Back to Journals » OncoTargets and Therapy » Volume 13
circKRT7-miR-29a-3p-COL1A1 Axis Promotes Ovarian Cancer Cell Progression
Authors An Q, Liu T, Wang M, Yang Y, Zhang Z, Lin Z, Yang B
Received 20 April 2020
Accepted for publication 28 July 2020
Published 9 September 2020 Volume 2020:13 Pages 8963—8976
DOI https://doi.org/10.2147/OTT.S259033
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Takuya Aoki
Qiang An, Ting Liu, Ming-yang Wang, Yu-jia Yang, Zhen-dong Zhang, Zhen-jiang Lin, Bing Yang
Department of Gynecology, Affiliated Hospital of Zunyi Medical University, Zunyi, Guizhou 563000, People’s Republic of China
Correspondence: Bing Yang
Department of Gynecology, Affiliated Hospital of Zunyi Medical University, Dalian Road, Zunyi City, Guizhou Province 563003, People’s Republic of China
Tel +86-851-28608162
Email [email protected]
Background: Circular RNA (circRNA) has emerged as an important regulator in the progression of human diseases. However, the role of circRNAs in ovarian cancer remains largely unknown.
Materials and Methods: DNA sequencing and PCR were used to identify the existence and expression of circKRT7. The targeting relationship between circKRT7/miR-29a-3p and miR-29a-3p/COL1A1 was verified by fluorescence reporter assay. In vitro, colony formation, transwell and wound healing assay were used to detect the effects of circKRT7 and miR-29a-3p on the proliferation, migration and invasion ability of ovarian cancer cells. In vivo, xenograft tumor model was performed to validate the role of circKRT7 and miR-29a-3p in tumor growth.
Results: We found that circKRT7 can promote the proliferation and metastasis of ovarian cancer cells by absorbing miR-29a-3p, which leads to the up-regulation of COL1A1. In vitro, knock-down of circKRT7 can inhibit the migration and invasion of ovarian cancer cells. This effect of circKRT7 is achieved by adsorbing miR-29a-3p and subsequently COL1A1 release. In vivo experiments, the reduction of circKRT7 expression can also slow tumor growth, and this inhibition was partly counteracted after miR-29a-3p repression.
Conclusion: Overall, circKRT7 promotes EMT-related cell progression by absorbing miR-29a-3p in ovarian cancer. This suggests the crucial role of circular RNA in the malignant evolution in cancer.
Keywords: ovarian cancer, KRT7, circular RNA, EMT
Introduction
Ovarian cancer is the fifth leading cause of cancer mortality in women. More than 200,000 people are diagnosed and about half of them die each year.1,2 One of the causes of high ovarian cancer mortality is that most patients have metastasized when diagnosed.3,4 In addition to BRCA1 and BRCA2 mutations increasing the risk of ovarian cancer,5,6 the occurrence of EMT was considered to be one of the main factors of tumor metastasis. Accumulating evidence suggested that matrix stiffness and tumor cell-extracellular matrix (ECM) interaction play an important role in regulating tumor cell invasion, metastasis and EMT progression.7
Keratin, as a component of epithelial cells, is the main defense of cells against stress and damage.8,9 It is not only important for normal tissue function, but also participates in the pathological and physiological processes of a series of diseases. As a member of the keratin family, KRT7 has also been found to be involved in the malignant evolution of colon, ovarian, and gastric cancer.10–12 Besides forming KRT7 mRNA, transcripts from the same genome can also form non-coding RNA, including lncRNAs, and circRNAs. KRT7-AS as lncRNA can promote KRT7 protein expression. In colon cancer and gastric cancer, KRT7-AS promotes the malignant evolution of tumors by up-regulating the expression of KRT7.12,13 However, so far, the role of KRT7 circular RNA in ovarian cancer is poorly understood.
Here we investigated the biological function of circKRT7 in ovarian cancer. We found that circKRT7 is abnormally highly expressed in ovarian cancer patients. In OVCAR3 and ES-2 cells, knocked-down of circKRT7 inhibited the proliferation and invasion of ovarian cancer cells. In vivo, the repression of circKRT7 also inhibited tumor growth in animals. In addition, we found that circKRT7 promoted the progression of ovarian cancer by adsorbing miR-29a-3p and further increasing COL1A1 protein level. These tests demonstrate the importance of circKRT7 in the EMT-related progression of ovarian cancer malignancy and suggest the possibility of circRNA as a tumor diagnostic and therapeutic target.
Materials and Methods
Cell Lines and Clinical Specimens
Ovarian cancer cell lines, SKOV3, ES-2, were obtained from Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (Shanghai, China). CoC1, Caov-3, and Caov-4 cells were obtained from Cell Resource Center of Chinese Academy of Medical Sciences (Beijing, China). All cells have been identified by STR before purchase. Meanwhile, we have performed a Mycoplasma test every 6 weeks and confirm that the cells are not contaminated. Cells were cultured in RPMI-1640 or DMEM medium with 10% FBS in a 5% CO2 humidified incubator at 37 °C. Ten fresh tumor tissues with adjacent normal tissues were collected immediately after surgery from patients in Affiliated Hospital of Zunyi Medical University. A written informed consent was obtained from each patient. Ethical approval from the Affiliated Hospital of Zunyi Medical University was received before experiments and studies were performed in accordance with the Declaration of Helsinki.
Scanning Electron Microscopy (SEM)
ES-2 and SKOV3 cells were transfected with miR-29a-3p mimics or circKRT7 Interference plasmid. 48h later, cells were fixed with glutaraldehyde, dehydrated in acetone/isoamyl acetate (1:1), and dried with a gradient concentration of acetonitrile. Then, the treated cells were sprayed with gold and treated with a scanning electron microscope (JEOL 6000, Japan) to record the morphological changes of the cells. Each experiment was performed in triplicate.
Invasion Assay
Cell invasion assay was performed using a 8μm Transwell chamber (Corning, USA).The upper chamber was coated with Matrigel (BD Biosciences) and added serum-free medium. At 48 hours after transfection, cells were trypsinized and added to the top chamber at a final concentration of 1×105 cells per well. The lower chamber medium contained 10% FBS. These cells were then incubated at 37°C for 24 hours. After removing the non-invasive cells, cells on the bottom of the chambers were fixed in 4% paraformaldehyde and stained with 0.1% crystal violet. Cells that invaded into the bottom surface were counted in at least five random fields. Each experiment was performed in triplicate.
Immunohistochemistry
Fresh tissue was embedded in paraffin and cut into 4-μm-thick sections. After heat treatment in a microwave oven and blocking the antigen with a 3% H2O2 solution, the sections were incubated with the following primary antibody at 4°C overnight, COL1A1 (Affinity, China, 1:50), E-cadherin (cell signaling, USA, 1:200), vimentin (Santa Cruz, USA, 1:100). After incubation with labeled secondary antibody (Affinity, China), the signal was detected by DAB method (Beyotime, China). Immunohistochemical staining scores were evaluated by two pathologists. Staining scores were evaluated as follows: no positive cells scored 0; positive cells scored 1–3 for yellow, light brown, and dark brown staining, respectively. Each experiment was performed in triplicate.
RNA Extraction and Real-Time PCR Analysis
Total RNA was isolated from cells and fresh tissues using TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. The obtained RNA is divided into three equal parts. One of them was treated with RNase R to remove linear RNA for circKRT7 detection, the other was used to detect miRNA, and the remaining part was used to detect the expression of internal reference. For circRNA detection, RNase R is used to remove linear RNA. With or without 3 U/μg of RNase R (Epicentre Technologies, USA), 2 μg of total RNA was treated at 37°C for 30 minutes, and then the treated RNA was purified using the RNeasy MinElute Cleanup Kit (Qiagen, Germany). Random primers were used to synthesize cDNA from the obtained RNA, and QPCR was performed on the LightCycler480® sequence detector (Roche, Switzerland) and Fast SYBR Green Master Mix kit (Tiangen). The PCR program is as follows: 94°C 5min; 94°C 30s, 58°C 30s, 72°C 30s for 35 cycles; 72°C 10min. For the detection of miRNA, we used the specific primers of miR29a-3p and U6 to synthesize cDNA and amplified the obtained cDNA as a template. The PCR program is as follows: 94°C 5min; 94°C 30s, 56°C 30s, 72°C 30s for 30 cycles; 72°C 10min. For the detection of GAPDH mRNA, we used Oligo dT to synthesize cDNA from the total RNA that had not been treated with RNase R, and then amplified the cDNA as a template. The PCR program is as follows: 94°C 5min; 94°C 30s, 56°C 30s, 72°C 40s for 25 cycles; 72°C 10min. Data were quantified using a 2-ΔΔct quantification method to analyze the results. All primer sequences are as follows: circKRT7-F: 5ʹ-CTGAGGTCAAGGCGCAGTAT-3ʹ, circKRT7-R: 5ʹ-TTGCTCATGTAGGCAGCATC-3ʹ, miR-29a-3p-RT: 5ʹ-GTCGTATCCAGTGCAGGGTCCGAGGTGCACTGGATACGACC TGAACAC-3ʹ, miR-29a-3p-F: 5ʹ-TGCGGACTGATTTCTTTTGG-3ʹ, miR-29a-3p-R: 5ʹ-CCAGTGCAGGGTCCGAGGT-3ʹ, U6-RT: 5ʹ-GTCGTATCCAGTGCAGGGTCCGAGG TGCACTGGATACGACAAAATATGGAAC-3ʹ, U6-F: 5ʹ-TGCGGGTGCTCGCTTCGGCA GC-3ʹ, U6-R: 5ʹ-CCAGTGCAGGGTCCGAGGT-3ʹ. GAPDH-F: 5ʹ-GGGAAACTGTGGC GTGAT-3ʹ, GAPDH-R: 5ʹ-GTGGTCGTTGAGGGCAAT-3ʹ. Each experiment was performed in triplicate.
Luciferase Reporter Assay
The dual-luciferase reporter experiment was used to verify the targeting between circKRT7 and miR-29a-3p and COL1A1 and miR-29a-3p. The wild-type or mutant circKRT7 and COL1A1 3ʹUTR sequences containing miR-29-a-3p binding sites were inserted into the pmirGLO vector. ES-2 cells were seeded into 96-well plates. After the cells grew to the logarithmic growth phase, the dual-luciferase reporter plasmid was co-transfected into the cells with miR-29-a-3p or control mimics. After 48 hours, cells were lysed with 100 μL of lysis buffer per well according to the instructions of Dual-luciferase reporter gene detection kit (Beyotime, China). Then, lysis was centrifuged at 10,000–15,000g for 3–5 minutes and supernatant was used for detection of firefly luciferase and Renilla luciferase activities. Each experiment was performed in triplicate.
Plasmid, Oligo and Transfection
In order to construct the circKRT7 interference plasmid, two synthetic hairpin nucleic acids were annealed and inserted into the pENTR/H1/TO vector. The primer sequences are as follows: Top Strand5ʹ-CACCGACCAAGGTACGAAGATGAAACGAATTTCATCTT CGTACCTTGGTC-3ʹ, Bottom Strand:5ʹ-AAAAGACCAAGGTACGAAGA TGAAATTCG TTTCATCTTCGTACCTTGGTC-3ʹ. The 322-base length circKRT7 nucleic acid sequence was synthesized and cloned into pLCDH-ciR vector and sequenced for identification. The antisense oligonucleotide sequence of miR-29a-3p was synthesized and used to block the level of miR-29a-3p. All plasmids and oligo were transfected with lipo8000 (Beyotime Biotechnology, China) into SKOV3 and ES-2 cells according to the manufactures.
Western Blot
Cells were lysed on ice using RIPA lysate (50mM Tris (pH 7.4), 150mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, 500mM EDTA, and a mixture of 100 × PMSF protease inhibitors). Total protein obtained was separated on a 10% SDS-PAGE gel and transferred to a PVDF membrane. After blocking in 5% BSA for 2 hours, membranes were incubated with the following primary antibodies for 2 hours at room temperature: COL1A1 (Affinity, China, 1:50), E-cadherin (cell signaling, USA, 1:1000), vimentin (Santa Cruz, USA, 1:500). Finally, after incubation with HRP-labeled secondary antibody (Affinity, China, 1:2000) at room temperature for 2 hours, the membranes were exposed and protein expression were detected. Each experiment was performed in triplicate.
Immunofluorescence FISH
Cells were seeded onto slides in 24-well plates and incubated overnight. After fixing the cells with 4% paraformaldehyde, cells were washed with PBS for 5 minutes. Alex 488 labeled miR-29a-3p probe and Cy5 labeled circKRT7 probe were used to detect their localization. All procedures were in accordance with the instructions of the original hybridization detection kit (D-0010, EXONBIO, China). Probes are diluted 100-fold with the hybridization solution. After prehybridization, the cells were incubated with the probe-containing hybridization solution at 37°C for 24 hours. Finally adding 20ul DAPI solution and standing for 15 min. Samples were photographed with a confocal microscope (Nikon A1, Japan). Each experiment was performed in triplicate.
Wound Healing Assay
Transfected cells were seeded in 24-well plates containing 10% FBS culture medium at a final concentration of 5x105 cells per well. Twenty-four hours later, when the cell density reached about 90, the cell layer was scraped with a pipette tip to form a straight linear wound. After removing suspended cells with PBS, the culture was grown by adding complete medium. After 48 hours, a picture of the wound area was taken under a microscope. Each experiment was performed in triplicate.
Colony Formation Assay
Cells were seeded into a 25-cm2 culture flask. When the cells entered the logarithmic growth phase, the cells were transfected with exogenous plasmids and oligos. After culture for 48 hours, the culture medium was removed and the cells were treated with trypsin into single cells, and then seeded into 6-well plates at a final concentration of 1000 cells per well. The cells were placed in a 5% CO2 environment at 37°C for 15 days, and the medium was changed every 3 days. After 15 days, cells were fixed with absolute ethanol and stained with 1% crystal violet for 10 min. After washing with PBS and drying, the colonies of the cells were counted. Each experiment was performed in triplicate.
In vivo Experiment
Eighteen Five-week-old BALB/c mice were purchased from Charles River (Beijing China), and mice were randomly divided into three groups. Untreated cells and stable cell lines knocking down circKRT7 were inoculated subcutaneously with 5 × 106 cells per mouse. Tumor size was measured every 3 days. Tumor volume was calculated using the formula: Tumor volume = (length×width2)/2. When the tumor size reached 50 to 100 mm3, miR-29a-3p inhibitor was intratumorally injected into circKRT7+ 29a-3p inhibitor groups. Thirty-one days after injection, all animals were euthanized through the intravenous injection of barbiturate at a final concentration of 100 mg/kg. Then, the solid tumors were harvested from the mice by surgery. All tissues were fixed in 4% formalin and embedded in paraffin for H&E histological and immunohistochemical staining. Ethical approval was obtained from Zunyi Medical University prior to the commencement of the study. All animal experiments were performed in accordance with the ethical standards of the Institutional Animal Care and Use Committee (IACUC) at Zunyi Medical University for the welfare of the laboratory animals.
Statistical Analysis
All statistical analyses were performed using SPSS 19.0 (SPSS, Chicago, IL, USA). Significant differences among groups were compared using Student’s t-test. Comparisons among three or more groups were conducted using ANOVA with Dunnett’s post-test. The correlation between KRT7 expression and clinicopathological factors was estimated by Fisher’s exact test. Statistical significance was considered at P < 0.05 and labeled with *.
Results
Highly Expressed circKRT-7 Positively Correlates with Tumor Growth in Ovarian Cancer
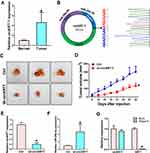
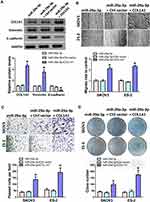
Our previous research found that KRT7 was abnormally highly expressed in ovarian cancer and plays a role in the progression of ovarian cancer. Here we further study the biological function of circular RNA of KRT7 in ovarian cancer. The prediction results of circbank (http://www.circbank.cn/) and circbase (http://www.circbase.org/) databases showed that circKRT7 (hsa_circ_0026360, 322nt) contains exons 3, 4, 5 and the exon 3 and exon 5 are spliced together. First, we verified the expression of circKRT7 in 10 ovarian cancer tissues and paired adjacent tissues using qRT-PCR. The results showed that the expression of circKRT7 in ovarian cancer tissues was higher than that of normal tissues (Figure 1A). After confirming circKRT7 through DNA sequencing, we predicted the miRNAs adsorbed by circKRT7 and found multiple miR-29a-3p binding sites (Figure 1B). ES-2 cells were transfected with the circKRT7 inhibitory plasmid pENTR/H1/sh-circKRT7 and obtained stable cell lines. We inoculated subcutaneously with 5×106 cells per mouse. The same amount of cells transfected with the control plasmid pENTR/H1/TO was inoculated into nude mice as a control. After 30 days, tumor tissues were obtained and the expressions of circKRT7 and miR-29a-3p were detected. The results showed that tumor growth was inhibited after knocking down circKRT7 (Figure 1C and D). In tumors with circKRT7 repression, the expression of miR-29a-3p was increased (Figure 1E and F). Resistance to digestion by RNase R exonuclease further confirmed that this RNA species is circular (Figure 1G). This suggests that the biological function of circKRT7 in ovarian cancer may be mediated by miR-29a-3p.
Down-Regulation of circKRT7 Inhibits Proliferation and Invasion of Ovarian Cancer Cells
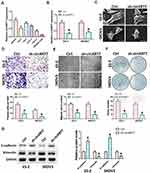
We first detected the expression of circKRT7 in 5 kinds of ovarian cancer cells, SKOV3, CoC1, ES-2, Caov-3, Caov-4 (Figure 2A). Then, ES-2 and SKOV3 cells with the highest expression of circKRT7 were selected to knock down circKRT7 and detect its effect on the cell phenotype. After detection of the decreasing of circKRT7 by PCR (Figure 2B), electron microscopy was performed and found that lamellar pseudopods increased to limit cell migration (Figure 2C). Meanwhile, we used transwell and wound healing assay to detect the cell invasion and migration ability, respectively. The results show that knocking down circKRT7 could inhibit cell invasion (Figure 2D) and migration (Figure 2E). Colony formation results indicated that circKRT7 knocked down also inhibited cell proliferation (Figure 2F). In addition, we also used Western blot to detect the expression of EMT-related markers. The results showed that E-cadherin expression was up-regulated, while vimentin expression was reduced (Figure 2G).
miR-29a-3p Adsorbed by circKRT7 While Targeting COL1A1
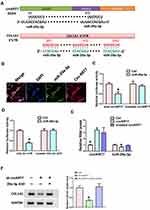
In order to verify the targeting relationship between circKRT7 and miR-29a-3p, we first used the circbank database to predict their binding sites. Next, we used the Targetscan database (www.targetscan.org) to predict the binding of miR-29a-3p and COL1A1 (Figure 3A). In order to verify whether circKRT7 adsorbed miR-29a-3p, immunofluorescence was performed and found that circKRT7 and miR-29a-3p co-localized in ES-2 cells (Figure 3B). Then, we inserted the wild and mutated binding sequence of miR-29a-3p in circKRT7 to the luciferase reporter plasmid, which was co-transfected with control mimics or miR-29a-3p mimics. The results showed that miR-29a-3p can bind wild-type circKRT7 and inhibit luciferase activity, but had weak binding to mutant binding sequences (Figure 3C). We next verified the targeting relationship between miR-29a-3p and COL1A1 using the luciferase reporter assay. The results show that miR-29a-3p can indeed bind to COL1A1 and inhibit its translation (Figure 3D). In addition, we overexpress wild-type circKRT7 and mutant circKRT7, in which miR-29a-3p binding sites were mutated. We observed that miR-29a-3p expression was only inhibited in wild circKRT7-overexpressing cells (Figure 3E). Moreover, Western blot analysis was performed to detect COL1A1 protein level after transfected with circKRT7 shRNA alone or co-transfected with miR-29a-3p ASO. The results showed that the expression of COL1A1 was down-regulated after knocking down circKRT7, but ASO could reverse the inhibition of sh-circKRT7 (Figure 3F).
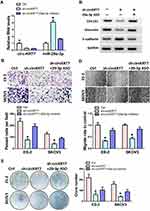
Overexpression of miR-29a-3p Inhibits Ovarian Cancer Cell Invasion and Proliferation
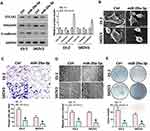
We transfected miR-29a-3p mimics into ovarian cancer cells SKOV3 and ES-2 and then detected the expression of COL1A1, E-cadherin and vimentin. Results showed that the expressions of COL1A1 and vimentin expression were suppressed after miR-29a-3p mimics treatment, while E-cadherin expression was up-regulated (Figure 4A). The morphology of cells was also observed to be changed by an electron microscope (Figure 4B). At the same time, we used transwell and wound healing tests to detect cell invasion and migration ability. Results showed that over-expression of miR-29a-3p can inhibit ovarian cancer cell invasion and migration (Figure 4C and D). Moreover, colony formation assay indicated that over-expression of miR-29a-3p could also repress cell proliferation (Figure 4E).
COL1A1 Counteracts the Inhibitory Effect of miR-29a-3p in Ovarian Cancer Cells
To further confirm the targeting relationship between miR-29a-3p and COL1A1, we overexpressed COL1A1 and transfected control vectors in miR-29a-3p pre-transfected ES-2 cells. Western blot results showed that compared with the control group, the protein levels of COL1A1 and vimentin were up-regulated after overexpression of COL1A1, while the expression of E-cadherin was down-regulated (Figure 5A). Then, we performed transwell, wound healing and colony formation assay to detect changes in cell invasion, migration and proliferation ability. The results showed that overexpression of COL1A1 can indeed reverse the inhibitory effect of miR-29a-3p on cell migration (Figure 5B), invasion (Figure 5C) and proliferation (Figure 5D) ability to a certain extent.
miR-29a-3p Can Partially Reverse the Function of circKRT7 in Ovarian Cancer Cells
To verify that the function of circKRT7 in ovarian cancer was mediated by miR-29a-3p, we performed a rescue experiment. CircKRT7 was knocked down in ovarian cancer cells, and then miR-29a-3p inhibitor was transfected to inhibit the increase of miR-29a-3p caused by circKRT7 downregulation. After detecting the expressions of circKRT7 and miR-29a-3p (Figure 6A), Western blot test was used to detect the expression of COL1A1, E-cadherin and vimentin in ES-2 cells. The results showed that inhibition of circKRT7 could release miR-29a-3p, which caused the down-regulation of COL1A1, vimentin and E-cadherin up-regulation. After blocking with miR-29a-3p antisense oligonucleotides, the expression of COL1A1 and vimentin was restored (Figure 6B). We then used transwell, wound healing and colony formation experiments to verify whether miR-29a-3p ASO could counteract the effect of cicKRT7 knock-down. Results suggested that inhibition of miR-29a-3p could indeed restore the cell invasion (Figure 6C), migration (Figure 6D) and proliferation ability (Figure 6E) that inhibited by circKRT7 down-regulation in ES-2 and SKOV3 cells.
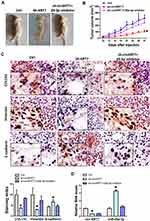
Suppression of circKRT7 Inhibits Ovarian Cancer Tumorigenesis in vivo
The above results have initially demonstrated the positive role of circKRT7 in ovarian cancer cells. Here we further confirm whether the effect of circKRT7 on ovarian cancer was mediated by miR-29a-3p in vivo. We subcutaneously inoculated ES-2 cells with stable low expression of circKRT7 in nude mice, which were evenly divided into 3 groups of 6 mice each. When the tumor reached 2 mm in diameter, we injected miR-29a-3p inhibitor into the tumor. Tumor size was regularly monitored. Tumor was harvested after euthanasia and found that tumor growth was inhibited after circKRT7 was knocked down, while inhibition of miR-29a-3p could reverse this role of circKRT7 (Figure 7A and B). After fresh solid tumors were embedded and cut into 4um sections, we used immunohistochemistry to detect the expression changes of COL1A1 and EMT marker. The results showed that the protein expression of COL1A1 and vimentin was inhibited, while the expression of adhesion protein E-cadherin was up-regulated after circKRT7 was knocked down. Similarly, miR-29a-3p inhibitor could restore the expression of COL1A1 and vimentin in the circKRT7 low expression group, while inhibiting the expression of E-cadherin (Figure 7C). The expressions of circKRT7 and miR-29a-3p were also detected by qRT-PCR (Figure 7D).
Discussion
With the development of bioinformatics and high-throughput sequencing technology, more and more circRNAs are discovered and concerned. CircRNA is conserved among species and is stable in blood, saliva, and exosomes. These characteristics may enable circRNA to be used in the diagnosis of diseases, including cancer. Although a lot of research is focusing on circRNA, many questions remain unanswered. CircKRT7 is derived from KRT7 gene, which is frequent findings in multiple cancers and constitutes a prognostic factor. However, no research has focused on circKRT7 and its biological role in ovarian cancer. In this study, we found that circKRT7 is abnormally highly expressed in ovarian cancer. In order to verify the function and mechanism of circKRT7 in ovarian cancer, we used cell and animal experiments to verify. It was found that overexpression of circKRT7 can block the level of miR-29a-3p and promote the proliferation and invasion of ovarian cancer cells. As a tumor suppressor, miR-29a-3p has been shown to inhibit the progression of thyroid tumors, hepatocellular carcinoma, and glioma.14–17 In addition, circFOXO3 could promote prostate cancer progression by sponging miR-29a-3p.18 Here we found that miR-29a-3p can inhibit the expression of COL1A1, and circKRT7 can precisely release COL1A1 by adsorbing miR-29a-3p, which ultimately urges the proliferation and metastasis of ovarian cancer cells.
Most malignant tumors derived from epithelial tissues. Epithelial tissue-derived tumors endow mesenchymal properties through EMT pathway and become more malignant and metastatic.19,20 Here we find that the expression of vimentin was significantly up-regulated after over-expression of circKRT7, while the expression of E-cadherin was suppressed. This means that cell adhesion was reduced and exercise ability was enhanced. We also found similar results in animal experiments, that is, circKRT7 can promote vimentin and inhibit the expression of E-cadherin, so that tumor cells have stronger metastatic ability. These results indicated that the promotion effect of circKRT7 on ovarian cancer was probably through the EMT pathway.
ECM is crucial for EMT and tumor metastasis, and collagen is one of the main components of ECM.21,22 As a member of the collagen I family, the relationship between COL1A1 and tumor cell proliferation and invasion has been reported in many cancers, such as pancreatic cancer, hepatocellular carcinoma, and gastric cancer.23–25 In this study, we found that miR-29a-3p can bind to COL1A1 and inhibit its protein expression, while circKRT7 can restore the expression of COL1A1 by adsorbing miR-29a-3p, thereby promoting metastasis of ovarian cancer cells. Of course, whether this specific expression of circKRT7 is caused by genetics or the tumor microenvironment remains to be further studied.
In summary, we have verified the important role of KRT7 circular RNA in the malignant progression of ovarian cancer. The high expression of circKRT7 in ovarian cancer patients was first confirmed. In addition, circKRT7 has been shown to promote the proliferation and metastasis of ovarian cancer cells by absorbing miR-29a-3p through EMT pathway. These results not only explain the biological function of circKRT7, but also provide some theoretical support for the clinical diagnosis and treatment of ovarian cancer.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Torre LA, Trabert B, DeSantis CE, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68(4):284–296. doi:10.3322/caac.21456
2. Eisenhauer EA. Real-world evidence in the treatment of ovarian cancer. Ann Oncol. 2017;28(suppl_8):viii61–viii65. doi:10.1093/annonc/mdx443
3. Smith-Bindman R, Poder L, Johnson E, Miglioretti DL. Risk of malignant ovarian cancer based on ultrasonography findings in a large unselected population. JAMA Intern Med. 2019;179(1):71–77. doi:10.1001/jamainternmed.2018.5113
4. Pignata S, C Cecere S, Du Bois A, Harter P, Heitz F. Treatment of recurrent ovarian cancer. Ann Oncol. 2017;28(suppl_8):viii51–viii56. doi:10.1093/annonc/mdx441
5. George A, Kaye S, Banerjee S. Delivering widespread BRCA testing and PARP inhibition to patients with ovarian cancer. Nat Rev Clin Oncol. 2017;14(5):284–296. doi:10.1038/nrclinonc.2016.191
6. Hoskins PJ, Gotlieb WH. Missed therapeutic and prevention opportunities in women with BRCA-mutated epithelial ovarian cancer and their families due to low referral rates for genetic counseling and BRCA testing: a review of the literature. CA Cancer J Clin. 2017;67(6):493–506. doi:10.3322/caac.21408
7. Kaur A, Ecker BL, Douglass SM, et al. Remodeling of the collagen matrix in aging skin promotes melanoma metastasis and affects immune cell motility. Cancer Discov. 2019;9(1):64–81. doi:10.1158/2159-8290.CD-18-0193
8. Ehrlich F, Fischer H, Langbein L, et al. Differential evolution of the epidermal keratin cytoskeleton in terrestrial and aquatic mammals. Mol Biol Evol. 2019;36(2):328–340.
9. Guldiken N, Kobazi Ensari G, Lahiri P, et al. Keratin 23 is a stress-inducible marker of mouse and human ductular reaction in liver disease. J Hepatol. 2016;65(3):552–559. doi:10.1016/j.jhep.2016.04.024
10. Chen S, Su T, Zhang Y, et al. Fusobacterium nucleatum promotes colorectal cancer metastasis by modulating KRT7-AS/KRT7. Gut Microbes. 2020;1–15. doi:10.1080/19490976.2020.1782158
11. Wang P, Magdolen V, Seidl C, et al. Kallikrein-related peptidases 4, 5, 6 and 7 regulate tumour-associated factors in serous ovarian cancer. Br J Cancer. 2018;119(7):1–9. doi:10.1038/s41416-018-0260-1
12. Huang B, Song JH, Cheng Y, et al. Long non-coding antisense RNA KRT7-AS is activated in gastric cancers and supports cancer cell progression by increasing KRT7 expression. Oncogene. 2016;35(37):4927–4936. doi:10.1038/onc.2016.25
13. Chen S, Su T, Zhang Y, et al. Fusobacterium nucleatum promotes colorectal cancer metastasis by modulating KRT7-AS/KRT7. Gut Microbes. 2020;11(3):511–525. doi:10.1080/19490976.2019.1695494
14. Ma Y, Sun Y. miR-29a-3p inhibits growth, proliferation, and invasion of papillary thyroid carcinoma by suppressing NF-kappaB signaling via direct targeting of OTUB2. Cancer Manag Res. 2019;11:13–23. doi:10.2147/CMAR.S184781
15. Wang X, Liu S, Cao L, et al. miR-29a-3p suppresses cell proliferation and migration by downregulating IGF1R in hepatocellular carcinoma. Oncotarget. 2017;8(49):86592–86603. doi:10.18632/oncotarget.21246
16. Shao NY, Wang DX, Wang Y, et al. MicroRNA-29a-3p downregulation causes Gab1 upregulation to promote glioma cell proliferation. Cell Physiol Biochem. 2018;48(2):450–460. doi:10.1159/000491776
17. Catanzaro G, Sabato C, Russo M, et al. Loss of miR-107, miR-181c and miR-29a-3p promote activation of notch2 signaling in pediatric high-grade gliomas (pHGGs). Int J Mol Sci. 2017;18(12):2742. doi:10.3390/ijms18122742
18. Kong Z, Wan X, Lu Y, et al. Circular RNA circFOXO3 promotes prostate cancer progression through sponging miR-29a-3p. J Cell Mol Med. 2020;24(1):799–813. doi:10.1111/jcmm.14791
19. Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29(3):212–226. doi:10.1016/j.tcb.2018.12.001
20. Shibue T, Weinberg RA. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat Rev Clin Oncol. 2017;14(10):611–629. doi:10.1038/nrclinonc.2017.44
21. Gilkes DM, Semenza GL, Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer. 2014;14(6):430–439. doi:10.1038/nrc3726
22. Wei SC, Fattet L, Tsai JH, et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway. Nat Cell Biol. 2015;17(5):678–688. doi:10.1038/ncb3157
23. Chakravarthy D, Munoz AR, Su A, et al. Palmatine suppresses glutamine-mediated interaction between pancreatic cancer and stellate cells through simultaneous inhibition of survivin and COL1A1. Cancer Lett. 2018;419:103–115. doi:10.1016/j.canlet.2018.01.057
24. Ma HP, Chang HL, Bamodu OA, et al. Collagen 1A1 (COL1A1) is a reliable biomarker and putative therapeutic target for hepatocellular carcinogenesis and metastasis. Cancer (Basel). 2019;11(6):786. doi:10.3390/cancers11060786
25. Shi Y, Duan Z, Zhang X, Zhang X, Wang G, Li F. Down-regulation of the let-7i facilitates gastric cancer invasion and metastasis by targeting COL1A1. Protein Cell. 2019;10(2):143–148. doi:10.1007/s13238-018-0550-7
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.