Back to Journals » OncoTargets and Therapy » Volume 13
circPDSS1 Stimulates the Development of Colorectal Cancer via Activating the Wnt/β-Catenin Signaling
Authors Fang Q, Yang A, Dong A, Zhao L
Received 14 February 2020
Accepted for publication 6 May 2020
Published 30 June 2020 Volume 2020:13 Pages 6329—6337
DOI https://doi.org/10.2147/OTT.S249853
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Leo Jen-Liang Su
This paper has been retracted.
Qun Fang, 1,* Aijie Yang, 2,* Anshan Dong, 3 Ligang Zhao 4
1Department of Emergency, Yidu Central Hospital of Weifang, Weifang, People’s Republic of China; 2Department of Radiotherapy, Qilu Hospital (Qingdao), Cheeloo College of Medicine, Shandong University, Qingdao, People’s Republic of China; 3Department of Gastroenterology, Weifang Second People’s Hospital, Weifang, People’s Republic of China; 4Department of General Surgery, PLA Rocket Force Characteristic Medical Center, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Anshan Dong
Department of Gastroenterology, Weifang Second People’s Hospital, 7 Yuanxiao Street, Weifang, Shandong 261000, People’s Republic of China
Tel +8613573654782
Email [email protected]
Objective: This study aims to illustrate the role of circPDSS1 and the Wnt/β-catenin signaling in the development of colorectal cancer (CRC).
Patients and Methods: Cancerous mucosa and normal paracancerous mucosa tissues more than 5 cm away from the tumor were surgically collected from 56 CRC patients. circPDSS1 levels in collected tissues and CRC cell lines were detected by quantitative real-time polymerase chain reaction (qRT-PCR). The influence of circPDSS1 on clinical features of CRC patients was analyzed. After knockdown of circPDSS1 in HCT-8 and HCT-116 cells, phenotype changes were examined by Transwell, tube formation and wound healing assay. Western blot and rescue experiments were finally performed to uncover the role of circPDSS1 and the Wnt/β-catenin signaling in the development of CRC.
Results: circPDSS1 was upregulated in CRC mucosa tissues than controls. High level of circPDSS1 predicted high rates of lymphatic metastasis and distant metastasis, and poor prognosis in CRC patients. Knockdown of circPDSS1 attenuated migratory ability and angiogenesis in CRC cells. Protein levels of key genes in the Wnt/β-catenin signaling, including β-catenin, GSK-3β, c-Myc, MMP-9 and cyclin D1 were downregulated in CRC cells transfected with sh-circPDSS1. Overexpression of β-catenin reversed the role of circPDSS1 in attenuating migratory ability and angiogenesis in CRC cells.
Conclusion: Upregulated circPDSS1 in CRC is closely linked to lymphatic metastasis, distant metastasis and overall survival. It stimulates the migratory ability and angiogenesis in CRC cells via activating the Wnt/β-catenin signaling.
Keywords: circPDSS1, Wnt/β-catenin, CRC, metastasis
Introduction
Colorectal cancer (CRC) is a popular malignant disease throughout the world, which seriously endangers health, and it is estimated that the incidence of CRC ranks third and its mortality ranks second place in malignancies.1–3 With changes in lifestyle and diet structure (high-fat, high-protein, low-fiber diet), male sex, accelerated aging population, inflammatory bowel disease and a positive family history, the incidence of CRC remains high.4–6 In China, the increased trends of incidence and mortality of CRC pose a great burden on families and the society.7 It is estimated that medical cost on CRC is the most expensive in tumor diseases and its non-medical expense is hard to evaluate.8,9 Therefore, the screening, diagnosis and treatment of CRC in the early phase significantly could improve the clinical outcomes of CRC. Nevertheless, the diagnostic methods of early-stage CRC are limited, and the therapeutic efficacies of surgery and adjuvant chemotherapy for advanced CRC are not ideal.10,11 The main reason for this dilemma is that the pathogenesis of CRC has not been fully elucidated.11 It is necessary to reveal the specific pathogenesis of CRC, thus benefiting patients in middle or late stage.12,13
circRNAs differ from linear RNAs, which are closed loop RNAs formed by covalent bonds, containing exon and intron sequencing.14,15 circRNAs are abundantly expressed in eukaryotes.15,16 The majority of circRNAs are highly conserved and resistant to RNAases, showing tissue or stage-specificity.16,17 Previous studies have shown that circRNAs are involved in the development and metastasis of some malignant tumors, including bladder cancer, gastric cancer, hepatocellular carcinoma, esophageal squamous cell carcinoma, and ovarian cancer.18–21 circPDSS1, which reported with aberrant expressions in many tumors, could suggest the possibility to be a promising tumor hallmark.22,23 Meanwhile, we found the abnormal expression and the identified biological functions of circPDSS1 in CRC by data analysis.
The role of circPDSS1 in CRC is largely unknown. This study mainly explored the biological functions of circPDSS1 in influencing malignant phenotypes of CRC cells and the involvement of the Wnt/β-catenin signaling. Our findings may provide a new idea in the diagnosis and treatment of CRC.
Patients and Methods
CRC Patients and Samples
Cancerous mucosa and normal paracancerous mucosa tissues more than 5 cm away from the tumor were surgically collected from 56 CRC patients, which were pathologically confirmed by H&E staining. None of them had pre-operative anti-cancer treatment. Samples were immediately frozen in liquid nitrogen and stored at −80°C. This study got approval by Ethics Committee of Yidu Central Hospital of Weifang and it was conducted after informed consent of each subject. This study complied with the Declaration of Helsinki.
Cell Lines and Reagents
CRC cell lines (HT29, HCT-8, HCT-116, SW480) and the intestinal epithelial cell line (FHC) were purchased from ATCC (Manassas, VA, USA). HT29 and SW480 were cultured in Dulbecco’s modified eagle medium (DMEM) (Thermo Fisher Scientific, Waltham, MA, USA), and the others were in Roswell Park Memorial Institute 1640 (RPMI 1640). 10% fetal bovine serum (FBS) (Gibco, Rockville, MD, USA), 100 U/mL penicillin and 100 μg/mL streptomycin were applied in culture medium. Cell passage was conducted until cells were grown to 80–90% confluence.
Transfection
Cells were inoculated in 6-well plates and cultured to 30–50% confluence. They were transfected with plasmids constructed by GenePharma (Shanghai, China) using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Transfection efficacy was tested by quantitative real-time polymerase chain reaction (qRT-PCR) at 48 h.
Tube Formation Assay
Cells incubated in the mixture of 300 μL of TCM and 300 μL of culture medium were applied per well in a 24-well plate where 300 μL of Matrigel per well was pre-coated. After 2-day cell culture, angiogenesis was assessed by calculating the total branch points per sample under a microscope (Nikon Corporation, Tokyo, Japan).
Wound Healing Assay
Cells were inoculated in 6-well plates and grown to 90% confluence. After creation of an artificial wound in cell monolayer, medium with 1% FBS was replaced. 24 hours later, wound closure was captured for calculating the percentage of wound healing (magnification 40×).
Transwell Assay
Matrigel was pre-coated on the bottom of the Transwell chamber (Millipore, Billerica, MA, USA) for 24 h. 200 μL of suspension (2.0×105/mL) was applied in the upper side of the chamber inserted in a 24-well plate with 560 μL of medium containing 10% FBS in the bottom. After 48 h of incubation, cells in the bottom were fixed in methanol for 15 min, dyed with crystal violet for 20 min and counted using a microscope. Migratory cell number was counted in 5 randomly selected fields per sample (magnification 40×).
Quantitative Real-Time PCR (qRT-PCR)
Extracted RNAs by TRIzol reagent (Invitrogen, Carlsbad, CA, USA) were purified by DNase I treatment, and reversely transcribed into cDNAs using Primescript RT Reagent (Takara, Otsu, Japan). The obtained cDNAs underwent qRT-PCR using SYBR®Premix Ex Taq™ (Takara, Japan). Each sample was performed in triplicate, and relative level was calculated by 2−ΔΔCt and normalized to that of β-actin. circPDSS1: forward: 5ʹ-GTGGTGCATGAGATCGCCT-3ʹ, reverse: 5ʹ-GGGTTGTGTGATGAAACCTG-3ʹ; β-actin: forward: 5ʹ-CCTGGCACCCAGCACAAT-3ʹ, reverse: 5ʹ-GCTGATCCACATCTGCTGGAA-3ʹ.
Western Blot
Cells were lysed on ice for isolating proteins. After detection of protein concentration by BCA method, protein samples were separated by 10% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE). They were subsequently loaded on polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). Non-specific antigens were blocked in 5% skim milk for 2 hours. Membranes were reacted with primary and secondary antibodies for indicated time. Band exposure and analyses were finally conducted.
In vivo Xenograft Model
In vivo xenograft model was established after approval of the Animal Ethics and Use Committee. A total of 10 male nude mice were randomly assigned into two groups (n=5). They were subcutaneously administrated with HCT-8 cells transfected with sh-NC or sh-circPDSS1, respectively. Tumor size was recorded every 7 days. Six weeks later, tumor tissues were collected and weighed after sacrifice. Tumor volume = (width2×length)/2.
Luciferase Assay
Cells were pre-inoculated in 24-well plates overnight. They were co-transfected with pcDNA-NC/pcDNA-β-catenin and circPDSS1-WT/circPDSS1-MUT, respectively. Luciferase vectors used here were constructed based on the predicted binding sequences in the 3ʹUTR of β-catenin. After 48 h cell co-transfection, luciferase activity was measured.
Statistical Analysis
SPSS 22.0 (SPSS IBM, Armonk, NY USA) was used for data analyses. Data were expressed as mean ± standard deviation. Differences between groups were analyzed by the t-test. The influences of circPDSS1 on clinical features of CRC patients were analyzed by Chi-square analysis. Kaplan-Meier curves were depicted for survival analysis. P < 0.05 was considered as statistically significant.
Results
Highly Expressed circPDSS1 in CRC Tissues and Cell Lines
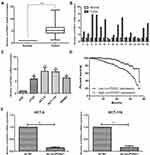
Compared with paracancerous mucosa, circPDSS1 was upregulated in mucosa tissues of CRC (Figure 1A and B). Similarly, circPDSS1 was upregulated in CRC cell lines, especially HCT-8 and HCT-116 cells (Figure 1C). These two cell lines were selected for establishing the in vitro circPDSS1 knockdown model by transfection of sh-circPDSS1 (Figure 1E).
circPDSS1 Expression Was Correlated with Lymph Node and Distance Metastasis in CRC Patients
Recruited 56 CRC patients were divided into two groups according to the median level of circPDSS1 in cancerous mucosa tissues collected from them. By analyzing their clinical data, it is found that circPDSS1 level was positively correlated to rates of lymphatic metastasis and distant metastasis, while it was unrelated to age, sex and tumor staging in CRC patients (Table 1). In addition, follow-up data were collected for assessing the relationship between circPDSS1 level and prognosis in CRC. Kaplan-Meier method yielded the conclusion that high level of circPDSS1 indicated a poor prognosis in CRC (Figure 1D).
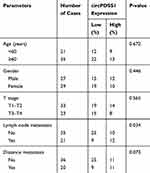 |
Table 1 Association of circPDSS1 Expression with Clinicopathologic Characteristics of Colorectal Cancer |
Knockdown of circPDSS1 Inhibited Cell Metastasis and Angiogenesis in CRC
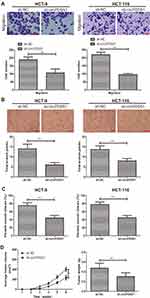
Transwell assay revealed fewer migratory cells after knockdown of circPDSS1 in HCT-8 and HCT-116 cells, indicating the weakened migratory ability (Figure 2A). Similarly, decreased percentage of wound closure in CRC cells transfected with sh-circPDSS1 also confirmed the role of circPDSS1 in triggering CRC metastasis (Figure 2C). Compared with those of controls, CRC cells transfected with sh-circPDSS1 had lower total branch points, suggesting the inhibited angiogenesis (Figure 2B). The above evidences have proven the in vitro functions of circPDSS1 in CRC cells. Subsequently, in vivo influences of circPDSS1 on CRC progression were explored by establishing the xenograft model. Compared with those of sh-NC, in vivo knockdown of circPDSS1 resulted in smaller tumor volume and lower tumor weight, indicating the inhibited CRC progression (Figure 2D).
Knockdown of circPDSS1 Inhibited the Activity of the Wnt/β-Catenin Signaling in CRC
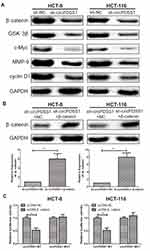
After transfection of sh-circPDSS1, protein levels of β-catenin, GSK-3β, c-Myc, MMP-9 and cyclin D1 were downregulated in CRC cells, indicating the regulatory effect of circPDSS1 on the activation of the Wnt/β-catenin signaling (Figure 3A). To further uncover the involvement of the Wnt/β-catenin signaling in CRC, we constructed pcDNA-β-catenin. Transfection of pcDNA-β-catenin markedly upregulated β-catenin in CRC cells with circPDSS1 knockdown (Figure 3B). In addition, the Bioinformatics and luciferase reporter gene analysis was applied to assess whether circPDSS1 act on β-catenin. The results showed that compared with NC group, the luciferase activity of cells transfected with β-catenin overexpression vector decreased significantly, which suggested that β-catenin was the target gene of circPDSS1 (Figure 3C).
β-Catenin Was Responsible for Malignant Phenotypes of CRC Regulated by circPDSS1
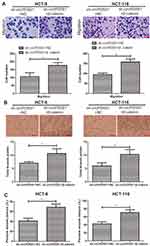
We thereafter explored the potential function of β-catenin in the malignant development of CRC. Higher migratory cell number was seen in CRC cells co-transfected with sh-circPDSS1 and pcDNA-β-catenin than those with solely knockdown of circPDSS1 (Figure 4A). As expected, overexpression of β-catenin enhanced wound closure percentage in CRC cells with circPDSS1 knockdown (Figure 4C). The similar trend was also found in tube formation assay (Figure 4B). It is concluded that overexpression of β-catenin abolished the role of circPDSS1 in suppressing CRC metastasis and angiogenesis.
Discussion
The occurrence and development of CRC are complicated involving multiple factors.1–3 Theories of gene regulation, tumor stem cells and microenvironmental inflammation are well concerned in the field of CRC research.24,25 In addition, the cancer-associated fibroblasts and endothelial cells, as well as tumor-associated macrophages, which construct this favourable for the tumor growth and invasiveness microenvironment.25 Gene regulation theory highlights the involvement of oncogene activation, tumor suppressor inactivation and signaling transduction in tumor development.10–13 circRNAs, which are newly discovered RNAs, are promising tumor hallmarks.14,15 They are structurally formed through back splicing of the 3ʹ and 5ʹ terminals.15,16 The specific structure of circRNAs results in their characteristics of extension, conservatism and tissue specificity.16,17
circRNAs have been hot topics in the research of human diseases, including Alzheimer’s disease, osteoarthritis and atherosclerosis.16,17 Differentially expressed circRNAs have been identified between tumor tissues and paracancerous ones, and they may be promising therapeutic targets.18,19 A great number of circRNAs with vital biological functions require to be explored.14–17 We collected 56 pairs cancerous mucosa and normal paracancerous mucosa tissues in CRC patients. It is found that circPDSS1 was upregulated in CRC samples. Its level was positively linked to lymphatic metastasis and distant metastasis rates in CRC patients, suggesting the potential tumor-promoting role in CRC. In addition, circPDSS1 knockdown model was established in HCT-8 and HCT-116 cells because of their highest abundance of circPDSS1 in the tested CRC cell lines. Transwell, tube formation and wound healing assay all suggested the role of circPDSS1 in stimulating metastasis and angiogenesis in CRC. Knockdown of circPDSS1 markedly downregulated vital genes in the Wnt/β-catenin signaling.
circRNAs not only compete with pre-mRNAs that target on gene regulations, but also regulate parental gene expressions.26,27 circRNAs are vital regulators in the development of malignant tumors.18,19 Specifically, the predictive role of cirRNAs should be further discussed. They regulate the Wnt pathway in CRC, while activation of the Wnt pathway leads to the increase of expression of mRNAs that directly bind to their gene promoters.28 Our findings have already confirmed the involvement of circPDSS1 and the Wnt/β-catenin signaling in the malignant development of CRC. Subsequently, we further explored how circPDSS1 and β-catenin synergistically drove the malignant phenotypes of CRC cells. Overexpression of β-catenin remarkably abolished the inhibitory effects of circPDSS1 on CRC cell metastasis and angiogenesis. As a result, it is suggested that circPDSS1 drove the malignant development of CRC via activating the Wnt/β-catenin signaling. Therefore, we need to further expand the sample size and more clinical data to reveal the circPDSS1 in the development of CRC patients.
Conclusions
Upregulated circPDSS1 in CRC is closely linked to lymphatic metastasis, distant metastasis and overall survival. It stimulates the migratory ability and angiogenesis in CRC cells via activating the Wnt/β-catenin signaling.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383(9927):1490–1502. doi:10.1016/S0140-6736(13)61649-9
2. Rawla P, Sunkara T, Barsouk A. Epidemiology of colorectal cancer: incidence, mortality, survival, and risk factors. Prz Gastroenterol. 2019;14(2):89–103. doi:10.5114/pg.2018.81072
3. Gini A, Jansen E, Zielonke N, et al. Impact of colorectal cancer screening on cancer-specific mortality in Europe: a systematic review. Eur J Cancer. 2020;127:224–235. doi:10.1016/j.ejca.2019.12.014
4. Thanikachalam K, Khan G. Colorectal cancer and nutrition. Nutrients. 2019;11:1. doi:10.3390/nu11010164
5. Azeem S, Gillani SW, Siddiqui A, Jandrajupalli SB, Poh V, Syed SS. Diet and colorectal cancer risk in Asia–a systematic review. Asian Pac J Cancer Prev. 2015;16(13):5389–5396. doi:10.7314/apjcp.2015.16.13.5389
6. Zarkavelis G, Boussios S, Papadaki A, Katsanos KH, Christodoulou DK, Pentheroudakis G. Current and future biomarkers in colorectal cancer. Ann Gastroenterol. 2017;30(6):613–621. doi:10.20524/aog.2017.0191
7. Gu MJ, Huang QC, Bao CZ, et al. Attributable causes of colorectal cancer in China. BMC Cancer. 2018;18(1):38. doi:10.1186/s12885-017-3968-z
8. Wong MC, Ding H, Wang J, Chan PS, Huang J. Prevalence and risk factors of colorectal cancer in Asia. Intest Res. 2019;17(3):317–329. doi:10.5217/ir.2019.00021
9. Tsoi KK, Hirai HW, Chan FC, Griffiths S, Sung JJ. Cancer burden with ageing population in urban regions in China: projection on cancer registry data from World Health Organization. Br Med Bull. 2017;121(1):83–94. doi:10.1093/bmb/ldw050
10. Issa IA, Noureddine M. Colorectal cancer screening: an updated review of the available options. World J Gastroenterol. 2017;23(28):5086–5096. doi:10.3748/wjg.v23.i28.5086
11. Maida M, Macaluso FS, Ianiro G, et al. Screening of colorectal cancer: present and future. Expert Rev Anticancer Ther. 2017;17(12):1131–1146. doi:10.1080/14737140.2017.1392243
12. Villeger R, Lopes A, Veziant J, et al. Microbial markers in colorectal cancer detection and/or prognosis. World J Gastroenterol. 2018;24(22):2327–2347. doi:10.3748/wjg.v24.i22.2327
13. Lech G, Slotwinski R, Slodkowski M, Krasnodebski IW. Colorectal cancer tumour markers and biomarkers: recent therapeutic advances. World J Gastroenterol. 2016;22(5):1745–1755. doi:10.3748/wjg.v22.i5.1745
14. Patop IL, Kadener S. circRNAs in cancer. Curr Opin Genet Dev. 2018;48:121–127. doi:10.1016/j.gde.2017.11.007
15. Patop IL, Wust S, Kadener S. Past, present, and future of circRNAs. EMBO J. 2019;38(16):e100836. doi:10.15252/embj.2018100836
16. Fischer JW, Leung AK. CircRNAs: a regulator of cellular stress. Crit Rev Biochem Mol Biol. 2017;52(2):220–233. doi:10.1080/10409238.2016.1276882
17. Zhang Q, Wang W, Zhou Q, et al. Roles of circRNAs in the tumour microenvironment. Mol Cancer. 2020;19(1):14. doi:10.1186/s12943-019-1125-9
18. Xu H, Wang C, Song H, Xu Y, Ji G. RNA-Seq profiling of circular RNAs in human colorectal cancer liver metastasis and the potential biomarkers. Mol Cancer. 2019;18(1):8. doi:10.1186/s12943-018-0932-8
19. Hao S, Cong L, Qu R, Liu R, Zhang G, Li Y. Emerging roles of circular RNAs in colorectal cancer. Onco Targets Ther. 2019;12:4765–4777. doi:10.2147/OTT.S208235
20. Yin Y, Long J, He Q, et al. Emerging roles of circRNA in formation and progression of cancer. J Cancer. 2019;10(21):5015–5021. doi:10.7150/jca.30828
21. Wan L, Zhang L, Fan K, Cheng ZX, Sun QC, Wang JJ. Circular RNA-ITCH suppresses lung cancer proliferation via inhibiting the Wnt/β-Catenin pathway. Biomed Res Int. 2016;2016:1579490. doi:10.1155/2016/1579490
22. Yu Q, Liu P, Han G, Xue X, Ma D. CircRNA circPDSS1 promotes bladder cancer by down-regulating miR-16. Biosci Rep. 2020;40:1. doi:10.1042/BSR20191961
23. Ouyang Y, Li Y, Huang Y, et al. CircRNA circPDSS1 promotes the gastric cancer progression by sponging miR-186-5p and modulating NEK2. J Cell Physiol. 2019;234(7):10458–10469. doi:10.1002/jcp.27714
24. Pretzsch E, Bosch F, Neumann J, et al. Mechanisms of metastasis in colorectal cancer and metastatic organotropism: hematogenous versus peritoneal spread. J Oncol. 2019;2019:7407190. doi:10.1155/2019/7407190
25. Francescangeli F, De Angelis ML, Zeuner A. Dietary factors in the control of gut homeostasis, intestinal stem cells, and colorectal cancer. Nutrients. 2019;11:12. doi:10.3390/nu11122936
26. Liu J, Li D, Luo H, Zhu X. Circular RNAs: the star molecules in cancer. Mol Aspects Med. 2019;70:141–152. doi:10.1016/j.mam.2019.10.006
27. Welden JR, Stamm S. Pre-mRNA structures forming circular RNAs. Biochim Biophys Acta Gene Regul Mech. 2019;1862(11–12):194410. doi:10.1016/j.bbagrm.2019.194410
28. Boussios S, Ozturk MA, Moschetta M, et al. The developing story of predictive biomarkers in colorectal cancer. J Pers Med. 2019;9:1. doi:10.3390/jpm9010012
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.