Back to Journals » Journal of Healthcare Leadership » Volume 9
Collaborative public–private initiatives targeting multidrug-resistant tuberculosis (MDR-TB) supported by the Lilly MDR-TB Partnership: experiences in 2012–2016
Authors Shelby PW, Lia MP, Israel A
Received 14 December 2016
Accepted for publication 23 March 2017
Published 6 June 2017 Volume 2017:9 Pages 47—57
DOI https://doi.org/10.2147/JHL.S130207
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Russell Taichman
Peter W Shelby, Maria Paola Lia, Amy Israel
Lilly Global Health Programs, Geneva, Switzerland
Abstract: Since 2003, the Lilly Foundation has supported the noncommercial Lilly MDR-TB Partnership, which involves more than 45 local, national, global, and nongovernmental organizations and governments. The aim of the Lilly MDR-TB Partnership is to achieve significant global impact on multidrug-resistant tuberculosis (MDR-TB) by addressing a series of important local health care needs in highly affected countries: China, India, Russia, and South Africa. The main focus of activities during 2012–2016 was on community needs in primary care. Supported projects seek to make meaningful and measurable progress toward global and national TB objectives. The partnership programs share an overall conceptual approach known as “research, report, advocate”, based on the piloting of novel approaches on a small scale, with outcomes assessed at early stages. The results are analyzed and communicated to governments, health-policy experts, and local and national stakeholders, including those in other countries facing similar MDR-TB challenges. For successful, cost-effective initiatives, the analysis is used as support when advocating for the scaling up of initiatives to regional or national levels. This article discusses representative examples of projects supported by the Lilly MDR-TB Partnership in the time period 2012–2016. The examples illustrate the potential for globally informed, locally designed primary-care collaborations to strengthen health care systems and support TB policies and offer observations to inform future health care public–private partnerships.
Keywords: public–private partnership, nongovernmental organization, public health, community programs, primary care, prevention
Background
As a transmissible, airborne disease that disproportionally affects resource-limited populations, tuberculosis (TB) has for decades been a neglected disease worldwide. With more than 9 million new cases annually, TB kills an estimated 1.5 million individuals every year.1,2 Among the multifactorial drivers of the TB epidemic are shortcomings of sanitary and public health systems, inadequate living standards, stigma, a limited number of effective treatments – particularly for multidrug-resistant TB (MDR-TB) – and market failure, leading to a long hiatus in the development of medicines. In consequence, many tools for the prevention, diagnosis, and treatment of TB and MDR-TB are obsolete.3
Public–private partnerships (PPPs) are joint efforts of governments, nongovernmental organizations, companies, foundations, academia, and other stakeholders. PPPs can be particularly important for low- and middle-income countries grappling with the issue of how to control diseases such as TB.4 By harnessing the collective capacity and combining unique capabilities, expertise, and assets from each organization, PPPs aim to replace variable-quality, inefficient, and potentially expensive health care provision with more rapid, affordable, and correct prevention, diagnosis, and treatment. However, this is a tall order, and the sheer number of activities targeting TB ensures a large variation in quality and outcomes among different projects.5,6
Since 2003, the Lilly Foundation has supported the Lilly MDR-TB Partnership, which involves the company and a number of local, national, global, and nongovernmental organizations and governments. The partnership operates in four high-burden MDR-TB countries: China, India, Russia, and South Africa. Although the work has involved technology transfer of medicines and support for drug discovery and research-and-development programs, the main focus of activities during 2012–2016 was on community needs in primary care. Projects seek to make meaningful and measurable progress toward global and national TB objectives.
Partnership programs share an overall conceptual approach known as “research, report, advocate” (RRA). In RRA, novel approaches are piloted on a small scale, with outcomes assessed as rigorously as possible at early stages. The results of pilot projects are analyzed and communicated to governments, health-policy experts, and local and national stakeholders. For successful, cost-effective initiatives, the analysis of the outcomes is used as support by the partnerships in advocating for the scaling up of initiatives to regional or national levels. Transparency is maintained in communications and lessons from failures communicated openly for the benefit of partners and policy makers.
In this communication, we survey the experiences from representative examples of projects supported by the Lilly MDR-TB Partnership, with a focus on the time period 2012–2016. The degree of success was not a criterion for the selection; rather, the examples illustrate the potential for globally informed, locally designed primary-care collaborations to strengthen health care systems and support policies to improve TB rates through lessons and conclusions from the projects that may be valuable in future policy considerations. While not all country-based projects are highlighted in this paper, it is important to underscore that many more projects are conducted under the Lilly MDR-TB Partnership involving collaborative work with numerous health care organizations.
Materials and methods
The Lilly MDR-TB Partnership is part of Lilly’s corporate responsibility efforts to strengthen health care systems, expand access to health care, and find new evidence-based solutions that can be adapted, replicated, and scaled up (www.lillyglobalhealth.com). The Lilly MDR-TB Partnership seeks to help address the global MDR-TB problem by focusing on, addressing, and sharing lessons about specific local and national health care issues. In a multidimensional approach, major programs include MDR-TB medicine-manufacturing technology transfer, support for research and development, and a series of locally tailored partnership projects, such as training health care providers on effective TB care, building local health-system capacity, raising public awareness, and improving access to safe, effective medicines. The partnership focuses on the four countries most severely affected by the MDR-TB epidemic: China, India, Russia, and South Africa.
All projects are aligned with local government priorities. As Lilly is no longer engaged commercially in the field of anti-infective agents, no donations are involved, and this is the company’s largest ever philanthropic effort. The Lilly MDR-TB technology-transfer program started in 2003, before the period of focus for the current survey, but as it was completed in 2015, it is formally included in the Lilly MDR-TB Partnership. This project is described in detail in a publicly available report, which is included in Supplementary material.
Likewise, the current publication does not go into details of the Lilly TB Drug Discovery Initiative, which was founded in 2007. This is a not-for-profit PPP between Lilly, the Infectious Disease Research Institute, and the National Institute of Allergy and Infectious Diseases of the US National Institutes of Health. In 2009, Academia Sinica (National Academy of Sciences of Taiwan) joined the initiative. Lilly was also a founding partner of the TB Drug Accelerator consortium, set up in 2012, which has been described extensively elsewhere7,8 and is not a focus of the present discussion. An overview of the projects, organizations, goals, and core activities is provided in Table 1.
China
The objective of the partnership’s activities in China is to help address the shortage of qualified health care professionals and capacity addressing MDR-TB. To support health care-capacity building in line with the national TB-control policy and ensure a sustainable benefit, the partnership is working with national health organizations (listed in Table 1). In a cascading of competence, each nurse trained within the training for transformation program is in turn required to train additional 20 nurses and allied health professionals.
India
In India, as in many Asian countries, pharmacies are often the first point of contact with the health care system, and in many areas they act as “de facto primary health care providers”.9 The reasons are many, including the accessibility of providers and a perception that private care provides higher quality than public services.10–12 As pharmacists are operating in an intensely competitive commercial environment, they may not have full knowledge on various diseases and could thus unknowingly not recognize the signs and symptoms of TB.
To engage pharmacists in identifying and referring people who have symptoms of TB to testing and treatment facilities, a number of small-scale projects were supported in different Indian states: West Bengal, Telangana, Andhra Pradesh, Maharashtra, and Tamil Nadu. A defined process was used, as outlined in Table 1. A key part of the preparation was the development and provision of creative and visually appealing communication materials, presenting information in a format and vocabulary accessible to nonspecialist audiences. Results were presented at scientific conferences and in discussions with other stakeholders at local and national levels.
Russian Federation
In the Russian Federation, MDR-TB is a large and growing problem.13 The Lilly MDR-TB Partnership supported the nonprofit organization Partners in Health in a number of activities with two overall goals: improving access to high-quality MDR-TB care and training of health care providers and researchers.
It has been estimated that the risk of infection by MDR-TB in hospitalized patients is six times greater than that of patients treated as outpatients.14 Most cases of MDR-TB arise through transmission between a carrier and untreated individuals.15 The transmission of MDR-TB is greatly facilitated in settings such as hospitals and clinics, in which patients with undiagnosed DR-TB often share wards and rooms with patients with drug-sensitive disease.14,16 The Lilly MDR-TB Partnership supported the implementation of the FAST intensified administrative approach to TB transmission control15 at hospitals in Voronezh Oblast and Petrozavodsk (Karelia). FAST has four components: Find cases Actively (rapid diagnosis); Separate safely to reduce exposure to drug-resistant strains (by nosocomial transmission) and Treat effectively (with second-line drugs where appropriate). With FAST, the aim was to shorten the time to diagnosis and treatment allocation, and in consequence the time undiagnosed patients with MDR-TB spend in close proximity to other susceptible individuals.
In addition to transmission-reduction efforts, the Lilly MDR-TB Partnership and Partners in Health have furthered capacity building through training and support of health care providers and patients. Regional School of Patient programs were established in TB hospitals to provide patients with training on TB and the need to complete treatment courses. Russia is a vast country, and in an effort to overcome distances, a number of Internet-based solutions are being piloted. Projects such as Skype Supervision in Rural TB Control and Skype Observed Therapy routines aim to provide quality of care to TB patients in remote and isolated districts.
South Africa
With the highest burden of MDR-TB in Africa, South Africa faces a number of significant barriers to health care. These range from structural limitations to too few trained personnel and limited expertise in selected areas. It is national policy to decentralize treatment to communities, in order to increase access to care. By placing TB care more closely to home, TB-care success rates are thought to increase, and loss of income can be avoided.
The Lilly MDR-TB Partnership supported decentralization in KwaZulu-Natal and Eastern Cape, the two provinces with the highest MDR-TB rates. The King Dinuzulu Hospital Complex focused mainly on improving the care and management of patients with DR-TB, while the Red Cross worked at community level in supporting all those affected by DR-TB by providing psychosocial support and ensuring continued linkage to care at primary-health care level. In Eastern Cape, the partner Donald Woods Foundation helped the National Department of Health to decentralize TB care by working at hospitals, primary-health care clinics, prisons, schools, and taxi ranks and offering psychosocial support and strengthening linkages to care.
The nonprofit human development organization FHI 360 assisted the National Department of Health as technical advisor in preparing sites to decentralize. Details of resources and costs analyzed have been published elsewhere.17 The total cost per patient was the cost of patient-specific resource usage plus the daily ward-specific and shared costs for inpatients.
Ethical issues
Ethical approvals for individual projects were secured by the respective local responsible partner organizations as appropriate.
Results
China
At the time of writing, the program has trained 204 TB nurses working mainly in the TB and HIV fields. Training had led to significant behavioral changes. A majority of the trained nurses (>75%) had improved ventilation in their center and also provided surgical masks to coughing patients. Trained nurses had further implemented changes to how sputum was collected and transported to the lab. These changes led to improvements in the quality of sputum samples. Nurse training also improved patient relations, since trained nurses were able to provide psychosocial and emotional support and education to patients and their family members. The surveyed, trained nurses also reported improved treatment adherence among their patients, although no numbers were provided.
The 19 surveyed nurses had in their turn trained 4,163 health care workers, including 1,926 nurses, 146 doctors, 1,785 allied health workers, nursing students and community staff in their hospitals, and community members. An important corollary of the increased knowledge was a sense of empowerment among the nurses. After training, they experienced increased confidence in their ability to improve their practice and to negotiate with superiors and colleagues to effect improvements to the services.
Resource optimization was further enhanced by partner activities. The Chinese Center for Disease Control and Prevention developed a systematic training tool kit that is adaptable to different needs and skills among health care providers. The China Medical Association TB Society has set up a nationwide teleconsultancy and an Internet-based training network. Each of the two partners has established six provincial-level TB health care centers of excellence that account for one-third of China’s provincial-level administrative regions.
India
All Indian pharmacist-engagement projects provided measurable outcomes for assessments, although not all projects provided the same measures. Overall, the projects led to significant increases in the number of individuals with suspected TB referred for testing. Most (>90%) of the referred patients were tested, indicating that the referrals were based on realistic concerns. Of those tested, 11% were diagnosed as having TB (Table 2). In areas where pharmacists were involved in referrals, they contributed around 5%–8% of the total number of people testing positive in TB units. Pharmacists have also become successfully engaged in the provision of directly observed treatment, short course (DOTS) treatments to patients. Initial results indicate positive impact on treatment adherence.
A qualitative evaluation of patients’ experiences was performed in Telangana state by interviewing 42 people with diagnosis of TB who had been referred through the pharmacist program. More than half (n=26; 62%) had sought out the pharmacist as first point of contact to obtain cough and fever medication. Referrals were efficient: 62% of those referred were tested on the day after referral. Most people (n=29; 69%) stated that the referral had shortened delays in care-seeking behavior (by around 109 days), and 74% (n=31) of respondents voiced satisfaction with pharmacists’ services. None of the individuals referred for diagnosis had incurred out-of-pocket costs for the tests.
The results from the pilot studies were taken into account by the Indian government when deciding in 2014 to scale up support for training of pharmacists in provision of DOTS and other therapies for TB.18 The positive response from pharmacists was notable, even though there were no direct financial incentives to take part. Informal surveys indicated that many pharmacists were motivated by a sense of communality. Pharmacists were overall aware of being seen as primarily businesspeople, and considered the project an opportunity to deepen their interaction with their local community.
Russia
Preliminary results from the implementation of FAST strategies for rapid diagnosis of MDR-TB in Voronezh have been published.15 The data show a significant reduction in the time to identification of MDR-TB patients compared with the traditional patient pathways (Table 3). Similar numbers were reported from the Petrozavodsk center (Table 3). Of 1,165 patients with suspected pulmonary TB, 93.5% were tested within 2 days of admission. Of 202 patients identified with rifampin-resistant strains, 99% were started on MDR-TB treatment within 3 working days of receiving the test results. The protocol thus limited exposure of other patients and staff to the MDR-TB carriers to ≤5 days.
  | Table 3 Number of patients with MDR-TB identified and put on appropriate treatment regimens within 3 working days using FAST in hospitals in Voronezh and Petrozavodsk, respectively Notes: *Seven patients did not enroll; 99% of enrolled patients were assigned treatment within 3 working days. Data from Barrera et al.15 Abbreviations: MDR-TB, multidrug-resistant tuberculosis; FAST, Find cases Actively, Separate safely and Treat effectively. |
At the time of writing, no systematic analysis has been performed of transmission rates in the hospitals before and after implementation of the FAST strategy. However, although a direct correlation with the measures undertaken has not been formally established, prevalence of MDR-TB in Voronezh fell from 31.7 per 100,000 in 2010 to 18.8 per 100,000 in 2014. Mortality from TB in the region was reduced by half between 2000 and 2015 (1.8 in the city and 4.5 per 100,000 population in the oblast) and is currently lower than the average rate in the Russian Federation. The incidence of TB in Voronezh city is currently (2015) 18.7 per 100,000 population, which is comparable to rates in the European Union, and in the Voronezh oblast it is 29.6 per 100,000 population.
The models pioneered locally are being expanded nationally and included in the national policy for organizing TB-care practices in the Russian Federation. Among lessons learned to date is the importance of coordination of local projects and to ensure that local staff support the activities. If several parallel processes are followed, this will weaken commitment and efficiency. Similarly, if staff feel surveyed and managed from outside without the sense of being active, valued participants, this will lead to weak dedication and a squandering of resources and efforts. It is also important to provide sufficient support for participants to attend conferences and professional events to present the design and results of projects. Such presentations are crucial to advocating successes for possible scaling up at the national level.
South Africa
Analyses in KwaZulu-Natal have shown that community-based care for people with MDR-TB may lead to more successful treatment outcomes than care in a central, specialized hospital, but with large local variations.19 Decentralization has yielded improved case findings, shorter periods of treatment initiation, and better follow-up at community levels.19 It also reduces transport costs and time away from family.20
The follow-up study showed that for the year 2012, both centralized and decentralized hospital care were more expensive per person treated than community-based care, whether provided in local clinics or by the mobile-care units set up by the government (Table 4). When the costs of treatment success per patient were analyzed, the difference between the most and least expensive care models was greater still.
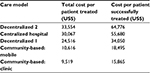  | Table 4 Cost-effectiveness of different care models for people with MDR-TB in KwaZulu-Natal province, South Africa, in 201220 Note: Reproduced from Loveday M. Cost-effectivess of different models of care for MDR-TB patients in South Africa. Presented at: the 68th session of the World Health Assembly; May 20th, 2015; Geneva, Switzerland.25 Abbreviation: MDR-TB, multidrug-resistant tuberculosis. |
The results of the cost-effectiveness studies provided the National Department of Health with important information for deciding and implementing the policy of making treatment available closer to patients’ homes. As a result, decentralization initiation in all provinces has been prioritized. In addition, this has led to the transfer of skills to primary-health care settings and infrastructure revamping.
Discussion
The global epidemic of TB/MDR-TB, by and large a disease of the poor, is partly an example of market failure, leading to lack of resources and tools for the prevention, diagnosis, and treatment of illness. This overview has shown examples from innovative collaborations supported by the Lilly MDR-TB Partnership, which focus on local public health measures. The examples illustrate the potential for a multifaceted PPP approach to help address the global MDR-TB problem by focusing on specific local and national barriers, gaps, and challenges. They also provide lessons for future projects.
Many projects were carried out in primary-care settings. The reasons for this are several. Often, the need for support is greatest in such settings. There are important benefits involved in bringing care more closely to peoples’ homes, such as avoiding the extra burden of travel and loss of work on already burdened individuals and families. Further, local expertise can be leveraged and lessons learned from pilot projects in primary care are directly related to the conditions of actual care or “real-world” settings.
Common to all primary-care projects described here is a focus on mobilizing existing untapped resources. Although new diagnostic tools and new drugs will be instrumental in overcoming the MDR-TB epidemic, the collaborations within the Lilly MDR-TB Partnership show the significant positive effects that can be achieved with small-budget projects when targeted at underutilized health-system capacity within a country. Indian pharmacists, local health centers in South Africa, underappreciated diagnostic strategies in Russia, and Chinese health care workers in need of technical TB training are all examples of local resources with the potential to become lifesaving, long-term strategic assets in the fight against MDR-TB.
Strengthening health care systems
Many of the efforts were directed toward long-term strengthening of health care systems. The experience in China illustrates the importance of keeping sustainability in mind, by focusing resources on building skills of health care providers. This need was recognized before the start of the projects, and it was mutually recognized that the Lilly MDR-TB Partnership could make a difference by providing necessary funds. Supporting the training of health care workers proved to be an effective means of promoting the spread of TB information and positive behavioral changes. Although the sustainability of projects will need to be demonstrated over the longer term, there are strong synergies between the different supported initiatives in China. The training tool kits, teleconsultancy, and Internet-based training network developed by partner organizations the Chinese Center for Disease Control and Prevention and the China Medical Association TB Society provide significant scope to leverage all capacity-building activities for sustainable long-term impact on quality of care and patient outcomes. Such synergies stretch funds and ensure that successful changes are maintained over the longer horizon.
The Indian pilot projects sought to strengthen the health care system by involving previously underutilized resources, in this case commercial pharmacists. An interesting finding here was the willingness of pharmacists to engage, even though there were no direct financial incentives offered. As they act within an intensely competitive environment, some Indian pharmacists are expected to take little interest in promoting public health causes, perceived as a nonprofit activity that will not bring commercial business. However, in the local programs, many pharmacists reported a willingness to help their local communities as a motivating factor. It is interesting to note this positive experience, as it is in contrast to reports elsewhere of pharmacists providing low-quality services. The realization that greater numbers of identified patients may increase their catchment area for services may of course have been an added incentive for pharmacists, independently of considerations of the public good. The high rate of people on pharmacist-provided DOTS who completed their course of therapy may also indicate that trained pharmacists recognized the commercial benefits from keeping customers on their medication for the necessary period of time. Whatever their commercial interests, turning pharmacists from mere sellers of medicinal products into active health care providers in the wider sense seems a good example of how sustainability can be achieved.
Informing and supporting policy making
In addition to strengthening local health care systems, successful PPPs that gather program and impact metrics and that are carried out in alignment with government priorities can provide practical support for policies aimed at wider-spread implementation. This was seen in all four countries. In low- to middle-income countries, limited budgets are often driving “frugal innovation” in commercial businesses but also increasingly in the provision of health care. Although the development of novel devices and technologies has tended to receive the greatest attention by media,21 the concept is highly suitable for adaptation to processes and strategies, as illustrated by a number of PPP projects, including those described here.
In health care systems with limited resources, innovative models need to be subjected to careful cost-effectiveness analysis, eg, as done for the South African models of care. While this is important, it is also necessary to bear in mind that what matters to public health is treatment outcome. Costs for MDR-TB are driven by various elements, especially hospitalization, which is more likely for patients in whom treatment has failed. Therefore, improved treatment success can be expected to reduce costs. In the South African example, the overall economic benefits from the decentralized health care models remained when costs were calculated per successfully treated patient. This indicates that the economic benefits over conventional hospital-based models did not come at the cost of success rates. Such analyses support the revisions in 2011 to the South African guidelines, which now recommend less strict hospitalization of people with MDR-TB, focusing on people with extensive drug-resistant-TB.22
For maximal impact, policies need to be implementable on supraregional or national levels. In the case of the Indian projects, a number of small-scale pilots were run testing similar strategies, in order to capture potential differences between local conditions. The low variation in results means that conclusions can be generalized and indicates that the projects could be scaled up successfully across regions with similar systems of local health care providers. In Russia, two hospitals implemented a similar strategy with minor variations, and the large impact on early measures in both centers suggests that any differences in local implementation had only a minor impact on the outcomes.
The Russian experience likewise shows how a successful pilot study can support the feasibility of relatively simple and low-budget approaches to improved treatment strategies and reduced risk of transmission of disease. The results are in stark contrast to what was typically achieved under normal operating conditions. Without use of the FAST approach, treatment failure was often identified several months after admission, meaning that other patients and staff were commonly exposed to MDR-TB for a very long time before treatment was initiated in patients with MDR-TB. The clearly demonstrated benefits with the locally pioneered models support the ongoing changes in national policy for organizing TB-care practices in the Russian Federation. At the time of writing, no systematic analysis has been performed of transmission rates in the hospitals before and after implementation of the FAST strategy. Such data would be important to support scaling up and wider uptake of the approach in other hospitals and regions, including outside the Russian Federation. To achieve this, adequate training is necessary, and the ongoing activities are expected to provide a foundation for rapid, efficient uptake of successfully tested strategies.
The experiences from the Lilly MDR-TB Partnership showed the need for adequate support for the “report” component of RRA to provide support and influence policies. Activities that present the design and results of projects to wider audiences can be resource-intense. Capacity planning also needs to include adequate support for project participants to attend scientific conferences and professional events. Such exposure increases the potential for scaling up successful programs at the national level. The “report” component is thus essential to prepare the ground for “advocate” activities. In addition, the exposure and recognition confer a stronger sense of ownership and motivation to project teams.
Further lessons and implications
The experiences from the Lilly MDR-TB Partnership provide several broad lessons. First is the need for long-term overall commitment. Drug development typically needs a horizon of a decade or longer, but local partners in public health initiatives also depend on long-term support, financial predictability, and commitment to overcome initial problems with new projects and to ensure that initiatives are kept running until reliable results can be obtained. This is not a new finding, but it remains difficult to achieve for some corporate organizations, where decisions can be driven by quarterly or annual reporting requirements. Corporate foundations with long-term horizons and budgets can balance this need.
A second lesson is that long-term commitment should be combined with a willingness to adapt and to drop unsuccessful approaches early, to avoid misallocating resources. For projects in the Lilly MDR-TB Partnership, this is embodied in the research and report components of RRA: small-scale pilots, assessed early, with results communicated to governments, health-policy experts, and other stakeholders. Openness and readiness to cancel projects are an essential prerequisite of the approach, since unsuccessful initiatives also provide lessons and may save money by avoiding misallocation of resources if evaluated at an early stage. Sharing lessons from even unsuccessful approaches can be valuable to others facing similar challenges.
Research-driven companies are familiar with failure: the vast majority of promising molecules fail to reach the stage of human trials, and most of those that do fail before the approval stage.23,24 If clear measures of success can be predefined, it is possible to winnow out unsuccessful approaches and free up resources for other pilots. This increases the willingness of sponsors to take risk, as money is less likely to be sunk into long-term failures. The Lilly MDR-TB Partnership attempted to apply this open mind-set to innovative projects suggested by partners, supporting a number of small pilot projects as proof-of-concept studies. The freedom to tinker with strategies and details gives partners an independence not always available for projects with strict guidance from funding bodies. The feedback from partners has been very positive. In the words of one Indian organization, the partnership provided “. . . the freedom to test things which others would not have allowed us to try”.
Finally, adopting a willingness to identify and address a series of major challenges that may occur in various locales – and to identify appropriate and able partners – can make meaningful global impact. The local projects in the Lilly MDR-TB Partnership have generated important contributions to the MDR-TB epidemic globally through both direct changes in communities where the partnership operates and through the dissemination of project findings to other TB stakeholders around the world.
Outlook
The projects described herein suggest that PPPs can be used effectively to pilot a number of small-scale projects with successful innovations presented to governments and public health providers for potential scaling up. As TB is a global problem, the involvement of global companies and foundations in PPPs can add value and increase the speed of dissemination of successes and lessons.
TB has been with humanity for thousands of years and will not disappear overnight. A willingness to try out new solutions and an openness to rapid exchange of information on projects are necessary mind-sets for both private and public stakeholders if the world is to reach the goal of freedom from TB within the foreseeable future.
Acknowledgment
The authors are grateful to Pelle Stolt for editorial help with this manuscript.
Disclosure
All authors are employees of Lilly Global Health Programs. The authors report no other conflicts of interest in this work.
References
World Health Organization. Global Tuberculosis Report 2015. 20th ed. Geneva: WHO; 2015. Available from: apps.who.int/iris/bitstream/10665/191102/1/9789241565059_eng.pdf. Accessed April 3, 2017. | ||
Dheda K, Barry CE, Maartens G. Tuberculosis. Lancet. 2016;387:1211–1226. | ||
Nathanson E, Nunn P, Uplekar M, et al. MDR tuberculosis: critical steps for prevention and control. N Engl J Med. 2010;363:1050–1058. | ||
World Health Organization. Multidrug-Resistant Tuberculosis (MDR-TB) Indicators: A Minimum Set of Indicators for the Programmatic Management of MDR-TB in National Tuberculosis Control Programmes. Geneva: WHO; 2010. Available from: http://whqlibdoc.who.int/hq/2010/WHO_HTM_TB_2010.11_eng.pdf. Accessed April 3, 2017. | ||
Lal SS, Uplekar M, Katz I, et al. Global Fund financing of public-private mix approaches for delivery of tuberculosis care. Trop Med Int Health. 2011;16:685–692. | ||
Wells WA, Uplekar M, Pai M. Achieving systemic and scalable private sector engagement in tuberculosis care and prevention in Asia. PLoS Med. 2015;12:e1001842. | ||
Nathan C. Cooperative development of antimicrobials: looking back to look ahead. Nat Rev Microbiol. 2015;13:651–657. | ||
Mundel T. Honing the priorities and making the investment case for global health. PLoS Biol. 2016;14:e1002376. | ||
Seeberg J, Pannarunothai S, Padmawati RS, Trisnantoro L, Barua N, Pandav CS. Treatment seeking and health financing in selected poor urban neighbourhoods in India, Indonesia and Thailand. Soc Sci Med. 2014;102:49–57. | ||
Sudha G, Nirupa C, Rajasakthivel M, et al. Factors influencing the care-seeking behaviour of chest symptomatics: a community-based study involving rural and urban population in Tamil Nadu, South India. Trop Med Int Health. 2003;8:336–341. | ||
Fochsen G, Deshpande K, Diwan V, Mishra A, Diwan VK, Thorson A. Health care seeking among individuals with cough and tuberculosis: a population-based study from rural India. Int J Tuberc Lung Dis. 2006; 10:995–1000. | ||
Gautham M, Binnendijk E, Koren R, Dror DM. “First we go to the small doctor”: first contact for curative health care sought by rural communities in Andhra Pradesh and Orissa, India. Indian J Med Res. 2011;134:627–638. | ||
Russian Academy of Medical Science [webpage on the Internet]. Drug-resistant tuberculosis in the Russian Federation. 2011; Available from: http://www.ncbi.nlm.nih.gov/books/NBK62453. Accessed April 25, 2016. | ||
Gelmanova IY, Keshavjee S, Golubchikova VT, et al. Barriers to successful tuberculosis treatment in Tomsk, Russian Federation: non-adherence, default and the acquisition of multidrug resistance. Bull World Health Organ. 2007;85:703–711. | ||
Barrera E, Livchits V, Nardell E. F-A-S-T: a refocused, intensified, administrative tuberculosis transmission control strategy. Int J Tuberc Lung Dis. 2015;19:381–384. | ||
Gandhi NR, Moll A, Sturm AW, et al. Extensively drug-resistant tuberculosis as a cause of death in patients co-infected with tuberculosis and HIV in a rural area of South Africa. Lancet. 2006;368:1575–1580. | ||
Schnippel K, Rosen S, Shearer K, et al. Costs of inpatient treatment for multi-drug-resistant tuberculosis in South Africa. Trop Med Int Health. 2013;18:109–116. | ||
Gauri A [webpage on the Internet]. Government ropes in pharmacists to check tuberculosis. 2014. Available from: http://timesofindia.indiatimes.com/city/indore/Government-ropes-in-pharmacists-to-check-tuberculosis/articleshow/40369653.cms. Accessed April 3, 2017. | ||
Loveday M, Wallengren K, Brust J, et al. Community-based care vs. centralised hospitalisation for MDR-TB patients, KwaZulu-Natal, South Africa. Int J Tuberc Lung Dis. 2015;19:163–171. | ||
Loveday M, Padayatchi N, Wallengren K, et al. Association between health systems performance and treatment outcomes in patients co-infected with MDR-TB and HIV in KwaZulu-Natal, South Africa: implications for TB programmes. PloS One. 2014;9:e94016. | ||
Arasaratnam A, Humphreys G. Emerging economies drive frugal innovation. Bull World Health Organ. 2013;91:6–7. | ||
Cox H, Ramma L, Wilkinson L, Azevedo V, Sinanovic E. Cost per patient of treatment for rifampicin-resistant tuberculosis in a community-based programme in Khayelitsha, South Africa. Trop Med Int Health. 2015; 20:1337–1345. | ||
Hughes J, Rees S, Kalindjian S, Philpott K. Principles of early drug discovery. Br J Pharmacol. 2011;162:1239–1249. | ||
Morgan S, Grootendorst P, Lexchin J, Cunningham C, Greyson D. The cost of drug development: a systematic review. Health Policy. 2011;100:4–17. | ||
Loveday M. Cost-effectivess of different models of care for MDR-TB patients in South Africa. Presented at: the 68th session of the World Health Assembly; May 20th, 2015; Geneva, Switzerland |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


