Back to Journals » Clinical Ophthalmology » Volume 9
Clinical outcomes of wavefront-guided laser in situ keratomileusis to treat moderate-to-high astigmatism
Authors Schallhorn S, Venter J, Hannan S, Hettinger K
Received 4 May 2015
Accepted for publication 17 June 2015
Published 13 July 2015 Volume 2015:9 Pages 1291—1298
DOI https://doi.org/10.2147/OPTH.S87887
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Scott Fraser
Steven C Schallhorn,1,2 Jan A Venter,2 Stephen J Hannan,2 Keith A Hettinger2
1University of California, San Francisco, CA, USA; 2Optical Express, Glasgow, UK
Purpose: The purpose of this study was to evaluate the refractive and visual outcomes of wavefront-guided laser in situ keratomileusis (LASIK) in eyes with myopic astigmatism and cylindrical component ≥2.0 diopter (D).
Methods: In this retrospective study, 611 eyes that underwent LASIK for simple or compound myopic astigmatism were analyzed. Preoperative refractive cylinder ranged from -2.00 D to -6.00 D (mean -2.76±0.81 D), and the sphere was between 0.00 D and -9.75 D (mean -2.79±2.32 D). Predictability, visual outcomes, and vector analysis of changes in refractive astigmatism were evaluated.
Results: At 3 months after LASIK, 83.8% of eyes had uncorrected distance visual acuity of 20/20 or better, 90.3% had manifest spherical equivalent within ±0.50 D, and 79.1% had residual refractive cylinder within ±0.50 D of intended correction. The mean correction ratio for refractive cylinder was 0.92±0.14, the mean error of angle was -0.45°±2.99°, and the mean error vector was 0.37±0.38 D. A statistically significant correlation was found between the error of magnitude (arithmetic difference in the magnitudes between surgically induced refractive correction and intended refractive correction) and the intended refractive correction (r=0.26, P<0.01).
Conclusion: Wavefront-guided LASIK for the correction of myopic astigmatism is safe, effective, and predictable.
Keywords: myopic astigmatism, wavefront-guided LASIK, Hartmann–Shack aberrometer
Introduction
Refractive errors with some degree of astigmatism are relatively frequent. In a recent population-based study, 32.3% of adult participants were found to have refractive cylinder of >0.50 diopter (D).1 However, the prevalence of moderate or high degree of astigmatism is significantly lower, with as little as 4.61% of patients having cylindrical error of 2.50 D or more.2 Surgical correction of astigmatism by means of an excimer laser ablation is technically more difficult and less effective than the treatment of plain spherical refraction.3 Studies reporting outcomes of moderate-to-high astigmatic treatment are relatively scarce,4–9 often combine different degrees of preoperative refractive cylinder, or lack rigorous analysis of vector parameters of astigmatic refraction.
Several factors can play a role in the inaccuracy of astigmatic surgical correction with excimer lasers. These include precise determination of preoperative magnitude and axis of refractive cylinder, the technology involved in adequate centration of the elliptical ablation profile during surgery,10–13 as well as understanding the biomechanics of corneal healing14 and incorporating it into the treatment nomogram. In this study, we evaluated the results of wavefront-guided (WFG) laser in situ keratomileusis (LASIK) in a large cohort of patients with simple or compound myopic astigmatism and refractive cylinder ≥2.00 D. To our knowledge, this is the first study reporting WFG excimer laser astigmatic correction for such large dataset.
Patients and methods
This retrospective, noncomparative study was deemed exempt from full review by the Committee of Human Research at the University of California, San Francisco, because it used only retrospective, de-identified patient data. Informed consent to undergo LASIK procedure was obtained from all patients.
Data were extracted from electronic database using the following criteria: primary LASIK procedures targeted for emmetropia; successful completion of 1 week, 1 month, and 3 months postoperative exam; preoperative simple or compound myopic astigmatism between 2.0 D and 6.0 D with no more than manifest spherical equivalent (MSE) of −12.0 D; surgeries performed with the VISX STAR S4 IR excimer laser (Abbott Medical Optics Inc., Santa Ana, CA, USA) using a WFG ablation profile derived from the iDesign Advanced WaveScan System (Abbott Medical Optics Inc.); visual acuity correctable to 20/32 or better prior to LASIK in both eyes; and age of 18 years or older. Data extraction techniques have been previously described.15
Exclusion criteria were active ophthalmic diseases, abnormal corneal shape, concurrent medications, or medical conditions that could impair healing of the ocular surface, and calculated postoperative corneal stromal bed thickness <250 μm in each eye. Soft contact lens wearers were asked to discontinue use at least 1 week prior to the procedure. Hard contact lens users (polymethylmethacrylate or rigid gas-permeable lenses) removed their lenses at least 3 weeks prior to baseline measurements and had two central keratometry readings and two manifest refractions taken at least 1 week apart that did not differ by >0.50 D in either meridian.
The preoperative ophthalmic examination included manifest and cycloplegic refraction, monocular and binocular uncorrected distance visual acuity (UDVA), corrected distance visual acuity (CDVA) using a calibrated projected eye chart, low-light pupil diameter, slit lamp biomicroscopy, dilated fundus examination, applanation tonometry, corneal topography, ultrasound pachymetry, and wavefront aberration measurement.
Postoperative visits were scheduled for 1 day, 1 week, 1 month, and 3 months. On the first postoperative visit, a detailed slit lamp examination was performed to evaluate flap position and the integrity of the cornea. At the remaining visits, manifest refraction, UDVA, CDVA, and slit lamp examinations were performed.
Surgery
All LASIK procedures were performed by experienced surgeons. Superior-hinge flaps were created by a femtosecond laser (iFS, Abbott Medical Optics Inc.). The diameter of the femtosecond flaps ranged from 8.0 mm to 9.2 mm, and the programmed depth ranged from 100 μm to 120 μm. Preoperative aberrometry was measured with the iDesign System, and the calculated ablation profile incorporated both higher order and lower order aberrations. The features of this aberrometer have been previously described.16 A nomogram was used to adjust the sphere according to the magnitude of the aberrometer-derived cylinder16 to avoid overcorrection of sphere. After the flaps were lifted, the programmed treatment was applied once iris registration was achieved. All surgical procedures were performed under topical anesthesia. Standard topical postoperative treatment was administered to all patients, consisting of a topical antibiotic and steroid drops four times a day for 1 week and preservative-free artificial tear drops, as necessary.
Statistical analysis
Parametric statistics were used to analyze differences between preoperative and postoperative outcomes (paired Student’s t-test), and Pearson correlation coefficients were used to assess the correlation between different variables. Vector analysis of change in refractive cylinder was performed, using a previously described technique.17 Prior to calculations, axes of left eye refractions were transformed (180° minus the original axis) to avoid certain errors when averaging data for right and left eyes.17 Astigmatic data were displayed on a double-angle plot. In a double-angle plot, the data for preoperative and postoperative refractive cylinder and axis are converted to an orthogonal x, y coordinate system, and the axis of refractive cylinder (ranging from 0° to 180°) is doubled to traverse a circle of 0°–360°. Additionally, a normalized version of double-angle plot was constructed by setting the preoperative axis of refractive cylinder as zero, and modifying the postoperative axis in relation to the preoperative axis. Basic definitions of all vector parameters used in this study are presented in Table 1. All data were analyzed using Microsoft Office Excel 2007 program (Microsoft Corporation) and STATISTICA (StatSoft Inc.) on a personal computer. A level of significance of P=0.05 was used.
  | Table 1 Definitions of vector parameters17 |
Results
This study included 611 eyes of 442 patients treated between December 2013 and July 2014. The mean patient age was 34.0±9.5 years (range: 18–65 years). Forty-eight percent of patients were male, and 52% were female. The mean keratometry in the study group was 44.0±1.3 D (range: 40.50–47.00 D). Preoperative and postoperative visual and refractive outcomes are summarized in Table 2.
Visual acuity
Significant improvement in UDVA and CDVA was observed comparing preoperative and 3 months postoperative data (P<0.01; Table 2). Figure 1 shows postoperative UDVA against preoperative CDVA; there was no statistically significant difference found between the two variables (P=0.06). The percentages of eyes achieving UDVA of 20/20 or better were 80.9% (494 eyes), 83.0% (507 eyes), and 83.8% (512 eyes) at 1 week, 1 month, and 3 months, respectively. The changes in CDVA between preoperative and 3 months postoperative visit were as follows: loss of two lines or more, 0.8% (five eyes); loss of one line, 6.1% (37 eyes); unchanged, 65.1% (398 eyes); gain of one line, 23.6% (144 eyes); and gain of two lines or more, 4.4% (27 eyes).
  | Figure 1 Comparison of postoperative cumulative monocular UDVA and preoperative CDVA. |
Refractive outcomes
Figure 2 plots attempted MSE against achieved MSE at 3 months postoperatively. The linear regression of the scattergram has a slope of 0.98 and the intercept of −0.07. At 3 months, 90.3% (n=552) of eyes were within ±0.50 D, and 99.2% (n=606) were within ±1.0 D of emmetropia. The percentage of eyes with residual refractive cylinder of ≤0.50 D, ≤0.75 D, and ≤1.00 D was 79.1% (483 eyes), 88.5% (541 eyes), and 95.7% (585 eyes), respectively. Table 3 shows the stability of refractive error between 1 week and 1 month, and between 1 month and 3 months visits. There was a small, but statistically significant change in refractive cylinder between 1 month and 3 months (−0.03±0.34 D, P=0.01), while sphere and MSE remained stable between the two visits.
Vector analysis of refractive cylinder
Figure 3 plots the intended refractive correction (IRC) versus surgically induced refractive correction (SIRC) at 3 months, postoperatively. Table 4 summarizes all vector parameters, stratified by the magnitude of preoperative refractive cylinder. The correction ratio for the whole study group was 0.92±0.14, indicating a slight undercorrection. There was a statistically significant correlation between IRC and error vector (r=0.29, P<0.01) and between IRC and error of magnitude (r=0.26, P<0.01). The percentage of eyes with error of angle within 5° and 10° was 89.2% (545 eyes) and 98.4% (601 eyes), respectively.
Figure 4 plots preoperative and 3 months postoperative refractive cylinder in a double-angle plot. The centroid of postoperative cylinder moved closer to the null point, and the shape of the ellipse (standard deviation of x and y data points) reduced significantly. Figure 5 depicts the normalized error vector. At 3 months, 71.0% and 84.1% of eyes had axis shift within ±15° and ±30°, respectively.
Discussion
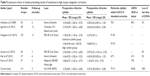
Findings of this study indicate that WFG LASIK in myopic eyes with high cylinder is safe, efficient, and predictable. Three months postoperatively, 90.3% (n=552) of eyes had MSE within ±0.50 D of intended correction, and 83.8% (n=512) had postoperative UDVA 20/20 or better. There was a statistically significant improvement in CDVA from −0.04±0.07 logMAR preoperatively to −0.07±0.06 logMAR 3 months postoperatively. The comparison with other studies is difficult mainly because in most of the studies of myopic astigmatism, there is no differentiation between different degrees of refractive cylinder. Studies summarized in Table 54–9 present results specifically for myopic eyes with moderate-to-high cylinder. We achieved favorable outcomes in comparison to the literature review. Although the mean preoperative refractive cylinder in our study group is slightly lower than in most of the other studies, Table 4 shows error vector (which essentially equals to the magnitude of residual refractive cylinder) stratified by preoperative cylinder. The highest error vector we achieved was 0.74±0.55 D in the subgroup of eyes with preoperative refractive cylinder between 5.00 D and 6.00 D.
We observed a small, but statistically significant, regression in refractive cylinder between 1-week and 1-month visits (−0.08±0.39 D, P<0.01), and 1-month and 3-month visits (−0.03±0.34, P=0.01), while a small hyperopic shift in sphere between 1-month and 3-month exams was not statistically significant (+0.02±0.30, P=0.10). This contradicts the study of Igarashi et al5 where changes in spherical and cylindrical error were longitudinally assessed over a 1-year period following LASIK for moderate-to-high myopic astigmatism. The authors found significant regression in sphere but no statistically significant change in cylinder between 1-week and 1-year exams (mean cylinder −0.67±0.54 D at 1 week and −0.63±0.63 D at 1 year).5 The study was conducted on 48 eyes with preoperative refractive cylinder of −2.74±0.99 D. Similar findings were presented in the study of Hasegawa et al6 where refractive cylinder remained stable up to 12 months postoperatively in patients with moderate-to-high myopic astigmatism. A small change in refractive cylinder in our study showed statistical significance probably due to the large sample size (compared to other studies); however, it would be interesting to evaluate whether there was a further change beyond the 3-month postoperative visit.
The vector analysis of refractive cylinder revealed slight undercorrection with the correction ratio (ratio of the magnitude of SIRC to IRC) of 0.92±0.14 at 3 months postoperatively. The mean error of magnitude (arithmetic difference in the magnitudes between SIRC and IRC) was 0.22±0.40 D, and this variable was correlated to the IRC (r=0.26, P<0.01). The error of angle was minimal and slightly clockwise (−0.45°) with a relatively low standard deviation of 2.99°. In comparison to other studies reporting vector analysis of high myopic astigmatism, Alió et al8 found a similar mean error of angle of −0.49° but higher standard deviation (4.41°) in a group of patients with the mean preoperative refractive cylinder of −3.64±0.55 D. Wavefront-optimized ablation profile with cyclotorsion control was used in this study, and similar undercorrection was seen (correction ratio 0.91±0.13). Katz et al9 using a wavefront-optimized ablation profile, achieved a median correction ratio of 1.06, and the median error of angle of 3.60° (95% confidence interval 2.28°–6.28°) in eyes with preoperative refractive cylinder greater than 3.00 D. In our study, the absolute error of angle was <2° for each subgroup of preoperative refractive cylinder (Table 4), and the standard deviation was low in each category.
Astigmatic correction requires adequate centration of the treatment and control of cyclotorsional movements. Cyclotorsion that can occur when patients change from an upright to supine position18–22 is a known source of astigmatic undercorrection. On average, the cyclotorsional errors are approximately 2° but could be much higher in a small percentage of patients.20,22 A 4° and a 10° misalignment would theoretically result in a 14% and a 35% undercorrection of astigmatism, respectively.20 The misalignment can also result in induction of higher order aberrations (HOAs),23–25 which would defeat the object of WFG laser correction. In this study, we used a new Hartmann–Shack device (iDesign System) that uses an enhanced iris registration system with a high-definition camera and increased capture rate, which could have had an impact on the astigmatism accuracy achieved in this study. Postoperative HOAs were not assessed in this study, which would be interesting to evaluate, since a new diagnostic device was used. Currently, there is only one study reporting changes in postoperative HOAs of WFG LASIK linked to the iDesign System.26 The study found minimal induction of HOAs, which was poorly correlated to the level of preoperative myopia, and there was a decrease in HOAs recorded in 30.4% of eyes.26 However, it would be interesting to evaluate changes in HOAs, specifically in patients with high refractive cylinder, which could be a subject of future prospective studies.
The limitation of this study was that we concentrated on accuracy of astigmatic correction in patients with high preoperative cylinder, but we were unable to comment on changes in postoperative HOAs, which would prove difficult in such a large cohort. Since we observed a change in refractive cylinder, it would be of interest to assess the refractive change over a longer period of time, which was not possible with our retrospective data.
Although our study confirms that correction of cylinder is not as effective as the spherical component and slight undercorrection was seen in our dataset, clinically acceptable results were achieved in a large cohort of patients with a significant portion of eyes achieving 20/20 visual acuity. Despite some drawbacks, this study is, to the best of our knowledge, one of the few studies presenting results of WFG LASIK in patients with high refractive cylinder.
Disclosure
Steven C Schallhorn, MD, is a consultant of Abbott Medical Optics Inc. and Zeiss, and a Global Medical Director of Optical Express. The other authors report no conflicts of interest in this work. None of the other authors have a financial or proprietary interest in the products and materials presented in this paper.
References
Wolfram C, Höhn R, Kottler U, et al. Prevalence of refractive errors in the European adult population: the Gutenberg Health Study (GHS). Br J Ophthalmol. 2014;98(7):857–861. | ||
Khan MI, Muhtaseb M. Prevalence of corneal astigmatism in patients having routine cataract surgery at a teaching hospital in the United Kingdom. J Cataract Refract Surg. 2011;37(10):1751–1755. | ||
Randleman JB, White AJ Jr, Lynn MJ, Hu MH, Stulting RD. Incidence, outcomes and risk factors for retreatment after wavefront-optimized ablations with PRK and LASIK. J Refract Surg. 2009;25:273–276. | ||
Arbelaez MC, Vidal C, Arba-Mosquera S. Excimer laser correction of moderate to high astigmatism with a non-wavefront-guided aberration-free ablation profile: six-month results. J Cataract Refract Surg. 2009;35(10):1789–1798. | ||
Igarashi A, Kamiya K, Shimizu K, Komatsu M. Time course of refractive and corneal astigmatism after laser in situ keratomileusis for moderate to high astigmatism. J Cataract Refract Surg. 2012;38(8):1408–1413. | ||
Hasegawa A, Kojima T, Isogai N, Tamaoki A, Nakamura T, Ichikawa K. Astigmatism correction: laser in situ keratomileusis versus posterior chamber collagen copolymer toric phakic intraocular lens implantation. J Cataract Refract Surg. 2012;38(4):574–581. | ||
Ivarsen A, Næser K, Hjortdal J. Laser in situ keratomileusis for high astigmatism in myopic and hyperopic eyes. J Cataract Refract Surg. 2013;39(1):74–80. | ||
Alió JL, Plaza-Puche AB, Martinez LM, Torky M, Brenner LF. Laser in situ keratomileusis using optimized aspheric profiles and cyclotorsion control to treat compound myopic astigmatism with high cylinder. J Cataract Refract Surg. 2013;39(1):28–35. | ||
Katz T, Wagenfeld L, Galambos P, Darrelmann BG, Richard G, Linke SJ. LASIK versus photorefractive keratectomy for high myopic (>3 diopter) astigmatism. J Refract Surg. 2013;29(12):824–831. | ||
Chernyak DA. Iris-based cyclotorsional image alignment method for wavefront registration. IEEE Trans Biomed Eng. 2005;52(12):2032–2040. | ||
Ghosh S, Couper TA, Lamoureux E, Jhanji V, Taylor HR, Vajpayee RB. Evaluation of iris recognition system for wavefront-guided laser in situ keratomileusis for myopic astigmatism. J Cataract Refract Surg. 2008;34(2):215–221. | ||
Khalifa M, El-Kateb M, Shaheen MS. Iris registration in wavefront-guided LASIK to correct mixed astigmatism. J Cataract Refract Surg. 2009;35(3):433–437. | ||
Shen EP, Chen WL, Hu FR. Manual limbal markings versus iris-registration software for correction of myopic astigmatism by laser in situ keratomileusis. J Cataract Refract Surg. 2010;36(3):431–436. | ||
Azar DT, Chang JH, Han KY. Wound healing after keratorefractive surgery: review of biological and optical considerations. Cornea. 2012;31(suppl 1):S9–S19. | ||
Hettinger KA. The role of biostatistics in the quality improvement of refractive surgery. J Refract Surg. 2009;25(7 suppl):S651–S654. | ||
Schallhorn S, Brown M, Venter J, Teenan D, Hettinger K, Yamamoto H. Early clinical outcomes of wavefront-guided myopic LASIK treatments using a new-generation hartmann-shack aberrometer. J Refract Surg. 2014;30(1):14–21. | ||
Eydelman MB, Drum B, Holladay J, et al. Standardized analyses of correction of astigmatism by laser systems that reshape the cornea. J Refract Surg. 2006;22(1):81–95. | ||
Smith EM Jr, Talamo JH, Assil KK, Petashnick DE. Comparison of astigmatic axis in the seated and supine positions. J Refract Corneal Surg. 1994;10:615–620. | ||
Smith EM Jr, Talamo JH. Cyclotorsion in the seated and supine patient. J Cataract Refract Surg. 1995;21:402–403. | ||
Swami AU, Steinert RF, Osborne WE, White AA. Rotational malposition during laser in situ keratomileusis. Am J Ophthalmol. 2002;133:561–562. | ||
Ciccio AE, Durrie DS, Stahl JE, Schwendeman F. Ocular cyclotorsion during customized laser ablation. J Refract Surg. 2005;21:S772–S774. | ||
Chernyak DA. Cyclotorsional eye motion occurring between wavefront measurement and refractive surgery. J Cataract Refract Surg. 2004;30:633–638. | ||
Bueeler M, Mrochen M, Seiler T. Maximum permissible lateral decentration in aberration-sensing and wavefront-guided corneal ablation. J Cataract Refract Surg. 2003;29(2):257–263. | ||
Bueeler M, Mrochen M, Seiler T. Maximum permissible torsional misalignment in aberration-sensing and wavefront-guided corneal ablation. J Cataract Refract Surg. 2004;30(1):17–25. | ||
Wang L, Koch DD. Residual higher-order aberrations caused by clinically measured cyclotorsional misalignment or decentration during wavefront-guided excimer laser corneal ablation. J Cataract Refract Surg. 2008;34(12):2057–2062. | ||
Smadja D, De Castro T, Tellouck L, et al. Wavefront analysis after wavefront-guided myopic LASIK using a new generation aberrometer. J Refract Surg. 2014;30(9):610–615. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.