Back to Journals » Drug Design, Development and Therapy » Volume 9
Clinical evaluation of the efficacy and safety of a medical device in various forms containing Triticum vulgare for the treatment of venous leg ulcers – a randomized pilot study
Authors Romanelli M , Macchia M, Panduri S, Paggi B, Saponati G, Dini V
Received 12 February 2015
Accepted for publication 2 April 2015
Published 27 May 2015 Volume 2015:9 Pages 2787—2792
DOI https://doi.org/10.2147/DDDT.S82712
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Shu-Feng Zhou
Marco Romanelli,1 Michela Macchia,1 Salvatore Panduri,1 Battistino Paggi,1 Giorgio Saponati,2 Valentina Dini1
1Wound Healing Research Unit, Dermatology Division, Department of Clinical and Experimental Medicine, University of Pisa, 2ISPharm srl, Lucca, Italy
Abstract: This study was carried out to assess the efficacy and tolerability of the topical application of an aqueous extract of Triticum vulgare (TV) in different vehicles (cream, impregnated gauzes, foam, hydrogel, and dressing gel) for the treatment of venous lower leg ulcers. Fifty patients were randomized to receive one of the five investigational vehicles. Treatment was performed up to complete healing or to a maximum of 29 days. The wound size reduction from baseline was the primary efficacy variable, which was measured by means of a noninvasive laser scanner instrument for wound assessment. In all groups, apart from the foam group, a similar trend toward the reduction of the surface area was observed. The cream showed the greatest effect on the mean reduction of the lesion size. At last visit, six ulcers were healed: two in the cream group, three in the gauze group, and one in the dressing gel group. In the patients treated with the cream, the gauzes, the hydrogel, and the dressing gel, the reduction of lesion size was 40%–50%; the reduction was smaller in the foam group. No impact in terms of age on the healing process was found. The Total Symptoms Score decreased in all groups during the study; a greater efficacy in terms of signs/symptoms was observed in the patients treated with the gauzes. In the dressing gel group, one patient had an infection of the wound after 3 weeks of treatment and 2 of colonization, leading to a systemic antibiotic treatment. The events were judged as nonrelated to the device used. On the basis of the results, it could be argued that the medical device may be useful in the treatment of chronic venous ulcers.
Keywords: venous leg ulcers, Triticum vulgare, wound dressings
Introduction
Approximately 1% of the Western population suffers from venous leg ulcers;1 they are more common among women and in the elderly.2 These figures may be underestimated as a result of inadequate assessment, which fails to identify venous ulcers, and as a result of underreporting.3 The primary risk factors are old age, obesity, previous leg injuries, deep venous thrombosis, and phlebitis.4 Venous leg ulcers are often recurrent, and open ulcers can persist from weeks to many years.5 Severe complications include cellulitis, osteomyelitis, and malignant change. The refractory nature of these ulcers increases the risk of morbidity and mortality and can have a significant impact on patient quality of life and on costs.6–9 Compression therapy is the standard care for venous ulcers and chronic venous insufficiency;10,11 various types of compression therapy are available.12–14 Compression reduces edema, improves venous reflux, enhances healing of ulcers, and reduces pain. Success rates range from 30% to 60% at 24 weeks and from 70% to 85% after 1 year.15 After an ulcer has healed, lifelong maintenance of compression therapy may reduce the risk of recurrence.16 However, adherence to the therapy may be limited by pain, amount of exudate, dressing change difficulties, and physical limitations, including obesity and contact dermatitis.17,18 Dressings are often used under compression bandages to promote faster healing and prevent adherence of the bandage to the ulcer bed. A wide range of dressings are available, including hydrocolloids, foams, hydrogels, ointments, and simple nonadherent dressings.19,20 No definitive evidence exists of a clinically significant difference among dressing types.19
A new medical device produced in the pharmaceutical forms of cream, soaked gauzes, foam, hydrogel, and dressing gel, intended for the topical treatment of cutaneous wounds, is currently under development by the manufacturer. The device, containing an aqueous extract of Triticum vulgare, acts by removing the excess exudate from the wound surface and providing a moist, sterile microenvironment, a barrier to microorganisms, and thermal insulation. The aim of this study was to evaluate the efficacy and the local tolerability of the various forms of the medical device in the topical treatment of venous leg ulcers and to identify differences in the activity – if any – between the various pharmaceutical forms. This was a preliminary pilot study conducted on a small group of patients.
Methods
Patient population
The study protocol included patients of both sexes aged 18–85 years, affected by a single venous ulcer of the lower limb lasting no more than 6 months. To be eligible for the study, the lesion was required to have clinical and instrumental signs of venous insufficiency, with the edema capable of being controlled by compression and a lesion surface area not smaller than 5 cm2 and not greater than 100 cm2. Patients with the following conditions at the screening had to be excluded from study participation: pregnancy or breast-feeding; obstructive chronic arteriopathies involving the affected area of the study lesion; concomitant treatment with antibiotics, NSAID, narcotics, corticosteroids, antineoplastics, or immunosoppressants; or presence of other important medical conditions that may interfere with wound healing.
Study design and treatments
The study plan included a baseline visit, wherein the patients eligible for the study were randomized using the block randomization method which is designed to randomize subjects into groups that result in equal sample sizes. This method is used to ensure a balance in sample size across groups over time. Patients were assigned to receive one of the following five treatments: cream, soaked gauzes, foam, hydrogel, and dressing gel. The soaked gauzes and the dressing gel were applied onto the lesion which had previously been cleansed with a sterile saline solution and covered with a sterile gauze and a secondary absorbent dressing in the case of particularly exuding lesions. A two-layer bandaging system was applied after every dressing change. In the patient group treated with the cream, foam, and hydrogel, the products were uniformly distributed on a sterile gauze and then applied onto the lesion, as with the gauzes. All the study devices were applied at the study site every 3 days, starting from the baseline visit. When the dressing was replaced, any devitalized tissue was removed using a clip or buffer with saline solution. Follow-up visits were scheduled at weekly intervals of therapy, for a maximal observational period of 29 days and a maximum of five visits. Leg compression was continued through the study. The use of paracetamol (acetaminophen, 500 mg tablets) was permitted as a painkiller.
Outcome measures
The reduction from baseline of the ulcer surface area was the primary efficacy variable.21 The surface area was measured by means of a laser scanner (Shilouette®, Aranz Medical, Christchurch, New Zealand). Signs (perilesional erythema, bleeding) and symptoms (burning, pain, itching) due to the lesion were measured using a 4-point scale (0–3: absent, mild, moderate, severe) at each visit, and in each patient the scores were added up to obtain the Total Symptoms Score (TSS). Other efficacy variables included the presence/absence of granulation tissue and the use of relief paracetamol. Adverse events were recorded at all times throughout the study.
Ethics
The study protocol was approved by the reference Ethical Committee. Informed written consent was signed by all participants prior to any study-related procedure being started. The study was conducted according to the Declaration of Helsinki (Fortaleza) and the Good Clinical Practice rules.
Statistical analysis
Being a pilot study, a calculation of the sample size was not performed. The following elements were considered for data analysis: safety, ie, all randomized patients who received at least one application of study devices; intention-to-treat (ITT), ie, all patients of the safety population who did not violate major inclusion–exclusion criteria; per protocol (PP), ie, all patients of the efficacy population who completed the study without major violations of study procedures. The analysis of the primary efficacy variables was planned on both the ITT and PP populations, whereas the analysis of secondary variables was planned on the ITT population only. Moreover, no major protocol deviations were found, and all analyses were conducted on the ITT population. All analyses were performed separately inside the treatment groups. The primary analysis of efficacy was performed on the reduction of the ulcer surface area at last visit compared to baseline. Parametric (Student’s paired t-test) and nonparametric (Wilcoxon test) methods were used. The interval confidence (IC)95% of the mean of differences was calculated at time points following the baseline visit. The same methods described for the primary efficacy analysis were used for the reduction of the TSS compared to baseline. ANOVA was applied to evaluate the impact of age on the healing process. The LOCF (last observation carried forward) method was adopted for missing data. As a consequence of a marked right-skewness of the surface area values, in addition to the arithmetic mean, the quadratic mean (root mean square, RMS) was calculated at the time points as a descriptive statistic.
Results
Patient disposition and baseline characteristics
A total of 50 patients were enrolled and randomized to receive the assigned treatments (ten patients in each treatment group). All randomized patients entered the safety and ITT population. Table 1 shows the demographic and main baseline characteristics in the five groups. The mean lowest age was found in the hydrogel group, and the highest in the dressing gel group. The foam group showed the largest mean ulcer size, and the hydrogel group the smallest.
Efficacy
Table 2 shows the differences vs baseline of the ulcer surface area throughout the study, as well as the P-values. Figure 1 shows the ulcer size (quadratic mean) in the groups at the study time points.
  | Figure 1 Quadratic mean of ulcer surface area (cm2) in all the groups during the study. |
In all the groups, apart from the foam group, a similar trend toward a reduction of the ulcer surface area was observed. The cream showed the greatest effect on the mean reduction of lesion size. At last visit, six ulcers were healed: two in the cream group, three in the gauzes group, and one in the dressing gel group. In one patient of the gauzes group, the size of the ulcer remained almost unchanged. The percentage reduction of the ulcer surface area at last visit is shown in Figure 2. In the patients treated with the cream, the gauzes, the hydrogel, and the dressing gel, the reduction of lesion size was 40%–50%; the reduction was smallest in the foam group.
Apparently, age did not significantly influence the healing process.
The TSS decreased in all groups throughout the study (Figure 3): the greatest efficacy on signs/symptoms was observed in the patients treated with the gauzes (P-values: Student’s t, 0.031; Wilcoxon, 0.023).
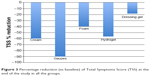  | Figure 3 Percentage reduction (vs baseline) of Total Symptoms Score (TSS) at the end of the study in all the groups. |
The presence of granulation tissue was observed in almost all the patients (cream =8, gauzes =8, foam =9, hydrogel =10, dressing gel =8).
No patients took paracetamol. None of patients in any group required surgical cleaning or debridement of the lesion at any time during the study.
Safety
In the dressing gel group after 3 weeks of treatment and 2 of colonization, one patient showed an infection of the wound, leading to treatment with systemic antibiotics.22 The events were judged as nonrelated to the device used.
Discussion
The etiology of venous ulcers is complex and not fully understood, but is thought to be due to chronic venous hypertension resulting from venous insufficiency caused by a deficient calf-muscle pump.23 People presenting with venous leg ulcers have venous insufficiency due to problems with the calf-muscle pump as a result of venous occlusion or incompetence of the valves in the deep, superficial, or perforating veins.24 Secondary venous insufficiency occurs when damage to the valves is caused by an identifiable condition, such as deep vein thrombosis.25 Damage to the venous valves results in the blood flowing in the inverse direction, pooling of blood in the veins, and exposure of the veins to high pressure. Venous hypertension results in localized edema, producing localized tissue damage. As a consequence, tissues may fail to heal after even minor trauma, resulting in an ulcer. Once a venous ulcer has occurred it often follows a cyclical pattern of healing and recurrence. Recurrence rates between 45% and 70% at 1 year are reported.26 The time to healing can be very long, and a significant number of patients do not complete ulcer healing, or do so only after many years.27 Compression bandaging has been identified as the gold standard in the treatment of venous leg ulcers. The compression system is applied in a graduated fashion, with the pressure decreasing from the toe to the knee. The reduction of venous pressure and edema allows healing of the ulcer to occur. The healing rate can vary, depending on initial ulcer size and duration; a mean healing rate of 70% has been reported for recent smaller ulcers,28 while a randomized controlled clinical trial that included ulcers of various sizes and duration reported a healing rate of 34%.29 The use of dressing placed over the ulcer prior to the application of compression to improve healing is widespread, and the number of available products is increasing.30,31 However, no clear evidence-based efficacy difference exists among dressing types; therefore, new products are currently under development. The various pharmaceutical forms of the medical device evaluated in this study are intended for topical treatment of cutaneous lesions of different origin (wounds, burns, ulcers, etc). In the composition of the device, the watery extract of T. vulgare appears, which has been proven to improve the healing of superficial damaged tissues.32 In a recent study it has been shown to have a pro-proliferative effect of the fractions ST-98 and K >1,000 in NIH-3T3 fibroblasts. Moreover, these fractions formulated as cream preparations were effective in in vivo models of skin lesion also.33 In this study, we evaluated the effects of the medical device as dressing in addition to compression in patients suffering from venous leg ulcers. In all groups, a reduction in the lesion surface area and clinical signs/symptoms of the disease was observed. The best results were recorded in the patients treated with the cream (two ulcers healed), the gauzes (three ulcers healed), and the hydrogel; moreover, it cannot be excluded that on a larger treatment time-span and/or in selected patients the dressing gel (one ulcer healed) and foam also can show greater activity. Indeed, a similar frequency of granulation tissue was observed in all groups. Peculiarly, the effects of the different pharmaceuticals on the clinical signs/symptoms and surface area reduction did not correspond; in fact, the greatest effect on surface area was observed in the cream and the hydrogel groups, whereas the reduction of TSS was more noticeable in the gauzes group. This could be due to the higher baseline score in the latter group, but other explanations cannot be excluded. No safety concerns arose during the study. Some intentional limitations affected the study. They were mainly due to the small number of patients in the groups, the relatively short period of observation (4 weeks), and the absence of a control group. This was a pilot study aimed at evaluating the usefulness of the medical device in the treatment of venous ulcers and at speculatively examining different activities (if any) in the pharmaceutical forms. Therefore, a formal sample size calculation was not performed and ten patients in each group were judged as an adequate number in order to detect activity and possible differences in such activity. This seemed reasonable to us, but, as a consequence, the significance (P-value) of the statistical tests should be interpreted very cautiously. The healing of venous ulcers usually requires a month-long treatment; however, the primary variable of the study was not the time to healing, but rather the reduction of the ulcer surface area within a fixed time, nevertheless, six ulcers healed. Finally, the absence of a control group appeared to be justified by the preliminary nature of the study; the results of the study may prove to be the starting point for a larger, longer-lasting, controlled, randomized clinical trial.
On the basis of the results, it can be argued that the medical device can be useful in the treatment of chronic venous ulcers. Since the device acts as a protective dressing, it may have a greater healing effect on acute skin injuries also, such as traumatic wounds and superficial burns.
Acknowledgment
Damor Pharmaceuticals granted the Dermatology Division for conducting the study and ISPharm srl for managing the data and revising the manuscript.
Disclosure
The product was codeveloped in partnership with a pharmaceutical institute responsible for designing, supervising, and coordinating the study. Otherwise, the authors report no conflicts of interest in this work.
References
White-Chu EF, Conner-Kerr TA. Overview of guidelines for the prevention and treatment of venous leg ulcers: a US perspective. J Multidiscip Healthc. 2014;7:111–117. | ||
Abbade LP, Lastoria S. Venous ulcer: epidemiology, physiopathology, diagnosis and treatment. Int J Dermatol. 2011;44(6):449–456. | ||
Phillips TJ. Chronic cutaneous ulcers-aetiology and epidemiology. J Invest Dermatol. 1994;42:S38–S41. | ||
Nelson EA, Bell-Syer SE, Cullum NA. Compression for preventing recurrence of venous ulcers. Cochrane Database Syst Rev. 2000;(4):CD0023037. | ||
Briggs M, Nelson EA. Topical agents or dressing for pain in venous leg ulcers. Cochrane Database Syst Rev. 2003;(1):CD001177. | ||
Ruckley CV. Socioeconomic impact of chronic venous insufficiency and leg ulcers. Angiology. 1997;48(1):67–69. | ||
Green J, Jester R. Health-related quality of life and chronic venous leg ulceration: part 1. Br J Community Nurs. 2009;14(12):S12, S14, S16–S17. | ||
Lozano Sanchez FS, Marinel Io Roura J, Carrasco Carrasco E, et al. Venous leg ulcer in the context of chronic venous disease. Phlebology. 2014;29(4):220–226. | ||
Etufug CN, Phillips TJ. Venous ulcers. Clin Dermatol. 2007;25(1):121–130. | ||
Fletcher A, Cullum N, Sheldon TA. A systematic review of compression treatment for venous leg ulcers. BMJ. 1997;315(7108):576–580. | ||
Collins L, Seraj S. Diagnosis and treatment of venous ulcers. Am Fam Physician. 2010;81(8):989–996. | ||
Dolibog P, Franek A, Taradaj J, et al. A comparative clinical study on five types of compression therapy in patients with venous leg ulcers. Int J Med Sci. 2014;11(1):34–43. | ||
Partsch H. Compression for the management of venous leg ulcers: which material do we have? Phlebology. 2014;29(1 Suppl):140–145. | ||
Mauch KF, Asi N, Elraiyah TA, et al. Comparative systematic review and meta-analysis of compression modalities for the promotion of venous ulcer healing and reducing ulcer recurrence. J Vasc Surg. 2014;60:71S–90S. | ||
Margolis DJ, Berlin JA, Strom BL. Which venous leg ulcers will heal with limb compression bandage? Am J Med. 2000;109(1):15–19. | ||
Phillips TJ, Machado F, Trout R, et al. Prognostic indicators in vnous ulcers. J Am Acad Dermatol. 2000;43(4):627–630. | ||
Raju S, Neglén P. Clinical practice. Chronic venous insufficiency and varicose veins. N Engl J Med. 2009;360(22):2319–2327. | ||
Baindbridge P. Why don’t patients adhere to compression therapy? Br J Community Nurs. 2013;(Suppl):S35–S36, S38–S40. | ||
Palfreyman S, Nelson EA, Michaels JA. Dressing for venous leg ulcers: systematic review and meta-analysis. BMJ. 2007;335(7613):244. | ||
Warriner RA, Carter MJ. The current state of evidence-based protocols in wound care. Plast Reconstr Surg. 2011;127(Suppl 1):144S–153S. | ||
Margolis DM, Berlin JA, Strom BL. Risk factors associated with failure of a venous ulcer to heal. Arch Dermatol. 1999;135:920–926. | ||
European Wound Management Association. Identifying criteria for wound infection. London, UK: Medical Education Partnership Ltd; 2005. | ||
Grey JF, Harding KG, Enoch S. Venous and arterial leg ulcers. BMJ. 2006;332(7537):347–350. | ||
Bauersachs J, Fleming I, Busse R. Pathophysiology of chronic venous insufficiency. Phlebology. 1996;11(1):16–22. | ||
Angle N, Bergan JJ. Chronic venous ulcer. BMJ. 1997;314(7086):1019. | ||
Lees TA, Lambert D. Prevalence of lower limb ulceration in an urban health district. Br J Surg. 1992;79(10):1032–1034. | ||
Rukley CV. Caring for patients with chronic leg ulcer. BMJ. 1998;316(7129):407–408. | ||
Royal College of Nursing. The management of patients with venous leg ulcers. London, UK: Royal College of Nursing; 2006. | ||
Morrel CJ, Walters S, Dixon S, et al. Cost-effectiveness of community leg ulcer clinics: a randomised clinical trial. BMJ. 1998;316(3):1487–1491. | ||
Maggio G, Armenio A, Ruccia F, et al. A new protocol for the treatment of the chronic venous ulcers of the lower limb. Clin Exp Med. 2012;12(1):55–60. | ||
Salazar-Alvareza AE, Riera-Del Moral LF, Garcia-Arranz M, et al. Use of platelet–rich plasma in the healing of chronic ulcers of the lower extremity. Actas Dermosifiliogr. 2014;105(6):597–604. | ||
Martini P, Mazzatenta C, Saponati G. Efficacy and tolerability of Fitostimoline in two different forms (soaked gauzes and cream) and Citrizan gel in the topical treatment of second-degree superficial cutaneous burns. Dermatology Res Pract. 2011;634:24–28. | ||
Sanguigno L, Minale M, Vannini E, et al. Oligosaccharidic fractions derived from Triticum vulgare extract accelerate tissutal repairing processes in in vitro and in vivo models of skin lesions. J Ethnopharmacol. 2015;159:198–208. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



