Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Comparison of Characteristics Between ICS-Treated COPD Patients and ICS-Treated COPD Patients with Concomitant Asthma: A Study in Primary Care
Authors Savran O , Godtfredsen NS , Sørensen T, Jensen C , Ulrik CS
Received 9 December 2019
Accepted for publication 21 March 2020
Published 28 April 2020 Volume 2020:15 Pages 931—937
DOI https://doi.org/10.2147/COPD.S241561
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Osman Savran,1 Nina Skavlan Godtfredsen,1,2 Torben Sørensen,3 Christian Jensen,4 Charlotte Suppli Ulrik1,2
1Department of Respiratory Medicine, Hvidovre Hospital, Hvidovre, Denmark; 2Institute of Clinical Medicine, University of Copenhagen, Copenhagen,Denmark; 3Værløse Lægehus, Værløse, Denmark; 4Lægehuset Remisen, Præstø, Denmark
Correspondence: Charlotte Suppli Ulrik
Respiratory Research Unit, Department of Respiratory Medicine, Hvidovre Hospital, Hvidovre DK-2650, Denmark
Email [email protected]
Background and Objective: Inhaled corticosteroids (ICS) for COPD has been much debated. Our aim was to identify characteristics associated with prescribing ICS for patients with COPD alone compared to those with concomitant asthma in general practice.
Patients and Methods: Participating general practitioners (GPs) (n=144) recruited patients with COPD (ICPC 2nd ed. code R95) currently prescribed ICS (ACT code R03AK and R03BA). Data, if available, on demographics, smoking habits, spirometry, COPD medication, dyspnea score, and exacerbation history were retrieved from the medical records. Logistic regression analysis was used to identify possible differences in characteristics between patients with COPD alone compared to those having a concomitant diagnosis of asthma.
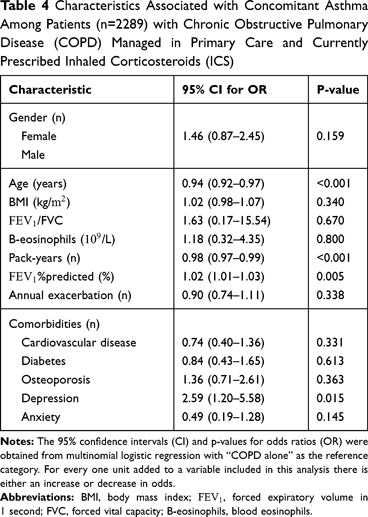
Results: A total of 2.289 (45% males) COPD patients on ICS were recruited. Compared to patients with COPD alone (n=1.749), those with COPD and concomitant asthma (n=540) were younger (p< 0.001), had higher BMI, higher FEV1/FVC ratio, higher blood eosinophil count and less life-time tobacco exposure (36 and 26 pack-years, respectively). Compared to COPD alone, logistic regression analysis showed that COPD with concomitant asthma was significantly associated to age (OR 0.94; CI 0.92 to 0.97; p< 0.001), pack-years of smoking (OR 0.98; CI 0.97 to 0.99; p< 0.001), %pred (OR 1.02; CI 1.00 to 1.03; p=0.005), and doctor-diagnosed depression (OR 2.59; CI 1.20 to 5.58; p=0.015).
Conclusion: In COPD patients currently prescribed ICS, the presence of concomitant asthma was associated with being younger, having less tobacco exposure, more preserved lung function and a higher likelihood of doctor-diagnosed depression compared to COPD alone.
Keywords: COPD, concomitant asthma, inhaled corticosteroids (ICS), general practice
Introduction
Long-acting bronchodilators are the cornerstone of pharmacological treatment of chronic obstructive pulmonary disease (COPD).1 However, inhaled corticosteroids (ICS) have been shown to reduce exacerbations, hospitalizations and have also been linked to a reduction in mortality in COPD patients.2–5 Global Initiative of Chronic Obstructive Lung Disease (GOLD) has recommended using ICS in patients with severe airflow limitation and/or patients at high risk of COPD exacerbations.1 However, there is evidence of widespread use of ICS in patients with less severe COPD not fulfilling the GOLD criteria for ICS use.6–8
A meta-analysis of available clinical trials suggested that ICS treatment in patients with COPD may reduce the annual decline in lung function.2 Furthermore, improvements in health status has been demonstrated in COPD patients prescribed monotherapy with LABA or LAMA,9,10 but add-on ICS to long-acting bronchodilator treatment has been demonstrated to improve health status even further.11
ICS-therapy may be associated with potentially severe side effects as studies have demonstrated an increased risk of fractures and pneumonia.12–14 Furthermore, the distinction between side effects such as pneumonia and symptoms is difficult to define in a primary care setting.15 The adverse effects of ICS treatment in COPD patients points to the importance of more research on the risk-benefit profile of ICS.16,17 It is therefore crucial to understand what determines the use of ICS in COPD patients.
In recent years there has been an increasing interest in understanding the concomitant diagnosis of asthma in COPD patients and distinguishing clinical features associated with either COPD or asthma in patients presenting with obstructive lung diseases.18,19 This has given rise to treatment recommendations for COPD patients with concomitant asthma.20 However, there is no consensus on a definition of COPD patients with a concomitant asthma diagnosis, and it has proven challenging to properly characterize these patients. In a large cohort of patients with COPD managed in primary care and currently prescribed inhaled corticosteroids, our aim was to identify possible differences in characteristics associated with prescribing ICS for patients diagnosed with COPD alone and those with concomitant asthma.
Patients and Methods
All study participants had a confirmed diagnosis of COPD, were currently prescribed ICS (coded as International Classification of Primary Care, 2nd ed. code R95 in electronic patient journals and with the ACT code R03AK and R03BA, indicating ICS treatment), and were managed by general practitioners (GPs) in primary care, and for some patients, also specialists. The selection process of patients participating in the study is summarized in Figure 1, also illustrating the primary focus of the present study. Further details related to the selection procedure has been published previously.21 Participating GPs provided information, if available, on demographics, clinical characteristics, blood eosinophils, consultations for respiratory disease, prescribed medication, hospital admissions for COPD, previous COPD exacerbations, smoking status, selected comorbidities, and duration of COPD for all recruited patients. Each general practice included up to a maximum of 20 randomly selected patients. All patients were assigned a unique project ID number to assure anonymity with only the designated GP being unblinded to patient identity.
The study was approved by the Danish Data Protection Agency. The present study was a non-interventional, non-drug study, and, therefore, it was not mandatory to obtain approval from the ethical committee and the Danish Medicines Agency.
Assessment of Symptoms, Airflow Limitation and Acute Exacerbations
Severity of symptoms was assessed by the Medical Research Council (MRC) scale22 and/or the COPD assessment test (CAT). Spirometric findings were used to estimate airflow obstruction and categorize the severity of the disease, based on a cut-off for 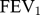 according to the classification provided by GOLD.23 Patients were also classified into four groups, i.e., A, B, C and D, according to the GOLD 2017 classification, based on COPD symptoms (MRC-scale score
according to the classification provided by GOLD.23 Patients were also classified into four groups, i.e., A, B, C and D, according to the GOLD 2017 classification, based on COPD symptoms (MRC-scale score 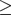 2 and/or CAT score
2 and/or CAT score  10) and the exacerbation history.
10) and the exacerbation history.
Data Analysis
Data are reported as mean values ± one standard deviation (SD). Clinical variables and characteristics of COPD alone were compared to COPD with concomitant asthma using independent sample t-test for continuous variables. The comparison between COPD alone and COPD patients with a concomitant asthma diagnosis in terms of possible clinically-relevant variables and characteristics was conducted using a multinomial logistic regression model and reported as odds ratios and 95% confidence intervals. A p-value <0.05 was considered statistically significant. Data were analyzed using the statistical program IBM SPSS version 25 (IBM Corporation, Armonk, NY, USA).
Results
Characteristics of Enrolled Patients with COPD
Of the COPD patients currently on ICS therapy (n=2289) included in the present study, 540 (24%) were classified by the GP as having concomitant asthma.
Slightly more female than male patients were recruited (55% vs. 45%), whereas no difference was found in mean age between sexes (71 years; p=0.83). Further characteristics of the patients are given in Table 1.
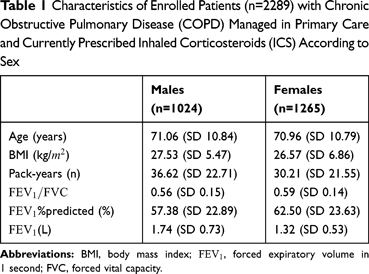 |
Table 1 Characteristics of Enrolled Patients (n=2289) with Chronic Obstructive Pulmonary Disease (COPD) Managed in Primary Care and Currently Prescribed Inhaled Corticosteroids (ICS) According to Sex |
COPD Alone and COPD Patients with a Concomitant Asthma Diagnosis
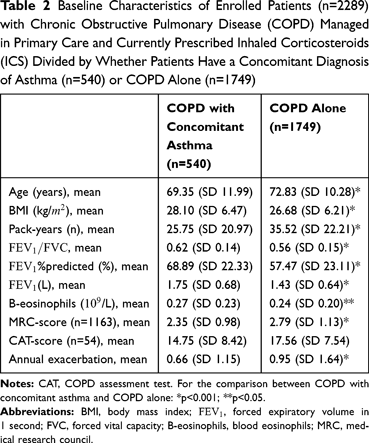
Baseline characteristics of the two groups, i.e., COPD alone and COPD with concomitant asthma, are outlined in Table 2. A statistically significant difference was found in mean MRC for COPD alone and for COPD patients with concomitant asthma (p<0.001). The mean number of pack years for COPD alone patients and for COPD patients with concomitant asthma, respectively, was 36 and 26 (p<0.001). COPD patients with concomitant asthma had higher blood-eosinophil count than patients with COPD alone (p=0.02). Further details are given in Table 2.
The distribution of severity of airflow limitation, based on the GOLD classification, among patients with COPD alone and COPD with concomitant asthma is presented in Figure 2, revealing a significant difference between the two groups (p<0.001). The distribution of the most common comorbidities and levels of blood eosinophils, respectively, in patients with COPD alone and COPD with concomitant asthma are presented in Figure 3 and Table 3.
 |
Figure 3 Comorbidities in patients with chronic obstructive pulmonary disease (COPD) alone compared to COPD patients with concomitant asthma. |
Compared to COPD alone, logistic regression analysis showed that COPD with concomitant asthma was significantly associated to age (OR 0.94; CI 0.92 to 0.97; p<0.001), pack-years of smoking (OR 0.98; CI 0.97 to 0.99; p<0.001), 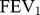 %pred (OR 1.02; CI 1.00 to 1.03; p=0.005), and doctor-diagnosed depression (OR 2.59; CI 1.20 to 5.58; p=0.015) (Table 4).
%pred (OR 1.02; CI 1.00 to 1.03; p=0.005), and doctor-diagnosed depression (OR 2.59; CI 1.20 to 5.58; p=0.015) (Table 4).
Discussion
This study shows that a concomitant asthma diagnosis in COPD patients treated with ICS in primary care is associated with younger age, less airflow limitation, but more likely to be associated with a doctor-diagnosis of depression compared to patients with COPD alone.
Reduced benefit from corticosteroids and increased inflammation in the airways has previously been suggested as a side effect of being a current smoker in patients with chronic airway diseases such as COPD and asthma.24 On the other hand, a recent study analyzing data from three randomized controlled trials of budesonide-formoterol studies in COPD patients concluded that currently smoking COPD patients on ICS/LABA therapy had reduced exacerbation rates compared to those on LABA alone treatment at greater blood-eosinophil counts.25 In line with this, the present study found that COPD patients with concomitant asthma and on average higher mean blood eosinophil count had a lower exacerbation rate compared to patients with COPD alone and a lower blood eosinophil count indicating that ICS is more beneficial in the COPD with concomitant asthma group of patients. COPD patients with concomitant asthma also had higher lung function, however, this might be due to the fewer pack-years smoked.
A quarter of a cohort of COPD patients have asthma characteristics and according to the strategy document by GOLD might be eligible for ICS treatment.20 In keeping with these facts, the respective GPs in this study have rightfully continued ICS prescription because of concomitant asthma in a quarter of included patients. Continued ICS prescription might also depend on whether patients presented with airflow limitation and/or severe symptoms, although these predictors alone should not determine ICS prescription according to the strategy document by GOLD.20
In this study, classification of participants to have COPD with concomitant asthma was based on the participating GPs interpretation of the overall case history, objective findings and response to therapy. As this was a non-interventional study, no formal criteria for a concomitant diagnosis of asthma was applied, which may be the background for the observation that the proportion of COPD patients in the present study with a concomitant diagnosis of asthma was a little higher compared to what has been reported from other studies. However, on the other hand, all patients in the present study were currently prescribed ICS and by that, it may be expected to observe a slightly higher proportion of patients classified as having concomitant asthma.
Similar to the present study, other studies have previously found higher prevalence of depressive symptoms among patients with chronic airway disease such as asthma.26,27 Another study also found prevalence of depression to be greater among patients with uncontrolled asthma compared to those with well-controlled asthma, suggesting that treatment is crucial in depression prevalence in chronic airway disease.28 In addition, another recent study found that the risk of depression is higher in patients presenting with symptoms of both asthma and COPD.29
Our study has strengths worth mentioning. First, we included a large cohort based on a real-world population of COPD patients followed by their designated GPs. This reflects information gathered by day-to-day clinical visits of patients in general practices and follow-up by GPs, including information on hospital admissions and respiratory outpatient visits. Additionally, plenty of data was available from this large population-based cohort, which at first sight makes it exceptionally relevant in finding different significances among different subsets of included COPD patients.
Certain limitations should be emphasized in the current study. The results presented here might resemble random significant associations between clinical variables and characteristics in subpopulations of COPD patients treated with ICS in primary care. We found a significant association between depression and concomitant asthma, which has also been reported in previous studies of the so-called asthma-COPD overlap. As shown in Table 4, the confidence interval for the association was wide, but based on previous findings, also in patient with more severe asthma, the overserved association in the present study is likely to be valid.
On the other hand, some of the significant associations found in this study might have been influenced by a possible lack of a thorough examination and diagnosis of COPD patients in general practice leading to less clinical important differences and findings between patients with COPD alone and COPD with concomitant asthma. In line with this, patients with COPD alone may have a history with more exacerbations primarily due to having more severe disease (Figure 2). Lastly, very few patients had information on CAT-score (2.4%), which might have made it difficult to demonstrate genuinely significant difference in symptoms amongst patients with COPD alone and COPD patients with concomitant asthma. Meanwhile, almost all patients had information on exacerbation rate (99.3%) and all had information on whether they had a concomitant diagnosis of asthma. This reflects a necessity for further use of symptom scores as part of the assessment of COPD patients in primary care.
In the current study investigating COPD patients prescribed ICS in primary care, a diagnosis of concomitant asthma indicated higher risk of depression, less exposure to tobacco smoke, and greater lung function. These main differences between patients with COPD alone and COPD with concomitant asthma indicate the need for investigation of detailed treatment strategies in primary care COPD patients, not least for those with concomitant asthma, and hopefully, reduce risk of unbeneficial ICS treatment in the future.
Disclosure
Dr Christian Jensen reports personal fees from Boehringer Ingelheim, personal fees from Astra Zeneca, personal fees from Novo Nordisk, and personal fees from Novartis, during the conduct of the study. The authors report no other conflicts of interest in this work.
References
1. Global strategy for the diagnosis, management and prevention of COPD, global initiative for chronic obstructive lung disease (GOLD); 2018. Available from: http://goldcopd.org/gold-reports/.
2. Yang IA, Clarke MS, Sim EH, Fong KM. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012;7:Cd002991.
3. Calverley PM, Anderson JA, Celli B, et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356(8):775–789. doi:10.1056/NEJMoa063070
4. Nannini LJ, Lasserson TJ, Poole P. Combined corticosteroid and long-acting beta(2)-agonist in one inhaler versus long-acting beta(2)-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012;9:Cd006829.
5. Nannini LJ, Poole P, Milan SJ, Kesterton A. Combined corticosteroid and long-acting beta(2)-agonist in one inhaler versus inhaled corticosteroids alone for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2013;8:Cd006826.
6. Bourbeau J, Sebaldt RJ, Day A, et al. Practice patterns in the management of chronic obstructive pulmonary disease in primary practice: the CAGE study. Can Respir J. 2008;15(1):13–19. doi:10.1155/2008/173904
7. Fitch K, Iwasaki K, Pyenson B, et al. Variation in adherence with global initiative for chronic obstructive lung disease (GOLD) drug therapy guidelines: a retrospective actuarial claims data analysis. Curr Med Res Opin. 2011;27(7):1425–1429. doi:10.1185/03007995.2011.583230
8. Lucas AE, Smeenk F, Smeele I, et al. Overtreatment with inhaled corticosteroids and diagnostic problems in primary care patients, an exploratory study. Fam Pract. 2008;25(2):86–91. doi:10.1093/fampra/cmn006
9. Dahl R, Chung KF, Buhl R, et al. Efficacy of a new once-daily long-acting inhaled beta2-agonist indacaterol versus twice-daily formoterol in COPD. Thorax. 2010;65(6):473–479. doi:10.1136/thx.2009.125435
10. Jones PW, Mahler DA, Gale R, et al. Profiling the effects of indacaterol on dyspnoea and health status in patients with COPD. Respir Med. 2011;105(6):892–899. doi:10.1016/j.rmed.2011.02.013
11. Wedzicha JA, Calverley PMA, Seemungal TA, et al. The prevention of chronic obstructive pulmonary disease exacerbations by salmeterol/fluticasone propionate or tiotropium bromide. Am J Respir Crit Care Med. 2008;177(1):19–26. doi:10.1164/rccm.200707-973OC
12. Janson C, Larsson K, Lisspers KH, et al. Pneumonia and pneumonia related mortality in patients with COPD treated with fixed combinations of inhaled corticosteroid and long acting beta2 agonist: observational matched cohort study (PATHOS). BMJ. 2013;346:f3306. doi:10.1136/bmj.f3306
13. Gonzalez AV, Coulombe J, Ernst P, et al. Long-term use of inhaled corticosteroids in COPD and the risk of fracture. Chest. 2018;153(2):321–328. doi:10.1016/j.chest.2017.07.002
14. Price DB, Russell R, Mares R, et al. Metabolic effects associated with ICS in patients with COPD and comorbid type 2 diabetes: a historical matched cohort study. PLoS One. 2016;11(9):e0162903. doi:10.1371/journal.pone.0162903
15. Chalmers JD, Tebboth A, Gayle A, et al. Determinants of initial inhaled corticosteroid use in patients with GOLD A/B COPD: a retrospective study of UK general practice. NPJ Prim Care Respir Med. 2017;27(1):43. doi:10.1038/s41533-017-0040-z
16. Vestbo J, Vogelmeier C, Small M, et al. Understanding the GOLD 2011 Strategy as applied to a real-world COPD population. Respir Med. 2014;108(5):729–736. doi:10.1016/j.rmed.2014.03.002
17. Suissa S, Barnes PJ. Inhaled corticosteroids in COPD: the case against. Eur Respir J. 2009;34(1):13–16. doi:10.1183/09031936.00190908
18. Bateman ED, Reddel HK, van Zyl-smit RN, et al. The asthma-COPD overlap syndrome: towards a revised taxonomy of chronic airways diseases? Lancet Respir Med. 2015;3(9):719–728. doi:10.1016/S2213-2600(15)00254-4
19. Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med. 2015;373(13):1241–1249. doi:10.1056/NEJMra1411863
20. Global strategy for the diagnosis, management and prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD); 2019. Available from: https://goldcopd.org/wp-content/uploads/2018/11/GOLD-2019-v1.6-FINAL-08Nov2018-wms.pdf.
21. Savran O, Godtfredsen N, Sørensen T, et al. COPD patients prescribed inhaled corticosteroid in general practice: based on disease characteristics according to guidelines? Chron Respir Dis. 2019;16:1479973119867949. doi:10.1177/1479973119867949
22. Bestall JC, Paul EA, Garrod R, et al. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax. 1999;54(7):581–586. doi:10.1136/thx.54.7.581
23. Global strategy for the diagnosis, management and prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD); 2017. Available from: http://goldcopd.org.
24. Tamimi A, Serdarevic D, Hanania NA. The effects of cigarette smoke on airway inflammation in asthma and COPD: therapeutic implications. Respir Med. 2012;106(3):319–328. doi:10.1016/j.rmed.2011.11.003
25. Bafadhel M, Peterson S, De Blas MA, et al. Predictors of exacerbation risk and response to budesonide in patients with chronic obstructive pulmonary disease: a post-hoc analysis of three randomised trials. Lancet Respir Med. 2018;6(2):117–126. doi:10.1016/S2213-2600(18)30006-7
26. Dyer CA, Sinclair AJ. A hospital-based case-control study of quality of life in older asthmatics. Eur Respir J. 1997;10(2):337–341. doi:10.1183/09031936.97.10020337
27. Seigel WM, Golden NH, Gough JW, et al. Depression, self-esteem, and life events in adolescents with chronic diseases. J Adolesc Health Care. 1990;11(6):501–504. doi:10.1016/0197-0070(90)90110-N
28. Woledesenbet MA, Shumet Mekonen S, Sori LM, et al. Epidemiology of depression and associated factors among asthma patients in Addis Ababa, Ethiopia. Psychiatry J. 2018;2018:5934872. doi:10.1155/2018/5934872
29. Yeh JJ, Lin C-L, Hsu W-H, et al. The relationship of depression in asthma-chronic obstructive pulmonary disease overlap syndrome. PLoS One. 2017;12(12):e0188017. doi:10.1371/journal.pone.0188017
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.