Back to Journals » OncoTargets and Therapy » Volume 15
Clinical Evidence and Selecting Patients for Treatment with Erdafitinib in Advanced Urothelial Carcinoma
Authors Sayegh N , Tripathi N, Agarwal N , Swami U
Received 9 May 2022
Accepted for publication 20 September 2022
Published 25 September 2022 Volume 2022:15 Pages 1047—1055
DOI https://doi.org/10.2147/OTT.S318332
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Leo Jen-Liang Su
Nicolas Sayegh, Nishita Tripathi, Neeraj Agarwal, Umang Swami
Huntsman Cancer Institute, University of Utah, Salt Lake City, UT, USA
Correspondence: Umang Swami, Division of Oncology, Huntsman Cancer Institute, University of Utah (NCI-CCC), 2000 Circle of Hope Drive Research North Bldg Suite No. 2121, Salt Lake City, UT, 84112, USA, Tel +1 801 587 4697, Email [email protected]
Abstract: Erdafitinib received accelerated approval on April 12, 2019, for patients with metastatic or locally advanced urothelial carcinoma with susceptible fibroblast growth factor receptor (FGFR) 3 or FGFR2 genetic alterations and who have progressed during or following at least one platinum-based chemotherapy. It thus became the first-ever targeted therapy to receive accelerated FDA approval for metastatic bladder cancer. In the BLC2001 trial, erdafitinib demonstrated an overall response rate of 40% in patients with urothelial carcinoma. Common adverse events include hyperphosphatemia and retinopathy and require regular monitoring. While the increase in serum phosphate levels has been determined to be a pharmacodynamic marker of response, further interrogation of other clinical, genomic, and transcriptomic biomarkers is warranted. Results of the ongoing Phase III trial, THOR, which is comparing erdafitinib to the standard of care (chemotherapy or immunotherapy), are expected to confer full approval. Establishing guidelines for optimal erdafitinib sequencing with immunotherapy and other approved targeted therapies (enfortumab vedotin and sacituzumab govitecan) remains an unmet need.
Keywords: advanced urothelial carcinoma, erdafitinib, fibroblast growth factor receptor, targeted therapy, patient selection
Introduction
Bladder cancer is the sixth most commonly diagnosed malignancy and the ninth most common cause of cancer-related deaths in the United States.1 In 2022, it is estimated to account for 81,180 new cases and 17,100 deaths.1 Urothelial carcinoma (UC) is the most common histologic type accounting for more than 90% of the cases.2 It can also arise in the urethra, renal pelvis, and ureter; in these cases, 17% will have simultaneous bladder cancer.3 Once metastatic, the 5-year survival declines drastically to 6% from 70% for localized disease.1
Platinum-based chemotherapy for 4–6 cycles followed by switch maintenance avelumab if no progression is the current first-line treatment for eligible patients with locally advanced or metastatic UC.4,5 For platinum ineligible patients or whose tumors express PD-L1, single-agent atezolizumab or pembrolizumab is an option.4 However, real-world studies show that more than half of the patients with advanced or metastatic bladder cancer never receive first-line therapy and only 17% receive second-line treatment.6 Therefore, there is an urgent, unmet need for effective systemic therapies. At present, second-line treatment options include single-agent immune checkpoint inhibitors directed against programmed cell-death protein-1 or its ligand, enfortumab vedotin-ejfv, and erdafitinib.4 Erdafitinib is the first targeted therapy to receive United States Food and Drug Administration (US FDA) accelerated approval for the treatment of adult patients with locally advanced or metastatic UC. To be eligible, UC must have susceptible fibroblast growth factor receptor (FGFR) 3 or FGFR2 genetic alterations and have progressed during or following at least one line of prior platinum-containing chemotherapy including within 12 months of neoadjuvant or adjuvant platinum-containing chemotherapy. This review discusses the molecular rationale and preclinical and clinical studies leading to its approval.
FGFR Mutations and Role of Erdafitinib
FGFRs belong to the receptor tyrosine kinase family. When ligand-bound or constitutively activated (as in some cancers), they cause downstream activation of RAS/mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3K)-Akt signaling pathways, thereby inducing cell proliferation, anti-apoptosis, differentiation, and angiogenesis (Figure 1).7 FGFR alterations occur in 20% of the patients with advanced UC. Of these, FGFR3 alterations (mutations and fusions) are significant from a therapeutic perspective8,9 and are more common in UTUC than UBC.10,11
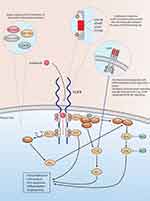 |
Figure 1 Mechanisms of resistance to fibroblast growth factor receptor (FGFR) signaling inhibition. |
After analyzing 412 high-grade chemo- and radiation-naïve samples, The Cancer Genome Atlas Program identified five expression subtypes of MIBC.12 The luminal-papillary subtype exhibits a lower expression of PD-L1 and lower immune signature; however, data concerning response to ICI remain conflictual.13–15 It has been characterized by a high rate of FGFR3 enrichment (44%; mutations (particularly FGFR3S249C), amplification, overexpression, and FGFR3-transforming acid coiled-coil containing protein 3 (TACC3) fusion), making it susceptible to FGFR tyrosine kinase inhibitors.12
Erdafitinib (JNJ-42756493) is an orally administered small molecule that is a potent and selective pan-FGFR (FGFR1-4) tyrosine kinase inhibitor first investigated by Janssen and Astrex pharmaceuticals. In vitro anti-proliferative effects have been demonstrated in cell lines from different cancer types (lung, bladder, breast, colorectal, etc.) through the inhibition of FGFR auto-phosphorylation. Antitumor activity has also been shown in xenograft mouse models involving bladder cancer cells harboring FGFR3 aberrations and manifesting ligand-independent activation of FGFR3. Inhibition of constitutively activated FGFR leads to decreased downstream mitogenic signaling.16
Clinical Evidence
Phase I
Clinical activity of erdafitinib was first demonstrated in a multicenter, first-in-human, phase I trial (EDI1001; NCT0103481), aiming to determine the recommended phase II dose (RP2D) as well as pharmacokinetics, pharmacodynamics, and safety. Sixty-five patients with advanced solid tumors and good performance status were enrolled, of which eight had a diagnosis of UC. A standard 3+3 dose-escalation schema was used. Doses ranging from 0.5 to 12 mg were administered on either a continuous or intermittent basis. Initial RP2D was determined to be 9 mg daily. However, maximum tolerated dose was not established. The most common treatment-emergent adverse events (TEAE) were hyperphosphatemia (65%), asthenia (55%), dry mouth (45%), and nail toxicity (35%). Grade ≥3 TEAEs occurred in 42% of the patients, the most frequent being liver function abnormalities. Recurrent hyperphosphatemia of 7mg/dL or higher was treated with dose reduction, interruption, or administration of oral phosphate binders. The intermittent regimen (7 days on – 7 days off) was associated with lower phosphate levels (<5.5 mg/dL) and fewer interruptions. No significant dose-related effect was noticed with calcium, parathyroid hormone, vitamin D, or FGF23 serum levels. Two of the five patients in the dose confirmation cohort had evaluable immunohistochemistry with a reduced phospho-ERK expression, demonstrating inhibition of the FGFR downstream signaling.17
One hundred and eighty-seven patients with advanced solid tumors were included in the final analysis, of whom 30 had UC. Patients with UC harboring pathogenic alterations in the FGFR genes (11 had fusions and 17 had mutations) had the highest ORR (46.2%, 95% CI 27–67) compared to patients with other solid tumors included in the cohort (27% for cholangiocarcinoma and <10% for other tumor types). Notably, all responders with UC had FGFR alterations. In these patients, the median duration of response was 5.6 months, and the median progression-free survival (PFS) was 5.1 months. The most common TEAEs remained hyperphosphatemia (64%), dry mouth (42%), and asthenia (28%).18
A significant correlation between phosphate and erdafitinib serum concentrations was observed. At 9 mg daily dosing and 10 mg intermittent dosing, the average change of serum phosphate from baseline was 58% and 64%, respectively. Maximum phosphate values ≥5.5 mg/dL by the end of the first cycle with daily dosing, chosen as pharmacodynamic values for drug effect, were achieved in 16 out of 21 responders (76%).18
Phase II
The BLC2001 was a single-arm, multicenter, open-label clinical trial that enrolled 99 patients with unresectable locally advanced or metastatic UC. Inclusion criteria comprised the presence of an FGFR3 mutation or FGFR2/3 fusion and an Eastern Cooperative Oncology Group (ECOG) performance-status score of 2 or less. Disease progression on or after at least one course of systemic chemotherapy was required unless cisplatin-ineligible (progression should have occurred within 12 months if chemotherapy was received in the neoadjuvant/adjuvant setting). Prior immunotherapy was allowed. There was no limit on the number of previous lines of systemic therapy. Patients with brain metastases, impaired kidney function, or persistent hyperphosphatemia were excluded. Treatment consisted of 8 mg of oral erdafitinib daily with possible escalation to 9 mg daily in case the target phosphate concentration of 5.5 mg/dL is not reached.19 A serum phosphate concentration ≥5.5 mg/dL is a pharmacodynamic indicator previously associated with better response rates, as discussed above.18 The primary endpoint was confirmed ORR, and secondary endpoints included PFS, duration of response (DOR), OS, and safety.19
Visceral metastases were present in 79% of the patients, and 12% were chemo naïve. The confirmed ORR was 40%, with 37% partial and 3% complete responses. ORR value was 43% in patients with UTUC and was highest (59%) in those previously treated with immunotherapy. The response was consistent, independent of metastatic site and number of prior therapies. Patients with FGFR3 mutations had a numerically better response than those with FGFR2/3 fusions (ORR of 49% vs 16%, respectively). At a median follow-up of 11 months, median PFS and OS were 5.5 and 13.8 months, respectively. The most common all-grade TEAEs were hyperphosphatemia (77%), stomatitis (58%), and diarrhea (51%).19 Most common grade ≥3 TEAEs were stomatitis (14%) and hyponatremia (11%).20 Grade ≥3 TEAEs were managed with dose reductions, and 13% of the patients discontinued treatment due to adverse events.19
At a median follow-up of 24 months, the study’s final analysis showed persistent clinical benefit regardless of FGFR alteration type, prior systemic treatment type, or presence of visceral metastases. PFS and OS were 5.5 and 11.3 months, respectively.20
Safety
Erdafitinib-associated hyperphosphatemia is due to the inhibition of the sodium-dependent phosphate co-transporter in the proximal renal tubule. It occurs in more than 75% of the cases and can be managed with dose reduction, treatment interruption, and phosphate binders such as sevelamer when phosphate levels are greater than 7mg/dL. Daily phosphate intake should be limited to 600–800 mg, and a nutritionist may be consulted. Concomitant use of phosphate elevating agents such as vitamin D, phosphate-based laxatives/antacids, and potassium phosphate should be avoided before dose escalation. Dry eye syndrome, central serous retinopathy, and retinal pigment epithelial detachment are common ocular toxicities. Therefore, a baseline ophthalmological examination should be performed and repeated monthly for the first 4 months of therapy and every 3 months thereafter. If retinal disorder does not respond to treatment withholding, erdafitinib should be permanently discontinued. Other common toxicities include diarrhea, stomatitis, xerosis, nail disorders (onycholysis and paronychia), and hand-foot syndrome.21
Patient Selection
As discussed above, erdafitinib is approved for patients with metastatic or locally advanced UC harboring susceptible FGFR3 or FGFR2 genetic alterations and who have progressed during or following at least one line of prior platinum-containing chemotherapy.21 Other approved therapies include (1) enfortumab vedotin (EV), a monoclonal antibody (mAb) targeting nectin-4 coupled with a microtubule inhibitor (monomethyl auristatin E), and (2) sacituzumab govitecan (SG) a tumor-associated calcium signal transducer 2 (Trop-2) mAb coupled with SN-38, the active metabolite of irinotecan.22
Antibody drug conjugates (EV, SG) and targeted therapies (erdafitinib) are generally considered when platinum-based regimens and PD-1/L1 inhibitors are no longer an option. However, the optimal treatment sequencing of these agents still needs to be determined. While EV and SG are approved after progression on both platinum-based chemotherapy and PD-1/L1 inhibitors, prior treatment with PD-1/L1 inhibitors is not required by the US FDA label for erdafitinib and EV.22–24 Nevertheless, most patients treated with erdafitinib in the real-world setting are pretreated with immunotherapy.25 Notably, data from the IMVigor 210 and CheckMate 275 trials show that ICIs are effective in patients harboring FGFR2/3 alterations, thus demonstrating that these alterations were not markers of ICI resistance.26 In the BLC2001 trial, the higher ORR in patients who previously progressed on ICIs might be an argument in favor of the sequential use of ICIs before erdafitinib.19 An exciting property of erdafitinib is the conservation of its efficacy in patients with visceral metastases, where ICIs are less effective.20 In a post hoc analysis of the BLC2001 trial, ORR remained similar in patients with ≥2 Bellmunt risk factors compared to low-risk patients.27 The final results of this trial confirmed the preservation of response in patients with poor performance status and visceral metastases.20
While selecting these novel agents, patient’s comorbidities, drug toxicity profile and available biomarkers must be evaluated. The use of erdafitinib is restricted to patients harboring susceptible FGFR 3 or FGFR 2 alterations, detected by therascreen® FGFR Rotor-Gene Q (RGQ) polymerase chain reaction (PCR) kit, which was approved as the companion diagnostic.21 It is worth noting that next-generation sequencing (NGS) has shown to be more sensitive than PCR for detecting FGFR alterations. In a multicancer analysis, only 20% of the FGFR alterations identified by NGS were detected by a PCR-based kit.28 Nevertheless, EV and SG should also be effective in patients with FGFR2/3 alterations.29 Another biomarker to account for is UGT1A1 genotype, as UGT1A1 *28 allele homozygosity is strongly correlated with increased SG toxicity due to reduced SN-38 metabolism.24,30 However, UGT1A1 genotyping is not required before starting SG. For patients receiving SG, special consideration should be given to the high rates of myelosuppression (namely neutropenia and anemia) and severe diarrhea.30,31 EV is the only agent with level 1 evidence showing OS benefit in this setting.32 Factors such as pre-existing neuropathy and hyperglycemia should be assessed before treatment with EV.31 Whether the efficacy of these three agents is influenced by sequencing remains to be investigated.
Potential Biomarkers of Response
In the BLC2001 trial, a differential response was observed between patients harboring FGFR2/3 point mutations or fusions (ORR of 49% and 16%, respectively). The used panel included R248C, S249C, G370C, and Y373C for point mutations, and FGFR2-BICC1, FGFR2-CASP7, FGFR3-TACC3, and FGFR3-BAIAP2L1 for fusions. While there was no difference in response between point mutation types, only patients with FGFR3: TACC3v1 fusion showed objective response.19
The EDI1001 phase I trial identified a phosphate serum level of 5.5 mg/dL as a pharmacodynamic marker, with a threshold of 7 mg/dL for dose adjustments.33 In addition, the correlation of phosphate serum levels with efficacy and safety was investigated in a retrospective study by Dosne et al, using data from the BLC2001 trial.34 Higher serum phosphate levels were significantly associated with better OS, PFS, and ORR, as well as higher rates of nail, dermatologic, and ocular adverse events.
Mechanisms of Resistance
Treatment with FGFR kinase inhibitors may induce the selection of resistant tumoral clones. Several mechanisms are involved in the resistance to FGFR kinase inhibitors and are not specific to erdafitinib or UC (Figure 1). They include bypass signaling, gatekeeper mutations, and tumor heterogeneity. External FGFR inhibition could lead to the activation of downstream signaling through alternate pathways known as bypass signaling. These pathways include MET, EGFR, PI3K/AKT, and RAS-MAPK and are usually caused by mutations or amplifications of their respective proteins.35 The EGFR/HER3-dependent PI3K-AKT signaling pathway overactivation occurs particularly in UC positive for FGFR3-TACC3 fusions.36,37 Gatekeeper mutations appear in the conserved valine residue and alter the interaction between the drug and the FGFR binding site. These mutations, namely FGFR2 V564F/I and FGFR3 V555M, occur either de novo or are treatment-induced. Other described mutations include FGFR1 V561M and FGFR4 V550E/L.35 Intratumoral heterogeneity depicts the differential clonal evolution within unresectable tumors. While the emergence of FGFR-independent and FGFR-dependant clones have been described in both UC and other tumor types, first-line treatment with cytotoxic agents and high tumor mutational burden are important factors contributing to increased intratumoral heterogeneity.38–40 Intrinsic FGFR3 signaling downregulation has been described to occur in UC cells harboring the FGFR3 S249C mutation, consequently rendering them FGFR3 signalling-independent and mainly EGFR signalling-dependant.36,37 Complementary serial comprehensive genomic profiling (CGP) of circulating tumor cells might be useful in overcoming the spatial and temporal limitations of tissue CGP in monitoring the emergence of resistant subclones.41
Ongoing Trials
THOR, a randomized open-label phase III trial is comparing erdafitinib to standard of care (chemotherapy or immunotherapy) in patients with metastatic or inoperable UC harboring prespecified FGFR3 mutations and/or FGFR2/3 fusions (NCT03390504). The target enrollment is 631 patients.42 Prior treatment with systemic therapy is required. Patients showing evidence of brain metastases or those with previous exposure to FGFR inhibitors are excluded. Cohort 1 includes patients pretreated with an anti-PD-L1 agent either alone (for cisplatin-ineligible patients) or with chemotherapy (in combination or maintenance). Cohort 2 includes patients pretreated with only one line of chemotherapy. Erdafitinib will be compared with chemotherapy (docetaxel or vinflunine) and immunotherapy (pembrolizumab) in cohort 1 and cohort 2, respectively. OS is the primary endpoint, while secondary endpoints include PFS, DOR, ORR, safety, and patient-reported outcomes. The results of this trial will be expected to determine the best option between erdafitinib and immunotherapy after progression on first-line chemotherapy in patients with FGFR alterations.
An inverse correlation between FGFR expression and tumoral immunogenicity might be able to explain the poor response of luminal-papillary UC to ICIs. A study by Robinson et al demonstrated that a high expression of FGFR3 was strongly associated with a T-cell-depleted microenvironment and decreased interferon-gamma response genes in UTUC.43 In experimental models, the combination of erdafitinib with anti-PD-1 agents appears to overcome ICI resistance compared to erdafitinib alone by enhancing the antitumor immunity.44 The NORSE study is an ongoing phase Ib/II trial investigating the combination of erdafitinib with cetrelimab, an anti-PD-1 antibody, in patients with advanced UC [NCT03473743]. The results of the phase Ib arm showed that the combination was well tolerated, with an ORR of 17% among the 15 participants.45 The preliminary results of the phase II arm demonstrated an ORR of 68% versus 33% in patients receiving the combination (n=27) and those receiving erdafitinib alone (n=26) respectively. TEAEs were concordant with the BLC2001 trial.46 Other ongoing trials are summarized in Table 1.
 |
Table 1 Ongoing Trials of Erdafitinib in Patients with Urothelial Carcinoma |
Conclusion
Erdafitinib, a small-molecule pan-FGFR inhibitor, has been showing clinical benefit in several trials, with acceptable safety and tolerability. In April 2019, it became the first FGFR kinase inhibitor to ever receive accelerated FDA approval for use in patients with platinum-refractory advanced UC. While it expands the array of treatments beyond progression on chemotherapy and/or immunotherapy, results of a phase III trial are needed to establish level 1 evidence of its utility. In addition, further investigation in determining its optimal sequencing with other antibody–drug conjugates and immunotherapy remains an unmet need.
Disclosure
Dr Neeraj Agarwal reports personal fees from Astellas, Astra Zeneca, Aveo, Bayer, Bristol Myers Squibb, Calithera, Clovis, Eisai, Eli Lilly, EMD Serono, Exelixis, Foundation Medicine, Genentech, Gilead, Janssen, Merck, MEI Pharma, Nektar, Novartis, Pfizer, Pharmacyclics, and Seattle Genetics, other from Astellas, Astra Zeneca, Bavarian Nordic, Bayer, Bristol Myers Squibb, Calithera, Celldex, Clovis, Eisai, Eli Lilly, EMD Serono, Exelixis, Genentech, Gilead, Glaxo Smith Kline, Immunomedics, Janssen, Medivation, Merck, Nektar, New Link Genetics, Novartis, Pfizer, Prometheus, Rexahn, Roche, Sanofi, Seattle Genetics, Takeda, and Tracon, during the conduct of the study. Dr Umang Swami reports personal fees from Astellas, Exelixis and Seattle Genetics, grants from Janssen, Exelixis and Astellas/Seattle Genetics, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7–33. doi:10.3322/caac.21708
2. Dahm P, Gschwend JE. Malignant non-urothelial neoplasms of the urinary bladder: a review. Eur Urol. 2003;44(6):672–681. doi:10.1016/S0302-2838(03)00416-0
3. Cosentino M, Palou J, Gaya JM, et al. Upper urinary tract urothelial cell carcinoma: location as a predictive factor for concomitant bladder carcinoma. World J Urol. 2013;31(1):141–145. doi:10.1007/s00345-012-0877-2
4. National Comprehensive Cancer Network. Bladder Cancer (Version 1.2022); 2022. Available from: https://www.nccn.org/professionals/physician_gls/pdf/bladder.pdf.
5. Powles T, Park SH, Voog E, et al. Avelumab maintenance therapy for advanced or metastatic urothelial carcinoma. N Engl J Med. 2020;383(13):1218–1230. doi:10.1056/NEJMoa2002788
6. Swami U, Grivas P, Pal SK, et al. Utilization of systemic therapy for treatment of advanced urothelial carcinoma: lessons from real world experience. Cancer Treat Res Commun. 2021;27:100325. doi:10.1016/j.ctarc.2021.100325
7. Katoh M, Nakagama H. FGF receptors: cancer biology and therapeutics. Med Res Rev. 2014;34(2):280–300. doi:10.1002/med.21288
8. Knowles MA, Hurst CD. Molecular biology of bladder cancer: new insights into pathogenesis and clinical diversity. Nat Rev Cancer. 2015;15(1):25–41. doi:10.1038/nrc3817
9. Di Martino E, Tomlinson DC, Knowles MA. A decade of fgf receptor research in bladder cancer: past, present, and future challenges. Adv Urol. 2012;2012(p):429213. doi:10.1155/2012/429213
10. Sfakianos JP, Cha EK, Iyer G, et al. Genomic characterization of upper tract urothelial carcinoma. Eur Urol. 2015;68(6):970–977. doi:10.1016/j.eururo.2015.07.039
11. Necchi A, Madison R, Pal SK, et al. Comprehensive genomic profiling of upper-tract and bladder urothelial carcinoma. Eur Urol Focus. 2021;7(6):1339–1346. doi:10.1016/j.euf.2020.08.001
12. Robertson AG, Kim J, Al-Ahmadie H, et al. Comprehensive molecular characterization of muscle-invasive bladder cancer. Cell. 2017;171(3):540–556e25. doi:10.1016/j.cell.2017.09.007
13. Siefker-Radtke A, Curti B. Immunotherapy in metastatic urothelial carcinoma: focus on immune checkpoint inhibition. Nat Rev Urol. 2018;15(2):112–124. doi:10.1038/nrurol.2017.190
14. Rosenberg JE, Hoffman-Censits J, Powles T, et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, Phase 2 trial. Lancet. 2016;387(10031):1909–1920. doi:10.1016/S0140-6736(16)00561-4
15. Kim J, Kwiatkowski D, McConkey DJ, et al. The cancer genome atlas expression subtypes stratify response to checkpoint inhibition in advanced urothelial cancer and identify a subset of patients with high survival probability. Eur Urol. 2019;75(6):961–964. doi:10.1016/j.eururo.2019.02.017
16. Perera TPS, Jovcheva E, Mevellec L, et al. Discovery and pharmacological characterization of JNJ-42756493 (erdafitinib), a functionally selective small-molecule FGFR family inhibitor. Mol Cancer Ther. 2017;16(6):1010–1020. doi:10.1158/1535-7163.MCT-16-0589
17. Tabernero J, Bahleda R, Dienstmann R, et al. Phase I dose-escalation study of JNJ-42756493, an oral pan-fibroblast growth factor receptor inhibitor, in patients with advanced solid tumors. J Clin Oncol. 2015;33(30):3401–3408. doi:10.1200/JCO.2014.60.7341
18. Bahleda R, Italiano A, Hierro C, et al. Multicenter phase I study of erdafitinib (JNJ-42756493), oral pan-fibroblast growth factor receptor inhibitor, in patients with advanced or refractory solid tumors. Clin Cancer Res. 2019;25(16):4888–4897. doi:10.1158/1078-0432.CCR-18-3334
19. Loriot Y, Necchi A, Park SH, et al. Erdafitinib in locally advanced or metastatic urothelial carcinoma. N Engl J Med. 2019;381(4):338–348. doi:10.1056/NEJMoa1817323
20. Siefker-Radtke AO, Necchi A, Park SH, et al. Efficacy and safety of erdafitinib in patients with locally advanced or metastatic urothelial carcinoma: long-term follow-up of a phase 2 study. Lancet Oncol. 2022;23(2):248–258. doi:10.1016/S1470-2045(21)00660-4
21. United States Food and Drug Administration. BALVERSA® prescribing information. Available from: https://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/BALVERSA-pi.pdf.
22. United States Food and Drug Administration. Hematology/oncology (cancer) approvals & safety notifications; 2022; Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/hematologyoncology-cancer-approvals-safety-notifications.
23. United States Food and Drug Administration. PADCEV® prescribing information Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/761137s000lbl.pdf
24. United States Food and Drug Administration. TRODELVY® prescribing information Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761115s009lbl.pdf
25. de Almeida Carvalho LM, de Oliveira Sapori Avelar S, Haslam A, et al. Estimation of percentage of patients with fibroblast growth factor receptor alterations eligible for off-label use of erdafitinib. JAMA Netw Open. 2019;2(11):e1916091 . doi:10.1001/jamanetworkopen.2019.16091
26. Wang L, Gong Y, Saci A, et al. Fibroblast growth factor receptor 3 alterations and response to PD-1/PD-L1 blockade in patients with metastatic urothelial cancer. Eur Urol. 2019;76(5):599–603. doi:10.1016/j.eururo.2019.06.025
27. Park SH, Loriot Y, Zhong B, et al. Erdafitinib in high-risk patients (pts) with advanced urothelial carcinoma (UC). J Clin Oncol. 2019;37(15_suppl):4543. doi:10.1200/JCO.2019.37.15_suppl.4543
28. Dios ID, Ma W, Antzoulatos S, et al. FGFR expression, fusion and mutation as detected by NGS sequencing of DNA and RNA. J Clin Oncol. 2020;38(15_suppl):e16061–e16061. doi:10.1200/JCO.2020.38.15_suppl.e16061
29. Adib E, Jain RK, Skelton WP, et al. Impact of FGFR2/3 activating genomic alterations on response to enfortumab vedotin in metastatic urothelial carcinoma (mUC). J Clin Oncol. 2021;39(6_suppl):472. doi:10.1200/JCO.2021.39.6_suppl.472
30. Bardia A, Messersmith WA, Kio EA, et al. Sacituzumab govitecan, a trop-2-directed antibody-drug conjugate, for patients with epithelial cancer: final safety and efficacy results from the phase I/II IMMU-132-01 basket trial. Ann Oncol. 2021;32(6):746–756. doi:10.1016/j.annonc.2021.03.005
31. Atiq S, Hirshman N, Shariff A, et al. The management of toxicities from immune, targeted and ADCs treatments in patients with urothelial cancer. Urol Oncol. 2021. doi:10.1016/j.urolonc.2021.10.002
32. Powles T, Rosenberg JE, Sonpavde GP, et al. Enfortumab vedotin in previously treated advanced urothelial carcinoma. N Engl J Med. 2021;384(12):1125–1135. doi:10.1056/NEJMoa2035807
33. Yu D, Fan K, Zhang H, et al. PizzaText: text entry for virtual reality systems using dual thumbsticks. IEEE Trans Vis Comput Graph. 2018;24(11):2927–2935. doi:10.1109/TVCG.2018.2868581
34. Dosne AG, Valade E, Goeyvaerts N, et al. Exposure-response analyses of erdafitinib in patients with locally advanced or metastatic urothelial carcinoma. Cancer Chemother Pharmacol. 2022;89(2):151–164. doi:10.1007/s00280-021-04381-4
35. Katoh M. Fibroblast growth factor receptors as treatment targets in clinical oncology. Nat Rev Clin Oncol. 2019;16(2):105–122. doi:10.1038/s41571-018-0115-y
36. Herrera-Abreu MT, Pearson A, Campbell J, et al. Parallel RNA interference screens identify EGFR activation as an escape mechanism in FGFR3 -mutant cancer. Cancer Discov. 2013;3(9):1058–1071. doi:10.1158/2159-8290.CD-12-0569
37. Wang L, Šuštić T, Leite de Oliveira R, et al. A functional genetic screen identifies the phosphoinositide 3-kinase pathway as a determinant of resistance to fibroblast growth factor receptor inhibitors in FGFR mutant urothelial cell carcinoma. Eur Urol. 2017;71(6):858–862. doi:10.1016/j.eururo.2017.01.021
38. Shi JY, Xing Q, Duan M, et al. Inferring the progression of multifocal liver cancer from spatial and temporal genomic heterogeneity. Oncotarget. 2016;7(3):2867–2877. doi:10.18632/oncotarget.6558
39. Gao Q, Wang Z-C, Duan M, et al. Cell culture system for analysis of genetic heterogeneity within hepatocellular carcinomas and response to pharmacologic agents. Gastroenterology. 2017;152(1):232–242. doi:10.1053/j.gastro.2016.09.008
40. Yarchoan M, Hopkins A, Jaffee EM. Tumor mutational burden and response rate to PD-1 inhibition. N Engl J Med. 2017;377(25):2500–2501. doi:10.1056/NEJMc1713444
41. Barata PC, Koshkin VS, Funchain P, et al. Next-generation sequencing (NGS) of cell-free circulating tumor DNA and tumor tissue in patients with advanced urothelial cancer: a pilot assessment of concordance. Ann Oncol. 2017;28(10):2458–2463. doi:10.1093/annonc/mdx405
42. Loriot Y, Necchi A, Park SH, et al. Erdafitinib compared with vinflunine or docetaxel or pembrolizumab in patients (pts) with metastatic or surgically unresectable (M/UR) urothelial carcinoma (UC) and selected fgfr gene alterations (FGFRalt): the phase III THOR study. Ann Oncol. 2018;29:viii327–viii328. doi:10.1093/annonc/mdy283.129
43. Robinson BD, Vlachostergios PJ, Bhinder B, et al. Upper tract urothelial carcinoma has a luminal-papillary T-cell depleted contexture and activated FGFR3 signaling. Nat Commun. 2019;10(1):2977. doi:10.1038/s41467-019-10873-y
44. Palakurthi S, Kuraguchi M, Zacharek SJ, et al. The combined effect of FGFR inhibition and PD-1 blockade promotes tumor-intrinsic induction of antitumor immunity. Cancer Immunol Res. 2019;7(9):1457–1471. doi:10.1158/2326-6066.CIR-18-0595
45. Moreno V, Loriot Y, Valderrama BP, et al. Does escalation results from phase Ib/II Norse study of erdafitinib (ERDA) + PD-1 inhibitor JNJ-63723283 (Cetrelimab [CET]) in patients (pts) with metastatic or locally advanced urothelial carcinoma (mUC) and selected fibroblast growth factor receptor (FGFR) gene alterations. J Clin Oncol. 2020;38(6_suppl):511.
46. Powles TB, Chistyakov V, Beliakouski V, et al. LBA27 - erdafitinib (ERDA) or ERDA plus cetrelimab (CET) for patients with metastatic or locally advanced urothelial carcinoma (mUC) and Fibroblast Growth Factor Receptor alterations (FGFRa): first phase (Ph) II results from the NORSE study. Ann Oncol. 2021;32(suppl_5):S1283–S1346. doi:10.1016/annonc/annonc74
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
