Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 10
Clinical efficacy of emollients in atopic dermatitis patients – relationship with the skin microbiota modification
Authors Seité S , Zelenkova H, Martin R
Received 8 September 2016
Accepted for publication 2 November 2016
Published 12 January 2017 Volume 2017:10 Pages 25—33
DOI https://doi.org/10.2147/CCID.S121910
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jeffrey Weinberg
Sophie Seité,1 Hana Zelenkova,2 Richard Martin3
1La Roche-Posay Dermatological Laboratories, Asnières, France; 2DOST, Private Clinic of Dermatovenereology, Svidnik, Slovakia; 3L’Oréal Research and Innovation, Tours, France
Background: We speculated that an emollient supplemented with a biomass of nonpathogenic bacteria such as Vitreoscilla filiformis (Vf), grown in a medium containing thermal spring water (LRP-TSW); (LRP-Vitreoscilla filiformis biomass [LRP-VFB]), could have a beneficial effect for patients with atopic dermatitis (AD).
Patients and methods: This double-blind, randomized, comparative study was conducted with 60 patients with moderate AD. Before starting the study, participants were pretreated for 15 days with drug therapy to improve their SCORing Atopic Dermatitis (SCORAD) by at least 25%. On Day 1, the eligible patients were randomized to either the emollient containing LRP-VFB associated with mannose (Product A) or another emollient (product B) and were treated twice daily for 1 month. Recurrence of flare-ups and microbial communities were characterized from swabs taken at Day 1 and Day 28, under axenic conditions, from affected (AF) and proximal unaffected (UAF) skin areas.
Results: At Day 1, the average SCORAD of each group and the microbial communities of AF and UAF areas for each participant were similar. One month after the end of the therapeutic treatment (Day 28), the average evolution of SCORAD at Day 28 compared to Day 1 of patients treated with product A was significantly lower than that of the patients treated with product B. A significantly increased level of Xanthomonas genus was noticed in the group treated with product A (versus product B). On the other hand, the level of Staphylococcus genus increased between Day 1 and Day 28 in the group treated with product B, but not in the group treated with product A. Interestingly, these differences were more pronounced for patients in relapse, and the associated SCORAD worsening was less in the group treated with product A versus the group treated with product B.
Conclusion: This study demonstrated that a specific emollient containing a biomass of non-pathogenic bacteria Vf grown in a medium containing TSW and associated with a selected carbon source is able to normalize skin microbiota and significantly reduce the number and severity of flare-ups compared with another emollient.
Keywords: skin microbiota, atopic dermatitis, emollient, Vitreoscilla filiformis, thermal spring water, Xanthomonas genus
Introduction
Changes in the composition of microbial communities that colonize skin have been linked to several dermatological diseases, in particular atopic dermatitis (AD).1,2 Staphylococci colonization or infection has been associated with this pathology. It has been described that the proportion of Staphylococcus sequences, particularly Staphylococcus aureus, is greater during disease flares than at baseline or post-treatment.3 However, the change in the bacterial communities associated with AD and their responses to therapy remain poorly understood.
We previously demonstrated that bacterial communities vary between closest affected (AF) or lesional and unaffected (UAF) or non-lesional skin from the same individual, thus providing a deeper insight into the bacterial communities involved in the skin dysbiosis associated with AD.4 We confirmed that staphylococci and a lower microbial diversity dominate lesional skin compared with adjacent UAF skin.3,4 We also showed that applying an emollient can return the compositional balance of an AF area to a state similar to that of a nearby UAF area.4 Last, but not the least, the proportion of a bacterial family such as Xanthomonadaceae with a keratolytic activity, which is naturally present on the skin, increased due to better access to keratin, which is favorable to their growth.4,5
Numerous clinical trials have attempted to demonstrate that supplementation with preventive probiotics (living non-pathogenic bacteria) has an action on the initial development and/or flares and severity of AD, but the results have been controversial.6–11 However, applying prebiotics (ie, nonliving extracts from nonpathogenic bacteria) onto the skin, as it has been reported for some acute intestinal diseases and based on previous publications,12–14 has been suggested as an interesting therapeutic approach to modulate or balance the immune system and manipulate the different populations of the cutaneous microbiota.15 This hypothesis was based on results from in vitro, animal and clinical studies. An in vitro model on a reconstructed epidermis revealed that, surprisingly, lysate from the Gram-negative bacterium, Vitreoscilla filiformis (Vf), stimulated the β-defensin production and other innate immune defense mechanisms through activation of toll-like receptor 2 (TLR2).14 A mouse model of AD revealed that the Vf lysate is able to reduce clinical inflammatory symptoms and the inflammatory reaction to an allergen when it was applied, suggesting a significant modulation of the immune response.13 In addition, a randomized trial demonstrated that the severity of AD significantly improved by using a formula containing Vf lysate.12
Therefore, we speculated that an emollient supplemented with a biomass of Vf grown in a medium containing La Roche-Posay (LRP) selenium-rich thermal spring water (TSW; LRP-Vitreoscilla filiformis biomass [LRP-VFB]) could have a clinical value in the management of AD. Furthermore, we associated a specific carbon source, mannose, with LRP-VFB to promote the growth of members of Xanthomonadaceae family. This sugar used in laboratory culture medium has been shown to boost the growth of this bacterial family unlike other families such as Staphylococcaceae, Streptococcaceae, Corynebacteriaceae, and Propionibacteriaceae.16
Patients and methods
Ethics statement
This study protocol complied with the ethical guidelines of the 1975 Declaration of Helsinki, was approved by the DOST ethical committee and was conducted according to ICH Guidelines for Good Clinical Practice. Written informed consent and photography consent were obtained from each subject before enrollment.
Emollients
The emollient A (product A) was a lipophilic cream containing 20% shea butter, 4% niacinamide, LRP thermal spring water (LRP-TSW), mannose, and a biomass of Vf grown in a medium containing TSW (LRP-VFB), (Lipikar Balm AP+; La Roche-Posay Dermatological Laboratories, Asnières, France). The emollient B (product B) was a commercial product prescribed for AD and containing triglyceride, glycerin, shea butter, and ceramide. Patients were randomized to apply either emollient A or emollient B. They were then instructed to apply their allocated product to their entire body twice daily, once in the morning and once in the evening. They were also instructed not to change their hygiene practices or to apply any other emollient (or any drugs, including corticotherapy or antibiotherapy) during the study.
Patient recruitment and sampling
This double-blind, randomized, comparative study included 60 patients (27 males and 33 females; aged from 6 months to 63 years) diagnosed with moderate AD. Clinical assessment of disease severity and skin microbiota sampling were conducted on three separate visits, a screening visit on January 6 or 7, 2014 (Day -15) and two visits on January 21 or 22, 2014 (Day 1) and February 18 or 19, 2014 (Day 28). At the screening visit, patients received a medical treatment (either corticoids, immunosuppressive, or phototherapy), according to the discretion of the dermatologist, to improve their SCORing Atopic Dermatitis (SCORAD) by at least 25% between Day -15 and Day 1 (Figure 1). At Day -15, at the first visit (Day 1) and at study end (Day 28), the same investigating dermatologist evaluated the severity of symptoms using the SCORAD index and clinical signs of erythema, dryness, and desquamation of one or more typical lesional (AF) skin areas and a proximal non-lesional (UAF) site (scored as absent =0, light =1, moderate =2, or severe =3). Each area was identified and photographed to ensure that the same area was sampled at each visit. Only individuals with SCORAD values between 0 and 40 at Day 1 and with at least a reduced SCORAD score of 25% between Day –15 and Day 1 were included in the study (n=53).
Skin microbiota samples were collected by the same sampler using aseptic techniques as previously described.4 Swabs were rubbed firmly for 20 seconds over 1 cm2 skin areas identified as being the most representative of AF. Similarly, samples were also collected from the closest UAF area. The cotton tip samples were stored at -80°C until being shipped on dry ice to the University of Colorado for processing.
DNA extraction, polymerase chain reaction (PCR) amplification, and sequencing
Genomic DNA was extracted from each swab, and the V1–V2 region of 16S rRNA genes was amplified using PCR with the primer set (27F/338R).4 All sequences were processed as previously described.4
Statistical analysis of clinical data
Statistical analysis of clinical data was performed using StatView 5.0 (StataCorp LP, College Station, TX, USA). Continuous variables were described by mean and standard deviation (SD) or standard error of the mean (SEM). Qualitative variables were described by absolute numbers and corresponding percentages. Features of AD were compared at Day 1 and Day 28 using the McNemar’s chi-square test. For each feature, the number of cases with improvement was described. For quantitative values of the SCORAD, comparisons between Day 1 and Day 28 were performed by the Wilcoxon rank-sum test for paired observations. To determine the differences between product A and product B at Day 1 and Day 28, SCORAD and delta SCORAD values were compared using the Wilcoxon rank-sum test for paired samples.
Statistical analysis of sequence data
Statistical analysis was performed using R-2.15. Statistical tests were done on the raw count of rarefied sample (10000 reads per sample). Stratification of samples was performed using the following parameters: UAF and AF zones under A or B treatment (four groups), patient status at Day 28 compared to Day 1 (remission if SCORAD decreased or was equal between Day 1 and Day 28 (two groups) and relapse if SCORAD increased) and treatment (A or B).
Results
This study enrolled 60 patients (27 males and 33 females; 13±14 years [0.5–63 years]) diagnosed with moderate form of AD (SCORAD =21±8). At Day 1, 53 patients presented with a SCORAD of 11±6 and were included in the study (Table S1). Seven patients presented with an improvement <25% and were excluded from the study.
After eliminating those individuals who lacked paired samples (SCORAD and microbiota) from both visits, 30 paired samples remained and were analyzed for their relation between SCORAD evolution and microbial community. Thirty-four paired samples were analyzed for microbiota communities at Day 1 and 48 paired samples at Day 28 (n=22 for A treatment and n=24 for B treatment). Interestingly, the weight of the product used during the study was on average 192±76 g for product A and 224±102 g for product B (p=0.0871).
Microbiota of UAF and AF skin at Day 1
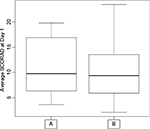
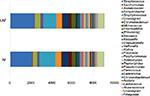
Before any emollient treatment (at Day 1), the average SCORAD of each treatment group was similar (11±5 and 10±6, respectively; p-value =0.8658; Figure 1). The microbial communities of AF areas and closest UAF areas for each participant were also similar (Figure 2).
Microbiota of UAF and AF skin at Day 28
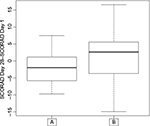
One month after the end of the therapeutic treatment (Day 28), the average SCORAD of the patients treated with product A was lower than that of the patients treated with product B (SCORAD =9.3±6.1 and 13.2±9.6, respectively; p-value =0.182). SCORAD variation between Day 1 and Day 28 was significantly different between treatments (p-value =0.01884; Figure 3) corresponding to -11% for product A versus +35% for product B. Interestingly, in the product A-treated group, nine of the 26 patients (30%) showed an aggravation (relapsing patients) versus 18 of the 27 (60%) in the product B-treated group, and 17 of the 26 remained in remission or improved in the product A-treated group versus only nine of the 27 in the product B-treated group. Of particular note, the associated SCORAD in relapsing patients worsened less in the group treated with product A (46%) versus the group treated with product B (79%).
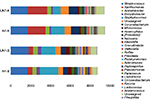
At Day 28, the microbial landscape of AF and UAF areas was similar in both groups but very different between each treatment (Figure 4). Particularly, a significantly increased level of Xanthomonas genus (p-value =2.57e-02) was noticed in the group treated with product A (versus product B; Figure 5). On the other hand, the level of Staphylococcus genus increased between Day 1 and Day 28 in the group treated with product B (p-value =0.1534 on UAF and p-value =0.2642 on AF), but was not observed in the group treated with product A (p-value =0.7697 on UAF and p-value =0.9729 on AF). Interestingly, these differences in the microbial landscape were more pronounced for patients in relapse (patients with an increased SCORAD at Day 28 versus Day 1).
Discussion
Using a high-throughput sequencing approach that targets two portions of the 16S rRNA gene (V1–V2), as recommended by Meisel et al,17 to study cutaneous bacterial communities and dysbiosis associated with the severity of AD and comparing an AF skin area to the closest UAF area per patient is a sensitive method to evaluate product efficacy. With this approach, we have previously shown that an emollient can equalize the bacterial composition of an AF area to a state similar to that of an adjacent UAF area.4 Very recently, the same strategy was used and exactly the same results were obtained after a 4-week treatment period with a topical corticosteroid.18
Prebiotics were initially defined as nondigestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria.19 Prebiotics that might be included in topically applied skin products also have the potential to support normal skin microbiota maintenance or normalize dysbiosis if it occurs.20 Relatively little is known about the benefits of this approach, but it has been shown that applying a biomass lysate of the nonpathogenic Gram-negative proteobacterium, Vf, helped to improve symptoms in patients with atopic or seborrheic dermatitis.12,13,21 It is interesting to note that VFB prepared from organisms grown in a medium enriched with TSW (LRP-VFB) resulted in more potent stimulation of mRNA expression for antimicrobial peptides and other innate immune defense mechanisms through activation of TLR2 in reconstructed epidermis.14 This original biological activity should be mainly explained by the specific structure of the lipid A moiety of the lipopolysaccharide part of Vf (Vf-LPS).22
In the present study, treating the symptoms of AD with an emollient containing LRP-VFB and mannose as a carbon source versus another recommended emollient yielded greater clinical improvement. This was associated with a significantly increased population of Xanthomonas genus, a Gram-negative proteobacteria (Figures 4 and 5). In contrast, the comparator product was associated with an increase in Staphylococcus genus bacteria. For the first time, this study demonstrated that the topical application of a prebiotic (ie, nonliving extracts from nonpathogenic bacteria) is an interesting therapeutic approach to modulate or balance the immune system and so normalize the cutaneous microbiota for at least 1 month after the end of a therapeutic treatment.
It is known that staphylococci are present on both healthy and AF skin of >90% of patients with AD. This presence is at least partially explained by an antimicrobial peptide deficiency and the poor quality of the skin barrier, which allows easy access to complex substrates.23,24 Furthermore, during flares, the loss of bacterial diversity and the predominance of Staphylococcus species, Staphylococcus epidermidis and S. aureus, worsen in correlation with severity,2–4 as shown in this study in relapsing patients. In this study as in the two previously published ones, we highlighted that an increase in bacteria from the Xanthomonadaceae family was induced by LRP-TSW either alone25 or in high concentration in an emollient4 or with LRP-VFB and mannose. This was associated with a pathology improvement and a decrease in the proportion of staphylococci. Nevertheless, to distinguish the singular effect of each component of the product, it would be necessary to perform other in vivo and/or in vitro studies.
Conclusion
This study highlights the long-lasting efficacy for managing AD (reducing flare-ups and decreasing the flare severity) of a moisturizer containing emollient, humectant, moisturizing, or occlusive agents that restore skin barrier function; having acceptable cosmetic quality and excellent dermatological tolerance and supplemented with ingredients that provide nutritional supplements to revive natural cutaneous defenses and normalize the cutaneous microbiota.
These data suggest that a prebiotic made of a nonliving extract of a specific Gram-negative proteobacterium, with an original nontoxic LPS structure (ie, Vf) grown in a controlled medium and associated with a selected carbon source (mannose), onto the skin is a new therapeutic approach to modulate or balance the immune system and would enable normalization of the cutaneous microbiota that could be beneficial in various immune-mediated skin diseases. This could be useful in the treatment of AD and potentially other chronic inflammatory skin diseases such as acne, rosacea, and psoriasis.
Acknowledgments
DNA extraction, PCR amplification, and sequencing were performed by Jessica B. Henley, Cooperative Institute for Research in Environmental Sciences, University of Colorado, Boulder, CO, USA. Technical support for the clinical part of the study was provided by Guénaëlle Le Dantec and Martine Fortuné. Statistical analyses were performed by Samuel Mondy.
Disclosure
This study was funded by La Roche-Posay Dermatological Laboratories and L’Oréal Research and Innovation, France. S. Seité and R. Martin are employees of L’Oréal. No conflicts of interest exists for Professor H. Zelenkova.
References
Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol. 2011;9(4):244–253. | ||
Grice EA. The skin microbiome: potential for novel diagnostic and therapeutic approaches to cutaneous disease. Semin Cutan Med Surg. 2014;33(2):98–103. | ||
Kong HH, Oh J, Deming C, et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22(5):850–859. | ||
Seite S, Flores GE, Henley JB, et al. Microbiome of affected and unaffected skin of patients with atopic dermatitis before and after emollient treatment. J Drugs Dermatol. 2014;13(11):1365–1372. | ||
Yamamura S, Morita Y, Hasan Q, Yokoyama K, Tamiya E. Keratin degradation: a cooperative action of two enzymes from Stenotrophomonas sp. Biochem Biophys Res Commun. 2002;294(5):1138–1143. | ||
Panduru M, Panduru NM, Salavastru CM, Tiplica GS. Probiotics and primary prevention of atopic dermatitis: a meta-analysis of randomized controlled studies. J Eur Acad Dermatol Venereol. 2015;29(2):232–242. | ||
Muraro A, Halken S, Arshad SH, et al. EAACI food allergy and anaphylaxis guidelines. Primary prevention of food allergy. Allergy. 2014;69(5):590–601. | ||
Kim SO, Ah YM, Yu YM, Choi KH, Shin WG, Lee JY. Effects of probiotics for the treatment of atopic dermatitis: a meta-analysis of randomized controlled trials. Ann Allergy Asthma Immunol. 2014;113(2):217–226. | ||
Wang IJ, Wang JY. Children with atopic dermatitis show clinical improvement after Lactobacillus exposure. Clin Exp Allergy. 2015;45(4):779–787. | ||
Drago L, De Vecchi E, Toscano M, Vassena C, Altomare G, Pigatto P. Treatment of atopic dermatitis eczema with a high concentration of Lactobacillus salivarius LS01 associated with an innovative gelling complex: a pilot study on adults. J Clin Gastroenterol. 2014;48(suppl 1):S47–S51. | ||
Niccoli AA, Artesi AL, Candio F, et al. Preliminary results on clinical effects of probiotic Lactobacillus salivarius LS01 in children affected by atopic dermatitis. J Clin Gastroenterol. 2014;48(suppl 1):S34–S36. | ||
Gueniche A, Knaudt B, Schuck E, et al. Effects of nonpathogenic gram-negative bacterium Vitreoscilla filiformis lysate on atopic dermatitis: a prospective, randomized, double-blind, placebo-controlled clinical study. Br J Dermatol. 2008;159(6):1357–1363. | ||
Volz T, Skabytska Y, Guenova E, et al. Nonpathogenic bacteria alleviating atopic dermatitis inflammation induce IL-10-producing dendritic cells and regulatory Tr1 cells. J Invest Dermatol. 2014;134(1):96–104. | ||
Mahe YF, Perez MJ, Tacheau C, et al. A new Vitreoscilla filiformis extract grown on spa water-enriched medium activates endogenous cutaneous antioxidant and antimicrobial defenses through a potential toll-like receptor 2/protein kinase C, zeta transduction pathway. Clin Cosmet Investig Dermatol. 2013;6:191–196. | ||
Seite S, Bieber T. Barrier function and microbiotic dysbiosis in atopic dermatitis. Clin Cosmet Investig Dermatol. 2015;8:479–483. | ||
Garrity G. Phylum Al. Crenarchaeota. In: Whitman W, editor. Bergey’s Manual of Systematic Bacteriology. Vol. 1. New York, Berlin, Heidelberg: Springer; 2012. | ||
Meisel JS, Hannigan GD, Tyldsley AS, et al. Skin microbiome surveys are strongly influenced by experimental design. J Invest Dermatol. 2016;136(5):947–956. | ||
Gonzalez ME, Schaffer JV, Orlow SJ, et al. Cutaneous microbiome effects of fluticasone propionate cream and adjunctive bleach baths in childhood atopic dermatitis. J Am Acad Dermatol. 2016;75(3):481.e–493.e. | ||
Gibson GR, Roberfroid MB. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr. 1995;125(6):1401–1412. | ||
Al-Ghazzewi FH, Tester RF. Impact of prebiotics and probiotics on skin health. Benef Microbes. 2014;5(2):99–107. | ||
Gueniche A, Cathelineau AC, Bastien P, et al. Vitreoscilla filiformis biomass improves seborrheic dermatitis. J Eur Acad Dermatol Venereol. 2008;22(8):1014–1015. | ||
Mahe Y, Martin R, Inventors. Lipopolysaccharide fractions of vitreoscilla filiformis useful for stimulating the synthesis of anti-microbial peptides of the skin. US Patent App. 12/055,975, 2008. | ||
Ong PY, Ohtake T, Brandt C, et al. Endogenous antimicrobial peptides and skin infections in atopic dermatitis. N Engl J Med. 2002;347(15):1151–1160. | ||
Nakatsuji T, Gallo RL. Antimicrobial peptides: old molecules with new ideas. J Invest Dermatol. 2012;132(3 pt 2):887–895. | ||
Martin R, Henley JB, Sarrazin P, Seite S. Skin microbiome in patients with psoriasis before and after balneotherapy at the Thermal Care Center of La Roche-Posay. J Drugs Dermatol. 2015;14(12):1400–1405. |
Supplementary material
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.