Back to Journals » Clinical Ophthalmology » Volume 11
Combined intrastromal injection of amphotericin B and topical fluconazole in the treatment of resistant cases of keratomycosis: a retrospective study
Authors Nada WM, Al Aswad MA, El-Haig WM
Received 19 February 2017
Accepted for publication 5 April 2017
Published 5 May 2017 Volume 2017:11 Pages 871—874
DOI https://doi.org/10.2147/OPTH.S135112
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Waled Mahdy Nada, Mahmoud A Al Aswad, Wael M El-Haig
Ophthalmology Department, Faculty of Medicine, Zagazig University, Zagazig, Egypt
Purpose: To evaluate the efficacy of combination therapy of a single intrastromal injection of amphotericin B and topical fluconazole in resistant cases of fungal keratitis, and also topical amphotericin B as monotherapy in terms of the duration of the recovery period and toxic drug effects.
Methods: This retrospective 2-year study reviewed 68 cases of unilateral fungal keratitis diagnosed by clinical features and positive laboratory culture results. Forty-one cases were resistant and did not respond to monotherapy with an antifungal agent. Thus, they were treated with a single intrastromal injection of amphotericin B in addition to topical fluconazole as combined antifungal therapy, representing group A. Twenty-seven cases were treated with topical amphotericin B as antifungal monotherapy, representing group B. Topical atropine 1% and different antibiotic eye drops were added to the antifungal agents in both groups. Follow-up of patient records was performed monitoring cure rate, duration of recovery period, and toxic drug effects such as pain, burning sensation, and corneal melting.
Results: The results revealed that group A, treated with combination therapy, showed recovery of 34 cases (82.9%) with a mean duration of 24±6.42 days, significantly different from group B which showed recovery of 16 cases (59.3%) with a mean duration of 39.66±13.6 days. Group A also showed less manifestation of drug toxicity than group B.
Conclusion: Combined intrastromal injection of amphotericin B and topical fluconazole can provide a good modality in the treatment of resistant cases of fungal keratitis, exhibiting highly potent antifungal effects, shorter recovery period, and reduced corneal toxicity.
Keywords: fungal keratitis, intrastromal injection, amphotericin B, fluconazole, combined therapy
Introduction
Cases of keratomycosis are still considered challenging to ophthalmologists due to the difficulties in management of such cases, starting from the availability of and susceptibility to antifungal agents, to penetration of corneal tissues, as fungal pathogens tend to deeply penetrate corneal tissues and may reach the anterior chamber. Thus, intrastromal and intracameral injection of antifungal agents, especially polyenes such as amphotericin B, constitutes the best chance for management of such infections, because these routes result in higher concentrations of the antifungal agents in corneal tissue and aqueous humor than topical use, enhancing the antifungal effects of the drugs.1,2
Also, the topical application of azoles, especially fluconazole and voriconazole, has been proven to be safe for ocular tissues and has good penetration into corneal tissue. Therefore, these eye drops are considered suitable for ocular use from a toxicological point of view.3 Combination therapy of antifungal agents has been shown to have potent antifungal activity and produce the best results in treating keratomycosis, especially in mild and moderate cases.4 Gatifloxacin is a potent antibiotic with additional antifungal effects, and has therefore been preferred for topical application in keratomycosis, as combined bacterial and fungal infection has been reported in many cases, thus, many studies preferred the use of antibiotics which have some antifungal effects.5
Methods
This hospital-based retrospective observational study was performed at the Zagazig University Hospital, Zagazig, Egypt from January 2015 to December 2016. The study reviewed 68 cases of unilateral fungal keratitis that were diagnosed clinically and confirmed by laboratory culture. Data were collected from the hospital records of fungal keratitis cases, including history, therapy, and follow-up data. The study was performed according to the World Medical Association Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects. The study was approved by the ethics committee of the Faculty of Medicine, Zagazig University, the patients’ informed consent for publishing their photos and to review their medical records, was obtained. Diagnosis of cases was based on the clinical features of fungal keratitis:
- history of trauma involving plant or vegetable material;
- history of surgical trauma;
- history of diabetes mellitus or corticosteroid use;
- history of resistant corneal ulcer;
- slit-lamp examination showing features of fungal keratitis: thick elevated keratitis, feathery edges of the ulcer, surrounding satellites, irregular hypopyon in the anterior chamber, and area of staining of the cornea by fluorescein stain smaller than the area of infiltration.
Additionally, laboratory culture was obtained after stopping medications for 48 hours, via corneal scraping with a sterile keratome under topical anesthesia (benoxinate Hcl 0.4%). The specimen was cultured in Sabouraud dextrose agar media and growth was observed up to 14 days. The study reviewed only 68 eyes with positive culture results, group A included 41 eyes of 41 patients with resistant cases which did not respond to monotherapy of antifungal agent for more than 40 days, (12 eyes received topical natamycin 2.5%, eleven eyes received topical amphotericin B 0.3 mg/mL, 13 eyes received topical itraconazole 2%, and five eyes received topical fluconazole 2%). The response to treatment was judged based on the healing of the ulcer, ie, disappearance of the symptoms and signs, and absence of fluorescein staining of the cornea. These cases were treated with a single intrastromal injection of amphotericin B (Fungizone 50 mg vial; Bristol-Myers Squibb, New York, NY, USA) 0.02 mg/mL in distilled water, using a 28-gauge needle, at 2–3 sites around the ulcer with midstromal injection of 0.1–0.15 mL containing 2–3μg of amphotericin B under topical anesthesia (benoxinate Hcl 0.4%), in addition to topical fluconazole 2 mg/mL eye drops five times daily (Diflucan® infusion, Pfizer, Inc., New York, NY, USA).
Group B, which included 27 eyes of 27 patients, was treated with topical amphotericin B (Fungizone 50 mg vial) 0.3 mg/mL 5% dextrose five times daily, in addition to regular debridement of the ulcer every 48 hours to facilitate penetration of the drug. They also received topical atropine 1% eye drops three times daily, in addition to different topical antibiotic eye drops five times a day (35 eyes received gatifloxacin 0.3%, 22 eyes received moxifloxacin 0.1%, and eleven eyes received tobramycin 0.3%) in both groups. Follow-up of patients was performed every 3 days in the first month and weekly for another 2 months. A slit-lamp was used to evaluate the healing characteristics (absence of epithelial defect and disappearance of inflammatory signs) and duration, and toxic drug effects, such as pain, burning sensation, and corneal melting were recorded.
Results
The demographic data of patients in group A revealed that 26 patients (63.4%) were male and 15 patients (36.6%) were female. Age of the patients ranged from 22 to 72 years with a mean age of 49.2±11.64 years. In group B, 16 patients (59.3%) were male and eleven patients (40.7%) were female, age of the patients ranged from 34 to 69 years with a mean age of 51.2±10.07 years.
The culture results of group A’s eyes revealed 16 (39%) cases of Candida, six (14.6%) cases of Aspergillus niger, seven (14%) cases of Aspergillus fumigatus, four (9.8%) cases of Penicillium, and eight (19.5%) cases of Fusarium. The culture results of group B revealed 15 (55.5%) cases of Candida, four (14.8%) cases of A. fumigatus, five (18.5%) cases of Alternaria, and three (11.1%) cases of Fusarium.
In group A, treated with combination therapy of antifungal agents, 34 (82.9%) of 41 eyes showed complete healing with a mean duration of healing of 24±6.42 days, and seven eyes showed no healing up to 40 days (Figure 1), these cases were recorded as treatment failure and other treatment modalities were prescribed (intrastromal voriconazole). In group B, 16 (59.3%) of 27 eyes showed complete healing with a mean duration of healing of 39.66±13.6 days, and eleven eyes showed no healing up to the same duration as in the first group (Figure 2), these cases were prescribed combination therapy similar to group A (Table 1).
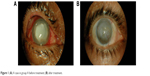  | Figure 1 (A) A case in group A before treatment; (B) after treatment. |
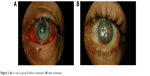  | Figure 2 (A) A case in group B before treatment; (B) after treatment. |
  | Table 1 Demographic and clinical findings and outcome in both groups |
Among the eight cases of Fusarium infection in group A, seven cases failed to show any signs of healing up to 40 days of treatment and were prescribed other treatment modalities such as intrastromal voriconazole injection.
Only four patients in group A (9.8%) suffered from burning sensation in the eye compared to eleven patients (40.7%) in group B. No toxic effects of intrastromal injection of amphotericin B, such as persistent corneal haze, edema, corneal erosion, and corneal melting were reported. Also, no other toxic effects of topical amphotericin B use in group B were reported.
Discussion
The study evaluated the use of a combination of intrastromal injection of amphotericin B with topical fluconazole, representing two groups of antifungal agents aiming to potentiate their effects against fungal pathogens. The choice of amphotericin intrastromal injection was based on the study of Qu et al1 who stated that intracorneal injection of amphotericin B is considered one of the most potent routes of administration in treating fungal keratitis, and this route minimizes the side effects of topical application of the drug, and shortens the duration of treatment, it also has very potent effects against fungal pathogens which usually deeply penetrate the corneal tissues. This study reported 82.9% healing in cases treated with combination therapy with a mean duration of 24±6.42 days, which was shorter than in the study of Mahdy et al6 which reported a mean duration of healing of 31 days in treatment with a combination of topical amphotericin B and subconjunctival injection of fluconazole, but the other study was different – they used fluconazole injection rather than the topical route which the present study used.
Many authors prefer the combination therapy of antifungal agents with one from the polyenes group and the other from the azole group for the management of fungal keratitis, such as Gong and Gong,4 who used natamycin and fluconazole, and Mahdy et al,6 who used amphotericin B and fluconazole. All of these studies reported that the combination therapy of antifungal agents produced better results in treating cases of keratomycosis compared to monotherapy, due to the broad spectrum antifungal effects.
This study reported healing failure in cases of Fusarium infection in group A (despite involving the failed cases of the other group) due to resistance of the organism to the treatment, and these cases were prescribed other treatment modalities, as explained by the study of Kalaiselvi et al7 and Khater et al8 who treated Fusarium keratitis with intrastromal injection of voriconazole, which has been proven to be the best choice for the treatment of Fusarium keratitis and resistant keratomycosis.
This study demonstrated the high efficacy of the combination of intrastromal injection of amphotericin B 0.02 mg/mL and topical fluconazole, and did not report any toxic effects associated with intrastromal injection. This finding is in contrast to the study by Qu et al9 which reported that the safe concentration of amphotericin B intrastromal injection was 0.01 mg/mL, so the present study may add another safe concentration of amphotericin B intrastromal use of 0.02 mg/mL as treatment for keratomycosis, leading to shorter duration of treatment and hence, healing of ulcers.
Conclusion
Except for Fusarium, the combined single intrastromal injection of amphotericin B and topical fluconazole can constitute a good modality for the treatment of resistant cases of fungal keratitis, exhibiting highly potent antifungal effects, shorter recovery period, and reduced corneal toxicity.
Acknowledgment
The authors would like to thank Dr Tamer Gamal, Ophthalmology Department, Zagazig University for his revision of the paper.
Disclosure
The authors report no conflicts of interest in this work.
References
Qu L, Li L, Xie H. Corneal and aqueous humor concentrations of amphotericin B using three different routes of administration in a rabbit model. Ophthalmic Res. 2010;43(3):153–158. | ||
Garcia-Valenzuela E, Song CD. Intracorneal injection of amphothericin B for recurrent fungal keratitis and endophthalmitis. Arch Ophthalmol. 2005;123(12):1721–1723. | ||
Fernández-Ferreiro A, González Barcia M, Gil Martínez M, Blanco Mendez J, Lamas Díaz MJ, Otero Espinar FJ. [Analysis of ocular toxicity of fluconazole and voriconazole eye drops using HET-CAM]. Farm Hosp. 2014;38(4):300–304. Spanish. | ||
Gong H, Gong X. Combined application of 5% natamycin and 0.2% fluconazole for the treatment of fungal keratitis. Eye Sci. 2013;28(2):84–87. | ||
Shams S, Ali B, Afzal M, Kazmi I, Al-Abbasi FA, Anwar F. Antifungal effect of Gatifloxacin and copper ions combination. J Antibiot (Tokyo). 2014;67(7):499–504. | ||
Mahdy RA, Nada WM, Wageh MM. Topical amphotericin B and subconjunctival injection of fluconazole (combination therapy) versus topical amphotericin B (monotherapy) in treatment of keratomycosis. J Ocul Pharmacol Ther. 2010;26(3):281–285. | ||
Kalaiselvi G, Narayana S, Krishnan T, Sengupta S. Intrastromal voriconazole for deep recalcitrant fungal keratitis: a case series. Br J Ophthalmol. 2015;99(2):195–198. | ||
Khater MM, El-Shorbagy MS, Selima AA. Argon laser photocoagulation versus intrastromal voriconazole injection in treatment of mycotic keratitis. Int J Ophthalmol. 2016;9(2):225–229. | ||
Qu L, Li L, Xie H. Toxicity and pharmacokinetics of intrastromal injection of amphotericin B in a rabbit model. Curr Eye Res. 2014;39(4):340–347. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
