Back to Journals » Journal of Multidisciplinary Healthcare » Volume 13
Cognitive Factors Associated to Pediculosis Preventive Behaviors Among Mothers of School-Age Children in Chaldoran County, Iran
Authors Nezhadali A, Babazadeh T , Nadrian H , Allahverdipour H
Received 2 November 2019
Accepted for publication 20 December 2019
Published 10 January 2020 Volume 2020:13 Pages 19—26
DOI https://doi.org/10.2147/JMDH.S237101
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Ali Nezhadali, 1 Towhid Babazadeh, 2 Haidar Nadrian, 3 Hamid Allahverdipour 4
1Department of Health Education and Promotion, Faculty of Health Sciences, Tabriz University of Medical Sciences, Tabriz, Iran; 2Department of Public Health, Sarab Faculty of Medical Sciences, Sarab 14711, Iran; 3Department of Health Education & Promotion, Tabriz University of Medical Sciences, Tabriz 14711, Iran; 4Research Center of Psychiatry and Behavioral Sciences, Department of Health Education & Promotion, Tabriz University of Medical Sciences, Tabriz 14711, Iran
Correspondence: Hamid Allahverdipour
Health Education & Promotion Research Center of Psychiatry and Behavioral Sciences, Department of Health Education & Promotion, Tabriz University of Medical Sciences, Tabriz 14711, Iran
Tel/Fax +98-411-3344731
Email [email protected]
Background: Despite the improvement of living standards and public health as well as conducting the specific programs based on pediculosis control guidelines, Head Lice Infestation (HLI) is still a worldwide health concern. The purpose of this study was to investigate the cognitive factors associated to school-age children’s HLI among mothers in an urban community.
Subjects and Methods: In this cross-sectional study, a multi-stage cluster random sample of 619 mothers of students in elementary schools of Chaldoran County, located in North-West of Iran, was recruited to participate in the study. Data were collected applying a set of questionnaires including socio-demographic and cognitive-behavioral factors, including Pediculosis Preventive Behaviors (PPBs), HLI knowledge, threat appraisal, as well as HLI prevention perceived self-efficacy, response efficacy, and perceived collective family efficacy.
Results: Statistically significant differences were found in the children’s HLI by their mother’s age and performance in PPBs (p-value=0.001). Hierarchical multiple linear regressions were performed with PPBs as outcome variable. According to their natures, the predictors for this outcome variable were classified in two different blocks. Demographic characteristics (p > 0.05), and cognitive constructs (p< 0.001) explained 9% and 21.1% of the observed variance in PPBs, respectively. Pediculosis was found as an epidemic among the students, particularly female students, of Chaldoran County, which call for action by health stakeholders and policymakers.
Conclusion: The healthcare providers are recommended to develop HLI prevention programs, within which the mothers and their school-age children as well as school mangers and public places’ employers are considered as the core target groups for interventions. Besides PPBs, response efficacy and perceived collective family efficacy to perform the behaviors should be considered as the core categories while designing such prevention programs.
Keywords: pediculosis, family health, school health, head lice, collective efficacy
Introduction
Head Lice Infestation (HLI) is a worldwide health concern, affecting people of all ages with different socioeconomic status, particularly school-aged children.1,2 HLI might cause distress and anxiety in children and other family members, and impose additional costs on the family and the health system.3,4 It has also social and psychological negative consequences, and may lead to failure in academic performance.5
According to reports, the occurrence of HLI is varied in different populations. In the past two decades, the reported prevalence has a variation from 1% to 20% in the Europe.6 In developing countries, however, it has risen to over 40% among children.7 The rate of infestation is reported to be 1.2% in Turkey,8 4.8% in the Netherlands,9 35% in Brazil,9 29.7% in Argentina10 and 28.8% in Venezuela.11 In Iran, evidence has indicated the prevalence rate of 8.8% and the female to male gender ratio of 5:5, respectively.12 In a previous study by Ziaoddini et al13 in west Azerbaijan, the prevalence rate was reported to be 9.11%. The high level of HLI prevalence may be associated to the incorrect use of the pediculicides, non-adherence to treatment, lack of HLI diagnosis,14 and reduction in the efficiency of Permethrin shampoo from 97% in the 1990s to 30% in 2010.15
Numerous factors are associated with the increasing rate of HLI including high population density; insufficient health facilities, lack of healthcare providers in school, not washing hair regularly,16 the duration of HLI, and the nature of “hair to hair” contact.17 The most common age group infested with lice is 3- to 12-year-old children. Moreover, one of the possible reasons is related to culture and social norms of each community.18 Therefore, understanding the social context of populations can be beneficial in exploring the predisposing factors of pediculosis.19
Several cognitive-behavioral and environmental factors may also act as obstacles to control the HLI. Understanding and recognizing these barriers may be also helpful in HLI prevention programs.20 In this regard one of the factors that may affect the control of HLI is reported to be the perception of family members about pediculosis and its preventative behaviors.21 Additionally, unfavorable socioeconomic status, population congestion, lower living standards, poverty status, poor health, and gender are reported as contributing factors. In Iran, in recent decades, the infestation rate has been increasing due to uncontrolled population growth, migration to cities, and marginalization around the cities which all have resulted in the creation of small communities with inadequate health facilities.22
Despite the progress of healthcare programs in Iran and specific programs based on the pediculosis control guidelines, head lice are common and have progressive prevalence among school-age children. Our aim in the present study was to examine the associations between cognitive factors and PPBs among mothers of school age children in Chaldoran County, Iran.
Subjects and Methods
Participants and Sampling
This cross-sectional study was carried out among 619 mothers with school-aged children in Chaldoran County, North-West of Iran from September to October, 2017. Multistage random sampling was employed to recruit participants in the study. In the first step, from the 12 primary schools in the County, four schools with 739 students were randomly selected, and then the mothers of the students were invited to participate in the study. G*Power software was used to estimate the required sample size based on the following parameters: level of confidence = 95%, power = 80%, correlation coefficient = 0.145, and a design effect = 2. The required sample size was estimated to be about 739. In total, 619 mothers participated in the study and all mothers completed the written questionnaire (Response Rate = 83.7%). The participants aged from 20 to 40 years and the mean age of mothers was 34, SD = 5.85. Via phone calls, the mothers were invited to participate in an initial session in the schools. In a private room in the schools, they were provided with the questionnaires in a self-report manner.
This study was approved by Ethics Committee in Tabriz University of Medical Sciences (Ethics Code: IR.TBZMED.REC.1396.600). Informed written consent was obtained from all the participants prior to formal commencement of data collection.
Measures
In order to collect data, a researcher-made questionnaire based on cognitive-behavioral constructs was developed. The questionnaire was prepared by reviewing other questionnaires applied in similar studies.23–27 Validity of the questionnaires was assessed by an expert panel (six health educationists, a psychologist, an entomologist and an epidemiologist). In order to assess reliability, a pilot study was performed on 20 mothers not included in the final sample. A brief description of the questionnaire is as follows:
Demographic Characteristics
The following demographic data were obtained: age of mothers, educational grade of their children, gender and birth rank of their child, job, educational status, the frequency of bathing the child per week, the frequency of combing the hair of child per day, using personal health accessories on their children (yes/no), using personal equipment to cut the child’s hair (yes/no), regular cleansing of the child’s comb (yes/no), having children with shared sheet blanket (yes/no), having children with shared toys (yes/no), hair length of the child (perfectly shaved, less than 1cm, up to the ear, long as the hair comes to the child’s shoulders, long as the hairs come to the child’s back), history of lice infestation in the child in the last three months (yes/no), the history of lice infestation among family members in the previous season (yes/no), using anti-lice shampoo in the case of lice infestation among family members in the previous season (yes or no), the history of participation in lice infestation educational programs in the previous season (yes or no).
Cognitive-Behavioral Constructs
The mothers’ knowledge was measured using a fourteen-item scale on the ways of transmission (e.g., Head lice can be transferred from animals such as cats and birds), identification (e.g., Head lice are most often found in the nape of neck), prevention (e.g., in case of HLI, the whole house should be sprayed) and treatment of head lice (e.g., to treat HLI, the anti-lice shampoo should be used twice a week). A three-choice answer (Yes, No, and I do not know) was considered as a response format. The Cronbach alpha for the scale was 0.735.
Threat Appraisal About Infestation by Head Lice
The items about threat appraisal (thirteen items) were prepared with a Likert scale as the response format. Given the 5-point scale, the score of each descriptor ranged from 0 to 4 (from strongly disagree to strongly agree). Perceived susceptibility was assessed with six items (e.g., the likelihood of infestation with head lice is high in the school environment). The total score ranged from 0 to 24. Cronbach’s alpha for this scale was 0.811. Perceived severity (seven items) was investigated with seven items (e.g., HLIcauses a fall in my child’s educational status). The theoretical range for this scale was 0 to 28. The Cronbach alpha was found to be 0.816.
Parental Perceived Self-Efficacy of HLI Prevention
This scale included seven items assessing parent’s beliefs about their abilities: 1) to diagnose lice and nits (e.g., I can distinguish lice from other insects); 2) to manage and treat head lice in their children (e.g., I can treat head lice if my child affected by it); 3) to get information from different sources about head lice; and 4) to plan for their child, to combing hair and bathing regularly. Participants were asked to express their perceived self-efficacy using a scale that ranged from 0 to 4 (from strongly disagree to strongly agree). Cronbach alpha was 0.829.
Response Efficacy of Preventive Guidelines
This scale also included seven items reviewing the mother’s perception about the efficiency of preventive methods to eliminate head lice; 1) individual behaviors such as combing and cutting hair (combing hair a few times a day may prevent lice infestation); 2) appropriate treatment to prevent the onset of others; 3) performing recommendations of health care providers; and 4) participating in training programs in order to prevent of lice. Mothers had five options to express their views on this scale (ranging from 0 = strongly disagree to 4 = strongly agree). The Cronbach alpha was 0.872.
Collective Efficacy at the Family to Control Head Lice
This scale included seven items that measured the perceptions of families on their ability: 1) to manage new challenges in participation of all family members to prevent head lice; 2) to provide emotional support in difficult times; 3) to support each other to overcome head lice in family level; and 4) to have mutual trust to solve the problem (e.g., all of family members help each other if one of them is infested by head lice, or e.g., my child tells me if she/he would be infested with head lice). To express their perceived family efficacy beliefs, participants were provided with the same 5-point response format (ranging from 0 = strongly disagree to 4 = strongly agree). The Cronbach alpha for this scale was 0.901.
Head Lice Detecting Behaviors
The behavioral construct included eight items about mother’s performance when dealing with head lice. A dichotomous response format (Yes/No) was considered to answer the questions. The behaviors included the following items: checking the child’s hair for infestation; encouraging the child to comb her/his hair every day; cutting the child's hair, ironing the child’s clothes regularly; encouraging the child to clean her/his comb daily; encouraging the child to have a bath at least once a week, participating in educational programs at school about pediculosis prevention, and understanding the transmission and prevention methods of pediculosis.
Statistical Analysis
Data analysis was carried out using SPSS ver. 21. Descriptive statistics including frequency, mean and standard deviation, as well as the Pearson correlation coefficient test, multiple linear regressions with Inter method was used to analyze the data. The level of significance was considered to be 0.05, a priori.
Results
A majority of participants (88.8%) were housewives and 244 (39.4%) mothers were illiterate. No statistically significant difference was found in demographic variables by Pediculosis infestation. As shown in Table 1, a statistically significant difference was found in the HLI rate by the age of children’s mothers (p-value=0.001). The HLI rate was significantly lower among students who use personal accessories for cutting hair (P-value=0.001), and use a personal mattress at home (p-value=0.024), compared to their counterparts. Ten percent of mothers reported that their child has a history of infestation with lice. In terms of PPBs, 221 (35.8%) participants reported that they have never used personalaccessories when cutting their child’s hair, among which 35 (59.3%) of all Pediculosis infestations were identified. Additionally, only 169 (27.1%) participants reported children’s personal mattresses were used at home (Table 1).
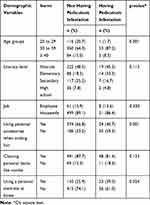 |
Table 1 The Relationship Between Demographic Variables with Having or Not Having Pediclousis Infestation (n = 619) |
Table 2 shows a comparison of PPBs between mothers with and without the history of Pediculosis infestation in their children. The frequency of regularly checking the child’s hair for HLI was 42 (71.2%) versus 329 (58.8%), and regular ironing of the child’s clothes (such as scarf, uniform, hats, etc.) was 35 (59.3%) versus 425 (75.9%) and was higher among mothers with non-infested children.
 |
Table 2 Comparison of Mother’s Behaviors in Last Month Among Infested and Non-Infested Children (n = 619) |
Table 3 shows bivariate correlations between the theoretical variables and PPBs. Applying a Pearson correlation coefficient test, statistically significant positive correlations were found between PPBs and all theoretical variables (Table 3). Hierarchical multiple linear regressions were performed on theoretical variables and PPBs, as the outcome variable.
 |
Table 3 Bivariate Correlations of Variables and PPBs |
As shown in Table 4 (step 1), all demographic and underlying variables were significant predictors of PPBs (p < 0.05, R2 total = 0.093), except for using a personal mattress at home and the mother’s job. In step 2, when the cognitive variables of the model were included in the model, age (p = 0.021), using personal accessories when cutting hair (p = 0.021), cleaning personal items like combs (p = 0.021), using a personal mattress at home (p = 0.021), the number of children with lice infestation (p = 0.021), response efficacy (p = 0.021), and perceived collective family efficacy (p = 0.01) were significant predictors of PPBs (R2 = 0.211). Among all variables, response efficacy and perceived collective family efficacy were the strongest predictors of PPBs.
 |
Table 4 Hierarchical Regression Analysis to Predict PPBs |
Discussion
Our aim was to investigate the cognitive-behavioral factors associated with head lice prevention among mothers with school-age children. Based on the findings, approximately 10% of families had children infested by head lice. Such a high prevalence rate of pediculosis is considered as an epidemic among the students studied, according to the definition provided by Centers for Disease Control and Prevention.28 Similar with several previous studies,29–31 the prevalence rate of pediculosis among the families with female school-age children was significantly higher than among their counterparts. A reason for this difference may be associated with the socio-cultural and religious factors. In some Islamic countries, like Iran, using headscarves (“hijab”) is obligatory for female students in the schools. Such head coverings predispose the female students, particularly the elementary students in the areas with high prevalence rates, to be infested by head lice. On the other hand, lice infestation is a social stigma in Iranian communities, and thus, mothers feel ashamed to announce their children’s infestation (particularly their daughters). All these problems may result in late diagnosis and treatment, and consequently escalating the distribution of infestation within the communities.
The results of our study showed that the cognitive and behavioral factors, altogether predicted 21% of total variance in PPBs. Among all variables, perceived collective family efficacy was the strongest significant predictor of the outcome variable. Perceived collective efficacy in families could act as a protective factor during crisis and stressful situations.32 In other words, efficacy among family members can provide a sense of effort and perseverance for conquering the problem.33 Therefore, improving collective efficacy within families may be considered an essential strategy to promote lice infestation preventive behaviors. In this regard, Lindeke et al (2018) declared that strengthening the power of families, results in better cooperation during their decision-making, which, consequently, may be helpful in protecting their children’s health.34 It seems that when family members have a comprehensive recognition of the problems suffered or have sick members of the family, they will try to improve his/her health and provide family members with more protection. In the instance that family members provide a supportive and protective environment for a lice infested individual, he/she may not be worried about expressing their problem with the family members. Therefore it is recommended that healthcare providers pay more attention to a family's abilities to solve health problems when designing pediculosis prevention programs.
Based on the findings of our study, the HLI rate in children with older mothers was more common than those in children with younger mothers. No evidence was found to explain why the children of older mothers are infested more when compared to their counterparts. In this regard, Rajabi et al (2013), in a study on children with asthma, showed that younger mothers provided a better quality of life for their children.35 As an explanation for this association we can say that by increase in the age of mother, an increase may also happen in the age gap between mother and child, and in the number of children that a mother may have, which may consequently decrease parent–child interactions or parent's attention to children’s health. Additionally, an older mother may not have sufficient time or ability, or good physical health to nurture the child, so she may unconsciously ignore some childcare responsibilities, such as personal health care.
Our results also indicated that HLI was significantly lower among the students who use personal equipment when caring for their hair (such as personal brush, scissors, etc.). Similar results were reported by Alborzi et al (2004) who studied the factors that determine head lice prevalence among elementary students.36 Honsen (2004), also in a study on the status of head lice control and management, emphasized personal health, including the use of personal belongings, as a substantial strategy to control lice infestation.37 In the case of only one HLI among individuals in a public place, using shared items (such as blanket, sheets, brushes, toys, clothes) may result in spreading the infestation among other members of the community. This claim has been also reported in several previous studies on rural students in Iran,38 and Jordan39 as well as a rapid review by Heukelbach and Feldmeier (2004).40 Therefore, in the settings with high prevalence rates of lice infestation, a community mobilization approach should be considered while planning HLI prevention programs, in a way that not only the family members of those infested are involved but also the managers and employers of public places such as hairdressing saloons and boarding dorms participate.
In addition, there was a significant association between ironing cloths and HLI in the present study. Similarly, Dehghanzadeh et al (2015) in a study conducted in the northwest of Iran, showed a high contribution between ironing cloths and the prevalence of head lice.41 This behavioral strategy may be proposed while family education programs are implemented in epidemic, endemic and hyper-endemic areas. For successful application of this strategy, it has been recommended to wash clothes for 20 mins in warm water (water and salt or chloride in water), and then ironing them.42
Our findings showed that the hair of lice-infested students were less frequently checked by their mothers, compared to their counterparts. In another Iranian study, the regular checks of daughters’ hair was not performed by their mothers.43 Riabi et al (2012) also found a significant association between the lack of regular hair check of daughters by mothers and HLI in the children.44 Previous studies have emphasized that parents should be more active than formal screening programs in recognition of lice infestation in their children.45,46 So, mothers should obtain the required ability to recognize head lice and nits, and should perform mechanical actions, like regular combing, to identify and remove any possible lice and nits, and prevent the infestation to be distributed among children and other family members.
Limitation
The data collection method was a self-report by mothers, thus recall bias is warranted. Also, we studied the predictors of PPBs in one urban area of Iran and thus our findings may have limited generalizability to other regions and cultures.
Conclusion
Our findings showed pediculosis as an epidemic among the students, particularly female students, of Chaldoran County, which call for action by health stakeholders and policymakers in Iran. The healthcare providers are recommended to develop HLI prevention programs, within which the mothers and their school-age children, as well as school mangers and public place employers, are considered as the core target groups for interventions. We found that response efficacy and perceived collective family efficacy are remarkably associated with PPBs in mothers. These findings indicated that response efficacy and perceived collective family efficacy to perform the behaviors should be considered as the core categories while designing such prevention programs. Community mobilization for fostering public cooperation across public and private sectors, including the families, may be considered as an effective strategy to improve partnership between the families and the healthcare providers and to strengthen community participation in sustainable pediculosis prevention and control.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethical approval to perform the study was obtained from the Ethics Committee in Tabriz University of Medical Sciences.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Acknowledgment
Authors would like to thank all personnel of Health vice-chancellery of Tabriz University of Medical Sciences for their cooperation.
Disclosure
The authors declare that they have no conflicts of interest regarding this study.
References
1. McNair CM. Ectoparasites of medical and veterinary importance: drug resistance and the need for alternative control methods. J Pharm Pharmacol. 2015;67(3):351–363. doi:10.1111/jphp.2015.67.issue-3
2. Durand R, Bouvresse S, Berdjane Z, Izri A, Chosidow O, Clark J. Insecticide resistance in head lice: clinical, parasitological and genetic aspects. Clin Microbiol Infect. 2012;18(4):338–344. doi:10.1111/j.1469-0691.2012.03806.x
3. Motovali-Emami M, Aflatoonian MR, Fekri A, Yazdi M. Epidemiological aspects of Pediculosis capitis and treatment evaluation in primary-school children in Iran. Pak J Biol Sci. 2008;11(2):260–264. doi:10.3923/pjbs.2008.260.264
4. Frankowski BL, Bocchini JA, Health CoS, Diseases CoI. Head lice. Pediatrics. 2010;126(2):392–403. doi:10.1542/peds.2010-1308
5. Doroodgar A, Sadr F, Sayyah M, Doroodgar M, Tashakkor Z, Doroodgar M. Prevalence and associated factors of head lice infestation among primary schoolchildren in city of Aran and Bidgol (Esfahan Province, Iran), 2008. Payesh J. 2011;10(4):439–e447.
6. Feldmeier H. Pediculosis capitis: new insights into epidemiology, diagnosis and treatment. Eur J Clin Microbiol Infect Dis. 2012;31(9):2105–2110. doi:10.1007/s10096-012-1575-0
7. Lesshafft H, Baier A, Guerra H, Terashima A, Feldmeier H. Prevalence and risk factors associated with pediculosis capitis in an impoverished urban community in Lima, Peru. J Glob Infect Dis. 2013;5(4):138. doi:10.4103/0974-777X.121994
8. Ciftci IH, Karaca S, Dogru O, Cetinkaya Z, Kulac M. Prevalence of pediculosis and scabies in preschool nursery children of Afyon, Turkey. Korean J Parasitol. 2006;44(1):95. doi:10.3347/kjp.2006.44.1.95
9. Borges R, Mendes J. Epidemiological aspects of head lice in children attending day care centres, urban and rural schools in Uberlandia, central Brazil. Memórias do Instituto Oswaldo Cruz. 2002;97(2):189–192. doi:10.1590/S0074-02762002000200007
10. Toloza A, Vassena C, Gallardo A, González-Audino P, Picollo MI. Epidemiology of Pediculosis capitis in elementary schools of Buenos Aires, Argentina. Parasitol Res. 2009;104(6):1295–1298. doi:10.1007/s00436-008-1324-6
11. Cazorla D, Ruiz A, Acosta M. Clinical and epidemiological study of pediculosis capitis in schoolchildren from Coro, Venezuela. Invest Clin. 2007;48(4):445–457.
12. Moosazadeh M, Afshari M, Keianian H, Nezammahalleh A, Enayati AA. Prevalence of head lice infestation and its associated factors among primary school students in Iran: a systematic review and meta-analysis. Osong Public Health Res Perspect. 2015;6(6):346–356. doi:10.1016/j.phrp.2015.10.011
13. Ziaoddini A, Riahi R, Heidari-Beni M, Ziaoddini H, Zamani S. National and provincial prevalence of Pediculus humanus capitis among Urban Students in Iran from 2014 to 2018. J Res Health Sci. 2019;19:4.
14. Burgess IF. Human lice and their control. Annu Rev Entomol. 2004;49.
15. Burgess IF, Kay K, Burgess NA, Brunton ER. Soya oil-based shampoo superior to 0.5% permethrin lotion for head louse infestation. Med Devices (Auckland, NZ). 2011;4:35.
16. Buczek A, Markowska-Gosik D, Widomska D, Kawa IM. Pediculosis capitis among schoolchildren in urban and rural areas of eastern Poland. Eur J Epidemiol. 2004;19(5):491–495. doi:10.1023/B:EJEP.0000027347.76908.61
17. Rukke BA, Birkemoe T, Soleng A, Lindstedt HH, Ottesen P. Head lice prevalence among households in Norway: importance of spatial variables and individual and household characteristics. Parasitology. 2011;138(10):1296–1304. doi:10.1017/S0031182011001004
18. Heukelbach J, Ugbomoiko US. Knowledge, attitudes and practices regarding head lice infestations in rural Nigeria. J Infect Devel Countries. 2011;5(09):652–657. doi:10.3855/jidc.1746
19. Redman-MacLaren M, MacLaren DJ, Harrington H, et al. Mutual research capacity strengthening: a qualitative study of two-way partnerships in public health research. Int J Equity Health. 2012;11(1):79. doi:10.1186/1475-9276-11-79
20. Hamidzadeh Y, Hashemiparast M, Hassankhani H, Allahverdipour H. Local-level challenges to implementing health education programs in rural settings: a qualitative study. Fam Med Prim Care Rev. 2019;21(1):30–34. doi:10.5114/fmpcr.2019.82976
21. Alhani F. Design and Evaluation of Family-Centered Empowerment Model to Prevent Iron Deficiency Anemia. Tehran: Tarbiat modarres University; 2003.
22. Vahabi A, Shemshad K, Sayyadi M, et al. Prevalence and risk factors of Pediculus (humanus) capitis (Anoplura: pediculidae), in primary schools in Sanandaj City, Kurdistan Province, Iran. Trop Biomed. 2012;29(2):207–211.
23. Madke B, Khopkar U. Pediculosis capitis: an update. Ind J Dermatol Venereol Leprol. 2012;78(4):429. doi:10.4103/0378-6323.98072
24. ALBashtawy M. Knowledge, attitudes, and practices of parents/guardians regarding pediculosis in the Umm el-Jimal district of Jordan. J Res Nurs. 2014;19(5):390–399. doi:10.1177/1744987112465882
25. Costa M, Faria L, Alessandri G, Caprara GV. Measuring parental and family efficacy beliefs of adolescents’ parents: cross‐cultural comparisons in Italy and Portugal. Int J Psychol. 2016;51(6):421–429. doi:10.1002/ijop.2016.51.issue-6
26. Madeira NG, de Souza PAT, da Silva Diniz RE. Perception and action of teachers and head lice in school. Revista Electrónica de Enseñanza de las Ciencias. 2015;14(2):119–130.
27. Sidoti E, Bonura F, Paolini G, Tringali G. A survey on knowledge and perceptions regarding head lice on a sample of teachers and students in primary schools of north and south of Italy. J Prev Med Hyg. 2009;50:3.
28. Nutanson I, Steen C, Schwartz R, Janniger C. Pediculus humanus capitis: an update. Acta Dermatovenerol Alp Panonica Adriat. 2008;17(4):147–159.
29. Doroodgar A, Sadr F, Doroodgar M, Doroodgar M, Sayyah M. Examining the prevalence rate of Pediculus capitis infestation according to sex and social factors in primary school children. Asian Pac J Trop Dis. 2014;4(1):25–29. doi:10.1016/S2222-1808(14)60308-X
30. Al-Maktari M. Head louse infestations in Yemen: prevalence and risk factors determination among primary schoolchildren, Al-Mahweet Governorate, Yemen. J Egypt Soc Parasitol. 2008;38(3):741–748.
31. El-Sayed MM, Toama MA, Abdelshafy AS, Esawy AM, El-Naggar SA. Prevalence of pediculosis capitis among primary school students at Sharkia Governorate by using dermoscopy. Egypt J Dermatol Venerol. 2017;37(2):33. doi:10.4103/ejdv.ejdv_47_16
32. Pepe S, Sobral J, Gómez-Fraguela JA, Villar-Torres P. Spanish adaptation of the adolescents’ perceived collective family efficacy scale. Psicothema. 2008;20:1.
33. Fulgencio AB, David AP. Reliability and validity of a measure of perceived collective family efficacy beliefs for Filipino adolescents. Asian J Social Sci Humanities. 2013;2(3):443–450.
34. Lindeke LL, Leonard BJ, Presler B, Garwick A. Family-centered care coordination for children with special needs across multiple settings. J Pediatr Health Care. 2002;16(6):290–297. doi:10.1016/S0891-5245(02)00063-9
35. Rajabi R, Sabzevari S, Borhani F, Haghdoost A, Bazargan N. The effect of family-centered empowerment model on quality of life school age children with asthma. J Health Promotion Manage. 2013;2(4):7–15.
36. Alborzi M, Shekarriz-Foumani R. The prevalence of Pediculus Capitis among primary schools of Shahriar County, Tehran province, Iran, 2014. Novelty Biomed. 2016;4(1):24–27.
37. Hansen RC. Overview: the state of head lice management and control. Am J Manag Care. 2004;10(9 Suppl):S260–263.
38. Noori A, Ghorban Pour M, Adib M, Noori AV, Niazi S. Head lice infestation (Pediculosis) and its associated factors in the rural school students of Kalaleh, in the academic 1392–93. Jorjani Biomed J. 2014;2(1):56–60.
39. Al Bashtawy M, Hasna F. Pediculosis Capitis among Primary-School Children in Mafraq Governorate, Jordan. East Mediterr Health J. 2012; 18(1):43–8.
40. Heukelbach J, Feldmeier H. Ectoparasites—the underestimated realm. The Lancet. 2004;363(9412):889–891. doi:10.1016/S0140-6736(04)15738-3
41. Dehghanzadeh R, Asghari-Jafarabadi M, Salimian S, Hashemi AA, Khayatzadeh S. Impact of family ownerships, individual hygiene, and residential environments on the prevalence of pediculosis capitis among schoolchildren in urban and rural areas of northwest of Iran. Parasitol Res. 2015;114(11):4295–4303. doi:10.1007/s00436-015-4670-1
42. Eisenhower C, Farrington EA. Advancements in the treatment of head lice in pediatrics. J Pediatr Health Care. 2012;26(6):451–461. doi:10.1016/j.pedhc.2012.05.004
43. Goodarzvand CP, Anoosheh M, Kazemnejad A The effectiveness of educating mothers on preventive behaviors of pediculosis on morbidity rate of their daughters. 2017.
44. Riabi HRA, Atarodi A. Epidemiological and clinical study of infested cases with pediculus capitis and P. corporis in Khorasan-e-Razavi, Iran. Iran J Parasitol. 2012;7(1):85.
45. Stafford G. Head lice: evidence-based guidelines based on the Stafford Report. J Fam Health Care. 2002;12(5 Suppl):1.
46. Mumcuoglu KY, Barker SC, Burgess I, et al. International guidelines for effective control of head louse infestations. J Drugs Dermatol. 2007;6(4):409–414.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
