Back to Journals » Cancer Management and Research » Volume 12
Combined Diagnostic Significance of Preoperative Serum β2-Microglobulin and Routine Blood Test in Patients with High-grade Glioma and Solitary Brain Metastasis
Authors Li L, Bu X, Wu B, Zhang S, Jin K, Xia L, Sun C
Received 23 June 2020
Accepted for publication 12 October 2020
Published 17 November 2020 Volume 2020:12 Pages 11735—11742
DOI https://doi.org/10.2147/CMAR.S268990
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Bilikere Dwarakanath
Liwen Li, 1,* Xiaomin Bu, 2,* Bin Wu, 1 Shuyuan Zhang, 1 Kai Jin, 1 Liang Xia, 1 Caixing Sun 1
1Department of Neurosurgery, Cancer Hospital of the University of Chinese Academy of Sciences (Zhejiang Cancer Hospital), Institute of Basic Medicine and Cancer (IBMC), Chinese Academy of Sciences, Hangzhou, Zhejiang 310022, People’s Republic of China; 2Department of Clinical Laboratory, Cancer Hospital of the University of Chinese Academy of Sciences (Zhejiang Cancer Hospital), Institute of Basic Medicine and Cancer (IBMC), Chinese Academy of Sciences, Hangzhou, Zhejiang 310022, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Caixing Sun; Liang Xia Tel +86-571-88122222
; Tel +86-13777820122
Email [email protected]; [email protected]
Background: High-grade glioma (HGG) and solitary brain metastasis (sBM) patients show similar symptoms in clinical practice, and accurately differential diagnosis directly affects the management and prognosis of patients. The aim of this study was to distinguish two entities by preoperative serum β 2-microglobulin (β 2-m) and routine blood test-associated inflammatory indexes including, white blood cell (WBC), neutrophils, lymphocytes, monocytes, and platelets count, red cell distribution width (RDW), platelet distribution width (PDW), neutrophil/lymphocyte ratio (NLR) and monocyte/lymphocyte ratio (MLR).
Patients and Methods: A retrospective analysis was performed in the Cancer Hospital of the University of Chinese Academy of Sciences from January 2015 to December 2019, including 127 patients of newly pathologically diagnosed with HGG and 174 patients with sBM. Clinical information including age, gender, pathological diagnosis, preoperative serum β 2-m and routine blood tests were collected, and NLR and MLR were calculated. The diagnostic significance of these markers for HGG and sBM was assessed by receiver operating characteristic (ROC) curves.
Results: The patients with sBM had significantly higher values of preoperative age, β 2-m, NLR and MLR as well as lower lymphocytes count than patients with HGG. Besides, the area under the curve (AUC) in differentiating HGG from sBM was 0.625 (95%CI: 0.561– 0.689) for age, 0.655 (0.594– 0.717) for β 2-m, 0.634 (0.571– 0.698) for NLR and 0.622 (0.559– 0.686) for MLR, and the combination of Age+β 2-m+NLR+MLR showed the best diagnostic performance with AUC of 0.731 (0.675– 0.788) and 0.048*Age+0.001*β 2-m+0.201*NLR+0.594*MLR> 5.813 could indicate sBM rather than HGG.
Conclusion: The Age+β 2-m+NLR+MLR combination was revealed as an inexpensive and noninvasive biomarker for differentiating between HGG and sBM before surgery.
Keywords: solitary brain metastasis, β 2-microglobulin, neutrophil/lymphocyte ratio, monocyte/lymphocyte ratio, routine blood test, high-grade glioma
Erratum for this paper has been published
Introduction
High-grade glioma (HGG) and brain metastasis (BM) are the most common malignant brain tumors in adults.1 Generally, glioma is solitary and BM is multifoci. However, it is difficult to differentiate HGG from solitary BM (sBM) since both tumors may have similar symptoms (including headache, vomiting, blurred vision, sensory deficit, dyskinesia and so forth) and display similar radiological characteristics: including peripheral contrast enhancement, central necrosis and surrounding edema.2,3 There is high clinical importance to distinguish these two intracranial pathologies before an operation, as the natural history, treatment, prognosis, and therapeutic implications are different, and it can be rewarding for clinical management of patients because it directly determines the choice of treatment options and affects the prognosis of patients.4,5
It is widely acknowledged that tumor-related systemic inflammatory response is closely correlated with various cancers.6,7 In previous studies, preoperative neutrophil/lymphocyte ratio (NLR) and lymphocyte/monocyte ratio (LMR) have been recognized as good diagnostic factors with high sensitivity and specificity in differential diagnosis of glioma from other benign brain tumors.8 Recently, the clinical application values of serum β2-microglobulin (β2-m) in predicting prognosis and monitoring of therapeutic efficacy has been rapidly highlighted.9 Several studies have shown an increased level of β2-m in solid tumors, including lung cancer and gastrointestinal cancer.10–12 However, the expression of β2-m in HGG and sBM remains unknown.
Therefore, the purpose of the present study was to determine whether the preoperative serum β2-m and hematological inflammatory indexes could differentiate HGG from sBM.
Patients and Methods
Patients Selection
The patients newly diagnosed with HGG and sBM from the Department of Neurosurgery of Cancer Hospital of the University of Chinese Academy of Sciences from January 2015 to December 2019 were recruited and retrospectively analyzed. All the patients included in our study had to meet the following inclusion standards: (1) patients newly diagnosed with HGG (grades III and IV according to the 2007 WHO classification) and sBM histologically verified in pathologic specimens from surgical resection or biopsy; (2) no prior antitumor treatment, such as radiotherapy or chemotherapy; (3) patients aged ≥18 years. Exclusion criteria: (1) patients with hematological disease, infectious disease, hyperpyrexia, other cancers, hypertension, diabetes mellitus, serious vital organ dysfunction, autoimmune diseases or HIV; (2) patients who had taken antibiotics or anti-inflammatory drugs in the prior three months; (3) patients with incomplete information of abovementioned preoperative serum. Finally, 127 patients with HGG and 174 patients with sBM were included in the analysis. This retrospective analysis was approved by the Ethics Committee of Cancer Hospital of the University of Chinese Academy of Sciences.
Data Collection
Demographic, clinical and histopathological data such as age, gender, pathological types, pre-existing conditions and medication history were collected from medical records. After hospitalization, fasting peripheral venous blood samples were taken for a blood count and biochemistry function test as a routine preoperative examination. The levels of β2-m were collected from the preoperative blood biochemistry test, the preoperative WBC, neutrophils, lymphocytes, monocytes, and platelets counts RDW (%) and PDW (%) were collected from the routine blood test. Moreover, operative NLR (quotient of neutrophil count to lymphocyte count), MLR (quotient of monocyte count to lymphocyte count) were calculated.
Statistical Analysis
Statistical analysis was carried out using SPSS version 25.0 (IBM Corporation, Armonk, NY, USA). The Kolmogorov–Smirnov test was used to determination the normal distribution of the variables. All data in this analysis were subject to non-normal distributions and were shown as median (25th, 75th percentiles). Comparisons between the two groups were conducted using the Mann–Whitney U-test. The categorical variables between groups were compared by a Pearson chi-squared test. The predictive probability of combined biomarkers for differentiating HGG from sBM was calculated using a binary logistic regression. The diagnostic significance of the preoperative indexes for distinguishing HGG from sBM was evaluated by the area under the curve (AUC) acquired from the receive operating characteristic (ROC) curves. An AUC >0.9 represented excellent diagnostic efficacy. An AUC >0.7 indicated good diagnostic efficacy. An AUC between 0.5 and 0.7 is considered poor diagnostic efficacy. When the Youden index is maximum, the best cutoff value is determined. A probability value (p-value)<0.05 is considered statistically significant.
Results
Study Population
A total of 301 patients newly diagnosed with HGG or sBM were enrolled in this study. The HGG group included 127 patients who received surgical resection or biopsy of glioma, including 59 with grade III and 68 with GBM. This group had 67 males (52.76%) and 60 females (47.24%) with a median age (25th, 75th percentile) of 56 (50, 65) years. The sBM group was composed of 174 patients who underwent surgical resection or biopsy of intracranial metastasis. This group had 102 males (58.62%) and 72 females (41.38%) with a median age (25th, 75th percentile) of 63 (53, 68) years.
Comparison of Preoperative Hematological Markers Between the Patients with HGG and sBM
Based on the K–S test, the p-values in the two groups were <0.05, which indicates the data were not normally distributed. Therefore, the value of each marker was represented as median (25th, 75th percentile). Table 1 and Figure 1 show the concentrations of these markers in patients with HGG and sBM. The patients with HGG were younger than those with sBM, and the difference was significant (p<0.001). No significant statistical difference was found regarding gender between the HGG and sBM groups (p>0.05). Besides, compared with the HGG patients, significantly higher level of β2-m, NLR and MLR as well as lower level of lymphocytes counts were observed in the sBM patients. Although the sBM patients have relatively higher level of median value of neutrophils count than HGG patients, no significant difference was observed (p>0.05). Furthermore, there were no statistically significant differences in the preoperative levels of WBC, monocytes, platelets counts, RDW and PDW between the groups (p>0.05).
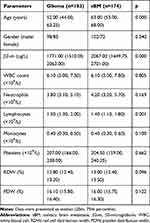 |
Table 1 Clinical Characteristics of the Patients with Glioma and sBM |
Diagnostic Efficacy of Single Preoperative Hematological Markers in Differentiating HGG from sBM
As shown in Figure 2A, diagnostic values of preoperative age, β2-m, NLR, and MLR in differentiating HGG from sBM were evaluated by the AUC acquired from ROC curves. The best cutoff value of age was found to be 56.5 years, and the AUC, sensitivity, specificity were 0.625, 70.70%, 52.85%. ROC curve analysis of β2-m individually showed the best cutoff value of 1932.50 (AUC=0.655, 95%CI: 0.594–0.717, sensitivity=58.05%, specificity=72.36%). ROC curve analysis of NLR had the best cutoff value of NLR 2.45 (AUC=0.634, 95%CI: 0.571–0.698, sensitivity=71.26%, specificity=52.85%). ROC curve analysis of MLR had the best cutoff value of MLR 0.28 (AUC=0.622, 95%CI: 0.559–0.686, sensitivity=56.32%, specificity=71.54%). According to the ROC curve analysis, the β2-m showed the best diagnostic performance in separating sBM from HGG, followed by NLR.
Diagnostic Efficacy of the Combined Preoperative Hematological Markers in Differentiating HGG from sBM
As shown in Figure 2B–C, ROC curves for the combined detection of these makers for differentiating HGG from sBM were constructed base on binary logistic regression. Table 2 shows the value of the combined detection of multiple markers combination (Age+β2-m, Age+NLR, Age+MLR, β2-m+NLR, β2-m+MLR, NLR+MLR, Age+β2-m+NLR, Age+β2-m+MLR, Age+NLR+MLR, β2-m+NLR+MLR, Age+β2-m+NLR+MLR). The results showed that the AUCs of five combinations were greater than 0.7 (β2-m+NLR, Age+β2-m+NLR, Age+β2-m+MLR, β2-m+NLR+MLR, Age+β2-m+NLR+MLR). For the combined detection of these indexes, the highest accuracy was obtained from Age+β2-m+NLR+MLR (AUC=0.731, 95%CI: 0.675–0.788, sensitivity=60.92%, specificity=77.24%), and the positive cutoff for sBM was 0.048*Age+0.001*β2-m+0.201*NLR+0.594*MLR>5.813.
 |
Table 2 Diagnostic Value of Preoperative Age, β2-m, Lymphocytes and Their Combinations in Patients with Glioma and sBM |
Discussion
Numerous research has shown that the systemic inflammation response in the tumor microenvironment may play a critical role in the development and progression of the tumor.13,14 In previous studies, the hematologic inflammatory markers have demonstrated significantly predictive factors for diagnosis, disease staging, and prognostic evaluation for various cancers.15,16 For instance, it was reported that a high level of preoperative peripheral blood NLR and MLR ratio was predictive of a poor prognosis for patients with glioma and BM.17–19 Besides, the preoperative NLR may be used to differentiate glioma from the benign brain tumors and healthy populations.8 However, few studies focus on the diagnostic value of inflammatory markers and their paired combinations for differentiating HGG from sBM. In this analysis, we investigated the levels of preoperative NLR and MLR and explored their diagnostic significance among patients with HGG and sBM.
It is well known that β2-m is a housekeeping gene and a nonglycosylated protein with a small light chain weight of 12-kDa, which is an important component of the major histocompatibility complex (MHC) class Iβ chain on the surface of nucleated cells.20 As an important structural protein, β2-m plays an important role in regulating host immune recognition by CD8+ T lymphocytes, and participates in iron metabolism and immunoglobulin transport.21 Besides, mounting evidence has highlighted the role of β2-m in host immune surveillance, which may regulate the survival, proliferation, migration, invasion, apoptosis, angiogenesis and metastasis of various types of cancer cell.22,23 Furthermore, overexpression of serum or urine β2-m was verified as an independent prognostic factor in many malignant tumors such as lymphoma, multiple myeloma and solid tumors.24,25 In addition, β2-m was also demonstrated to support the metastasis of bone malignant tumor by promoting the proliferation and survival of stromal cells.26 Moreover, multiple lines of evidence show that the serum β2-m is a significantly independent survival predictor and prognostic factor for several malignant tumor including renal cell carcinoma, multiple myeloma and leukemia.27–29 However, little is known about the levels of β2-m in glioma and BM. In this analysis, we investigated the levels of β2-m as well as several preoperative routine blood test-associated inflammatory markers in patients with HGG and sBM, including WBC, neutrophils, lymphocytes, monocytes, and platelets counts, NLR, MLR, RDW, and PDW, and assessed the diagnostic value for HGG and sBM.
According to our data, we demonstrated that patients with sBM have significantly higher values of age, preoperative serum β2-m, NLR, and MLR as well as lower level of preoperative lymphocyte count than patients with HGG. This finding suggests that age, β2-m, NLR, and MLR may have the ability to differentiate HGG from sBM. Unfortunately, the detailed mechanisms underlying the high level of serum β2-m, NLR, and MLR and low level of lymphocyte count in patients with sBM remain unknown. Some scholars thought the lymphocyte infiltration around brain tumors may mediate antitumor immune reaction and mean better prognosis, whereas the infiltration of neutrophils around tumors may result from the stimulation of neutrophil chemotactic factors and reactive oxygen species induced by tumor cells, and associates with a poor prognosis.30,31 Lower lymphocyte counts and higher neutrophil counts suggest reduced lymphocyte-dependent antitumor immune response and increased neutrophil-dependent inflammatory reactions.32
Due to the similar clinical symptoms and imaging characteristics, accurate differentiation between HGG and sBM is a common differential diagnosis problem in clinical practice.33 In general, pathological examination of suspicious tissue obtained from a biopsy or resection is considered as the gold standard procedure for definitive diagnosis when the intracranial lesion is uncertain, whereas CT and MRI are the important supplemental diagnostic approaches for assistance of diagnosis, tumor staging, and evaluation of therapeutic efficacy.9 However, both the neuroimaging tests and pathological examination are expensive with limited sensitivity, even at risk of hemorrhage and neurologic impairment, which have greatly limited their application for the clinical differential diagnosis.10 Thus, there is an urgent need for the low cost, easily obtained, and minimally invasive markers with improved diagnostic sensitivity and specificity for patients with HGG and sBM.
Therefore, we further investigated the diagnostic performance of preoperative age, β2-m, NLR, and MLR in patients with HGG against patients with sBM using ROC curve analysis. For single factor, β2-m demonstrated the best differential diagnostic performance, followed by NLR. For combined testing of these markers, the highest diagnostic accuracy was obtained in the combination of Age+β2-m+NLR+MLR. Routine blood tests and blood biochemistry test are inexpensive, minimally invasive and widely used in clinical practice for differential diagnosis of diseases.
This study has several limitations. First, as a retrospective analysis, some unavoidable biases may have occurred during data collection. Second, the number of patients enrolled in this study appeared to be limited and only from only a single center. Furthermore, we only evaluated the changes of hematological indexes and did not consider the differences in tumor immune microenvironment among patients. Therefore, prospective, multicenter and large-scale trials, continuous prognostic monitoring and deep research of the molecular mechanism are needed to further confirm our preliminary results. Moreover, it was reported that hematological markers had significant diagnostic value for distinguishing glioma from different brain pathologies, including acoustic neuroma, meningioma, nonlesional epilepsy and healthy individuals.8 However, the present study only focused on the differential diagnoses between HGG and sBM, without other neurological disorders.
Conclusion
We demonstrated that the combination detection of Age+β2-m+NLR+MLR was revealed as a promising, inexpensive and noninvasive biomarker in differentiating HGG from sBM, although the test showed high specificity (77.24%) and relatively low sensitivity (60.92%). We believe a larger patient sample study in the future may increase the diagnostic sensitivity. Our findings showed that 0.048*Age+0.001*β2-m+0.201*NLR+0.594*MLR>5.813 could indicate sBM rather than HGG.
Abbreviations
HGG, high-grade glioma; BM, brain metastasis; sBM, solitary brain metastasis; β2-m, β2-microglobulin; WBC, white blood cell; RDW, red cell distribution width; PDW, platelet distribution width. LMR, lymphocyte to monocyte ratio; LMR, monocyte to lymphocyte ratio; AUC, area under the curve; ROC, receiver operating characteristic. CT, computed tomography; MRI, magnetic resonance imaging.
Ethic Approval and Consent to Participate
This study was conducted in accordance with the regulations on patient confidentiality and the ethical standards of the Declaration of Helsinki. This research was approved by the Ethics Committee of Cancer Hospital of the University of Chinese Academy of Sciences. A waiver of patient consent was granted because the research involved no more than minimal risk to the participants and the waiver will not adversely affect the rights and welfare of the participants.
Acknowledgments
The present study was supported by the National Natural Science Foundation of China (Grant No. 81502147), the Natural Science Foundation of Zhejiang Province (LQ21H160006, LY21H160007), the Zhejiang Medical Science and Technology Project (2017KY260, 2018KY292, 2019RC127, 2018KY291), Chinese medicine science and technology plan of Zhejiang Province (2016ZA039), the Youth Scientific Innovation Foundation of Zhejiang Cancer Hospital (QN201902, QN201803), and the third term “new medical talents of Zhejiang province project”.
Disclosure
The authors report no conflicts of interest in this work or in the methods or materials used in this paper.
References
1. Davis ME Epidemiology and Overview of Gliomas. Sem oncol nursing. 2015;34:420–429, doi:10.1016/j.soncn.2018.10.001
2. Wu L, Wu M, Tian D, et al. Clinical and imaging characteristics of cerebral schistosomiasis. Cell Biochem Biophys. 2012;62(2):289–295. doi:10.1007/s12013-011-9294-1
3. Huang J, Luo J, Peng J, et al. Cerebral schistosomiasis: diffusion-weighted imaging helps to differentiate from brain glioma and metastasis. Acta Radiol. 2017;58(11):1371–1377. doi:10.1177/0284185116687173
4. Giese A, Westphal M. Treatment of malignant glioma: a problem beyond the margins of resection. J Cancer Res Clin Oncol. 2001;127(4):217–225. doi:10.1007/s004320000188
5. O’Neill BP, Buckner JC, Coffey RJ, Dinapoli RP, Shaw EG. Brain metastatic lesions. Mayo Clinic Proc. 1994;69(11):1062–1068. doi:10.1016/s0025-6196(12)61374-3
6. Cho A, McKelvey KJ, Lee A, Hudson AL. The intertwined fates of inflammation and coagulation in glioma. J Neurosurg. 2018;29:806–816. doi:10.1007/s00335-018-9761-8
7. Schulz M, Salamero-Boix A, Niesel K, Alekseeva T, Sevenich L. Microenvironmental Regulation of Tumor Progression and Therapeutic Response in Brain Metastasis. Front Immunol. 2019;10:1713. doi:10.3389/fimmu.2019.01713
8. Zheng SH, Huang J-L, Chen M, et al. Diagnostic value of preoperative inflammatory markers in patients with glioma: a multicenter cohort study. J Neurosurg. 2018;129(3):583–592. doi:10.3171/2017.3.jns161648
9. Bento L, et al. New prognosis score including absolute lymphocyte/monocyte ratio, red blood cell distribution width and beta-2 microglobulin in patients with diffuse large B-cell lymphoma treated with R-CHOP: spanish Lymphoma Group Experience (GELTAMO). J Nat. 2020;188:888–897. doi:10.1111/bjh.16263
10. Sasaki T, et al. Gastric cancer progression may involve a shift in HLA-E profile from an intact heterodimer to β2-microglobulin-free monomer. Int j Cancer. 2014;134:1558–1570. doi:10.1002/ijc.28484
11. Pokrass MJ, Liu MF, Lindorfer MA, Taylor RP. Activation of complement by monoclonal antibodies that target cell-associated β₂-microglobulin: implications for cancer immunotherapy. Mol Immunol. 2013;56:549–560. doi:10.1016/j.molimm.2013.05.242
12. Chen Y, et al. Prognostic significance of baseline peripheral absolute neutrophil, monocyte and serum β2-microglobulin level in patients with diffuse large b-cell lymphoma: a new prognostic model. J Neurosurg. 2016;175:290–299. doi:10.1111/bjh.14237
13. Sfanos KS, Yegnasubramanian S, Nelson WG, De Marzo AM. The inflammatory microenvironment and microbiome in prostate cancer development. Nat Rev Urol. 2018;15(1):11–24. doi:10.1038/nrurol.2017.167
14. Monkkonen T, Debnath J. Inflammatory signaling cascades and autophagy in cancer. Autophagy. 2018;14(2):190–198. doi:10.1080/15548627.2017.1345412
15. Ethier JL, Desautels D, Templeton A, Shah PS, Amir E. Prognostic role of neutrophil-to-lymphocyte ratio in breast cancer: a systematic review and meta-analysis. Breast Cancer Res. 2017;19(1):2. doi:10.1186/s13058-016-0794-1
16. Haram A, et al. The prognostic value of neutrophil-to-lymphocyte ratio in colorectal cancer: A systematic review. J Surg Oncol. 2017;115:470–479. doi:10.1002/jso.24523
17. Mitsuya K, Nakasu Y, Kurakane T, et al. Elevated preoperative neutrophil-to-lymphocyte ratio as a predictor of worse survival after resection in patients with brain metastasis. J Neurosurg. 2017;127(2):433–437. doi:10.3171/2016.8.jns16899
18. Bao Y, Yang M, Jin C, et al. Preoperative Hematologic Inflammatory Markers as Prognostic Factors in Patients with Glioma. World Neurosurg. 2018;119:e710–e716. doi:10.1016/j.wneu.2018.07.252
19. Zhang L, et al. Pre-stereotactic radiosurgery neutrophil-to-lymphocyte ratio is a predictor of the prognosis for brain metastases. J Nat. 2020;147:691–700. doi:10.1007/s11060-020-03477-w
20. Nomura T, Huang W-C, Zhau H, et al. β2-Microglobulin-mediated Signaling as a Target for Cancer Therapy. Anticancer Agents Med Chem. 2014;14(3):343–352. doi:10.2174/18715206113139990092
21. Feder JN et al. The hemochromatosis gene product complexes with the transferrin receptor and lowers its affinity for ligand binding.
22. Shi C, Zhu Y, Su Y, Chung LW, Cheng T. Beta2-microglobulin: emerging as a promising cancer therapeutic target. Drug Discov Today. 2009;14(1–2):25–30. doi:10.1016/j.drudis.2008.11.001
23. Yang J, Yi Q. Killing tumor cells through their surface β 2 -microglobulin or major histocompatibility complex class I molecules. Cancer. 2010;116(7):1638–1645. doi:10.1002/cncr.24953
24. Kim BR, Lee EJ, Seo SH, Lee SH, Rho SB. Dickkopf-3 (DKK-3) obstructs VEGFR-2/Akt/mTOR signaling cascade by interacting of β2-microglobulin (β2M) in ovarian tumorigenesis. Cell Signal. 2015;27:2150–2159. doi:10.1016/j.cellsig.2015.08.008
25. Sliker BH, et al. Beta 2-microglobulin regulates amyloid precursor-like protein 2 expression and the migration of pancreatic cancer cells. Cancer Biol Ther. 2019;20:931–940. doi:10.1080/15384047.2019.1580414
26. Josson S, Nomura T, Lin J-T, et al. β2-microglobulin induces epithelial to mesenchymal transition and confers cancer lethality and bone metastasis in human cancer cells. Cancer Res. 2011;71(7):2600–2610. doi:10.1158/0008-5472.can-10-3382
27. Sadamori N, et al. Clinical significance of beta 2-microglobulin in serum of adult T cell leukemia. Leukemia. 1995;9:594–597.
28. Rasmuson T, Grankvist K, Ljungberg B. Serum β 2 -Microglobulin and Prognosis of Patients with Renal Cell Carcinoma. Acta oncol. 1996;35(4):479–482. doi:10.3109/02841869609109926
29. Durie BG, et al. Prognostic value of pretreatment serum beta 2 microglobulin in myeloma: a Southwest Oncology Group Study. Blood. 1990;75:823–830.
30. Yue X, Lan F, Hu M, et al. Downregulation of serum microRNA-205 as a potential diagnostic and prognostic biomarker for human glioma. J Neurosurg. 2016;124(1):122–128. doi:10.3171/2015.1.jns141577
31. Kayhan A, et al. Preoperative Systemic Inflammatory Markers in Different Brain Pathologies: an Analysis of 140 Patients. Turk Neurosurg. 2019;29:799–803. doi:10.5137/1019-5149.jtn.24244-18.2
32. Zadora P, Dabrowski W, Czarko K, et al. Preoperative neutrophil-lymphocyte count ratio helps predict the grade of glial tumor - a pilot study. Neurol Neurochir Pol. 2015;49(1):41–44. doi:10.1016/j.pjnns.2014.12.006
33. Hochberg FH, et al. Glioma diagnostics and biomarkers: an ongoing challenge in the field of medicine and science. Expert Rev Mol Diagn. 2014;14:439–452. doi:10.1586/14737159.2014.905202
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


