Back to Journals » Therapeutics and Clinical Risk Management » Volume 12
Clinical features of brain metastases in breast cancer: an implication for hippocampal-sparing whole-brain radiation therapy
Authors Wu S, Sun J, Tong Q, Li F, He Z
Received 8 October 2016
Accepted for publication 9 November 2016
Published 13 December 2016 Volume 2016:12 Pages 1849—1853
DOI https://doi.org/10.2147/TCRM.S124212
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Deyun Wang
San-Gang Wu,1,* Jia-Yuan Sun,2,* Qin Tong,3 Feng-Yan Li,2 Zhen-Yu He2
1Department of Radiation Oncology, Xiamen Cancer Hospital, The First Affiliated Hospital of Xiamen University, Xiamen, 2Department of Radiation Oncology, Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center of Cancer Medicine, Guangzhou, 3Department of Radiation Oncology, The First Affiliated Hospital of University of South China, Hengyang, People’s Republic of China
*These authors contributed equally to this work
Objective: The objectives of this study were to describe the distribution of brain metastases (BM) in breast cancer patients and investigate the risk factors for perihippocampal metastases (PHM).
Patients and methods: Retrospective analysis of the clinicopathological characteristics and patterns of BM was performed. Associations between clinicopathological characteristics and PHM (the hippocampus plus 5 mm margin) were evaluated using logistic regression analyses.
Results: A total of 1,356 brain metastatic lesions were identified in 192 patients. Patients with 1–3 BM, 4–9 BM, and ≥10 BM accounted for 63.0%, 18.8%, and 18.2%, respectively. There were only 7 (3.6%) patients with hippocampal metastases (HM) and 14 (7.3%) patients with PHM. On logistic regression, the number of BM was an independent risk factor for PHM. Patients with ≥10 BM had a significantly higher risk of PHM compared with those with <10 BM. Breast cancer subtype (BCS) was not associated with PHM. The number of BM was significantly correlated with various BCSs. Patients with hormone receptor (HR)+/human epidermal growth factor receptor 2 (HER2)+, HR-/HER2+, and HR-/HER2- subtypes had a higher probability of ≥10 BM, relative to patients with an HR+/HER2- subtype.
Conclusion: Our study suggests that a low incidence of PHM may be acceptable to perform hippocampal-sparing whole-brain radiation therapy for breast cancer patients. Patients with extensive diffuse metastases (≥10 BM) were associated with higher odds of PHM.
Keywords: breast cancer, hippocampal metastases, brain metastasis, whole-brain radiation therapy
Introduction
With the development of comprehensive therapy and advances in the diagnostic imaging of breast cancer, brain metastases (BM) have been documented in approximately one-quarter of breast cancer patients.1 Whole-brain radiation therapy (WBRT) remains the main treatment for patients with four or more BM in breast cancer. For patients with oligometastatic disease (defined as 1–3 BM), stereotactic radiosurgery (SRS) with or without WBRT should be considered for local disease control and symptomatic palliation. Existing studies recommend delaying WBRT to prevent neurocognitive decline secondary to the observation of no benefit in the overall survival.2,3 The median overall survival time for breast cancer patients with BM treated with WBRT or SRS was 4–6 months and up to 16 months if solitary BM could be removed surgically.4,5
WBRT is associated with a range of toxicities, including significant deterioration in neurocognitive function and quality of life (QoL).6,7 The hippocampus is sensitive to radiation. The main clinical manifestation of the hippocampal injury secondary to radiotherapy was cognitive decline.8,9 Previous studies have reported that intracranial metastases of malignant tumors occurred rarely in the hippocampus relative to other anatomical brain locations.10–14 These studies provided evidence supporting hippocampal-sparing whole-brain radiation therapy (HS-WBRT).10–14 In a study by the Radiation Therapy Oncology Group (RTOG) 0933, HS-WBRT was reported as being protective against adverse neurocognitive outcomes and was associated with an improved QoL in patients. However, this study was limited by a small sample size of breast cancer patients.15 In a previous study, we reported a potential increase in hippocampal metastases (HM) and perihippocampal metastases (PHM) in breast cancer patients (n=56), relative to other malignant tumors.11 In this study, we collected data from two academic cancer centers for further analysis of HM and PHM in breast cancer patients to investigate potential risk factors for PHM.
Patients and methods
Breast cancer patients diagnosed with BM at the time of initial diagnosis or during the follow-up were retrospectively analyzed in the Sun Yat-sen University Cancer Center (SYSUCC) and the First Affiliated Hospital of Xiamen University (Xiamen Cancer Hospital [XMCH]) from January 1997 to September 2015. All study patients underwent magnetic resonance imaging (MRI) including T1-weighted, post-contrast, axial MRI image data at the time of BM occurrence. Patients with malignant tumors prior to breast cancer diagnosis and patients diagnosed with a secondary malignant tumor during follow-up were excluded from the analysis. All patients provided written consent for storage of their medical information in the hospital database and for use of their data in research including publication of that research. The study was approved by the ethics committee of the SYSUCC and the XMCH.
The hippocampus was delineated using enhanced, T1-weighted MRI according to the RTOG 0933 reference.15 The perihippocampal region was defined as a 5-mm margin around the hippocampus, accounting for measurement error and possible displacement following radiotherapy.14,16
Clinicopathological characteristics, including age, menopausal status, local treatment, tumor–node–metastasis (TNM) stage, estrogen receptor (ER) status, progesterone receptor (PR) status, human epidermal growth factor receptor 2 (HER2) status, breast cancer subtypes (BCSs), Ki-67 status, and the number of BM, were identified as potential risk factors for PHM. Hormone receptor (HR) positive status was defined as >1% ER or PR positive cells on immunohistochemistry. HER2 positivity was defined as an immunohistochemical grade of 3+ (in samples predating 2003) and an immunohistochemical grade of 3+ or 2+ using fluorescence in situ hybridization (FISH) on samples taken from 2003 onward. We did not define BCS of breast caner according to the St Gallen recommendation due to insufficient Ki-67 data.17 We defined four major groups of intrinsic BCS as follows: HR+/HER2− (ER+ and/or PR+, HER2−), HR+/HER2+ (ER+ and/or PR+, HER2+), HR−/HER2+ (ER−, PR−, and HER2+), and HR−/HER2− (ER−, PR−, and HER2−, triple negative breast cancer [TNBC]).18,19
The χ2 and Fisher’s exact tests were used to analyze group differences in categorical data. Univariable and multivariable binary logistic regression analyses were used to investigate clinicopathologic predictors of PHM. For all analyses, a P-value <0.05 was considered significant. All analyses were conducted using the SPSS statistical software package (version 16.0; SPSS Inc., Chicago, IL, USA).
Results
Clinicopathological characteristics
A total of 192 patients were identified for the analysis, including 136 (70.8%) patients from the SYSUCC and 56 (29.2%) patients from the XMCH. The median age at diagnosis of breast cancer was 46.0 years (range, 22–77 years). Thirty-six (18.7%) patients were initially diagnosed with M1 stage disease. Thirty of these M1 stage patients had not received surgical treatment. A total of 136 (70.8%) patients had extracranial metastatic disease. Of the available 166 patients with BCS, there were 26 (15.7%), 73 (44.0%), 25 (15.1%), and 42 (25.3%) patients with HR+/HER2−, HR+/HER2+, HR−/HER2+, and HR−/HER2− subtypes, respectively. Patient clinicopathological characteristics are summarized in Table 1.
Distribution of BM
A total of 1,356 brain metastatic lesions were identified across the 192 patients. The median number of BM lesions was 2 (range, 1–198). The most frequent anatomical locations of BM included the frontal lobe (29.3%, 397/1,356), cerebellum (26.0%, 353/1,356), parietal lobe (16.9%, 229/1,356), occipital lobe (12.7%, 172/1,356), temporal lobe (10.5%, 142/1,356), and brainstem (4.1%, 56/1,356; Table 2). There were 121 (63.0%), 36 (18.8%), and 35 (18.2%) patients with 1–3 BM, 4–9 BM, and ≥10 BM, respectively. Of all patients, only 7 (3.6%, 7/192) and 14 (7.3%, 14/192) patients had HM and PHM, respectively.
  | Table 2 The distribution of BM by location |
Risk factors for PHM
Logistic regression analyses for PHM were performed with the following variables: age at BM (<50 years vs ≥50 years; <60 years vs ≥60 years), local treatment, menopausal status, tumor size, nodal status, BCS, Ki-67 status, and the number of BM. On univariate logistic regression, the number of BM was associated with PHM. In multivariable analysis, the number of BM was the independent risk factor for PHM (Table 3). Patients with BM ≥10 was associated with 9.919 times the odds of PHM (odds ratio [OR] 9.919, 95% confidence interval [CI] 2.388–41.179, P=0.002) relative to those with 1–3 BM. There was no difference in the odds of PHM in patients with 4–9 BM compared to 1–3 BM. Age, menopausal status, surgical treatment, TNM stage, HR status, HER2 status, BCS, and Ki-67 status were not associated with PHM.
Association between BCS and the number of BM
The number of BM was significantly correlated with various BCSs (Table 4). Patients with HR+/HER2+, HR−/HER2+, and HR−/HER2− subtypes had a higher probability of ≥10 BM, which was significantly higher than patients with HR+/HER2− subtype (P=0.011).
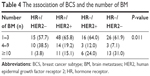  | Table 4 The association of BCS and the number of BM |
Discussion
Studies on the application of HS-WBRT in specific subtypes of malignant tumors are limited.10,14,20,21 In this study, we investigated the distribution of BM in breast cancer patients. Our results showed that HM and PHM occurred rarely in breast cancer patients, which was consistent with similar results in other studies.10–14 We also found that the number of BM was an independent risk factor for PHM and that BCS was not correlated with PHM.
Our previous single center study of 56 breast cancer patients observed an increase in risk of PHM relative to patients with other types of cancers, although the association was not statistically significant (P=0.084).11 In this study, we identified patients from two cancer centers, and the probabilities of HM (3.6%) and PHM (7.3%) were not significantly higher than the previous results (4.1% of patients developed HM and 5.5% of patients developed PHM in our previous study).11 In a previous study of 314 Chinese patients, the percentages of HM and PHM were 4.1% and 11.1%, respectively.10 In other studies, HM has been reported to be between 0 and 1.3% of total BM.12–14 The increase in the absolute risk of hippocampal recurrence with HS-WBRT was ~2%, which was considered acceptable.10
The majority of breast cancer patients with BM also have extracranial metastatic disease. Palliative treatment was the main therapy strategy for advanced breast cancer. A decrease in cognitive function has previously been observed in breast cancer patients receiving chemotherapy.22 This was related to a reduction in hippocampal volume and verbal memory performance.23 Therefore, a combination of WBRT and systemic chemotherapy may increase the risk of cognitive function decline in breast cancer patients with BM. However, there are no studies formally assessing the clinical value of HS-WBRT in breast cancer patients.
BCSs have previously been reported as being associated with distinct patterns of distant metastases in breast cancer. We found that the patients with the HR−/HER2− subtype were more likely to develop BM in our previous study.24 In this study, significant differences in the number of BM were observed across the different BCSs. The proportion of patients with >10 BM were higher in the HER2+ and TNBC groups relative to HR+/HER2− patients. In addition, patients with >10 HM had a significantly increased risk of PHM, although no association was found between BCS and PHM. In a study by Witt et al,25 73 patients with HER2+ subtype developed BM. The proportion of patients with HM and PHM were 6.8% (5/73) and 15.1% (11/73), respectively, the highest such proportions reported in the recent literature. Sun et al10 also found no correlation between BCS and PHM. Therefore, even though there was a relationship between BCS and the number of BM, BCS may not be the definitive factor in determining the implementation of HS-WBRT in breast cancer patients.
Knowledge around risk factors for PHM remains limited. Sun et al reported that only the number of BM was associated with PHM. Patients with more than 4 BM had significantly increased probability of PHM (OR 3.45 for 4–9 BM vs 1–3 BM, OR 10.50 for ≥10 BM vs 1–3 BM).10 In a study based on the small cell lung cancer, there was a potential association between the number of BM and HM risk, although this relationship was not significant (OR 1.4, P=0.09).21 The RTOG 0933 study excluded all patients with >10 BM due to diffuse distribution.26 In our study, the odds of PHM increased significantly in patients with >10 BM. The results of these studies suggest that a higher number of BM have a higher risk of PHM. In addition, we were unable to obtain accurate data of the number of BM and the time of PHM in the final stage of BM. Therefore, HS-WBRT may be not feasible in patients with a higher number of BM due to the potential risk of hippocampal recurrence.
There are currently no studies that assess the effects of HS-WBRT on breast cancer, and the benefits of HS-WBRT have not yet been validated. Given the low frequency of metastases within the hippocampus, the hippocampus could potentially be defined as a dose-limiting structure for WBRT. Dosimetric results suggest that it is now technically feasible to implement HS-WBRT.15,16,27
There are several limitations in our study that should be considered. First is the retrospective design, which is associated with selection bias. Second, BM was not the first site of distant metastases in most patients. Palliative therapies, including chemotherapy, targeting therapy, and endocrine therapy, may potentially impact on the patterns of BM. In addition, we did not collect data on HM and PHM following WBRT.
Conclusion
We observed a low incidence of HM and PHM in breast cancer patients with BM. A low incidence of PHM may be acceptable to perform HS-WBRT for breast cancer patients. Patients with extensive diffuse metastases (≥10 BM) were associated with higher odds of PHM.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No 81402527), the Sci-Tech Office of Guangdong Province (Nos 2013B021800157 and 2013B021800458), and the Natural Science Foundation of Fujian Province (No 2016J01635).
Disclosure
The authors report no conflicts of interest in this work.
References
Rostami R, Mittal S, Rostami P, Tavassoli F, Jabbari B. Brain metastasis in breast cancer: a comprehensive literature review. J Neurooncol. 2016;127(3):407–414. | ||
Leone JP, Leone BA. Breast cancer brain metastases: the last frontier. Exp Hematol Oncol. 2015;4:33. | ||
Chargari C, Campana F, Pierga JY, et al. Whole-brain radiation therapy in breast cancer patients with brain metastases. Nat Rev Clin Oncol. 2010;7(11):632–640. | ||
Lin NU, Bellon JR, Winer EP. CNS metastases in breast cancer. J Clin Oncol. 2004;22(17):3608–3617. | ||
Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(9422):1665–1672. | ||
Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol. 2009;10(11):1037–1044. | ||
Soffietti R, Kocher M, Abacioglu UM, et al. A European Organisation for Research and Treatment of Cancer phase III trial of adjuvant whole-brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: quality-of-life results. J Clin Oncol. 2013;31(1):65–72. | ||
Kempf SJ, Moertl S, Sepe S, et al. Low-dose ionizing radiation rapidly affects mitochondrial and synaptic signaling pathways in murine hippocampus and cortex. J Proteome Res. 2015;14(5):2055–2064. | ||
Greene-Schloesser D, Moore E, Robbins ME. Molecular pathways: radiation-induced cognitive impairment. Clin Cancer Res. 2013;19(9):2294–2300. | ||
Sun B, Huang Z, Wu S, et al. Incidence and relapse risk of intracranial metastases within the perihippocampal region in 314 patients with breast cancer. Radiother Oncol. 2016;118(1):181–186. | ||
Wu SG, Rao MY, Zhou J, et al. Distribution of metastatic disease in the brain in relation to the hippocampus: a retrospective single-center analysis of 6064 metastases in 632 patients. Oncotarget. 2015;6(41):44030–44036. | ||
Wan JF, Zhang SJ, Wang L, Zhao KL. Implications for preserving neural stem cells in whole brain radiotherapy and prophylactic cranial irradiation: a review of 2270 metastases in 488 patients. J Radiat Res. 2013;54(2):285–291. | ||
Marsh JC, Herskovic AM, Gielda BT, et al. Intracranial metastatic disease spares the limbic circuit: a review of 697 metastatic lesions in 107 patients. Int J Radiat Oncol Biol Phys. 2010;76(2):504–512. | ||
Harth S, Abo-Madyan Y, Zheng L, et al. Estimation of intracranial failure risk following hippocampal-sparing whole brain radiotherapy. Radiother Oncol. 2013;109(1):152–158. | ||
Gondi V, Pugh SL, Tome WA, et al. Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J Clin Oncol. 2014;32(34):3810–3816. | ||
Gondi V, Tolakanahalli R, Mehta MP, et al. Hippocampal-sparing whole-brain radiotherapy: a “how-to” technique using helical tomotherapy and linear accelerator-based intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2010;78(4):1244–1252. | ||
Goldhirsch A, Winer EP, Coates AS, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol. 2013;24(9):2206–2223. | ||
Sorlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci U S A. 2003;100(14):8418–8423. | ||
Arvold ND, Taghian AG, Niemierko A, et al. Age, breast cancer subtype approximation, and local recurrence after breast-conserving therapy. J Clin Oncol. 2011;29(29):3885–3891. | ||
Hong AM, Suo C, Valenzuela M, et al. Low incidence of melanoma brain metastasis in the hippocampus. Radiother Oncol. 2014;111(1):59–62. | ||
Kundapur V, Ellchuk T, Ahmed S, Gondi V. Risk of hippocampal metastases in small cell lung cancer patients at presentation and after cranial irradiation: a safety profile study for hippocampal sparing during prophylactic or therapeutic cranial irradiation. Int J Radiat Oncol Biol Phys. 2015;91(4):781–786. | ||
Jim HS, Phillips KM, Chait S, et al. Meta-analysis of cognitive functioning in breast cancer survivors previously treated with standard-dose chemotherapy. J Clin Oncol. 2012;30(29):3578–3587. | ||
Kesler S, Janelsins M, Koovakkattu D, et al. Reduced hippocampal volume and verbal memory performance associated with interleukin-6 and tumor necrosis factor-alpha levels in chemotherapy-treated breast cancer survivors. Brain Behav Immun. 2013;30(suppl):S109–S116. | ||
Wu SG, Sun JY, Yang LC, et al. Patterns of distant metastasis in Chinese women according to breast cancer subtypes. Oncotarget. 2016;7(30):47975–47984. | ||
Witt J, Pluard TJ, Ferraro DJ, et al. A study of the anatomic distribution of brain metastases in HER2+ breast cancer: implications for hippocampal avoidance PCI. J Clin Oncol. 2014;32(suppl):abstre11578. | ||
Mehta PM, Gondi V, Kanner A, et al. A Phase II Trial of Hippocampal Avoidance during Whole Brain Radiotherapy for Brain Metastases. Radiation Therapy Oncology Group 0933. Available from: https://www.rtog.org/ClinicalTrials/ProtocolTable/StudyDetails.aspx?action=openFile&FileID=7493. Accessed August 14, 2016. | ||
Rong Y, Evans J, Xu-Welliver M, et al. Dosimetric evaluation of intensity-modulated radiotherapy, volumetric modulated arc therapy, and helical tomotherapy for hippocampal-avoidance whole brain radiotherapy. PLoS One. 2015;10(4):e0126222. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


