Back to Journals » Clinical, Cosmetic and Investigational Dentistry » Volume 14
Clinical Management of Molar Incisor Hypomineralization Affected Molars in a Pediatric Patient Including Endodontic Treatment, Case Report and Review of the Literature
Authors Alfarraj JH , Alsaeed AA
Received 16 April 2022
Accepted for publication 21 June 2022
Published 28 June 2022 Volume 2022:14 Pages 183—189
DOI https://doi.org/10.2147/CCIDE.S371122
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Christopher E. Okunseri
Jawza H Alfarraj,1 AbdulAziz A Alsaeed2
1Pediatric Dentistry, King Fahad Medical City, Riyadh, Saudi Arabia; 2Endodontics, King Abdul-Aziz Medical City, Riyadh, Saudi Arabia
Correspondence: Jawza H Alfarraj, Tel +966 509948859, Email [email protected]
Abstract: The term molar incisor hypomineralization (MIH) describes the clinical manifestation of enamel hypomineralization of systemic origin affecting one or more permanent first molars and incisors. The purpose of this case report is to discuss the management of two mandibular molars with hypomineralization and to emphasize endodontic treatment for pediatric patients. An 8-year-old girl was referred to the pediatric dental clinic for restorations of multiple carious teeth. Upon clinical examination, based on the pattern of enamel hypomineralization and posteruptive breakdown presented, the diagnosis of MIH was established. As a part of the comprehensive treatment, one molar was restored with resin-based composite restorations. The other was endodontically involved and was treated with apexification and then covered with a stainless-steel crown. A two-year recall appointment revealed both molars to be free from signs and symptoms of inflammation. Management of MIH-affected molars should follow a conservative approach and endodontic treatment should be considered.
Keywords: case report, molar incisor hypomineralization, MIH, apexification, mineral trioxide aggregate, pediatric dentistry
Introduction
The term molar incisor hypomineralization (MIH) was first introduced in 2001 referring to the clinical form of enamel hypomineralization of systemic cause affecting one or more permanent first molars (PFMs) and permanent maxillary incisors.1 It can affect from one to all four PFMs.1 The risk of MIH affecting the permanent maxillary incisors increases with the increased number of PFMs affected.2 Reports have also indicated that the second primary molars, second permanent molars, and the tips of the permanent canines can be involved.3
MIH is a prevalent dental condition that effect up to 25% of children worldwide.3 Its exact etiology remains uncertain.1,3 In general, enamel hypomineralization is caused by a disturbance in the function of ameloblast cells during any of the amelogenesis stages. However, it is believed that MIH is due to multifactorial systemic factors that contribute to enamel hypomineralization.4 Studies show that children born premature or who had poor general health in their early life are at greater risk of developing MIH.3,4 Antibiotic use during early life has also been linked to MIH. However, due to the concurrence of disease and antibiotic therapy, it is difficult to determine whether the MIH was caused by the disease or the antibiotic.5
Clinical criteria for the diagnosis of MIH were established in 2001, which include: demarcated opacities, posteruption breakdown (PEB), atypical restorations, extraction of molars due to MIH, and failure of eruption of a molar or incisor.6 Demarcated opacities are enamel defects with altered translucency; the affected enamel is yellow to brown in color and has a clear boundary from normal enamel.7 The color change reflects differences in enamel hardness, porosity, and mineral content; the darker the color, the lower the hardness and greater the porosity.8 These opacities are usually limited to the incisal or cuspal third of the crown, rarely involving the cervical third.7
Surprisingly, the clinically sound enamel in hypomineralized molars is found to have an overall reduction in mineral concentration (of about 5%) and lower calcium/phosphorus ratios, meaning that the entire crown is affected to some extent.7
In normal teeth, the surface of intact enamel is typically hard and smooth. Also, the outer surface layer becomes hypermineralized with posteruptive maturation. Instead, MIH-affected enamel is soft and porous and susceptible to posteruptive breakdown (PEB).4 If not treated, PEB will be complicated by the formation of dental caries, which may lead to pulpal involvement.2 The purpose of the current case report is to discuss the treatment of MIH-affected teeth in a pediatric patient, with emphasis on the management of two mandibular first molars with hypomineralization defects and posteruptive breakdown. It also highlights the endodontic treatment approach for MIH-affected molars, especially for the immature apexes of pediatric patients.
Case Description
An 8-year-old girl referred from primary care to the Pediatric Dental Clinic at King Abdulaziz Medical City in Riyadh, Saudi Arabia, presented with her mother with the complaint of multiple carious teeth that sometimes led to pain. The mother denied any medical conditions or allergies for the child. The dental history revealed no dental trauma but the patient was seen three months prior in a primary-care hospital and fissure sealant of upper right first permanent molar was placed. Informed consent was obtained from the parent for all examinations, photographs, radiographs, and procedures. In addition, another informed consent was also obtained for publishing the case in a scientific journal including the photographs. This case was part of the pediatric dentistry and endodontics Saudi board training. No institutional approval was required to publish this case details.
Clinical examination revealed mixed dentition stages, extensive caries of all primary molars, and that the lower left first primary molar (#74) had been restored with defective amalgam restoration (Figure 1) (Canon EOS4000 digital camera was used for all photographic figures). The upper right permanent central incisor (#11) had a large demarcated yellow-to-brown opacity. The upper right and left first permanent molars (#16 and #26) had large demarcated opacities, yellowish-brown in color; lower right permanent first molar (#46) had posteruptive enamel breakdown and active caries; lower left permanent first molar (#36) showed posteruptive breakdown and deep mesial active caries (Figure 1), not tender to percussion and with a normal cold-test response (Endo-frost, coltene/whaledent, langenau Germany). Pulpal diagnosis for (#46) was normal pulp with normal apical tissue, and for (#36) was asymptomatic irreversible pulpitis with normal apical tissue.
 |
Figure 1 Pre-operative clinical photographs. |
Occlusion assessment showed class I molar relationship, and soft tissue examination was within normal limits (Figure 1). Radiographic examination showed multiple interproximal caries of primary molars with remaining roots; lower left permanent first molar (#36) with deep mesial caries and immature open apex (Figure 2).
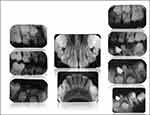 |
Figure 2 Pre-operative intra-oral radiographs. |
Based on the pattern of hypomineralization presented, clinical evaluation, and the related history and symptoms, the diagnosis of MIH was confirmed. Starting with the preventive phase, oral hygiene instructions were given including using fluoridated toothpaste, and topical fluoride varnish (Duraphat, Colgate Oral Care, Sydney, Australia) was applied on all hypomineralization spots.
For the restorative and surgical phase, multiple extractions and stainless-steel crowns were done for the primary molars to restore their function and eliminate any source of infection.
Regarding tooth (#46), caries and defective enamel were removed, pretreatment with 5.25% sodium hypochlorite was applied then the cavity was etched using 35% phosphoric acid (Ultra-Etch, Ultradent Products Inc., South Jordan, UT). Prime and adhesive (Adper ScotchbondTM Multi- purpose, 3M ESPE, St. Paul, MN) were applied and cured according to manufacturer instructions, and the cavity was restored with shade A2 of Z100TM composite material (3M ESPE, St. Paul, MN).
Regarding tooth (#36), consultation was sought from the orthodontic and endodontic departments, and treatment options were discussed with parents. It was planned to perform caries excavation then vital pulp therapy by MTA partial or full pulpotomy, but after removing the pulp chamber tissue, bleeding could not be controlled after 10 minutes of application of cotton pellets immersed in 5.25% of NaOCl, and blood was oozing from the distal canal and the tooth was found to require apexification. Thus, pulp extirpation was done during this visit. During the second visit, obturation of mesial root canals with gutta-percha and apexification with ProRoot white MTA (Dentsply Tulsa Dental, Oklahoma, USA) plug for the distal root canal were performed. Then the tooth was covered with a prefabricated stainless-steel crown SSC (3M ESPE, SSC, Germany) and cemented with glass ionomer cement (3M-ESPE, Ketac-Cem, Seefeld, Germany).
For the orthodontic phase, a lower lingual holding arch and upper band and loop were fabricated and cemented using (3M-ESPE, Ketac-Cem, Seefeld, Germany). The patient was seen at the six-month follow-up visit and was asymptomatic. Clinical examination showed a normal response of (#46) to percussion and cold test. The restoration was intact with no discoloration or signs of marginal leakage. Tooth (#36) showed a normal response to percussion. At the two-year follow-up, the patient had the same clinical findings as in the six-month follow-up (Figure 3). Radiographic imaging revealed that teeth (#36 and #46) had normal apical tissue, intact lamina dura, and periodontal ligament space, with total apical closure of both mesial and distal root canals of the tooth (#36) which confirmed a short-term success of the treatment (Figure 4C).
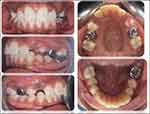 |
Figure 3 Two-year recall photographs. |
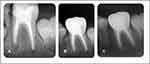 |
Figure 4 (A) Immediate post-operative radiographs, (B) six-months recall radiograph, (C) two- year recall radiographs. |
Discussion
Pediatric patients with MIH-affected teeth usually experience dental pain, hypersensitivity and aesthetic concern when their incisors are affected too.1,2 With MIH-affected molars, early detection and management are very important to prevent further enamel breakdown and pulpal involvement.2
Remineralization therapy should be initiated as soon as the defective surface is accessible, aiming to produce a mineralized surface layer.4 Topical fluoride, applied as varnishes or gels, can remineralize enamel, reduce sensitivity, and enhance resistance to demineralization.4 Fluoride has been the first remineralizing agent used in dentistry.8 In 1942, Cheyne hypothesized that dental hard tissues such enamel and dentin might respond to fluoride ion as bone hard tissue.8 He experimented his theory with aqueous solution of 0.05% potassium topical fluoride on preschool children; the application was for four minutes and was repeated at three to four months intervals.8,9 Then, after one year of investigations, he found that treated subjects had less the incidence of dental caries by 50% compared to untreated children.8 Fluoride varnish (Duraphat, Colgate Oral Care, Sydney, Australia) containing 50 mg NaF/mL (2.26% F, 22,600 ppm F) binds to enamel and plaque, acting as a reservoir for slow-release fluoride.9 There is a proven clinical value of topical fluoride application on hypomineralized molars, resulting in surface enamel hardening before dental restorations.9,10 In addition to conventional fluoride-based therapies also biomimetic hydroxyapatite and casein phosphopeptide-amorphous calcium phosphate have been recently introduced and showed promising results.10,11
Despite the well-known advantages of glass-ionomer-based restorative materials (GIC) such as fluoride release and chemical bonding, Resin composite (RC) still the material of choice for one-surface restorations with MIH affected molars.2 GIC-based restorations can be used as dentin replacement or as an interim restoration until RC can be placed.9
It has been suggested that pretreatment with 5.25% sodium hypochlorite can enhance the bond strength by removing protein coating the hydroxyapatite.12,13 Also, it has been recommended to remove all hypomineralized enamel before placing RC restorations.14
When managing first molars with severe MIH, especially with pulpal involvement, the first clinical consideration is whether to restore or extract the tooth. Restorations with composites, indirect restorations, or preformed metal crowns (PMC) are good options.14–16 The reported success rate of PMC in MIH-affected molars ranges from 85% to 100%.17
However, when extraction is considered, orthodontic assessment should be performed. Many factors should be evaluated such as the status of neighboring teeth and restorability, child cooperation, the underlying malocclusion, any hypodontia, the presence or absence of crowding, the presence of the third permanent molar, and the dental developmental stage of the child.15,16 If extraction is decided, the developmental stage between the ages of 8 to 10 years old has been recommended.18 However, a risk to developing a malocclusion should be accepted.17
In this case report, since the patient was cooperative and the tooth was restorable, an endodontist was consulted regarding the appropriate endodontic management. Vital pulp therapy is always recommended in pediatric patients due to the less invasive procedure and high success rate.19 MTA pulp capping, and partial or full pulpotomy are the treatment choices for immature permanent teeth with inflamed pulps.19,20 If homeostasis cannot be achieved, root apexification can be considered which is the process of creating a barrier with hard tissue at the root end. Traditionally, a calcium hydroxide-based apexification procedure has been advocated for treating an immature permanent tooth with an open apex.21 However, apexification with calcium hydroxide is associated with certain difficulties such as requiring a long-term application of calcium hydroxide, the need for multiple visits scheduled over many months, and the possibility of tooth fracture.22,23
An alternative for the multi-appointment apexification procedure is a single-step technique using an apical barrier. In 1999, Torabinejad & Chivian introduced the use of MTA as an apical plug.24 In a human-outcome study that compared the clinical and radiographic results of apexification with either MTA or calcium hydroxide, all cases obturated with MTA were successful at the 12-month recall, whereas 2 of the 15 calcium hydroxide cases had persistent disease.25 In a retrospective study, Jeeruphan et al compared the radiographic and survival outcomes of immature teeth treated with apexification methods. The success and survival rates were 95% and 77% for MTA apexification and calcium hydroxide apexification procedures, respectively.26
Preformed metal crowns (PMCs) can be used successfully in severely damaged MIH molars as a traditional treatment with high long-term survival rates until the final crown is served.9,26
Ideally, extraction of mandibular FPM should be avoided in pediatric patients with distal occlusion, deep bite, and increased overjet.27 It was reported that a 72% and 48% success rate of spontaneous space closure in the maxilla and mandible post extraction of first permanent molars, respectively.28
If the prognosis is poor and tooth eventually will be extracted, it might be wise to consider postponing extraction and save the tooth by endodontic treatment until the patient is a candidate for a dental implant. This would provide the patient with functional occlusion, avoid the need for future orthodontic treatment and preserve the bone for implant placement.15,16 Limitation of this case report that no long-term follow-up to evaluate the survival rate of endodontic treated molars in pediatric patients, which is an area of future research.
Conclusion
Management of severe MIH-affected teeth should follow a conservative approach. Endodontic treatment should be always considered, especially when early extraction will cause complex malocclusion.17
Clinical Significance
When managing permanent first molars with MIH and pulpal involvement, the first clinical consideration is whether to restore or extract. Most pediatric dentists tend to extract due to a widespread belief that the success rate of root canal treatment in immature root molars is poor. However, with new advances in endodontics, apexification can be done in a single visit with new materials such as MTA and success rates of up to 97%.22
Disclosure
The authors report no conflicts of interest in this work.
References
1. Weerheijm KL, Jalevik B, Alaluusua S. Molar-incisor hypomineralisation. Caries Res. 2001;35(5):390–391. doi:10.1159/000047479
2. William V, Messer LB, Burrow MF. Molar incisor hypomineralization: review and recommendations for clinical management. Pediatr Dent. 2006;28:3.
3. de Farias AL, Rojas-Gualdrón DF, Girotto Bussaneli D, et al. Does molar-incisor hypomineralization (MIH) affect only permanent first molars and incisors? New observations on permanent second molars. Int J Paediatr Dent. 2022;32(1):1–10. doi:10.1111/ipd.12780
4. Fearne J, Anderson P, Davis GR. 3D X-ray micro- scopic study of the extent of variations in enamel density in first permanent molars with idiopathic enamel hypomineralization. Br Dent J. 2004;196:634–638. doi:10.1038/sj.bdj.4811282
5. Jalevik B, Noren JG, Barregard L, Barregård L. Etiologic factors influencing the prevalence of demarcated opacities in permanent first molars in a group of Swedish children. Eur J Oral Sci. 2001;109:230–234. doi:10.1034/j.1600-0722.2001.00047.x
6. Weerheijm KL, Duggal M, Mejare I, et al. Judgement criteria for molar incisor hypomineralization (MIH) in epidemiologic studies: a summary of the European meeting on MIH held in Athens. Eur J Paediatr Dent. 2003;4:110–113.
7. Jalevik B, Odelius H, Dietz W, Noren JG. Secondary ion mass spectrometry and X-ray microanalysis of hypomineralized enamel in human permanent first molars. Arch Oral Biol. 2001;46:239–247. doi:10.1016/S0003-9969(00)00113-8
8. Zampetti P, Scribante A. Historical and bibliometric notes on the use of fluoride in caries prevention. Eur J Paediatr Dent. 2020;21(2):148–152. doi:10.23804/ejpd.2020.21.02.10
9. Fayle SA. Molar incisor hypomineralization: restorative management. Eur J Paediatr Dent. 2003;4:121–126.
10. Butera A, Pascadopoli M, Gallo S, et al. SEM/EDS evaluation of the mineral deposition on a polymeric composite resin of a toothpaste containing biomimetic zn-carbonate hydroxyapatite (microrepair®) in oral environment: a randomized clinical trial. Polymers. 2021;13(16):2740. doi:10.3390/polym13162740
11. Khanduri N, Kurup D, Mitra M. Quantitative evaluation of remineralizing potential of three agents on artificially demineralized human enamel using scanning electron microscopy imaging and energy-dispersive analytical X-ray element analysis: an in vitro study. Dent Res J. 2020;17(5):366–372. doi:10.4103/1735-3327.294329
12. Wright JT. The etch-bleach-seal technique for managing stained enamel defects in young permanent inci- sors. Pediatr Dent. 2002;24:249–252.
13. Venezie RD, Vadiakas G, Christensen JR, Wright JT. Enamel pretreatment with sodium hypochlorite to enhance bonding in hypocalcified amelogenesis imperfecta: case report and SEM analysis. Pediatr Dent. 1994;16:433–436.
14. Lygidakis NA, Chaliasou A, Siounas G. Evaluation of composite restorations in hypomineralized permanent molars: a four-year clinical study. Eur J Paediatr Dent. 2003;4:143–148.
15. Elhennawy K, Schwendicke F. Managing molar-incisor hypomineralization: a systematic review. J Dent. 2016;55:16–24. PMID: 27693779. doi:10.1016/j.jdent.2016.09.012
16. Somani C, Taylor GD, Garot E, Rouas P, Lygidakis NA, Wong FSL. An update of treatment modalities in children and adolescents with teeth affected by molar incisor hypomineralisation (MIH): a systematic review. Eur Arch Paediatr Dent. 2022;23(1):39–64. PMID: 34110615; PMCID: PMC8927013. doi:10.1007/s40368-021-00635-0
17. Oh N, Nam S, Lee J, Kim H. Retrospective study on the survival rate of preformed metal crowns in permanent first molars. J Korean Acad Pediatr Dent. 2020;47(2):140–147. doi:10.5933/JKAPD.2020.47.2.140
18. Ashley P, Noar J. Interceptive extractions for first permanent molars: a clinical protocol. Br Dent J. 2019;227(3):192–195. doi:10.1038/s41415-019-0561-7
19. Mente J, Hufnagel S, Leo M, et al. Treatment outcome of mineral trioxide aggregate or calcium hydroxide direct pulp capping: long-term results. J Endod. 2014;40:1746–1751. doi:10.1016/j.joen.2014.07.019
20. Simon S, Perard M, Zanini M, et al. Should pulp chamber pulpotomy be seen as a permanent treatment? Some preliminary thoughts. Int Endod J. 2013;46:79–87. doi:10.1111/j.1365-2591.2012.02113.x
21. Rafter M. Apexification: a review. Dent Traumatol. 2005;21:1–8. doi:10.1111/j.1600-9657.2004.00284.x
22. Sheehy EC, Roberts GJ. Use of calcium hydroxide for apical barrier formation and healing in non-vital immature permanent teeth: a review. Br Dent J. 1997;183:241–246. doi:10.1038/sj.bdj.4809477
23. Andreasen JO, Farik B, Munksgaard EC. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent Traumatol. 2002;18:134–137. doi:10.1034/j.1600-9657.2002.00097.x
24. Torabinejad M, Chivian N. Clinical applications of mineral trioxide aggregate. J Endod. 1999;25:197–205. doi:10.1016/S0099-2399(99)80142-3
25. El-Meligy OA, Avery DR. Comparison of apexification with mineral trioxide aggregate and calcium hydroxide. Pediatr Dent. 2006;28:248–253.
26. Jeeruphan T, Jantarat J, Yanpiset K, et al. Mahidol study 1: comparison of radiographic and survival outcomes of immature teeth treated with either regenerative endodontic or apexification methods—a retrospective study. J Endod. 2012;38:1330–1336. doi:10.1016/j.joen.2012.06.028
27. Randall RC. Preformed metal crowns for primary and permanent molar teeth: review of the literature. Pediatr Dent. 2002;24(5):489–500.
28. Alkhadra T. A systematic review of the consequences of early extraction of first permanent first molar in different mixed dentition stages. J Int Soc Prev Community Dent. 2017;7(5):223–226. doi:10.4103/jispcd.JISPCD_222_17
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
