Back to Journals » OncoTargets and Therapy » Volume 11
Clinical, dosimetric, and position factors for radiation-induced acute esophagitis in intensity-modulated (chemo)radiotherapy for locally advanced non-small-cell lung cancer
Authors Huang J , He TY , Yang RH, Ji TL, Li G
Received 18 May 2018
Accepted for publication 14 August 2018
Published 21 September 2018 Volume 2018:11 Pages 6167—6175
DOI https://doi.org/10.2147/OTT.S174561
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
Jin Huang,1 Tianyu He,1 Ronghui Yang,2 Tianlong Ji,1 Guang Li1
1Department of Radiotherapy, The First Hospital of China Medical University, Shenyang 110001, China; 2Department of Hematology, Shengjing Hospital of China Medical University, Shenyang 110000, China
Purpose: The purpose of this study was to estimate the relation between acute esophagitis (AE) and clinical, dosimetric, and position factors in patients with locally advanced non-small-cell lung cancer (NSCLC) receiving intensity-modulated (chemo)radiotherapy.
Materials and methods: A retrospective cohort analysis was performed to identify factors associated with Common Toxicity Criteria for Adverse Events grade 2 or worse AE (AE2+). A multivariable model was established including patient- and treatment-related variables and esophageal dose–volume histogram parameters. The esophagus was divided according to physiological anatomy, and logistic regression was used to analyze the position parameter for its correlation with AE2+.
Results: The incidence of AE2+ was 27.5%. All models included gender, concurrent chemoradiotherapy (CCRT), position parameter, and one of the dosimetric variables. The model with mean dose showed the best goodness of fit. Gender (OR=2.47, P=0.014), CCRT (OR=3.67, P=0.015), mean dose (OR=1.33, P<0.001), and maximum radiation position (OR=1.65, P=0.016) were significantly related to AE2+.
Conclusion: Gender, concurrent chemotherapy, maximum radiation position, and mean dose were independent risk factors for AE2+. The upper part of the esophagus showed a higher sensitivity to radiation toxicity.
Keywords: acute esophagitis, non-small-cell lung cancer, intensity-modulated radiation therapy, position parameter
Introduction
Concurrent chemoradiotherapy (CCRT) is the preferred treatment for patients with advanced-stage non-small-cell lung cancer (NSCLC). Although CCRT brings survival benefit compared with sequential chemoradiation or radiotherapy (RT) alone, the risk of post-therapy toxicity as acute esophagitis (AE) may increase.1 AE often occurs 2 weeks after the start of RT, including retrosternal pain, dysphagia, and odynophagia. Severe AE may require analgesics and parenteral nutrition and can even lead to treatment interruptions, which may reduce the quality of life of the patients and lower the chance of local tumor control.2,3
Previous studies have attempted to define clinical and dosimetric predictors of radiation-induced esophagitis. Factors found to correlate with AE include CCRT,4,5 lymphatic status,6,7 hyperfractionated RT (1.6 Gy/fraction twice daily),8 esophageal length,9 molecular markers,10 and a number of dosimetric parameters.11–16 However, the clinical applicability of these studies’ findings remains restricted because of their various study populations, different RT techniques, and different end points. In addition, there are differences between the classification systems of the Radiation Therapy Oncology Group (RTOG) acute radiation morbidity scoring criteria17 and the Common Toxicity Criteria for Adverse Events (CTCAE) version. Most studies focus on three-dimensional conformal radiotherapy (3D-CRT), and the impact of intensity-modulated radiotherapy (IMRT) on AE is less reported. Research has shown that 3D-CRT toxicity prediction models are not suitable for predicting toxicity after IMRT.18,19
New research has shown that different parts of the esophagus have different sensitivities to AE.20 However, the dosimetric predictors for AE in current studies are for the whole esophagus. In fact, most radiation areas for NSCLC cover only part of the esophagus. IMRT has advantages in reducing the exposure of organs at risk (OARs).21–23 However, it is difficult to constraint the dose of the whole esophagus and OARs together in some cases because of the anatomical positional relationship between OARs and tumors. Therefore, position factors should be regarded with care in the prediction of AE.
The objective of this study was to estimate the relation between AE and clinical, dosimetric, and position factors in patients with local regional advanced NSCLC receiving intensity-modulated (chemo)radiotherapy.
Materials and methods
Patients
In this retrospective study, we assessed a cohort of 193 consecutive patients who had undergone intensity-modulated (chemo)radiotherapy for stages IIa–IIIc NSCLC from March 2015 to December 2017 at a single center. This investigation was approved by the local institutional review board. The inclusion criteria histologically or cytologically confirmed NSCLC, with full dose–volume histogram (DVH) and clinical data available for evaluation. The exclusion criteria were postoperative radiation therapy for lung cancer, previous irradiation of chest, history of esophageal surgery, and esophageal cancer.
Radiation simulation, planning, and delivery
Patients were immobilized in the supine position and underwent a contrast-enhanced computed tomography (CT) scan with 5 mm slices from cricoid cartilage to mid-abdomen. The image data sets were transferred to the RayStation planning system (Raysearch Radiation Oncology Systems). Gross tumor volume (GTV) included the primary tumor and suspicious lymph nodes (confirmed by histopathology after endobronchial/endoesophageal ultrasonography, with malignant features on CT scan, and/or fluorodeoxyglucose–positron emission tomography positive). Clinical target volumes (CTVs) enclosed the GTV of the primary tumor and lymph nodes with 8-mm and 5-mm margins, respectively. The internal target volume (ITV) was delineated from four-dimensional CT images, which involved assessing the CTV on expiratory-phase images and then registering the outline to the images to create a union of target contours enclosing all possible positions of the target. The planning target volume (PTV) was created by an isotropic 5 mm expansion of the CTV. Patients were treated on a Synergy linac (Elekta Ltd, Crawley, UK). The prescribed dose to the PTV was 60 Gy in 30 (once-daily) fractions using IMRT with seven to nine coplanar fields or volumetric modulated arc therapy (VMAT). Weekly online image-guided RT for setup verification was required. The dose of the OARs was set to meet the following constraints: lung dose <15 Gy, V20 <30%; spinal cord, maximum dose <45 Gy; heart, V30 <40%; and esophagus, maximum dose <66 Gy.
Esophageal contours, grouping, and dosimetric data
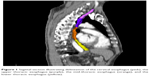
For each patient, the esophagus was delineated using the external esophageal contour from the cricoid cartilage to the gastroesophageal junction on the window of the planning CT scan (window width: 250–350 Hu; window level: 30–50 Hu). The esophagus was further divided into four subsites according to the seventh edition of the American Joint Committee on Cancer/Union for International Cancer Control (UICC) TNM staging system for esophageal carcinoma as follows: 1) the cervical esophagus was contoured from the cricoid cartilage to the sternal notch plane; 2) the upper thoracic esophagus was contoured from the sternal notch plane to the lower edge of the azygos vein; 3) the middle thoracic esophagus was contoured from the lower edge of the azygos vein to the lower pulmonary veins; and 4) the lower thoracic esophagus was contoured from the lower pulmonary veins to the gastroesophageal junction (Figure 1). Patients were classified into four subgroups according to the subsite of the maximum mean dose, defined as the cervical esophagus group, upper thoracic esophagus group, middle thoracic esophagus group, and lower thoracic esophagus group, respectively.
For consistency, the esophagus was redelineated in all patients by the same radiation therapist. DVHs were then generated. The esophageal dosimetric parameters of the entire esophagus and subsites were extracted from the dose data set in the treatment-planning system including volume, mean dose, and the percentage of the volume of esophagus receiving 10 Gy (V10) to 60 Gy (V60) (Figure S1).
Toxicity scoring
The end point was defined as AE grade ≥2 (AE2+) during or at 3 months after RT treatment. AE was assessed weekly according to CTCAE V5.0. Toxicities were recorded as maximum grade at any point during treatment or in the follow-up period.
Statistical analyses
Patient characteristics as well as clinical and dosimetric variables were summarized by descriptive statistics. One-way ANOVA with least significant difference was performed for analysis among the four subgroups. Univariate analyses were used to identify the significant risk factors among age (dichotomized by 60), Karnofsky Performance Status score (dichotomized by 90), gender, smoking history, pathology, tumor stage (dichotomized by T3), lymph node stage (dichotomized by N3), chemotherapy, maximum irradiated position, and dosimetric parameters, including mean dose and volumetric metrics. Because of the large intervariable correlation within the dosimetric variables, only one of the dosimetric variables was included in the pairs for each multivariable analysis. The Akaike information criterion (AIC) and Bayesian information criterion (BIC) were used for multivariable model selection, which balanced the goodness of fit of the model. The preferred model was chosen as the one resulting in the lowest AIC and BIC values. P<0.05 was considered as statistically significant. Statistical programming was performed using SPSS 24.0 software.
Results
Patient characteristics and clinical and dosimetric variables are summarized in Table 1. Of 193 patients treated with (chemo)radiotherapy, 90% received a platinum-containing regimen. AE was scored as grade 1 in 92 (47.7%) patients, as grade 2 in 44 (22.8%) patients, and as grade 3 in nine (4.7%) patients. No grade 4 or 5 AE was observed. The incidence of AE2+ in the cervical esophagus group, upper thoracic esophagus group, middle thoracic esophagus group, and lower thoracic esophagus group was 47.4% (9/19), 34.0% (18/53), 24.2% (22/91), and 13.3% (4/30), respectively. Baseline dosimetric characteristics were comparable between the four subgroups (Table 2).
Univariate logistic regression analysis revealed a significant increase in females, those with N3 stage, and those receiving concurrent chemotherapy. There was no statistical difference between the IMRT and VMAT (P>0.05). All dosimetric parameters, including mean dose, all relative volumetric variables, and maximum radiation positions, were significant (P<0.05; Table 3). N stage was not analyzed in the multivariate logistic regression model because it was highly associated with dosimetric parameters, which in turn correlated with esophagitis. Regarding the Spearman self-correlation matrix for the dosimetric variables, all variables were significantly correlated. An ANOVA analysis did not show any associations between gender, CCRT, position parameter, and dosimetric variables.
Each multivariate logistic regression model contained four independent variables: gender, CCRT, position parameter, and one of the dosimetric variables including mean dose and V10–V60. Gender, CCRT, maximum radiation position, and all the dosimetric variables demonstrated a significant correlation with AE2+ in all test models (Table S1). Goodness of fit was estimated by AIC and BIC values. The model with mean dose showed lower AIC and BIC values than models with other dosimetric variables (Table 4). We decided to include mean dose in the modeling procedure. The regression ORs for the optimal model indicated that patients had an increased risk of AE2+ with female (OR=2.47, P=0.014), with increasing mean dose (OR=1.33, P<0.001), when they received CCRT (OR=3.67, P=0.015), or with the maximum radiation position in the upper part of the esophagus (OR=1.65, P=0.016; Table 5).
Discussion
We constructed a multivariate model for AE2+ in patients with advanced-stage NSCLC treated with intensity-modulated (chemo)radiotherapy. Gender, CCRT, maximum radiation position, and mean dose were significantly associated with AE2+. Patients may be more sensitive to AE in the upper part of the esophagus in comparison with that in the lower part of the esophagus, which may contribute to a high risk of AE in patients with cervical lymph node metastases.
Research has shown that higher grade AE is the consequence of the accumulative effect of both chemotherapy and RT.4,5,24 Meta-analysis of Gong et al25 showed that patients receiving CCRT have an approximately fivefold increase in the risk of AE compared with those receiving sequential chemoradiotherapy (SCRT). The reason may be that cisplatin hampers the repair of DNA damage caused by the irradiation in esophageal epithelial cells. In some IMRT-based studies, CCRT was reported to result in a 27%–38% rate of grade 2 AE and an 8%–22% rate of grade 3 AE.19,24,26,27 In our study, the incidences of grade 2 and 3 AEs were 23.7% and 5.1%, respectively. The possible reason for the lower incidence is that the median RT dose was mostly 66 Gy in the other studies, whereas it was 60 Gy in ours.
Increased nodal stage has been identified as a predictor of AE. A relevant study pointed out that it is most likely a surrogate for larger tumor volumes and radiation fields.28 Most studies have shown that N3 is closely related to the incidence of AE.6,7 However, Uterlinde et al suggested that nodal stage is not correlated with tumor volume but that involved mediastinal nodes have more clinical significance.24 Therefore, N2–3 was selected for our analysis because of its increased correlation with mediastinal lymph nodes compared with N3. Gender was reported only occasionally in related studies.29,30 We found that female was statistically associated with esophagitis.
Various dosimetric parameters have shown a strong correlation with AE.11–16,31,32 However, these results were mostly based on 3D-CRT. The hypothesis that the toxicity prediction models of 3D-CRT are not suitable for predicting toxicity after IMRT was supported. In our study, the mean dose showed a better goodness of fit in the multivariate model. The relevant study by Wijsman et al26 also demonstrated that the mean dose was a good predictor of AE.
To the best of our knowledge, this is the second study suggesting the position parameter as related to AE. Pan et al20 proposed that the upper part of the esophagus is more sensitive to injuries because of the different distributions of the sensory axons.33 We divided the esophagus into four subsites according to physiological and anatomical structures. We found that the risk of AE in the upper esophagus was higher compared with that in the lower esophagus. Another possible reason is that the physiological structures of different parts of the esophagus are different. The epidermis of the upper esophageal mucous layer consists of squamous epithelial cells, and the muscularis consists of striated muscle. The epidermis of the lower esophageal mucous layer consists of columnar epithelial cells, and the muscularis consists of smooth muscle. The middle esophagus is a mixture of two different tissues above.34 This conclusion also suggests the importance of personalized sparing of esophageal dose. The esophagus is adjacent to the OARs, including lungs, spinal cord, and heart in the RT plan of NSCLC patients. In some cases, such as where the radiation field is large or the shape of the tumor is irregular, it is often difficult to simultaneously limit the dose of the whole esophagus and all other OARs. The esophagus may even be exposed to over radiation to meet the PTV-planning constraints. However, we found that the irradiation field often does not cover the entire esophagus in patients with advanced-stage NSCLC. A total of 80.3% of the cases had at least one part of the esophagus exposed to little or no radiation in this study (V10=0 Gy). If we can limit only the part of the esophagus covered in the radiation field, the concept of personalized treatment can be achieved. This finding also suggests that there may be differences in the optimal sparing dose for different parts of the esophagus. Moreover, patients with cervical lymph node metastases should be aware of the high risk of AE.
There are still many deficiencies in this study. First, single-center research may lead to regional bias. The model still requires internal and external validation. Second, the evaluation of AE was mainly reported by patients, which may result in deviation of the incidence of esophagitis. Furthermore, the CTCAE score is essentially variable. There is currently a lack of more objective and rigorous criteria to evaluate normal tissue complications. Third, using DVH parameters may result in loss of spatial dosimetric information and may also ignore the movements of esophagus. Currently available studies have attempted to evaluate the effectiveness of other 3D dosimetric parameters (such as the surface dose of esophagus).35
Conclusion
The most relevant prognostic variables in the multivariable model in patients with advanced-stage NSCLC undergoing intensity-modulated (chemo)radiotherapy are gender, concurrent chemotherapy, maximum radiation position, and mean dose. The sensitivity of AE varies in different parts of the esophagus. Validation in a large independent patient cohort is still warranted.
Abbreviations
CCRT, concurrent chemoradiotherapy; AE, acute esophagitis; NSCLC, non-small-cell lung cancer; RTOG, Radiation Therapy Oncology Group; CTCAE, Common Toxicity Criteria for Adverse Events; 3D-CRT, three-dimensional conformal radiotherapy; IMRT, intensity-modulated radiotherapy; OARs, organs at risk; UICC, Union for International Cancer Control; GTV, gross tumor volume; CTV, clinical target volume; ITV, internal target volume; PTV, planning target volume; DVHs, dose–volume histograms; SCRT, sequential chemoradiotherapy.
Ethics approval and consent to participate
We declare that this study was approved by the institutional review board at Medical Science Research Ethics Committee of The First Hospital of China Medical University (reference number: [2018]-148). Patients were required to provide written informed consent to participate in this research.
Availability of data and materials
All data generated or analyzed during this study are available from the corresponding author on reasonable request.
Disclosure
The authors report no conflicts of interest in this work.
References
Curran WJ, Paulus R, Langer CJ, et al. Sequential vs. concurrent chemoradiation for stage III non-small-cell lung cancer: randomized phase III trial RTOG 9410. J Natl Cancer Inst. 2011;103(19):1452–1460. | ||
Palma DA, Senan S, Oberije C, et al. Predicting esophagitis after chemoradiation therapy for non-small cell lung cancer: an individual patient data meta-analysis. Int J Radiat Oncol Biol Phys. 2013;87(4):690–696. | ||
Govaert SL, Troost EG, Schuurbiers OC, et al. Treatment outcome and toxicity of intensity-modulated (chemo) radiotherapy in stage III non-small cell lung cancer patients. Radiat Oncol. 2012;7:150. | ||
Caglar HB, Othus M, Allen AM. Esophagus in-field: a new predictor for esophagitis. Radiother Oncol. 2010;97(1):48–53. | ||
Bradley J, Deasy JO, Bentzen S, El-Naqa I. Dosimetric correlates for acute esophagitis in patients treated with radiotherapy for lung carcinoma. Int J Radiat Oncol Biol Phys. 2004;58(4):1106–1113. | ||
Zhang ZC, Xu J, Li BS, et al. Clinical and dosimetric risk factors of acute esophagitis in patients treated with 3-dimensional conformal radiotherapy for non-small-cell lung cancer. Am J Clin Oncol. 2010;33(3):271–275. | ||
Ahn SJ, Kahn D, Zhou S, et al. Dosimetric and clinical predictors for radiation-induced esophageal injury. Int J Radiat Oncol Biol Phys. 2005;61(2):335–347. | ||
Maguire PD, Sibley GS, Zhou SM, et al. Clinical and dosimetric predictors of radiation-induced esophageal toxicity. Int J Radiat Oncol Biol Phys. 1999;45(1):97–103. | ||
Hirota S, Tsujino K, Endo M, et al. Dosimetric predictors of radiation esophagitis in patients treated for non-small-cell lung cancer with carboplatin/paclitaxel/radiotherapy. Int J Radiat Oncol Biol Phys. 2001;51(2):291–295. | ||
De Ruyck K, Sabbe N, Oberije C, et al. Development of a multicomponent prediction model for acute esophagitis in lung cancerpatients receiving chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2011;81(2):537–544. | ||
Chapet O, Kong FM, Lee JS, Hayman JA, Ten Haken RK. Normal tissue complication probability modeling for acute esophagitis in patients treated with conformal radiation therapy for non-small cell lung cancer. Radiother Oncol. 2005;77(2):176–181. | ||
Zhu J, Zhang ZC, Li BS, et al. Analysis of acute radiation-induced esophagitis in non-small-cell lung cancer patients using the Lyman NTCP model. Radiother Oncol. 2010;97(3):449–454. | ||
Zhang Z, Xu J, Zhou T, et al. Risk factors of radiation-induced acute esophagitis in non-small cell lung cancer patients treated with concomitant chemoradiotherapy. Radiat Oncol. 2014;9:54. | ||
Kim TH, Cho KH, Pyo HR, et al. Dose-volumetric parameters of acute esophageal toxicity in patients with lung cancer treated with three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys. 2005;62(4):995–1002. | ||
Etiz D, Bayman E, Akcay M, Sahin B, Bal C. Dosimetric and clinical predictors of acute esophagitis in lung cancer patients in Turkey treated with radiotherapy. Asian Pac J Cancer Prev. 2013;14(7):4223–4228. | ||
Belderbos J, Heemsbergen W, Hoogeman M, Pengel K, Rossi M, Lebesque J. Acute esophageal toxicity in non-small cell lung cancer patients after high dose conformal radiotherapy. Radiother Oncol. 2005;75(2):157–164. | ||
Cox JD, Stetz J, Pajak TF. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995;31(5):1341–1346. | ||
Kwint M, Uyterlinde W, Nijkamp J, et al. Acute esophagus toxicity in lung cancer patients after intensity modulated radiation therapy and concurrent chemotherapy. Int J Radiat Oncol Biol Phys. 2012;84(2):e223–e228. | ||
Beetz I, Schilstra C, van Luijk P, et al. External validation of three dimensional conformal radiotherapy based NTCP models for patient-rated xerostomia and sticky saliva among patients treated with intensity modulated radiotherapy. Radiother Oncol. 2012;105(1):94–100. | ||
Pan Y, Brink C, Knap M, et al. Acute esophagitis for patients with local-regional advanced non small cell lung cancer treated with concurrent chemoradiotherapy. Radiother Oncol. 2016;118(3):465–470. | ||
Nielsen TB, Hansen O, Schytte T, Brink C. Inhomogeneous dose escalation increases expected local control for NSCLC patients with lymph node involvement without increased mean lung dose. Acta Oncol. 2014;53(1):119–125. | ||
Veldeman L, Madani I, Hulstaert F, De Meerleer G, Mareel M, De Neve W. Evidence behind use of intensity-modulated radiotherapy: a systematic review of comparative clinical studies. Lancet Oncol. 2008;9(4):367–375. | ||
Christian JA, Bedford JL, Webb S, Brada M. Comparison of inverse-planned three-dimensional conformal radiotherapy and intensity-modulated radiotherapy for non-small-cell lung cancer. Int J Radiat Oncol Biol Phys. 2007;67(3):735–741. | ||
Uyterlinde W, Chen C, Kwint M, et al. Prognostic parameters for acute esophagus toxicity in intensity modulated radiotherapy and concurrent chemotherapy for locally advanced non-small cell lung cancer. Radiother Oncol. 2013;107(3):392–397. | ||
Gong B, Jiang N, Yan G, et al. Predictors for severe acute esophagitis in lung cancer patients treated with chemoradiotherapy: a systematic review. Curr Med Res Opin. 2016;32(10):1–8. | ||
Wijsman R, Dankers F, Troost EG, et al. Multivariable normal-tissue complication modeling of acute esophageal toxicity in advanced stage non-small cell lung cancer patients treated with intensity-modulated (chemo-)radiotherapy. Radiother Oncol. 2015;117(1):49–54. | ||
Werner-Wasik M, Yorke E, Deasy J, Nam J, Marks LB. Radiation dose-volume effects in the esophagus. Int J Radiat Oncol Biol Phys. 2010;76(3 Suppl):S86–S93. | ||
Dehing-Oberije C, de Ruysscher D, Petit S, et al. Development, external validation and clinical usefulness of a practical prediction model for radiation-induced dysphagia in lung cancer patients. Radiother Oncol. 2010;97(3):455–461. | ||
De Ruyck K, Sabbe N, Oberije C, et al. Development of a multicomponent prediction model for acute esophagitis in lung cancer patients receiving chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2011;81(2):537–544. | ||
Takeda K, Nemoto K, Saito H, Ogawa Y, Takai Y, Yamada S. Dosimetric correlations of acute esophagitis in lung cancer patients treated with radiotherapy. Int J Radiat Oncol Biol Phys. 2005;62(3):626–629. | ||
Dehing-Oberije C, De Ruysscher D, Petit S, et al. Development, external validation and clinical usefulness of a practical prediction model for radiation-induced dysphagia in lung cancer patients. Radiother Oncol. 2010;97(3):455–461. | ||
Rose J, Rodrigues G, Yaremko B, Lock M, D’Souza D, D’Souza D. Systematic review of dose-volume parameters in the prediction of esophagitis in thoracic radiotherapy. Radiother Oncol. 2009;91(3):282–287. | ||
Collman PI, Tremblay L, Diamant NE. The central vagal efferent supply to the esophagus and lower esophageal sphincter of the cat. Gastroenterology. 1993;104(5):1430–1438. | ||
Que J. The initial establishment and epithelial morphogenesis of the esophagus: a new model of tracheal-esophageal separation and transition of simple columnar into stratified squamous epithelium in the developing esophagus. Wiley Interdiscip Rev Dev Biol. 2015;4(4):419–430. | ||
Dankers F, Wijsman R, Troost EG, Monshouwer R, Bussink J, Hoffmann AL. Esophageal wall dose-surface maps do not improve the predictive performance of a multivariable NTCP model for acute esophageal toxicity in advanced stage NSCLC patients treated with intensity-modulated (chemo-)radiotherapy. Phys Med Biol. 2017;62(9):3668–3681. |
Supplementary materials
  | Figure S1 Dose-volume histogram for four subgroups as the cervical esophagus (A), the upper thoracic esophagus (B), the mid-thoracic esophagus (C), and the lower thoracic esophagus (D). |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.