vascular closure devices from the model would decrease the savings with fast-track EVAR relative to the base-case results by approximately 9%—from $4000 to $3660 per patient.
A second sensitivity analysis was performed to estimate the impact associated with the inability to match groups on renal insufficiency and congestive heart failure. For this analysis, we excluded 32 fast-track EVAR patients with renal insufficiency or congestive heart failure. When comparing 218 (87%) fast-track EVAR patients without renal insufficiency or congestive heart failure to standard EVAR patients, total health care cost savings with fast-track EVAR increased by approximately 8%—from $4000 to $4310 per patient.
Ultimately, the base-case cost results in this study ($4000 savings with fast-track EVAR) were robust to modeling assumptions. The additional $340 cost related to exclusion of anesthesia and vascular access data from the model was balanced by an additional cost savings of $310 when adjusting for the greater frequency of renal insufficiency and congestive heart failure in the fast-track EVAR group.
A final sensitivity analysis evaluated the 216 (86%) patients who completed all elements of the fast-track EVAR protocol (bilateral PEVAR, local/regional anesthesia, no intensive care monitoring, and next-day hospital discharge). In this group, total health care cost savings relative to standard EVAR were $4650, with $3580 in savings to hospitals and $1060 in savings to health care payers (Figure 2).
Discussion
The results of this study demonstrate that a fast-track EVAR protocol using a 14 Fr endograft in well-selected patients results in shorter procedure time, lower utilization of intensive care monitoring, faster discharge, lower incidence of MAE, and lower readmission rates compared to a matched sample undergoing standard EVAR. Total perioperative health care costs were $4000 lower with fast-track EVAR, with cost reductions of $2980 to hospitals and $1030 to health care payers. Considering that EVAR without major complication or comorbidity has been reported to result in $4000 in negative operating margins per case,2 adoption of a fast-track EVAR program may help to achieve revenue neutrality within this subset of patients.
Several surgical specialties have introduced short-stay protocols for certain procedures with the goal of reducing complication rates, facilitating postoperative recovery, expediting hospital discharge, and reducing early readmissions.23,24 Yet few studies have investigated the financial implications of such treatment pathways in EVAR. Moscato et al,12 reported a $2400 savings in hospital costs from a retrospective review of patients who required only uncomplicated monitoring with same-day discharge after EVAR. Lachat et al,11 reported a $2800 hospital cost savings in matched patients with outpatient EVAR vs inpatient EVAR. Al-Zuhir et al,9 reported hospital savings of $2500 in patients undergoing short-stay EVAR with next-day discharge. Importantly, none of these studies were performed in the US and with a requirement of an overnight stay. Since EVAR reimbursement requires inpatient admission at most private institutions in the US, the design of the current study attempted to assess cost savings with fast-track EVAR within the framework of current reimbursement policy within the US. Hospital cost savings of $2980 with fast-track EVAR in the current study were in line with these previous studies, and were primarily attributable to shorter length of stay, shorter procedure time, and lower intensive care monitoring requirements.
To the author’s knowledge, no fast-track EVAR study has examined costs following hospital discharge. The fast-track EVAR protocol in the current study resulted in a $1030 savings to health care payers over the first 30 days following discharge, largely due to fewer MAEs requiring readmission. Hospital readmission has become the focus of quality improvement efforts owing to the added cost placed on patients and the health care system. In the fast-track, EVAR group, mean hospital stay was 1.2 days and the readmission rate was 1.6%. This in comparison to 1.7 days hospital stay and 6.8% readmission rate with standard EVAR. These results suggest that the hospital benefits of a fast-track EVAR protocol, which includes early discharge, are not offset by higher risk of postdischarge complications and associated readmissions.
Strengths of this research are a large sample of 1000 EVAR patients, patient matching to adjust for group differences in baseline characteristics, determination of utility and cost of EVAR using nationally representative data adjusted for primary health care payer mix, and robustness of conclusions to various sensitivity analysis assumptions. There are several limitations of this study that warrant additional discussion. First, since the fast-track EVAR study followed patients through 1-month, we were unable to determine if clinical benefits and cost savings with fast-track EVAR persisted over longer follow-up. Second, all patients who underwent fast-track EVAR were treated with a 14 Fr endograft and, therefore, these results may not be generalizable to other stent grafts or to other fast-track pathways. While endograft costs account for 38% to 54% of the hospital EVAR costs,21,25,26 endograft cost was not included in this study since stent graft manufacturer data were unavailable in the standard EVAR group and that these costs vary by institution. Regardless, the results of this study are robust to endograft cost assumptions since the 14 Fr endograft for fast-track EVAR remains less costly vs standard EVAR up to $4000 in additional endograft cost. Third, data for the standard EVAR group were derived from Medicare claims. The fast-track and standard EVAR groups were comprised of patients undergoing elective EVAR with comparable demographic and medical history after patient matching. However, despite this matching, other important variables such as aortoiliac morphology were not available within Medicare claims. Such unmeasured variables may have influenced patient outcomes and, thus, are important sources of possible bias. Finally, and perhaps most importantly, fast-track EVAR is only intended for well-selected patients. Patients with heavily calcified or extremely tortuous femoral arteries may be poor candidates for bilateral percutaneous vascular access and patients with major comorbidities may require intensive care monitoring or prolonged hospitalization. While it is plausible that certain elements of the fast-track EVAR protocol could be customized based on individual patient characteristics, the clinical utility of such an individualized strategy has not been studied.
Conclusions
The results of this study demonstrate that a fast-track EVAR protocol using a 14 Fr endograft in well-selected patients results in shorter procedure time, lower utilization of intensive care monitoring, faster discharge, lower incidence of MAE, and lower readmission rates compared to a matched sample undergoing standard EVAR. Further, total perioperative health care costs were $4000 lower with fast-track EVAR, with cost reductions of $2980 to hospitals and $1030 to health care payers. Additional studies with longer follow-up are needed to confirm that fast-track EVAR continues to offer significant clinical and cost benefits.
Data sharing statement
Individual deidentified participant data and study-related documents will not be made available.
Acknowledgment
Endologix, Inc. provided financial support for this study. Endologix, Inc. is the sponsor of the LIFE registry and was involved in study design and study management.
Disclosure
ZK, VGR, EAH, and LEM disclose consultancy with Endologix, Inc. WKN reports personal fees from Trivascular, outside the submitted work. LHA reports personal fees from Miller Scientific Consulting, during the conduct of the study. The authors report no other conflicts of interest in this work.
References
1. Brown LC, Powell JT, Thompson SG, et al. The UK EndoVascular Aneurysm Repair (EVAR) trials: randomised trials of EVAR versus standard therapy. Health Technol Assess. 2012;16:1–218. doi:10.3310/hta16090
2. Stone DH, Horvath AJ, Goodney PP, et al. The financial implications of endovascular aneurysm repair in the cost containment era. J Vasc Surg. 2014;59:283–290, 290 e281. doi:10.1016/j.jvs.2013.08.047
3. Humphries MD, Suckow BD, Binks JT, McAdam-Marx C, Kraiss LW. Elective endovascular aortic aneurysm repair continues to cost more than open abdominal aortic aneurysm repair. Ann Vasc Surg. 2017;39:111–118. doi:10.1016/j.avsg.2016.05.091
4. Centers for Medicare & Medicaid Services. CMS manual system - pub 100-20 one-time notification - transmittal 1494; 2015. Available from: https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/downloads/R1494OTN.pdf.
5. Fourie C, Biller-Andorno N, Wild V. Systematically evaluating the impact of diagnosis-related groups (DRGs) on health care delivery: a matrix of ethical implications. Health Policy. 2014;115:157–164. doi:10.1016/j.healthpol.2013.11.014
6. McIlvennan CK, Eapen ZJ, Allen LA. Hospital readmissions reduction program. Circulation. 2015;131:1796–1803. doi:10.1161/CIRCULATIONAHA.114.010270
7. Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152:292–298. doi:10.1001/jamasurg.2016.4952
8. Abularrage CJ, Sheridan MJ, Mukherjee D. Endovascular versus “fast-track” abdominal aortic aneurysm repair. Vasc Endovascular Surg. 2005;39:229–236. doi:10.1177/153857440503900303
9. Al-Zuhir N, Wong J, Nammuni I, Curran G, Tang T, Varty K. Selection, thirty day outcome and costs for short stay endovascular aortic aneurysm repair (SEVAR). Eur J Vasc Endovasc Surg. 2012;43:662–665. doi:10.1016/j.ejvs.2012.02.031
10. Dosluoglu HH, Lall P, Blochle R, Harris LM, Dryjski ML. Ambulatory percutaneous endovascular abdominal aortic aneurysm repair. J Vasc Surg. 2014;59:58–64. doi:10.1016/j.jvs.2013.06.076
11. Lachat ML, Pecoraro F, Mayer D, et al. Outpatient endovascular aortic aneurysm repair: experience in 100 consecutive patients. Ann Surg. 2013;258:
12. Moscato VP, O’Brien-Irr MS, Dryjski ML, et al. Potential clinical feasibility and financial impact of same-day discharge in patients undergoing endovascular aortic repair for elective infrarenal aortic aneurysm. J Vasc Surg. 2015;62:855–861. doi:10.1016/j.jvs.2015.04.435
13. Krajcer Z, Ramaiah V, Huetter M. Fast-track endovascular aneurysm repair: rationale and design of the multicenter Least Invasive Fast-Track EVAR (LIFE) registry. BMC Cardiovasc Disord. 2015;15:174. doi:10.1186/s12872-015-0167-1
14. Krajcer Z, Ramaiah V, Henao E, et al. Perioperative outcomes from the prospective multicenter Least Invasive Fast-Track EVAR (LIFE) registry. J Endovasc Ther. 2018;25:6–13. doi:10.1177/1526602817747871
15. Agency for Healthcare Research and Quality. HCUP databases; 2011. Available from: www.hcup-us.ahrq.gov/databases.jsp.
16. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383.
17. Stuart EA. Matching methods for causal inference: a review and a look forward. Stat Sci. 2010;25:1–21. doi:10.1214/09-STS313
18. Edwards ST, Schermerhorn ML, O’Malley AJ, et al. Comparative effectiveness of endovascular versus open repair of ruptured abdominal aortic aneurysm in the Medicare population. J Vasc Surg. 2014;59:575–582. doi:10.1016/j.jvs.2013.08.093
19. America’s Health Insurance Plans. National comparisons of commercial and Medicare fee-for service payments to hospitals. 2016. Available from: https://www.ahip.org/wp-content/uploads/2016/02/HospitalPriceComparison_2.10.16.pdf.
20. Edwards MS, Andrews JS, Edwards AF, et al. Results of endovascular aortic aneurysm repair with general, regional, and local/monitored anesthesia care in the American College of Surgeons National Surgical Quality Improvement Program database. J Vasc Surg. 2011;54:1273–1282. doi:10.1016/j.jvs.2011.04.054
21. O’Brien-Irr MS, Harris LM, Dosluoglu HH, et al. Factors that affect cost and clinical outcome of endovascular aortic repair for abdominal aortic aneurysm. J Vasc Surg. 2017;65:997–1005. doi:10.1016/j.jvs.2016.08.090
22. Manunga JM, Gloviczki P, Oderich GS, et al. Femoral artery calcification as a determinant of success for percutaneous access for endovascular abdominal aortic aneurysm repair. J Vasc Surg. 2013;58:1208–1212. doi:10.1016/j.jvs.2013.05.028
23. Brustia P, Peinetti F. Fast track aortic surgery: why not? Adv Sci Health. 2014;1:9–18.
24. Brustia P, Renghi A, Aronici M, et al. Fast-track in abdominal aortic surgery: experience in over 1,000 patients. Ann Vasc Surg. 2015;29:1151–1159. doi:10.1016/j.avsg.2015.02.012
25. Lemmon GW, Neal D, DeMartino RR, et al. Variation in hospital costs and reimbursement for endovascular aneurysm repair: a vascular quality initiative pilot project. J Vasc Surg. 2017;66:1073–1082. doi:10.1016/j.jvs.2017.02.039
26. Matsumura JS, Stroupe KT, Lederle FA, Kyriakides TC, Ge L, Freischlag JA. Costs of repair of abdominal aortic aneurysm with different devices in a multicenter randomized trial. J Vasc Surg. 2015;61:59–65. doi:10.1016/j.jvs.2014.08.003
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.