Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 11 » Issue 1
Comorbidities associated with COPD in the Middle East and North Africa region: association with severity and exacerbations
Authors MAHBOUB B , Al-Zaabi A, Iqbal M , Salhi H, Lahlou A, Tariq L, El Hasnaoui A
Received 17 June 2015
Accepted for publication 7 November 2015
Published 5 February 2016 Volume 2016:11(1) Pages 273—280
DOI https://doi.org/10.2147/COPD.S90626
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Bassam Mahboub,1 Ashraf Alzaabi,2 Mohammed Nizam Iqbal,3 Hocine Salhi,4 Aïcha Lahlou,5 Luqman Tariq,6 Abdelkader El Hasnaoui6
1Department of Pulmonary Medicine and Allergy, University of Sharjah, Sharjah, 2Respirology Division, Zayed Military Hospital, Abu Dhabi, 3Department of Pulmonary Medicine, Rashid Hospital, Dubai Health Authority, Dubai, United Arab Emirates; 4Foxymed, Paris, France; 5MS Health, Rabat, Morocco, 6GlaxoSmithKline, Dubai, United Arab Emirates
Objective: To assess the frequency of comorbidities in subjects with COPD and their association with respiratory symptom severity and COPD exacerbations.
Materials and methods: This was an analysis of the BREATHE study, a cross-sectional survey of COPD conducted in the general population of eleven countries in the Middle East and North Africa, including Pakistan. The study population consisted of a sample of subjects with COPD for whom the presence of comorbidities was documented. Three questionnaires were used. The screening questionnaire identified subjects who fulfilled an epidemiological case definition of COPD and documented any potential comorbidities; the detailed COPD questionnaire collected data on respiratory symptoms, COPD exacerbations, and comorbidities associated with COPD; the COPD Assessment Test collected data on the impact of respiratory symptoms on well-being and daily life.
Results: A total of 2,187 subjects were positively screened for COPD, of whom 1,392 completed the detailed COPD questionnaire. COPD subjects were more likely to report comorbidities (55.2%) than subjects without COPD (39.1%, P<0.0001), most frequently cardiovascular diseases. In subjects who screened positively for COPD, the presence of comorbidities was significantly (P=0.03) associated with a COPD Assessment Test score ≥10 and with antecedents of COPD exacerbations in the previous 6 months (P=0.03).
Conclusion: Comorbidities are frequent in COPD and associated with more severe respiratory symptoms. This highlights the importance of identification and appropriate management of comorbidities in all subjects with a diagnosis of COPD.
Keywords: comorbidity, COPD exacerbation, diabetes, cardiovascular disease, MENA region
Introduction
Comorbidities are frequent in subjects with COPD,1 and the incidence of these has been assessed in many studies.2–4 In a large study of 30,000 COPD patients in the UK, it was estimated that COPD patients presented a fivefold-higher risk of cardiovascular diseases, a threefold-higher risk of stroke, and a twofold-higher risk of diabetes.3 Given that the prevalence of both COPD and its principal comorbidities, such as cardiovascular diseases or diabetes, are rising around the world,5,6 the risk of coexistence of these chronic diseases is expected to increase. It is now recognized that the presence of comorbidities in COPD patients has a major impact on quality of life and on mortality.7 The majority of data regarding the burden of comorbidities associated with COPD has come from industrialized countries, where it has been shown that COPD comorbidities have an important impact on burden of disease and represent an important driver of cost.8,9
In the Middle East and North Africa (MENA), the prevalence of COPD has been estimated at around 4% of adults aged over 40 years in the general population.10 This prevalence is expected to grow rapidly, since smoking rates in the region are generally high and rising.11,12 In addition, the principal COPD comorbidities are also frequent in this region, the prevalence of diabetes in the MENA region being one of the highest in the world (11%).13,14 Furthermore, it was reported in 2000 that six of the ten countries with the highest prevalence of diabetes in the world are in this region. In the first decade of this century, the prevalence of diabetes in Saudi Arabia increased from 10% in 1999 to 30% in 2009,15 whereas the overall worldwide prevalence was estimated to be only 2.8% in 2000.16
Data on the prevalence and the impact of COPD-related comorbidities are limited in the MENA region. BREATHE was an international study conducted in ten countries of the MENA region and in Pakistan with the aim of estimating the prevalence of COPD symptoms in the general population, and of describing smoking habits, management, and burden of COPD and related health care utilization.17 The analysis described here addresses the presence of comorbidities in subjects with COPD identified in the BREATHE database. The objective of this analysis was to assess the frequency of comorbidities in subjects with COPD and their association with respiratory symptom severity and COPD exacerbations.
Materials and methods
This analysis was performed on data from the BREATHE study, the methodology of which has been described in detail elsewhere.17 Briefly, this study was a cross-sectional epidemiological survey of COPD conducted in a random sample of the general population of eleven countries (Algeria, Egypt, Jordan, Lebanon, Morocco, Pakistan, Saudi Arabia, Syria, Tunisia, Turkey, and the United Arab Emirates [UAE]) between June 2010 and December 2011. The present analysis concerned data related to comorbidities associated with COPD.
Study procedures and data collection
This study was based on the population analyzed in the BREATHE study,17 a population-based sample aged at least 40 years contacted by telephone. The overall response rate, defined as the ratio between the number of subjects who completed the screening questionnaire and the number of potentially eligible subjects, was 74.2%.
Three questionnaires were used in the study. The screening questionnaire, completed by 62,086 subjects,17 collected data on demographics, respiratory symptoms, smoking habits, and the presence of comorbidities. The objective of the screening questionnaire was to identify subjects who fulfilled an epidemiological case definition of COPD.10 In this epidemiological definition, COPD cases were defined as eligible subjects fulfilling both of the following criteria:
- either: 1) diagnosis criterion – already diagnosed with COPD, emphysema, or chronic bronchitis; or 2) symptom criterion – presenting either with symptoms that fulfill the definition of chronic bronchitis or with dyspnea.
- smoking criterion: lifetime smoking exposure of ≥10 pack-years.
All subjects positively screened for COPD were included in the present analysis.
The second questionnaire consisted of a detailed COPD questionnaire that was administered only to subjects screened positively for COPD (n=2,187), and collected data on symptom severity, COPD exacerbations, and comorbidities. A total of 1,392 subjects with COPD completed this questionnaire.18 The characteristics of this population were similar to those screened positively for COPD (n=2,187 subjects) in terms of demographics and COPD symptoms.18
Data relating to comorbidities were documented from replies to the following two questions of the detailed questionnaire.
- Aside from your respiratory condition, do you suffer from any other serious or chronic health conditions?
- Yes
- No
- If yes, what other serious or chronic health conditions do you have?
- Arthritis
- Asthma
- Cancer or tumor
- Depression
- Diabetes
- Cardiovascular disease
- High blood pressure/hypertension
- Other (specify)
The third questionnaire, which consisted of the COPD Assessment Test (CAT),18,19 was administered to subjects with COPD who had completed the detailed questionnaire.19 The CAT documents the impact of COPD on subjects’ well-being and daily life. A score <10 indicates a low impact, a score of 10–19 a medium impact, a score of 20–40 a high to very high impact. The scores were then crossed with the presence of comorbidities. The CAT questionnaire was completed by 1,015 of the 1,392 subjects completing the detailed COPD questionnaire (72.9%).
The presence of exacerbations was assessed over a period of 6 months. Since the number of exacerbations was not documented in the BREATHE study, the GOLD (Global initiative for chronic Obstructive Lung Disease) classification was adapted according to exacerbation history (yes or no). The definition of exacerbation used in the BREATHE study has been described elsewhere.18 An exacerbation was considered to have occurred in the previous 6 months if the subject fulfilled any of the following criteria:
- In the last 6 months, have you been told by your physician that you have had worsening of your “respiratory condition”? Yes
- In the last 6 months, have you been told by your physician that you have had acute bronchitis? Yes
- In the last 6 months, what aspects of your “respiratory condition” have worsened? (Cough during the daytime, cough during the night, phlegm, breathlessness or shortness of breath and fatigue, ability to perform regular activities): at least two symptoms cited.
The CAT score and exacerbation history were used to classify subjects with COPD into four groups depending on their symptoms and risk of exacerbations, according to the recommendations of the 2011 GOLD report.1 Group A was low risk (no exacerbations) and fewer symptoms (CAT score <10), group B low risk and more symptoms (CAT ≥10), group C to high risk (one or more exacerbations) and fewer symptoms, and Group D high risk and more symptoms.
Data analysis
The data presented in this study are principally descriptive. The occurrence of comorbidities was compared between subjects completing the screening questionnaire who fulfilled the criteria for COPD (n=2,187) and those that did not fulfill these criteria (n=59,707). Adjusted odds ratios were calculated controlling for potential confounding by age and sex using logistic regression analysis. The association between comorbidities on the one hand and occurrence of exacerbations, severity of COPD symptoms, and GOLD severity groups on the other was evaluated in COPD subjects who had completed the detailed COPD questionnaire (n=1,392). Study variables were compared using the χ2 test and the Mantel–Haenszel test as appropriate. All statistical tests were two-tailed, with a probability threshold of 0.05 taken as the upper boundary for significance. Analyses were performed using SPSS version 17 (SPSS Inc, Chicago, IL, USA).
Ethics
The study was conducted according to the principles of Good Epidemiological Practices. Participating subjects did not receive any financial compensation for their participation in the study. Because the survey had no repercussions on medical practice, ethics committee authorization was not required. All data collected were kept confidential and anonymous. Written informed consent was not obtained from participants as this was a telephone survey in the general population.
Study responsibilities
This study was initiated and funded by GlaxoSmithKline (the study sponsor). The study sponsor enlisted an academic steering committee to advise on the design and implementation of the study and on the analysis and interpretation of the results, for which the committee members received consultancy fees from the study sponsor. The committee members had full access to the study data, and were actively involved in the preparation of the present article. Operational management of the study and data analyses was delegated by the study sponsor to MS Health (Rabat, Morocco), an independent contract-research organization. The study sponsor funded editorial support from a medical writing agency (Foxymed, Paris, France) for the preparation of the present article.
Results
The analysis was performed on data pooled from all eleven participating countries.
Study sample
A total of 2,187 subjects fulfilled the epidemiological case definition of COPD (COPD population), of whom 1,392 completed the detailed COPD questionnaire. The latter population (n=1,392) represents the principal study population for the present analysis. The demographic characteristics of subjects screened positively or negatively for COPD are presented in Table 1. Subjects with COPD were on average older and more likely to be male than subjects without COPD.
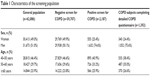  | Table 1 Characteristics of the screening population |
Distribution of comorbidities
The distribution of comorbidities among subjects screened positively or negatively for COPD is presented in Table 2. A total of 29,272 subjects in the screening population (47.1%) reported at least one comorbidity. The most frequently reported comorbidity was cardiovascular disease, followed by diabetes. The presence of all comorbidities was more frequent in women than in men, and notably for cardiovascular disorders, reported in 28.8% of all women and 18.3% of all men. In both men and women, the frequency of reported comorbidities increased with age.
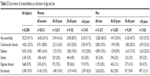  | Table 2 Occurrence of comorbidities as a function of age and sex |
Comorbidities were significantly (P<0.0001) more frequently reported in subjects screened positively for COPD (66.2%) than in controls who did not (46.5%). This corresponds to an odds ratio, adjusted for age and sex, of 2.63 (95% confidence interval 2.40–2.89). All individual comorbidities documented were also more frequent in the subjects screened positively for COPD (Table 3). Comorbidities were reported by 66.8% of COPD subjects who completed the detailed COPD questionnaire (930/1,392 subjects).
Presence of comorbidities according to CAT score
Of the COPD subjects who completed the detailed COPD questionnaire, 1,015 subjects provided CAT scores (72.9%). The majority of subjects (77.0%, n=782) reported a CAT score ≥10, indicating a high impact of respiratory symptoms on subject functioning. The distribution of comorbidities according to CAT score is presented in Figure 1. The probability of reporting a comorbidity was significantly (P=0.03) higher in subjects with a CAT score ≥10 compared to those with a CAT score <10.
  | Figure 1 Presence of comorbidities according to CAT scores. |
Presence of comorbidities according to the occurence of exacerbations
Of the 1,392 subjects with COPD who completed the detailed COPD questionnaire, a total of 661 (47.5%) subjects reported COPD exacerbations (Table 4). The association between the presence of comorbidities and the occurrence of exacerbations was assessed (Table 4). Overall, the proportion of subjects who reported exacerbations was significantly higher in the group of subjects with documented comorbidities than in those without (50.4% versus 44.6%, P=0.03 [χ2 test]). With respect to individual comorbidities, this association was no longer significant, except for asthma (P=0.003 [χ2 test]) and digestive diseases (P=0.02 [χ2 test]).
  | Table 4 Presence of comorbidities in COPD subjects with or without exacerbation |
Presence of comorbidities according to severity of symptoms and exacerbation history
A total of 1,015 subjects were classified into GOLD severity groups according to their CAT score and exacerbation history. Overall, half of the assessed population (n=549, 54.1%) were classified into group D, consisting of subjects with severe symptoms (CAT score ≥10) and a high risk of exacerbation (one or more exacerbations within the last 6 months of the study). The distribution of subjects with comorbidities (n=518) according to severity group is presented in Table 5. The majority of subjects with comorbidities (57.8%, n=299) were classified into the group with severe symptoms and high risk of exacerbation (group D). However, the distribution of subjects between GOLD severity grades did not differ significantly between those with comorbidities and those without comorbidities (P=0.08 [χ2 test]).
  | Table 5 Classification of subjects with or without comorbidities according to CAT score and exacerbation history |
The relationship between the presence of comorbidities on the one hand and the severity of COPD or the occurrence of exacerbation on the other was evaluated in the population of subjects classified according to GOLD severity group (n=1,015). The results are presented in Figure 2. The presence of comorbidities in COPD subjects was significantly associated with a high CAT score (≥10) (P=0.02) and GOLD group D (P=0.04).
  | Figure 2 Relationship between comorbidities, severity of COPD, and occurrence of COPD exacerbation. |
Discussion
This analysis of the BREATHE study revealed that two-thirds of COPD subjects reported comorbidities, principally cardiovascular diseases and diabetes, a significantly higher proportion than in subjects without COPD. The presence of comorbidities was significantly associated with COPD exacerbations and a higher impact of respiratory symptoms on functioning. The majority of subjects (57.8%, n=299) with comorbidities were assigned to group D of the GOLD severity classification, the most severe grade.
The coexistence of COPD and cardiovascular diseases or diabetes found in our study has been reported in several previous studies.2–4,20,21 A recent review of 25 studies reporting cardiovascular comorbidity in COPD subjects revealed that the relative risk of occurrence of cardiovascular comorbidities among COPD subjects ranged from 2.1 to 5.0 compared to subjects without COPD.4 This strong association between COPD and cardiovascular diseases may be explained by the fact that tobacco smoking is an important common risk factor for both COPD and cardiovascular diseases.22 In our study, the highest rate of COPD subjects reporting cardiovascular disease and diabetes was found in the UAE (71.4% and 50.0%, respectively; data not shown). This reflects the high prevalence of these diseases in the general populations of countries in the Gulf area, as illustrated by recent national statistics from Abu Dhabi showing that in 2011, 17% of the general population had hypertension and 21% had diabetes.23
This study revealed that the presence of comorbidities was significantly associated with a CAT score ≥10, which reflects a high impact of respiratory symptoms on functioning. This result is consistent with outcomes from other studies using different patient-reported outcome measures, which have highlighted the association between the presence of comorbidities and an impairment of quality of life or functioning in COPD subjects.24–26 The CAT threshold of ten was chosen, since this is the one recommended in the 2011 GOLD guidelines for the classification of patients with COPD,1 but it cannot be excluded that the strength of the association with comorbidities may differ if another CAT threshold were to be used.
We found that the presence of comorbidities was associated with an increased risk of experiencing COPD exacerbations. There are few data available concerning the relationship between the presence of comorbidities and COPD exacerbations in COPD, although a US study has reported that patients with comorbidities presented a higher risk of exacerbations than those without.27 Moreover, it is well documented that COPD exacerbations constitute a risk factor for subsequent development of cardiovascular disease.4,28 To our knowledge, our study is one of the first to use the GOLD severity-grade groups (A–D) in the classification of COPD subjects with comorbidities. We found that the majority of COPD subjects (57.8%) with comorbidities were classified in group D, corresponding to subjects who reported severe symptoms (CAT ≥10) and previous exacerbations. This is consistent with previous reports of more severe COPD symptoms in subjects with comorbidities.29
This study had certain limitations. Since data collection was based on self-report without medical ascertainment, it is possible that this may have introduced some anamnestic bias or underreporting of information on comorbidities. Given that this was a general population survey in which spirometry could not be performed systematically, there is likely to be some imprecision in the estimation of the proportion of subjects fulfilling the epidemiological definition of COPD used in the study. Secondly, in certain countries, such as the UAE, the number of subjects who respond to the COPD detailed questionnaire and who report comorbidities is low. This precludes the analysis of the data at an individual country level, which may lead to loss of information when the prevalence of individual comorbidities, such as diabetes, varies markedly between participating countries.
Conclusion
This study showed that the prevalence of comorbidities among COPD subjects is high in the MENA region. The presence of comorbidities was associated with increased severity of COPD symptoms and exacerbations of the disease. This highlights the importance of seeking out and managing appropriately comorbidities in all individuals with a diagnosis of COPD.
Acknowledgments
The authors would like to thank all members of the BREATHE Study Steering Committee for their contributions to this study: A Alzaabi (Abu Dhabi, UAE), M Beji (Tunis, Tunisia), A Ben Kheder (Tunis, Tunisia), M Idrees (Riyadh, Saudi Arabia), G Iraqi (Rabat, Morocco), A Javed (Peshawar, Pakistan), JA Khan (Karachi, Pakistan), A Khattab (Cairo, Egypt), ML Koniski (Beirut, Lebanon), B Mahboub (Sharjah, UAE), S Nafti (Algiers, Algeria), NM Obeidat (Amman, Jordan), M Polatli (Aydin, Turkey), A Sayiner (Izmir, Turkey), N Shahrour (Damascus, Syria), MA Tageldin (Cairo, Egypt), S Taright (Algiers, Algeria), E Uzaslan (Bursa, Turkey), S Wali (Jeddah, Saudi Arabia). Foxymed also provided medical writing support in the form of writing assistance, collating authors’ comments, grammatical editing, and referencing that was paid for by GlaxoSmithKline.
Disclosure
BM, AA, and MNI have received honoraria from GlaxoSmithKline Laboratories for their contribution to the BREATHE study. AL is an employee of MS Health, the clinical and epidemiological research company responsible for the implementation, collection of data, and statistical analysis of results of the BREATHE study. HS is an employee of Foxymed, a medical communication and consultancy company that participated in the exploration, interpretation of the results, and preparation of this manuscript on behalf of GlaxoSmithKline Laboratories. LT and AEH are employees of GlaxoSmithKline Laboratories, which funded the BREATHE study and markets a number of treatments for COPD, and own shares in the company.
References
GOLD (Global initiative for chronic Obstructive Lung Disease). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Bethesda (MD): GOLD; 2011. | ||
Mannino DM, Thorn D, Swensen A, Holguin F. Prevalence and outcomes of diabetes, hypertension and cardiovascular disease in COPD. Eur Respir J. 2008;32(4):962–969. | ||
Feary JR, Rodrigues LC, Smith CJ, Hubbard RB, Gibson JE. Prevalence of major comorbidities in subjects with COPD and incidence of myocardial infarction and stroke: a comprehensive analysis using data from primary care. Thorax. 2010;65(11):956–962. | ||
Müllerova H, Agusti A, Erqou S, Mapel DW. Cardiovascular comorbidity in chronic obstructive pulmonary disease: systematic literature review. Chest. 2013;144(4):1163–1178. | ||
World Health Organization. The Global Burden of Disease: 2004 Update. Geneva: WHO; 2008. | ||
World Health Organization. Global Surveillance, Prevention and Control of Chronic Respiratory Diseases: A Comprehensive Approach. Geneva: WHO; 2007. | ||
Barnes PJ, Celli BR. Systemic manifestations and comorbidities of COPD. Eur Respir J. 2009;33(5):1165–1185. | ||
Baty F, Putora PM, Isenring B, Blum T, Brutsche M. Comorbidities and burden of COPD: a population based case-control study. PloS One. 2013;8(5):e63285. | ||
de Miguel Diez J, Carrasco Garrido P, García Carballo M, et al. Determinants and predictors of the cost of COPD in primary care: a Spanish perspective. Int J Chron Obstruct Pulmon Dis. 2008;3(4):701–712. | ||
Tageldin MA, Nafti S, Khan JA, et al. Distribution of COPD-related symptoms in the Middle East and North Africa region: results of the BREATHE study. Respir Med. 2012;106 Suppl 2:S25–S32. | ||
Khattab A, Javed A, Iraqui G, et al. Smoking habits in the Middle East and North Africa region: results of the BREATHE study. Respir Med. 2012;106 Suppl 2:S16–S24. | ||
World Health Organization. Prevalence of tobacco use among adults and adolescents. 2012. Available from: http://gamapserver.who.int/gho/interactive_charts/tobacco/use/atlas.html. Accessed December 1, 2015. | ||
International Diabetes Federation. IDF Diabetes Atlas. 5th ed. Brussels: IDF; 2012. | ||
Sherif S, Sumpio BE. Economic development and diabetes prevalence in MENA countries: Egypt and Saudi Arabia comparison. World J Diabetes. 2015;6(2):304–311. | ||
Alqurashi KA, Aljabri KS, Bokhari SA. Prevalence of diabetes mellitus in a Saudi community. Ann Saudi Med. 2011;31(1):19–23. | ||
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27(5):1047–1053. | ||
El Hasnaoui A, Rashid N, Lahlou A, Salhi H, Doble A, Nejjari C. Chronic obstructive pulmonary disease in the adult population within the Middle East and North Africa region: rationale and design of the BREATHE study. Respir Med. 2012;106 Suppl 2:S3–S15. | ||
Idrees M, Koniski ML, Taright S, et al. Management of chronic obstructive pulmonary disease in the Middle East and North Africa region: results of the BREATHE study. Respir Med. 2012;106 Suppl 2:S33–S44. | ||
Jones PW, Shahrour N, Nejjari C, et al. Psychometric evaluation of the COPD Assessment Test (CAT): data from the BREATHE study in the Middle East and North Africa region. Respir Med. 2012;106 Suppl 2:S86–S99. | ||
Sin DD, Anthonisen NR, Soriano JB, Agusti AG. Mortality in COPD: role of comorbidities. Eur Respir J. 2006;28(6):1245–1257. | ||
Schnell K, Weiss CO, Lee T, et al. The prevalence of clinically-relevant comorbid conditions in patients with physician-diagnosed COPD: a cross-sectional study using data from NHANES 1999–2008. BMC Pulm Med. 2012;12:26. | ||
Cavaillès A, Brinchault-Rabin G, Dixmier A, et al. Comorbidities of COPD. Eur Respir Rev. 2013;22(130):454–475. | ||
Health Authority Abu Dhabi. Health Statistics 2011. Abu Dhabi: HAAD; 2012. | ||
Almagro P, Castro A. Helping COPD patients change health behavior in order to improve their quality of life. Int J Chron Obstruct Pulmon Dis. 2013;8:335–345. | ||
Fortin M, Dubois MF, Hudon C, Soubhi H, Almirall J. Multimorbidity and quality of life: a closer look. Health Qual Life Outcomes. 2007;5:52. | ||
Yeo J, Karimova G, Bansal S. Co-morbidity in older patients with COPD – its impact on health service utilisation and quality of life, a community study. Age Ageing. 2006;35(1):33–37. | ||
Rascon-Aguilar IE, Pamer M, Wludyka P, et al. Role of gastroesophageal reflux symptoms in exacerbations of COPD. Chest. 2006;130(4):1096–1101. | ||
Walter RE, Wilk JB, Larson MG, et al. Systemic inflammation and COPD: the Framingham Heart Study. Chest. 2008;133(1):19–25. | ||
Yawn BP, Kaplan A. Co-morbidities in people with COPD: a result of multiple diseases, or multiple manifestations of smoking and reactive inflammation? Prim Care Respir J. 2008;17(4):199–205. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

