Back to Journals » Journal of Pain Research » Volume 13
Clinical Comparison of Full-Endoscopic and Microscopic Unilateral Laminotomy for Bilateral Decompression in the Treatment of Elderly Lumbar Spinal stenosis: A Retrospective Study with 12-Month Follow-Up
Authors Yang F , Chen R, Gu D, Ye Q, Liu W , Qi J , Xu K, Fan X
Received 23 March 2020
Accepted for publication 22 May 2020
Published 11 June 2020 Volume 2020:13 Pages 1377—1384
DOI https://doi.org/10.2147/JPR.S254275
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Fei Yang,* Rigao Chen,* Dangwei Gu, Qingqing Ye, Wei Liu, Jianhua Qi, Kai Xu, Xiaohong Fan
Department of Spine Surgery, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, Sichuan, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaohong Fan Email [email protected]
Purpose: Although lumbar spinal stenosis (LSS) is the most common spinal disease in the elderly, there is still a confusion about the appropriate surgical treatment strategy. The aim of this study was to compare the safety and efficacy of full-endoscopic and microscopic unilateral laminotomy for bilateral decompression (ULBD) for LSS in elderly patients.
Patients and Methods: A retrospective analysis of 61 consecutive elderly patients with LSS who underwent either full-endoscopic (FE group) or microscopic (Micro group) unilateral laminotomy for bilateral decompression was performed. Clinical data were assessed before 2 weeks, 3 months, 6 months and 12 months after surgery using the Visual Analog Scale (VAS), the Oswestry Disability Index (ODI) and the modified MacNab criteria.
Results: There are no significant differences in VAS (back and leg) and ODI between the two groups. However, the VAS back pain in the FE group was significantly improved compared to the Micro group at 2 weeks. The rate of excellent or good outcomes was 87.88% and 85.71% in the FE and Micro group, respectively (P> 0.05). The hospital stay and early ambulation in FE group were shorter than those in Micro group, but the operation time was longer (P< 0.05). The complications between the FE group (18.18%) and the Micro group (17.86%) were minor (P> 0.05).
Conclusion: Both full-endoscopic and microscopic decompression have achieved favorable clinical results in treating elderly lumbar spinal stenosis, and the complications are minor. Full-endoscopic decompression has the advantages of small incision and rapid recovery, which can be used as an alternative for the treatment of lumbar spinal stenosis, especially the elderly with comorbidities.
Keywords: central stenosis, minimally invasive spinal surgery, MISS, comorbidity, complication
Introduction
With the increase of the elderly population and the prolongation of life expectancy, the prevalence of lumbar degenerative diseases, especially lumbar spinal stenosis, has increased significantly.1 Lumbar spinal stenosis usually begins with degeneration of the intervertebral disc and surrounding tissues, resulting in spinal canal narrowing and nerve compression, which is a common cause of low back and leg pain in the elderly.2 Because of its high incidence rate and negative impact on the quality of life, it is one of the most serious health problems.3 In order to improve mobility and quality of life, more elderly patients who have failed non-surgical treatment seek surgical treatment.4–6 However, there is much uncertainty in the surgical treatment of LSS, especially the elderly patients with comorbidities.7,8
The conventional approach for decompression is open laminectomy, which has been proved to be a surgical method with superior outcomes and satisfaction, compared to nonsurgical management.9,10 However, open decompression laminectomy can damage spinal structures such as paravertebral muscles, bones, and ligaments, resulting in muscle atrophy, low back pain, and postoperative low back syndrome.11–13 It is thus considered to promote iatrogenic instability and reoperation.14 Meanwhile, it requires decompression with fusion, the frequency of complex fusion procedures for spinal stenosis increased with higher risk of major complications, mortality, and medical utilization in the elderly.15,16 Minimally invasive spinal surgery (MISS) has been rapidly developed to minimize damage to the spine structure and overcome these problems.17 Microscopic unilateral laminotomy for bilateral decompression (MULBD) has gradually become the common surgical procedure with less blood loss, reducing pain, early ambulation, and shorter hospital stay.18–20 Nevertheless, the microscopic ULBD also have defects including the need for dissecting paravertebral muscles and the limitation of contralateral visual field.21 With the advancement of surgical technology and the improvement of instruments, several studies have reported favorable results in the treatment of LSS with full-endoscopic ULBD.22–26
So far, there have been few studies comparing full-endoscopic and microscopic ULBD in the treatment of elderly lumbar spinal stenosis. Thus, the objective of this study was to evaluate the safety and efficacy of full-endoscopic ULBD compared with that of microscopic ULBD for treating elderly lumbar spinal stenosis, who usually has comorbidities such as hypertension, diabetes, and heart disease.
Patients and Methods
Patient Population
From May 2017 to December 2018, 67 consecutive elderly patients with symptomatic LSS underwent unilateral laminotomy bilateral decompression (ULBD), full-endoscopic or microsurgical. Patients' data were entered into an electronic medical record system and their records were retrospectively analyzed. Six patients (9%) were lost during the follow-up period. Therefore, 33 patients were performed with full-endoscopic decompression (FE group) and 28 patients were performed with microscopic decompression (Micro group), all by a Surgeon with experience in minimally invasive spine surgery. The easiness or feasibility of the surgical technique did not play a role in the selection of surgery type. All participants provided written informed consent to participate in the study. The study was approved by the Ethics Committee of the Hospital of Chengdu University of Traditional Chinese Medicine and was performed in accordance with the Declaration of Helsinki.
Inclusion criteria included: 1) 65 years of age or older; 2) patients with neurogenic intermittent claudication with or without radiculopathy; 3) concordant imaging evidence of single-segment Central stenosis; 4) failed conservative treatment of at least 6 weeks. Exclusion criteria were as follows: 1) Isthmic spondylolisthesis or dynamic instability on the flexion/extension radiographs; 2) Pathological spinal diseases, such as infection, tumor, or previous spinal surgery.
Clinical Evaluation and Follow-Up
Age, sex, body mass index (BMI), Charlson Comorbidity Index (CCI),27 American Society of Anesthesiologists score (ASA), lumbar spinal stenosis grade28 and operation level were assessed between the two groups. Clinical efficacy was evaluated by calculating the Visual Analogue Scales (VAS)29 for back and leg pain, along with the Oswestry Disability Index (ODI)30 for functional status at preoperative,2-week,3-month, 6-month and 12-month postoperatively. Patient satisfaction was evaluated at the last follow-up using modified MacNab criteria.31 In addition, Perioperative data such as operative time, time to ambulation, hospital stay, and complications were documented. Complications were divided into major and minor, and major complications were defined as adversely affecting the patient’s recovery.7
Surgical Techniques
Microscopic UBLD
The patient was under general anesthesia and placed in a prone position with appropriate flexion. Taking the lesion segment as the center, a 4 cm incision was made along the standard median approach, the paravertebral muscles were dissected unilaterally and retracted by a Caspar, and the interlaminar windows were exposed. The cranial and caudal portion of the ipsilateral lamina were resected using kerrison rongeur, the ventral side of the hyperplastic process of the superior articular process was removed by drilling on the superficial surface of the lateral ligamentum flavum, and the narrow spinal canal in the medial pedicle of the Caudal vertebral body was completely decompressed. Tilting the operating table to the opposite side by 30–40°and adjusting the microscope angle, a burr and Kerrison rongeur were used to remove the base of the spinous process and a portion of contralateral lamina, followed by flavectomy. Finally, decompression of the contralateral recess was achieved by using a rongeur or a small drill to undercutting the medial part of the facet joint. After complete neural decompression was confirmed, hemostasis, negative pressure drainage was placed.
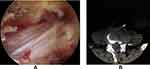
The full-endoscopic ULBD technique has been previously described.22–26 Briefly, patients were performed under general anesthesia and placed in the prone position with appropriate flexion on a C-arm fluororadiolucent table to widen the interlaminar window. The surgical segment and skin entry point were confirmed using anteroposterior radiographs. All operative procedures were performed with an endoscopic instrument system: Ilessys® Delta (joimax GmbH, Amalienbadstaβe 41, RaumFabrik 61,76,227 Karlsruhe, Germany), which is designed for posterior interlaminar technique. After making a paramedian incision of about 10 mm in length, Serial dilators were advanced until the Cranial lamina intersects with medial border of ipsilateral facet was palpated. The operation sheath was placed on the surface of the lamina and articular processes with its bevel facing the ligamentum flavum. The endoscope was then introduced and operated under continuous saline irrigation. After exposing the interlaminar window, the cranial and caudal portion of the ipsilateral lamina and the ventral side of the superior articular process were resected with drill and rongeur. Then the ligamentum flavum and its bone attachments were removed piece by piece. Subsequently, an adequate sublaminar space was created by tilting the endoscope towards the contralateral side and drilling off inner parts of the cranial lamina, and preserved the ligamentum flavum as much as possible to prevent dura injury. Resection of ligamentum flavum and the contralateral medial facetectomy were completed using drill and rongeur. Decompression was ended when it is confirmed that the bilateral nerves are completely decompressed (Figure 1). Hemostasis was achieved and drainage was performed postoperatively.
Postoperative Care
Both groups received intravenous non-steroidal anti-inflammatory analgesics daily for postoperative analgesia. Drainage was removed one day after the surgery. All patients were given a lumbar brace for 8 weeks.
Statistical Analysis
All statistical analyses were conducted using SPSS version 20.0 software (SPSS Inc., Chicago, IL, USA). Among the quantitative data, if the data conformed to normality and homogeneity of variance, the t-test was used for analysis, and it was expressed as mean and standard deviation; Those that did not conform to normality and homogeneity of variance were analyzed by Mann–Whitney U-tests, and were presented as median (interquartile range); the continuous measurement data were analyzed by Two-way Repeated measures ANOVA, and the data were expressed in the form of mean and standard deviation; Enumeration data were analyzed by Chi-square test, and Ridit analysis was used for ranked data. Take α=0.05 as the test level, and a value of P<0.05 was considered significant.
Results
The average age was 76.09±6.31 (range, 65–88) years in FE group and 72.96±5.85 (range, 65–85) years in Micro group. There were no significant differences in terms of baseline demographics, such as age, sex, BMI, CCI, ASA, lumbar spinal stenosis grade and operation level (Table 1).
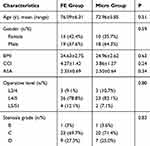 |
Table 1 Baseline Characteristics of the Included Patients |
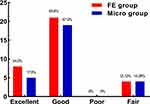
The mean (± standard deviation) VAS score for back pain improved from 4.70±0.88 to 2.21±0.78 in the FE group and from 4.68±0.94 to 2.50±0.75 in the Micro group (Figure 2A). The mean VAS score for leg pain improved from 5.97±0.85 to 2.39±1.12 in the FE group and from 6.07±0.81 to 2.68±1.09 in the Micro group (Figure 2B). The mean ODI improved from 60.58±5.85 to 29.42±8.84 in the FE group and from 61.86±7.32 to 28.75±7.06 in the Micro group (Figure 2C). There was no difference in the average back and leg VAS, ODI between the two groups preoperatively and at 2-week,6-month, and 12-month after surgery, while VAS back score in FE group was lower than that in Micro group at 2-week postoperative (2.55±0.75 vs 3.18±0.67, p<0.05).According to the modified MacNab criteria (Figure 3), excellent or good rate was 87.88% in FE group and 85.71% in Micro group at the 12-month review (p<0.05).
 |
Figure 2 Clinical outcomes preoperatively and at 2-week, 3-month, 6-month, and 12-month postoperatively. (A) VAS for back pain. (B) VAS for radicular leg pain. (C) Oswestry Disability Index (ODI). |
Perioperative data are shown in Table 2. The mean operation time of FE group was longer than that of Micro group (90.33 vs72.00, minutes, p<0.05). However, time to ambulation and hospital stay in FE group were shorter than that in Micro group.
 |
Table 2 The Perioperative Data and Complications |
No significant difference in the Complication rate was found between the groups (p<0.05, Table 2). No major complications such as pulmonary embolism, stroke, cardiac arrest, wound infections and peripheral nerve injury occurred. There were 2 cases of urinary retention in each group. In FE group, two patients of dural tear were treated with conservative treatment. One patient of acute exacerbation of chronic bronchitis was treated with third-generation cephalosporins. One patient had acute left heart failure the next day after surgery because of non-steroidal anti-inflammatory drugs. Although acute left heart failure is an acute and severe disease, the patient got out of bed quickly after treatment by a cardiologist and did not have adverse effects on recovery. Therefore, it was defined as a minor complication.7 In Micro group, one case of dural tear occurred, which was repaired during surgery. Two patients with transient delirium after surgery fully recovered after several days.
Discussion
In this retrospective study, which included 61 elderly patients with lumbar spinal stenosis, treated with unilateral laminotomy bilateral decompression (ULBD), full-endoscopic or microscopic, revealed no difference in clinical data at 12-month postoperatively. Although both groups of elderly patients relieved pain, improved function and quality of life, full-endoscopic technique had the advantages of less trauma and faster recovery. Complication rates were similar between the two groups, with no major complications occurring.
Making surgical intervention decisions is a formidable task for the elderly patients with multiple comorbidities. Some studies have reported a higher rate of complications after surgery in elderly patients.16,32,33 A study of patients aged 65 and older who underwent decompression and arthrodesis showed a complication rate of over 70% and a major complication rate of 23.6%.32 Another study involving 2320 elderly patients found that the elderly who underwent lumbar surgery had higher complications, with risk factors including reduced baseline functional status, longer and more complex procedures.16 However, in contrast, there have been quite a few reports that lumbar decompression without fusion may be performed safely and efficiently without undue risk to the elderly patient.34–38 A multicenter observational study shows that individuals aged 80 and older experienced similar and clinically important improvements in disease-specific self-reported outcomes after decompressive surgery for central LSS than younger participants, few complications in either.34 Another study of patients aged 75 and older showed that minimally invasive lumbar spine decompression is a safe and effective treatment, which should be considered good candidates for the elderly lumbar spinal stenosis.37 In this study, elderly patients rated as physical status I-III under ASA tolerated the surgical procedure well, many of them had comorbidities such as hypertension, diabetes, and chronic obstructive pulmonary disease. Although there were a few patients with acute heart failure, acute exacerbation of chronic bronchitis, urinary retention, transient delirium and dural tear, all complications were minor and did not adversely affect the recovery of patients. What needs to be vigilant is that elderly patients are more prone to perioperative complications, and careful perioperative monitoring is essential.
Although the concept of preserving the spinal structure is considered to be important in minimally invasive decompression surgery, there are still concerns about incomplete decompression in the treatment of LSS. The key to avoiding incomplete decompression is adequate resection of bone tissue under continuous visual control.22 The results of this study showed that both groups of patients had improved postoperative symptoms and maintained satisfactorily during a 12-month follow-up, which is comparable to those of other published studies of full-endoscopic technique,22–26 indicating that decompression was adequately performed. Our research showed that the good or excellent rate of the FE group and the Micro group were 87.88% and 85.71%, respectively, which are superior to previous studies of conventional laminectomy for the elderly, 67.1% or 69.1%.35,39 In addition, the clinical results of this study were similar to the studies of Microscopic unilateral laminotomy for bilateral decompression.18–20 However, VAS back pain scores in FE group were less than that of the Micro group at 2-week postoperative, and time to ambulation and hospital stay were also earlier in FE group. This result may be due to minimal tissue damage, rapid pain recovery, and improved quality of life. As with traditional lumbar laminectomy, the microscopic UBLD also needs to dissect the ipsilateral paravertebral muscle, which is an important cause of multifidus injury and low back pain.13 Compared with the literature, the results of these parameters were also in favor to the FE group.22,26,40,41
The average operation time in the FE group (90.33minutes) was longer than that in Micro group (72.00 minutes), which may be due to the steep learning curve of most minimally invasive techniques, especially in lumbar endoscopic surgery.42–46 The surgical field of endoscopic spine surgery is relatively narrow. Meanwhile, the difference between the actual surgical angle and the optical angle of the endoscope makes it difficult to manipulate the endoscopic surgical instruments. A recent study found that additional surgical experience may be needed to shorten the learning curve, and full-endoscopic techniques can be used as the main treatment for LSS.46 Although the average operation time of FE group was slightly longer, the results showed favorable clinical outcomes and no major surgical complications occurred. Additional experience of endoscopic surgery, attending workshops, and suitable patient selection can help shorten the operation time and decrease complications.
There are several limitations to the current study. First, it is a single-center retrospective study with a small sample size and short follow-up period. Second, analysis of postoperative imaging parameters is lacking in this study. Third, lack of evaluation of muscle injury after surgery, such as C-reactive protein, Creatine phosphokinase, among others. In addition, there may be selection bias in determining the surgical procedures. Thus, Adequate randomized prospective studies for full-endoscopic are needed to evaluate postoperative muscle injury, postoperative segmental instability, and long-term clinical outcomes.
Conclusion
Both full-endoscopic and microscopic decompression have achieved favorable clinical results in treating elderly lumbar spinal stenosis, and the complications are minor. Full-endoscopic technique has the advantages of small incision and rapid recovery, which can be used as an alternative for the treatment of lumbar spinal stenosis, especially the elderly with comorbidities.
Acknowledgments
We would like to thank the relevant staff for guidance and assistance for their support and collaboration in our hospital.
Disclosure
The authors report no conflicts of interest concerning the materials or methods used in these findings specified in this paper.
References
1. Fehlings MG, Tetreault L, Nater A, et al. The aging of the global population: the changing epidemiology of disease and spinal disorders. Neurosurgery. 2015;77(Suppl 4):S1–5. doi:10.1227/NEU.0000000000000953
2. Hilibrand AS, Rand N. Degenerative lumbar stenosis: diagnosis and management. J Am Acad Orthop Surg. 1999;7(4):239–249.
3. Otani K, Kikuchi S, Yabuki S, et al. Lumbar spinal stenosis has a negative impact on quality of life compared with other comorbidities: an epidemiological cross-sectional study of 1862 community-dwelling individuals. Sci World J. 2013;2013:590652. doi:10.1155/2013/590652
4. Kobayashi K, Ando K, Nishida Y, Ishiguro N, Imagama SJESJ. Epidemiological trends in spine surgery over 10 years in a multicenter database. Eur Spine J. 2018;27(8).
5. Aizawa T, Kokubun S, Ozawa H, et al. Increasing incidence of degenerative spinal diseases in Japan during 25 years: the registration system of spinal surgery in Tohoku University spine society. Tohoku J Exp Med. 2016;238(2):153–163. doi:10.1620/tjem.238.153
6. O’Lynnger TM, Zuckerman SL, Morone PJ, Dewan MC, Vasquez-Castellanos RA, Cheng JS. Trends for spine surgery for the elderly: implications for access to healthcare in North America. Neurosurgery. 2015;77(Suppl 4):S136–141. doi:10.1227/NEU.0000000000000945
7. Cloyd JM, Acosta FL, Ames CP. Complications and outcomes of lumbar spine surgery in elderly people: a review of the literature. J Am Geriatr Soc. 2008;56(7):1318–1327. doi:10.1111/j.1532-5415.2008.01771.x
8. Choi JM, Choi MK, Kim SB. Perioperative results and complications after posterior lumbar interbody fusion for spinal stenosis in geriatric patients over than 70 years old. J Korean Neurosurg Soc. 2017;60(6):684–690. doi:10.3340/jkns.2017.0203
9. Malmivaara A, Slatis P, Heliovaara M, et al. Surgical or nonoperative treatment for lumbar spinal stenosis? A randomized controlled trial. Spine. 2007;32(1):1–8. doi:10.1097/01.brs.0000251014.81875.6d
10. Atlas SJ, Keller RB, Wu YA, Deyo RA, Singer DE. Long-term outcomes of surgical and nonsurgical management of lumbar spinal stenosis: 8 to 10 year results from the maine lumbar spine study. Spine. 2005;30(8):936–943. doi:10.1097/01.brs.0000158953.57966.c0
11. Kim CW. Scientific basis of minimally invasive spine surgery: prevention of multifidus muscle injury during posterior lumbar surgery. Spine. 2010;35(Supplement):S281–286. doi:10.1097/BRS.0b013e3182022d32
12. Fan S, Hu Z, Zhao F, Zhao X, Huang Y, Fang X. Multifidus muscle changes and clinical effects of one-level posterior lumbar interbody fusion: minimally invasive procedure versus conventional open approach. Eur Spine J. 2010;19(2):316–324. doi:10.1007/s00586-009-1191-6
13. Hu ZJ, Fang XQ, Zhou ZJ, Wang JY, Zhao FD, Fan SW. Effect and possible mechanism of muscle-splitting approach on multifidus muscle injury and atrophy after posterior lumbar spine surgery. J Bone Joint Surg Am. 2013;95(24):
14. Guha D, Heary RF, Shamji MF. Latrogenic spondylolisthesis following laminectomy for degenerative lumbar stenosis: systematic review and current concepts. Neurosurg Focus. 2015;39(4):E9. doi:10.3171/2015.7.FOCUS15259
15. Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303(13):1259–1265. doi:10.1001/jama.2010.338
16. Saleh A, Thirukumaran C, Mesfin A, Molinari RW. Complications and readmission after lumbar spine surgery in elderly patients: an analysis of 2320 patients. Spine J. 2017;17(8):1106–1112. doi:10.1016/j.spinee.2017.03.019
17. Smith ZA, Fessler RG. Paradigm changes in spine surgery: evolution of minimally invasive techniques. Nat Rev Neurol. 2012;8(8):443–450. doi:10.1038/nrneurol.2012.110
18. Mobbs RJ, Li J, Sivabalan P, Raley D, Rao PJ. Outcomes after decompressive laminectomy for lumbar spinal stenosis: comparison between minimally invasive unilateral laminectomy for bilateral decompression and open laminectomy: clinical article. J Neurosurg Spine. 2014;21(2):179–186. doi:10.3171/2014.4.SPINE13420
19. Liu X, Yuan S, Tian Y. Modified unilateral laminotomy for bilateral decompression for lumbar spinal stenosis: technical note. Spine. 2013;38(12):E732–737. doi:10.1097/BRS.0b013e31828fc84c
20. Mayer HM. Microsurgical decompression of acquired (degenerative) central and lateral spinal canal stenosis. In: Mayer HM, editor. Minimally Invasive Spine Surgery: A Surgical Manual. Berlin, Heidelberg: Springer Berlin Heidelberg; 2006:397–408.
21. Thome C, Zevgaridis D, Leheta O, et al. Outcome after less-invasive decompression of lumbar spinal stenosis: a randomized comparison of unilateral laminotomy, bilateral laminotomy, and laminectomy. J Neurosurg Spine. 2005;3(2):129–141. doi:10.3171/spi.2005.3.2.0129
22. Komp M, Hahn P, Oezdemir S, et al. Bilateral spinal decompression of lumbar central stenosis with the full-endoscopic interlaminar versus microsurgical laminotomy technique: a prospective, randomized, controlled study. Pain Physician. 2015;18(1):61–70.
23. Hasan S, McGrath LB, Sen RD, Barber JK, Hofstetter CP. Comparison of full-endoscopic and minimally invasive decompression for lumbar spinal stenosis in the setting of degenerative scoliosis and spondylolisthesis. Neurosurg Focus. 2019;46(5):E16. doi:10.3171/2019.2.FOCUS195
24. Lee CW, Yoon KJ, Jun JH. Percutaneous endoscopic laminotomy with flavectomy by uniportal, unilateral approach for the lumbar canal or lateral recess stenosis. World Neurosurg. 2018;113:e129–e137. doi:10.1016/j.wneu.2018.01.195
25. Huang YH, Lien FC, Chao LY, Lin CH, Chen SH. Full endoscopic uniportal unilateral laminotomy for bilateral decompression in degenerative lumbar spinal stenosis: highlight of ligamentum flavum detachment and survey of efficacy and safety in 2 years of follow-up. World Neurosurg. 2020;134:e672–e681. doi:10.1016/j.wneu.2019.10.162
26. Ito F, Ito Z, Shibayama M, et al. Step-by-step sublaminar approach with a newly-designed spinal endoscope for unilateral-approach bilateral decompression in spinal stenosis. Neurospine. 2019;16(1):41–51. doi:10.14245/ns.1836320.160
27. Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. doi:10.1016/0895-4356(94)90129-5
28. Schizas C, Theumann N, Burn A, et al. Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images. Spine. 2010;35(21):1919–1924. doi:10.1097/BRS.0b013e3181d359bd
29. Chapman CR, Casey KL, Dubner R, Foley KM, Gracely RH, Reading AE. Pain measurement: an overview. Pain. 1985;22(1):1–31. doi:10.1016/0304-3959(85)90145-9
30. Lue YJ, Hsieh CL, Huang MH, Lin GT, Lu YM. Development of a Chinese version of the oswestry disability index version 2.1. Spine. 2008;33(21):2354–2360. doi:10.1097/BRS.0b013e31818018d8
31. Macnab I. Negative disc exploration. An analysis of the causes of nerve-root involvement in sixty-eight patients. J Bone Joint Surg Am. 1971;53(5):891–903. doi:10.2106/00004623-197153050-00004
32. Carreon LY, Puno RM, Dimar JR, Glassman SD, Johnson JR. Perioperative complications of posterior lumbar decompression and arthrodesis in older adults. J Bone Joint Surg Am. 2003;85(11):2089–2092. doi:10.2106/00004623-200311000-00004
33. Raffo CS, Lauerman WC. Predicting morbidity and mortality of lumbar spine arthrodesis in patients in their ninth decade. Spine. 2006;31(1):99–103. doi:10.1097/01.brs.0000192678.25586.e5
34. Giannadakis C, Solheim O, Jakola AS, et al. Surgery for lumbar spinal stenosis in individuals aged 80 and older: a multicenter observational study. J Am Geriatr Soc. 2016;64(10):2011–2018. doi:10.1111/jgs.14311
35. Arinzon ZH, Fredman B, Zohar E, et al. Surgical management of spinal stenosis: a comparison of immediate and long term outcome in two geriatric patient populations. Arch Gerontol Geriatr. 2003;36(3):273–279. doi:10.1016/S0167-4943(02)00172-3
36. Antoniadis A, Ulrich NH, Schmid S, Farshad M, Min K. Decompression surgery for lumbar spinal canal stenosis in octogenarians; a single center experience of 121 consecutive patients. Br J Neurosurg. 2017;31(1):67–71. doi:10.1080/02688697.2016.1233316
37. Rosen DS, O’Toole JE, Eichholz KM, et al. Minimally invasive lumbar spinal decompression in the elderly: outcomes of 50 patients aged 75 years and older. Neurosurgery. 2007;60(3):503–510. doi:10.1227/01.NEU.0000255332.87909.58
38. Shabat S, Arinzon Z, Folman Y, et al. Long-term outcome of decompressive surgery for lumbar spinal stenosis in octogenarians. Eur Spine J. 2008;17(2):193–198. doi:10.1007/s00586-007-0514-8
39. Best NM, Sasso RC. Outpatient lumbar spine decompression in 233 patients 65 years of age or older. Spine. 2007;32(10):
40. Xin Z, Huang P, Zheng G, Liao W, Zhang X, Wang Y. Using a percutaneous spinal endoscopy unilateral posterior interlaminar approach to perform bilateral decompression for patients with lumbar lateral recess stenosis. Asian J Surg. 2019.
41. Lv Z, Jin L, Wang K, et al. Comparison of effects of PELD and fenestration in the treatment of geriatric lumbar lateral recess stenosis. Clin Interv Aging. 2019;14:2187–2194. doi:10.2147/CIA.S226295
42. Ahn J, Iqbal A, Manning BT, et al. Minimally invasive lumbar decompression-the surgical learning curve. Spine J. 2016;16(8):909–916. doi:10.1016/j.spinee.2015.07.455
43. Wang B, Lu G, Patel AA, Ren P, Cheng I. An evaluation of the learning curve for a complex surgical technique: the full endoscopic interlaminar approach for lumbar disc herniations. Spine J. 2011;11(2):122–130. doi:10.1016/j.spinee.2010.12.006
44. Hsu HT, Chang SJ, Yang SS, Chai CL. Learning curve of full-endoscopic lumbar discectomy. Eur Spine J. 2013;22(4):727–733. doi:10.1007/s00586-012-2540-4
45. Park SM, Kim HJ, Kim GU, et al. Learning curve for lumbar decompressive laminectomy in biportal endoscopic spinal surgery using the cumulative summation test for learning curve. World Neurosurg. 2019;122:e1007–e1013. doi:10.1016/j.wneu.2018.10.197
46. Lee CW, Yoon KJ, Kim SW. Percutaneous endoscopic decompression in lumbar canal and lateral recess stenosis – the surgical learning curve. Neurospine. 2019;16(1):63–71. doi:10.14245/ns.1938048.024
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.