Back to Journals » Clinical and Experimental Gastroenterology » Volume 14
Chronic Antibiotic-Refractory Pouchitis: Management Challenges
Authors Outtier A, Ferrante M
Received 1 February 2021
Accepted for publication 6 May 2021
Published 14 June 2021 Volume 2021:14 Pages 277—290
DOI https://doi.org/10.2147/CEG.S219556
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Andreas M. Kaiser
An Outtier, Marc Ferrante
Department of Gastroenterology and Hepatology, University Hospitals Leuven, KU Leuven, Leuven, Belgium
Correspondence: Marc Ferrante
Department of Gastroenterology and Hepatology, University Hospitals Leuven, Herestraat 49, Leuven, 3000, Belgium
Tel +32 16 34 42 18
Fax +32 16 33 07 23
Email [email protected]
Background: Pouchitis is the most common long-term complication in patients with ulcerative colitis who underwent restorative proctocolectomy with ileal pouch-anal anastomosis. The incidence of acute pouchitis is 20% after 1 year and up to 40% after 5 years. Chronic antibiotic-refractory pouchitis develops in approximately 10% of patients.
Aim: To present a narrative review of published literature regarding the management of chronic antibiotic-refractory pouchitis.
Methods: Current relevant literature was summarized and critically evaluated.
Results: Clear definitions should be used to classify pouchitis into acute versus chronic, and responsive versus dependent versus refractory to antibiotics. Before treatment is started for chronic antibiotic-refractory pouchitis, secondary causes should be ruled out. There is a need for validated scoring systems to measure the severity of the disease. Because chronic antibiotic-refractory pouchitis is a rare condition, only small studies with often a poor study design have been performed. Treatments with antibiotics, aminosalicylates, steroids, immunomodulators and biologics have shown to be effective and safe for chronic antibiotic-refractory pouchitis. Also, treatments with AST-120, hyperbaric oxygen therapy, tacrolimus enemas, and granulocyte and monocyte apheresis suggested some efficacy.
Conclusion: The available data are weak but suggest that therapeutic options for chronic antibiotic-refractory pouchitis are similar to the treatment strategies for inflammatory bowel diseases. However, randomized controlled trials are warranted to further identify the best treatment options in this patient population.
Keywords: chronic antibiotic-refractory pouchitis, inflammatory bowel disease, biologics
Introduction
Pouchitis is the most common long-term complication in patients with ulcerative colitis (UC) and familial adenomatous polyposis (FAP) who underwent restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA). Symptoms may include increased stool frequency and fluidity, rectal bleeding, fecal urgency, incontinence, abdominal cramps, fever and extra-intestinal manifestations.1 Routine laboratory tests may be normal, or they may reveal anemia, an elevated C-reactive protein, electrolyte abnormalities, iron deficiency and vitamin D deficiency.2,3 Fecal calprotectin and lactoferrin appear to be significant predictors of pouchitis.4 A diagnosis of pouchitis can only be made after confirmation on endoscopy and eventual histological examination.
The incidence of acute pouchitis is estimated to be around 20% after 1 year and up to 40% after 5 years. Chronic antibiotic-refractory pouchitis develops in approximately 10% of patients.5 Besides differences in length and type of follow-up, an important reason for variation in incidence is the lack of universally accepted diagnostic criteria for pouchitis.1
Pouchitis can develop, based on the number of episodes and response to antibiotics, from acute antibiotic-responsive (one to three episodes a year) to chronic antibiotic-dependent (four or more antibiotic-responsive episodes a year or need for ongoing antibiotic use). In some patients, the symptoms persist despite a course of more than four weeks of antibiotic therapy (chronic antibiotic-refractory pouchitis or CARP).
In cases where chronic antibiotic-refractory pouchitis is suspected, it is important to rule out other diagnoses, such as a pouch outlet obstruction, strictures, pouch fistula, peripouch inflammation, cuffitis, prepouch ileitis, irritable pouch syndrome or secondary etiologies, such as infections (eg Clostridium difficile and cytomegalovirus), non-steroidal anti-inflammatory drug (NSAID) use, concomitant auto-immune disorders (eg coeliac disease) and pouch ischemia.6 A relook at the colectomy specimen is warranted to rule out a missed diagnosis of Crohn’s disease. Whether a pouch stricture, pouch fistula and prepouch ileitis are sufficient to diagnose Crohn’s disease is still debated.
The etiology of pouchitis is considered to be multifactorial and suggests an abnormal immune response to an imbalanced gut microbiome7 in genetically susceptible individuals8 following triggering events.9 Acute antibiotic-responsive pouchitis is predominately microbially mediated and chronic antibiotic-refractory pouchitis is predominately immune mediated. This may explain why chronic antibiotic-refractory pouchitis is more commonly seen in UC patients than FAP patients.10
Chronic antibiotic-refractory pouchitis is the most challenging form of pouchitis to treat. Because it is a rare condition, only small studies with often a poor study design are performed. Patients may end up therapy resistant and eventually need surgery with a permanent ileostomy.
Risk Factors
Assessing the presence of risk factors could eventually guide prophylaxis for pouchitis. These risk factors include the presence of primary sclerosing cholangitis (PSC)11 and other extra-intestinal manifestations,12 serological markers (IgG4, ANCA, ACCA and Omp),13–15 extensive colitis,16,17 backwash ileitis,18,19 corticosteroid exposure before proctocolectomy,20,21 periproctocolectomy thrombocytosis,20 young age,22–24 male sex,24 construction of a J-pouch,25 postoperative NSAID use12 and non-smoking20 (Table 1). Some genetic mutations (eg NOD2/CARD15 mutation26) are also associated with the development of pouchitis; however, testing is costly, not widely available and thus not routinely performed.
 |
Table 1 Risk Factors of Pouchitis |
Prevention
The use of probiotics (VSL#3,27,28 Lactobacillus rhamnosus GG29 or Clostridium butyricum MIYAIRI30) is suggested for the prophylaxis of antibiotic-dependent pouchitis; however, evidence is not strong and conflicting. Sulfasalazine may be an alternative to more expensive probiotics for the long-term prevention of pouchitis development.31 This should be combined with a diet balanced in fermentable fibers to establish a favorable microbiome. In a prospective cohort study including 172 patients with ileal pouches, lower fruit consumption was correlated with higher rates of pouchitis compared with higher fruit intake.32
Assessing Disease Activity of Pouchitis
When starting a treatment for pouchitis, we need to be able to measure the effect of the treatment in an objective way. That is why we need scoring systems that measure the severity of the disease and can be used for follow-up.
The most commonly used scoring systems are the pouch disease activity index (PDAI)33 (Table 2), including clinical, endoscopic and histological features, and a more simplified modified PDAI (mPDAI),34 not containing the histological criteria. Nevertheless, these scores are not validated and their routine use in clinical practice is rare. Also, these scoring systems do not take into account quality of life indicators that may be significant to the patient.
 |
Table 2 The Pouch Disease Activity Index (PDAI) |
The Oresland score evaluates the functional outcome of the pouch. It includes 11 parameters, evaluating the 24-h stool frequency, nighttime defecation, the urgency of defecation, dyschezia, soiling and the use of pads, perianal skin irritation, dietary restrictions, regular medication for diarrhea and social handicap.35 The value obtained in the Oresland score allows us to classify the pouch its functional outcome into three groups: good (0–4), moderate (5–7) and poor (8–15) (Table 3). Another score was presented by Brandsborg et al evaluating the function of the pouch and subsequently the quality of life of the patient.36 The score takes into account the feeling of incomplete emptying, number of bowel movements, uncontrolled loss of stools, use of anti-diarrheal medication, urgency and the ability to defer the urge to defecate (Table 4). Despite its importance, these scores do not seem to be routinely used in clinical practice yet.
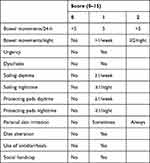 |
Table 3 The Oresland Score |
 |
Table 4 Score Evaluating the Function of the Pouch and Quality of Life Suggested by Brandsborg et al |
Treatment of Acute Pouchitis
Patients with acute pouchitis typically respond to antibiotic therapy. Ciprofloxacin is considered the first-line therapy for acute pouchitis. In a randomized clinical trial by Shen et al, the effectiveness and side effects of ciprofloxacin and metronidazole for treating acute pouchitis were compared.37 Both antibiotics were effective with a significant reduction in PDAI; however, ciprofloxacin produced a greater reduction in the PDAI and a greater improvement in symptom and endoscopy subscores, and was better tolerated than metronidazole. Rifaximin might also be useful in treating acute pouchitis. A randomized placebo-controlled pilot trial showed that clinical remission occurred more frequently in patients treated with rifaximin, however, the difference was not significant.38
Treatment of Chronic Antibiotic-Refractory Pouchitis
Due to the lack of randomized controlled trials, the treatment of chronic antibiotic-refractory pouchitis is largely empirical. Over the years, immunomodulators, biologics and small molecules have been introduced, similar to the treatment strategies for inflammatory bowel diseases, considering chronic antibiotic-refractory pouchitis is predominantly immune mediated.
Antibiotics
Oral vancomycin has been successfully used to induce and maintain remission in PSC-associated chronic antibiotic-refractory pouchitis at the Cleveland Clinic.39,40 Furthermore, it may also improve liver function tests.41–43 There are no published data on the long-term efficacy or safety yet.
Several studies investigated the use of a combination therapy with ciprofloxacin and rifaximin44,45 or tinidazole46 and showed its effectiveness in treating patients with chronic antibiotic-refractory pouchitis (Table 5).
 |
Table 5 Studies Investigating Antibiotics and Aminosalicylates for Chronic Antibiotic-Refractory Pouchitis |
A study by McLaughin et al used fecal coliform sensitivity testing for targeted antibiotic treatment that appears highly effective in treating patients with antibiotic-resistant pouchitis. They recommend that all pouchitis patients who fail standard antibiotic treatment or develop resistance while on long-term antibiotic treatment undergo fecal coliform sensitivity testing to guide antibiotic therapy.47 However, this is still not routinely used in clinical practice.
Aminosalicylates
While aminosalicylates form the basis for the treatment of ulcerative colitis and cuffitis, their efficacy in chronic antibiotic-refractory pouchitis is far from established. Topical and oral mesalazines have been tried in a small cohort of ten patients with chronic antibiotic-refractory pouchitis, demonstrating remission rates of 50%46 (Table 5).
Steroids
Oral and rectal budesonide48–50 and beclomethasone dipropionate51 have been used successfully in inducing remission in chronic antibiotic-refractory pouchitis (Table 6). Nonetheless, prolonged use of steroids is discouraged to prevent serious side effects.
 |
Table 6 Studies Investigating Corticosteroids for Chronic Antibiotic-Refractory Pouchitis |
Immunomodulators
Historically, immunomodulators including azathioprine, mercaptopurine and methotrexate have been used as subsequent therapy for chronic antibiotic-refractory pouchitis. There are, however, little data on the use of immunomodulators for this indication.52,53 The current position of immunomodulators in the treatment algorithm mainly depends on the availability and early access to biologics.
Biologics
Anti-TNF
In 2018, a systematic review with meta-analysis evaluated the efficacy of anti-tumor necrosis factor (TNF) therapy in patients with chronic antibiotic-refractory pouchitis. The rate of short-term clinical remission, evaluated at 8 weeks of induction therapy, was low (10%). However, the rate of long-term clinical remission at 12 months was 37%.54–75 A study from the Leuven group retrospectively assessed infliximab and adalimumab therapy in 23 and 13 chronic antibiotic-refractory pouchitis patients, respectively. At 14 weeks, clinically relevant remission was observed in 43.5% and 38.5% of patients in the infliximab and adalimumab group, respectively. At final follow-up, only 17.4% of patients in the infliximab group remained in clinically relevant remission. Anti-TNF therapy was discontinued in 40.7% of patients due to intolerance or drug reaction76 (Table 7).
 |
Table 7 Studies Investigating Anti-TNF Agents for Chronic Antibiotic-Refractory Pouchitis |
As the majority of UC patients who come to colectomy for refractory colitis nowadays have already been treated with anti-TNF therapy, the treatment of chronic antibiotic-refractory pouchitis with anti-TNF agents can always be hampered due to immunogenicity after a drug holiday. This may ultimately result in infusion reactions, primary non-response or loss of response. Therefore, biologics with another mode of action are probably indicated for the treatment of chronic antibiotic-refractory pouchitis in patients who previously failed both infliximab and adalimumab.
Vedolizumab
In the last few years, the efficacy and safety of vedolizumab has been reported more and more in case series and small observational studies for the treatment of chronic antibiotic-refractory pouchitis.76–80
A retrospective study from the USA included 19 chronic antibiotic-refractory pouchitis patients treated with vedolizumab and reported 32% clinical response after 3 months. Improvement of endoscopic and total mPDAI were seen in 74% of patients.77 The retrospective study from Leuven also included 15 chronic antibiotic-refractory pouchitis patients treated with vedolizumab. Clinically relevant remission was observed in 60% of patients at week 14 and 53.3% of patients at the last follow-up76 (Table 8).
 |
Table 8 Studies Investigating Vedolizumab for Chronic Antibiotic-Refractory Pouchitis |
We eagerly await the results of the EARNEST study (NCT02790138), a placebo-controlled study that evaluated the efficacy and safety of vedolizumab in the treatment of chronic pouchitis, to further decide if vedolizumab can be a beneficial treatment for this condition.
Ustekinumab
Only little is known about the role of ustekinumab in the treatment of chronic antibiotic-refractory pouchitis.
Weaver et al retrospectively described the largest pouchitis cohort of 56 patients treated with ustekinumab; however, only nine patients had chronic antibiotic-refractory pouchitis, while the remaining 47 patients had Crohn’s disease-related complications of the pouch.81 At 6 months, data were only available from six patients and five of them had a clinical response, but none were in clinical remission. Ollech et al performed a retrospective study including 24 patients with chronic antibiotic-refractory pouchitis who received ustekinumab treatment, leading to clinical improvement in 50% of patients, but none were in clinical remission82 (Table 9).
 |
Table 9 Studies Investigating Ustekinumab for Chronic Antibiotic-Refractory Pouchitis |
A prospective Belgian multicenter open-label study (NCT04089345) is being performed to confirm these findings.
Small Molecules
There is only one case report published where a 20-year-old woman was treated with tofacitinib for chronic antibiotic-refractory pouchitis. She had already received treatments with anti-TNF therapy, systemic steroids and vedolizumab, but the response was unsatisfactory. Because the patient refused a permanent ileostomy, tofacitinib was started as a last resort. The therapy led to clinical and endoscopic improvement with a reduced mPDAI from 10 to 3.83 Another case of chronic antibiotic-refractory pouchitis treated with tofacitinib has been reported in an observational study, however its details are unknown.84
A Phase 2 open-label prospective pilot study (NCT04580277) will start at Cedars-Sinai Medical Center to evaluate the effectiveness and safety of tofacitinib in subjects with chronic pouchitis.
Fecal Microbiota Transfer
Fecal Microbiota Transfer (FMT) may be an alternative approach to classical treatment of chronic antibiotic-refractory pouchitis given the role of bacteria in the pathogenesis.
Landy et al conducted a pilot study including eight patients with chronic antibiotic-refractory pouchitis who were treated with one single FMT via nasogastric administration.85 After 4 weeks, two patients had a clinical response (decrease in PDAI with ≥ 3 points) but none achieved clinical remission (PDAI < 7). Stallmach et al conducted an observational study including five patients with chronic antibiotic-refractory pouchitis.86 Administration of FMT was directly into the jejunum during esophagogastroduodenoscopy with an interval of 3–4 weeks. Within 4 weeks after the last FMT, all patients had clinical, endoscopic and histological responses. Selvig et al performed a pilot study including nineteen patients with chronic antibiotic-refractory pouchitis who were treated with a minimum of one FMT delivered via pouchoscopy.87 After 4 weeks, total PDAI, endoscopic PDAI and histologic PDAI did not decrease significantly. However, there was a significant improvement in stool frequency and trend for improvement in abdominal pain. Finally, Herfarth et al performed a placebo-controlled trial of endoscopic and oral FMT in patients with chronic antibiotic-dependent pouchitis.88 Patients were randomized to receive either active or placebo FMT via pouchoscopy followed by daily oral administration for 14 days. All six patients enrolled, failed to respond and needed antibiotic rescue therapy. In the open-label extension study, 1 out of 5 patients achieved antibiotic-free clinical remission (mPDAI < 4).
Although FMT is safe for chronic antibiotic-refractory pouchitis, the current data fail to prove efficacy. Because these data are limited by study heterogeneity, additional studies are required to guide the use of FMT in patients with chronic antibiotic-refractory pouchitis.
Miscellaneous
Several other treatment options were investigated for chronic antibiotic-refractory pouchitis; however, large randomized controlled trials are lacking to provide enough power to the data.
A study by Croagh et al explored the influence of a change in poorly absorbed short-chain carbohydrates (FODMAPs) in the diet, by virtue of their osmotic effect, on the frequency and quality of stool output from an ileal pouch.89 Patients were asked to strictly follow a low FODMAP diet for 6 weeks. No response was seen in patients with pouchitis, but this diet might be efficacious in reducing stool frequency in patients who do not have active pouchitis or other inflammation associated with the pouch. A recently initiated study (NCT04640155) will further investigate whether a low FODMAP diet in patients with chronic antibiotic-refractory pouchitis might improve symptoms and pouch inflammation.
AST-120 is an agent that is comprised of highly adsorptive, porous carbon microspheres with the ability to adsorb small-molecular-weight toxins, inflammatory mediators and harmful bile acids. Shen et al conducted a trial to evaluate the efficacy and safety of AST-120 in patients with active pouchitis.90 Twenty patients were included and received AST-120 2g three times a day for 4 weeks. Clinical response (reduction of PDAI with ≥ 3 points) was seen in 55% of patients and clinical remission (PDAI < 7) in 50% of patients. The agent was well tolerated.
Alicaforsen, an antisense enema to intercellular adhesion molecule-1, can improve the clinical symptoms, endoscopic mucosal appearance and histologic inflammation in patients with chronic antibiotic-refractory pouchitis. Miner et al carried out an open-label study including 12 patients with chronic pouchitis treated with one 240 mg Alicaforsen enema nightly for 6 weeks.91 By week 6, seven of the 12 patients (58%) were in remission (PDAI < 7) with a mean decrease of six points in PDAI. A retrospective study by Greuter et al identified 13 patients treated with at least one dose of Alicaforsen for chronic antibiotic-refractory pouchitis.92 At 2–3 months after therapy, clinical and endoscopic disease activity was significantly reduced. Finally, a randomized, placebo-controlled trial (NCT02525523) was performed including subjects with chronic antibiotic-refractory pouchitis, where patients were treated with the study drug (240 mg Alicaforsen or placebo enema) administered once nightly for 6 weeks. Results were disappointing with no significant reduction in relative stool frequency and no significant difference in endoscopic remission (absence of friability and ulceration).
Hyperbaric oxygen therapy (HBOT) for chronic antibiotic-refractory pouchitis was investigated in a retrospective case series of 28 patients treated with a median number of 30 HBOT sessions.93 There was a significant reduction in the mean clinical mPDAI from 3.19 to 1.91 and mean endoscopic mPDAI from 2.34 to 1.29. Despite minor adverse events, HBOT was well tolerated. A prospective open-label pilot study (NCT03526796) is being performed in China to further investigate this modality.
In a prospective pilot study, ten patients with chronic antibiotic-refractory pouchitis were treated for eight weeks with a tacrolimus enema.94 The mean PDAI decreased significantly from 15.9 to 7.8. Seven patients achieved complete remission of clinical symptoms, nine patients were clinical responders and three patients were in remission (PDAI < 7). No severe adverse events occurred. This small study suggested that the use of topical tacrolimus for the treatment of chronic antibiotic-refractory pouchitis is safe and effective in the short term for clinical symptoms and may have early rescue efficacy.
Granulocyte and monocyte apheresis (GMA) has shown therapeutic efficacy in patients with active UC by modulating systemic and local inflammatory activities. Furthermore, several studies have reported that depleting granulocytes and monocytes by GMA leads to a diminished inflammatory profile in the intestinal mucosa, and remission of clinical symptoms in patients with active UC.95–99 An open-label prospective study was done by Yamamoto et al to evaluate the efficacy of granulocyte and monocyte apheresis for chronic antibiotic-refractory pouchitis.100 Thirteen patients were included and received 10 GMA sessions at 2 sessions/week over five consecutive weeks. Six patients responded to the treatment (reduction of PDAI with ≥ 3 points) but none achieved remission (PDAI < 4). GMA has a good safety profile, but its efficacy appears to be limited in the management of chronic antibiotic-refractory pouchitis. A large controlled study should be conducted to evaluate the efficacy of GMA therapy in patients with pouchitis at an earlier clinical stage, before the disease has become refractory to conventional medical therapy.
Surgery
Surgery may be a last resort for patients with chronic antibiotic-refractory pouchitis who are refractory to all medical treatments. An end ileostomy, with or without pouch excision, will be performed.
Conclusion
Pouchitis can be suspected based on clinical symptoms and laboratory findings, but should be confirmed with endoscopy and histology. Clear definitions should be used to classify pouchitis into acute versus chronic, and responsive versus dependent versus refractory to antibiotics. Before treatment is started for chronic antibiotic-refractory pouchitis, secondary causes should be ruled out. Also, scoring the disease, taking into account the quality of life of the patient, should guide you in choosing the best treatment option for your patient.
Managing patients with chronic antibiotic-refractory pouchitis remains a challenge for the treating gastroenterologist or abdominal surgeon. Because chronic antibiotic-refractory pouchitis is mainly immune mediated, therapeutic options are similar to the treatment strategies for inflammatory bowel diseases. Treatments with antibiotics, aminosalicylates, steroids, immunomodulators and biologics has been shown to be effective for chronic antibiotic-refractory pouchitis. However, randomized controlled trials are warranted to further identify the best treatment options in this patient population.
Abbreviations
CARP, chronic antibiotic-refractory pouchitis; FAP, familial adenomatous polyposis; FODMAP, Fermentable Oligosaccharides, Disaccharides, Monosaccharides And Polyols; GMA, granulocyte and monocyte apheresis; HBOT, hyperbaric oxygen therapy; FMT, Fecal Microbiota Transfer; IPAA, ileal pouch-anal anastomosis; mPDAI, modified pouch disease activity index; NSAID, non-steroidal anti-inflammatory drug; PDAI, pouch disease activity index; PSC, primary sclerosing cholangitis; TNF, tumor necrosis factor; UC, ulcerative colitis.
Acknowledgments
Marc Ferrante is a Senior Clinical Investigator of the Research Foundation Flanders (FWO), Belgium.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval for the version to be published; and agreed to be accountable for all aspects of the work.
Disclosure
Marc Ferrante received financial support for research from AbbVie, Amgen, Biogen, Humira, Janssen, Pfizer, and Takeda; consultancy fees from AbbVie, Boehringer-Ingelheim, Celltrion, Janssen, Lilly, Medtronic, MSD, Pfizer, Sandoz, Takeda and Thermo Fisher; and speakers fees from AbbVie, Amgen, Biogen, Boehringer-Ingelheim, Falk, Ferring, Janssen, Lamepro, MSD, Mylan, Pfizer, Sandoz, Takeda and Truvion Healthcare. The authors report no other conflicts of interest in this work.
References
1. Mahadevan U, Sandborn WJ. Diagnosis and management of pouchitis. Gastroenterology. 2003;124(6):1636–1650. doi:10.1016/S0016-5085(03)00325-1
2. M’Koma AE, Wise PE, Schwartz DA, et al. Prevalence and outcome of anemia after restorative proctocolectomy: a clinical literature review. Dis Colon Rectum. 2009;52:726. doi:10.1007/DCR.0b013e31819ed571
3. Lu H, Lian L, Navaneethan U, Shen B. Clinical utility of C-reactive protein in patients with ileal pouch anal anastomosis. Inflamm Bowel Dis. 2010;16:1678. doi:10.1002/ibd.21239
4. Yamamoto T, Shimoyama T, Bamba T, et al. Consecutive monitoring of fecal calprotectin and lactoferrin for the early diagnosis and prediction of pouchitis after restorative proctocolectomy for ulcerative colitis. Am J Gastroenterol. 2015;110(6):881–887. doi:10.1038/ajg.2015.129
5. Lepistö A, Luukkonen P, Järvinen HJ, et al. Cumulative failure rate of ileal pouchanal anastomosis and quality of life after failure. Dis Colon Rectum. 2002;45:1289–1294. doi:10.1007/s10350-004-6412-9
6. Shen B. Diagnosis and management of postoperative ileal pouch disorders. Clin Colon Rectal Surg. 2010;23(4):259–268. doi:10.1055/s-0030-1268252
7. Hold GL. Role of the gut microbiota in inflammatory bowel disease pathogenesis: what have we learnt in the past 10 years? World J Gastroenterol. 2014;20:1192. doi:10.3748/wjg.v20.i5.1192
8. Lees CW, Barrett JC, Parkes M, et al. New IBD genetics: common pathways with other diseases. Gut. 2011;60:1739–1753. doi:10.1136/gut.2009.199679
9. Ananthakrishnan AN, Bernstein CN, Iliopoulos D, et al. Environmental triggers in IBD: a review of progress and evidence. Nat Rev Gastroenterol Hepatol. 2017;15:39–49. doi:10.1038/nrgastro.2017.136
10. Lohmuller JL, Pemberton JH, Dozois RR, et al. Pouchitis and extraintestinal manifestations of inflammatory bowel disease after ileal pouch-anal anastomosis. Ann Surg. 1990;211:622–627.
11. Wasmuth HH, Tranø G, Endreseth BH, et al. Primary sclerosing cholangitis and extraintestinal manifestations in patients with ulcerative colitis and ileal pouch–anal anastomosis. J Gastrointest Surg. 2010;14:1099–1104. doi:10.1007/s11605-010-1223-x
12. Achkar JP, Al-Haddad M, Lashner B, et al. Differentiating risk factors for acute and chronic pouchitis. Clin Gastroenterol Hepatol. 2005;3:60–66. doi:10.1016/S1542-3565(04)00604-4
13. Shen B, Remzi FH, Nutter B, et al. Association between immune-associated disorders and adverse outcomes of ileal pouch-anal anastomosis. Am J Gastroenterol. 2009;104:655–664. doi:10.1038/ajg.2008.76
14. Singh S, Sharma PK, Loftus EV
15. Ferrante M, Declerck S, Coopmans T, et al. Development of pouchitis following ileal pouch-anal anastomosis (IPAA) for ulcerative colitis: a role for serological markers and microbial pattern recognition receptor genes. J Crohns Colitis. 2008;2(2):142–151. doi:10.1016/j.crohns.2007.10.003
16. Lipman JM, Kiran RP, Shen B, et al. Perioperative factors during ileal pouch-anal anastomosis predict pouchitis. Dis Colon Rectum. 2011;54:311–317. doi:10.1007/DCR.0b013e3181fded4d
17. Hashavia E, Dotan I, Rabau M, et al. Risk factors for chronic pouchitis after ileal pouch-anal anastomosis: a prospective cohort study. Colorectal Dis. 2012;14:1365–1371. doi:10.1111/j.1463-1318.2012.02993.x
18. Abdelrazeq AS, Kandiyil N, Botterill ID, et al. Predictors for acute and chronic pouchitis following restorative proctocolectomy for ulcerative colitis. Colorectal Dis. 2008;10:805–813. doi:10.1111/j.1463-1318.2007.01413.x
19. White E, Melmed GY, Vasiliauskas EA, et al. A prospective analysis of clinical variables, serologic factors, and outcome of ileal pouch-anal anastomosis in patients with backwash ileitis. Dis Colon Rectum. 2010;53:987–994. doi:10.1007/DCR.0b013e3181dcb3f2
20. Fleshner P, Ippoliti A, Dubinsky M, et al. A prospective multivariate analysis of clinical factors associated with pouchitis after ileal pouch-anal anastomosis. Clin Gastroenterol Hepatol. 2007;5:952–958. doi:10.1016/j.cgh.2007.03.020
21. Okita Y, Araki T, Tanaka K, et al. Predictive factors for development of chronic pouchitis after ileal pouch-anal anastomosis in ulcerative colitis. Digestion. 2013;88:101–109. doi:10.1159/000353151
22. Stahlberg D, Gullberg K, Liljeqvist L, et al. Pouchitis following pelvic pouch operation for ulcerative colitis. Incidence, cumulative risk, and risk factors. Dis Colon Rectum. 1996;39:1012–1018. doi:10.1007/BF02054692
23. Uchino M, Ikeuchi H, Matsuoka H, et al. Clinical features and management of pouchitis in Japanese ulcerative colitis patients. Surg Today. 2013;43:1049–1057. doi:10.1007/s00595-012-0377-4
24. Wu X-R, Shen B, Lan P. Chapter 12 - risk factors for chronic inflammatory disorders of ileal pouch. In: Shen B, editor. Pouchitis and Ileal Pouch Disorders. Cambridge, MA: Academic Press; 2019:147–161.
25. Mukewar S, Wu X, Lopez R, et al. Comparison of long-term outcomes of S and J pouches and continent ileostomies in ulcerative colitis patients with restorative proctocolectomy-experience in subspecialty pouch center. J Crohns Colitis. 2014;8:1227–1236. doi:10.1016/j.crohns.2014.02.019
26. Meier CB, Hegazi RA, Aisenberg J, et al. Innate immune receptor genetic polymorphisms in pouchitis: is CARD15 a susceptibility factor? Inflamm Bowel Dis. 2005;11:965–971. doi:10.1097/01.MIB.0000186407.25694.cf
27. Gionchetti P, Rizzello F, Venturi A, et al. Oral bacteriotherapy as maintenance treatment in patients with chronic pouchitis: a double-blind, placebo-controlled trial. Gastroenterology. 2000;119:305–309. doi:10.1053/gast.2000.9370
28. Mimura T, Rizzello F, Helwig U, et al. Once daily high dose probiotic therapy (VSL#3) for maintaining remission in recurrent or refractory pouchitis. Gut. 2004;53:108–114. doi:10.1136/gut.53.1.108
29. Gosselink M, Schouten W, van Lieshout L, et al. Delay of the first onset of pouchitis by oral intake of the probiotic strain Lactobacillus rhamnosus GG. Dis Colon Rectum. 2004;47:876–884. doi:10.1007/s10350-004-0525-z
30. Yasueda A, Mizushima T, Nezu R, et al. The effect of Clostridium butyricum MIYAIRI on the prevention of pouchitis and alteration of the microbiota profile in patients with ulcerative colitis. Surg Today. 2016;46:939–949. doi:10.1007/s00595-015-1261-9
31. Scaioli E, Sartini A, Liverani E, et al. Sulfasalazine in prevention of pouchitis after proctocolectomy with ileal pouch-anal anastomosis for ulcerative colitis. Dig Dis Sci. 2017;62:1016–1024. doi:10.1007/s10620-017-4454-9
32. Godny L, Maharshak N, Reshef L, et al. Fruit consumption is associated with alterations in microbial composition and lower rates of pouchitis. J Crohns Colitis. 2019;13:1265–1272. doi:10.1093/ecco-jcc/jjz053
33. Sandborn WJ, Tremaine WJ, Batts KP, et al. Pouchitis after ileal pouch-anal anastomosis: a Pouchitis Disease Activity Index. Mayo Clin Proc. 1994;69(5):409–415. doi:10.1016/S0025-6196(12)61634-6
34. Shen B, Achkar JP, Connor J, et al. Modified Pouchitis Disease Activity Index: a simplified approach to the diagnosis of Pouchitis. Dis Colon Rectum. 2003;46(6):748–753. doi:10.1007/s10350-004-6652-8
35. Oresland T, Fasth S, Nordgren S, et al. The clinical and functional outcome after restorative proctocolectomy. A prospective study in 100 patients. Int J Colorectal Dis. 1989;4:50–56. doi:10.1007/BF01648551
36. Brandsborg S, Nicholls RJ, Montersen LS, et al. Restorative proctocolectomy for ulcerative colitis: development and validation of a new scoring system for pouch dysfunction and quality of life. Colorectal Dis. 2013;15(12):e719–25. doi:10.1111/codi.12425
37. Shen B, Achkar JP, Lashner BA, et al. A randomized clinical trial of ciprofloxacin and metronidazole to treat acute pouchitis. Inflamm Bowel Dis. 2001;7:301. doi:10.1097/00054725-200111000-00004
38. Isaacs K, Sandler R, Abreu M, et al. Rifaximin for the treatment of active pouchitis: a randomized, double-blind, placebo-controlled pilot study. Inflamm Bowel Dis. 2007;13(10):1250–1255. doi:10.1002/ibd.20187
39. Shen B. Chapter 29 - medical treatment of pouchitis. In: Shen B, editor. Pouchitis and Ileal Pouch Disorders. Cambridge, MA: Academic Press; 2019:343–356. doi:10.1016/B978-0-12-809402-0.00029-0
40. Thind K, Shen B, Farooqi R. Vancomycin in treatment of immune-mediated pouchitis. Am J Gastroenterol. 2018;113:S1180. doi:10.14309/00000434-201810001-02074
41. Buness C, Lindor KD, Miloh T. Oral vancomycin therapy in a child with primary sclerosing cholangitis and severe ulcerative colitis. Pediatr Gastroenterol Hepatol Nutr. 2016;19:210–213. doi:10.5223/pghn.2016.19.3.210
42. Damman JL, Rodriguez EA, Ali AH, et al. Review article: the evidence that vancomycin is a therapeutic option for primary sclerosing cholangitis. Aliment Pharmacol Ther. 2018;47:886–895. doi:10.1111/apt.14540
43. Rahimpour S, Nasiri-Toosi M, Khalili H, et al. A triple blinded, randomized, placebo-controlled clinical trial to evaluate the efficacy and safety of oral vancomycin in primary sclerosing cholangitis: a pilot study. J Gastrointestin Liver Dis. 2016;25:457–464. doi:10.15403/jgld.2014.1121.254.rah
44. Gionchetti P, Rizzello F, Venturi A, et al. Antibiotic combination therapy in patients with chronic, treatment-resistant pouchitis. Aliment Pharmacol Ther. 1999;13:713–718. doi:10.1046/j.1365-2036.1999.00553.x
45. Abdelrazeq A, Kelly S, Lund J, et al. Rifaximin-ciprofloxacin combination therapy is effective in chronic active refractory pouchitis. Colorectal Dis. 2005;7:182–186. doi:10.1111/j.1463-1318.2004.00746.x
46. Shen B, Fazio VW, Remzi FH, et al. Combined ciprofloxacin and tinidazole therapy in the treatment of chronic refractory pouchitis. Dis Colon Rectum. 2007;50:498–508. doi:10.1007/s10350-006-0828-3
47. McLaughlin SD, Clark SK, Shafi S, et al. Fecal coliform testing to identify effective antibiotic therapies for patients with antibiotic-resistant pouchitis. Clin Gastroenterol Hepatol. 2009;7:545–548. doi:10.1016/j.cgh.2009.01.002
48. Gionchetti P, Rizzello F, Poggioli G, et al. Oral budesonide in the treatment of chronic refractory pouchitis. Aliment Pharmacol Ther. 2007;25:1231–1236. doi:10.1111/j.1365-2036.2007.03306.x
49. Chopra A, Pardi D, Loftus E, et al. Budesonide in the treatment of inflammatory bowel disease: the first year of experience in clinical practice. Inflamm Bowel Dis. 2006;12:29–32. doi:10.1097/01.MIB.0000192323.82426.83
50. Sambuelli A, Boerr L, Negreira S, et al. Budesonide enema in pouchitis–a double-blind, doubledummy, controlled trial. Aliment Pharmacol Ther. 2002;16:27–34. doi:10.1046/j.1365-2036.2002.01139.x
51. Gionchetti P, Calabrese C, Calafiore A, et al. Oral beclomethasone diproprionate in chronic refractory pouchitis. J Chrohns Colitis. 2014;8:649–653. doi:10.1016/j.crohns.2013.12.001
52. Dalal RL, Shen B, Schwartz DA. Management of pouchitis and other common complications of the pouch. Inflamm Bowel Dis. 2018;24:989–996. doi:10.1093/ibd/izy020
53. Sandborn WJ, Pardi DS. Clinical management of pouchitis. Gastroenterology. 2004;127:1809–1814. doi:10.1053/j.gastro.2004.10.011
54. Huguet M, Pereira B, Goutte M, et al. Systematic review with meta-analysis: anti-TNF therapy in refractory pouchitis and Crohn’s disease-like complications of the pouch after ileal pouch-anal anastomosis following colectomy for ulcerative colitis. Inflamm Bowel Dis. 2018;24:261–268. doi:10.1093/ibd/izx049
55. Kelly OB, Rosenberg M, Tyler AD, et al. Infliximab to treat refractory inflammation after pelvic pouch surgery for ulcerative colitis. J Crohns Colitis. 2016;10:410–417. doi:10.1093/ecco-jcc/jjv225
56. Robbins L, Zaghiyan KN, Melmed GY, et al. Sa1152 do anti-TNF agents work in ulcerative colitis patients with de novo Crohn’s disease who failed anti-TNF therapy before ileal pouch-anal anastomosis? Gastroenterology. 2015;148:S–241. doi:10.1016/S0016-5085(15)30792-7
57. Iizuka M, Etou T, Yagisawa H, et al. One year of infliximab therapy successfully improved a case of refractory pouchitis without the use of antibiotics. Intern Med. 2014;53:2581–2583. doi:10.2169/internalmedicine.53.2793
58. Viazis N, Giakoumis M, Koukouratos T, et al. Long term benefit of one year infliximab administration for the treatment of chronic refractory pouchitis. J Crohns Colitis. 2013;7:e457–60. doi:10.1016/j.crohns.2013.02.018
59. Barreiro-de Acosta M, García-Bosch O, Souto R, et al. Efficacy of infliximab rescue therapy in patients with chronic refractory pouchitis: a multicenter study. Inflamm Bowel Dis. 2012;18:812–817. doi:10.1002/ibd.21821
60. Barreiro-de Acosta M, García-Bosch O, Gordillo J, et al. Efficacy of adalimumab rescue therapy in patients with chronic refractory pouchitis previously treated with infliximab: a case series. Eur J Gastroenterol Hepatol. 2012;24:756–758. doi:10.1097/MEG.0b013e3283525a7b
61. Li Y, Lopez R, Queener E, et al. Adalimumab therapy in crohn’s disease of the ileal pouch. Inflamm Bowel Dis. 2012;18:2232–2239. doi:10.1002/ibd.22933
62. Haveran LA, Sehgal R, Poritz LS, et al. Infliximab and/or azathioprine in the treatment of crohn’s disease-like complications after IPAA. Dis Colon Rectum. 2011;54:15–20. doi:10.1007/DCR.0b013e3181fc9f04
63. Yeates J, Rashid M. Successful long-term use of infliximab in refractory pouchitis in an adolescent. Gastroenterol Res Pract. 2010;2010:860394. doi:10.1155/2010/860394
64. Ferrante M, D’Haens G, Dewit O, et al.; Belgian IBD Research Group. Efficacy of infliximab in refractory pouchitis and crohn’s disease-related complications of the pouch: a Belgian case series. Inflamm Bowel Dis. 16;2010:243–249. doi:10.1002/ibd.21037
65. Gionchetti P, Straforini G, Tambasco R, et al. M1085 use of infliximab and adalimumab in refractory pouchitis. Gastroenterology. 2010;138:S–328. doi:10.1016/S0016-5085(10)61511-9
66. Akitake R, Nakase H, Tamaoki M, et al. Modulation of th1/th2 balance by infliximab rescues postoperative occurrence of small-intestinal inflammation associated with ulcerative colitis. Dig Dis Sci. 2010;55:1781–1784. doi:10.1007/s10620-009-0910-5
67. Calabrese C, Gionchetti P, Rizzello F, et al. Short-term treatment with infliximab in chronic refractory pouchitis and ileitis. Aliment Pharmacol Ther. 2008;27:759–764. doi:10.1111/j.1365-2036.2008.03656.x
68. Shen B, Remzi FH, Lavery IC, et al. Administration of adalimumab in the treatment of crohn’s disease of the ileal pouch. Aliment Pharmacol Ther. 2009;29:519–526. doi:10.1111/j.1365-2036.2008.03920.x
69. Molnar T, Farkas K, Nagy F, et al. Successful use of infliximab for treating fistulizing pouchitis with severe extraintestinal manifestation: a case report. Inflamm Bowel Dis. 2008;14:1752–1753. doi:10.1002/ibd.20485
70. Coburn LA, Wise PE, Schwartz DA. The successful use of adalimumab to treat active crohn’s disease of an ileoanal pouch during pregnancy. Dig Dis Sci. 2006;51:2045–2047. doi:10.1007/s10620-006-9452-2
71. Kooros K, Katz AJ. Infliximab therapy in pediatric crohn’s pouchitis. Inflamm Bowel Dis. 2004;10:417–420. doi:10.1097/00054725-200407000-00013
72. Viscido A, Habib FI, Kohn A, et al. Infliximab in refractory pouchitis complicated by fistulae following ileo-anal pouch for ulcerative colitis. Aliment Pharmacol Ther. 2003;17:1263–1271. doi:10.1046/j.1365-2036.2003.01535.x
73. Colombel JF, Ricart E, Loftus EV
74. Arnott ID, McDonald D, Williams A, et al. Clinical use of infliximab in crohn’s disease: the Edinburgh experience. Aliment Pharmacol Ther. 2001;15:1639–1646. doi:10.1046/j.1365-2036.2001.01092.x
75. Ricart E, Panaccione R, Loftus EV, et al. Successful management of crohn’s disease of the ileoanal pouch with infliximab. Gastroenterology. 1999;117:429–432. doi:10.1053/gast.1999.0029900429
76. Verstockt B, Claeys C, De Hertogh G, et al. Outcome of biological therapies in chronic antibiotic-refractory pouchitis: a retrospective single-centre experience. United European Gastroenterol J. 2009;7:1215–1225. doi:10.1177/2050640619871797
77. Singh A, Khan F, Lopez R, et al. Vedolizumab for chronic antibiotic-refractory pouchitis. Gastroenterol Rep (Oxf). 2019;7:121–126. doi:10.1093/gastro/goz001
78. Philpott J. Ashburn J and Shen B. Efficacy of vedolizumab in patients with antibiotic and anti-tumor necrosis alpha refractory pouchitis. Inflamm Bowel Dis. 2017;23:E5–E6. doi:10.1097/MIB.0000000000000992
79. Bar F, Kuhbacher T, Dietrich NA, et al. Vedolizumab in the treatment of chronic, antibiotic-dependent or refractory pouchitis. Aliment Pharmacol Ther. 2018;47:581–587. doi:10.1111/apt.14479
80. Gregory M, Weaver KN, Hoversten P, et al. Efficacy of vedolizumab for refractory pouchitis of the ileo-anal pouch: results from a multicenter US cohort. Inflamm Bowel Dis. 2019;25:1569–1576. doi:10.1093/ibd/izz030
81. Weaver K, Gregory M, Syal G, et al. Ustekinumab is effective for the treatment of Crohn’s disease of the pouch in a multicenter cohort. Inflamm Bowel Dis. 2018. doi:10.1093/ibd/izy302
82. Ollech J, Glick L, Weisshof R, et al. Ustekinumab is effective for the treatment of chronic antibiotic-refractory pouchitis. Dig Dis Sci. 2019;64:3596–3601. doi:10.1007/s10620-019-05697-1
83. Okano S, Yoshimura N, Sako M, et al. A case of refractory chronic pouchitis successfully treated with tofacitinib. Clin J Gastroenterol. 2020;13:560–563. doi:10.1007/s12328-020-01108-5
84. Weisshof R, Aharoni Golan M, Sossenheimer PH, et al. Real-world experience with tofacitinib in IBD at a tertiary center. Dig Dis Sci. 2019;64:1945–1951. doi:10.1007/s10620-019-05492-y
85. Landy J, Walker A, Li V, et al. Variable alterations of the microbiota, without metabolic or immunological change, following faecal microbiota transplantation in patients with chronic pouchitis. Sci Rep. 2015;5:12955. doi:10.1038/srep12955
86. Stallmach A, Lange K, Buening J, et al. Fecal Microbiota Transfer in patients with chronic antibiotic-refractory pouchitis. Am J Gastroenterol. 2016;111(3):411–413. doi:10.1038/ajg.2015.436
87. Selvig D, Piceno Y, Terdiman J, et al. Fecal microbiota transplantation in pouchitis: clinical, endoscopic, histologic, and microbiota results from a pilot study. Dig Dis Sci. 2020;65(4):1099–1106. doi:10.1007/s10620-019-05715-2
88. Herfarth H, Barnes E, Long M, et al. Combined endoscopic and oral fecal microbiota transplantation in patients with antibiotic-dependent pouchitis: low clinical efficacy due to low microbial engraftment. Inflamm Intest Dis. 2019;4(1):1–6. doi:10.1159/000497042
89. Croagh C, Shepherd SJ, Berryman M, Muir JG, Gibson PR. Pilot study on the effect of reducing dietary FODMAP intake on bowel function in patients without a colon. Inflamm Bowel Dis. 2007;13(12):1522–1528. doi:10.1002/ibd.20249
90. Shen B, Pardi DS, Bennett AE, et al. The efficacy and tolerability of AST-120 (spherical carbon adsorbent) in active pouchitis. Am J Gastroenterol. 2009;104(6):1468–1474. doi:10.1038/ajg.2009.138
91. Miner P, Wedel M, Bane B, Bradley J. An enema formulation of alicaforsen, an antisense inhibitor of intercellular adhesion molecule-1, in the treatment of chronic, unremitting pouchitis. Aliment Pharmacol Ther. 2004;19(3):281–286. doi:10.1111/j.1365-2036.2004.01863.x
92. Greuter T, Rogler G. Alicaforsen in the treatment of pouchitis. Immunotherapy. 2017;9(14):1143–1152. doi:10.2217/imt-2017-0085
93. Hasan B, Yim Y, Ur Rashid M, et al. Hyperbaric oxygen therapy in chronic inflammatory conditions of the pouch. Inflamm Bowel Dis. 2020;izaa245. doi:10.1093/ibd/izaa245
94. Uchino M, Ikeuchi H, Matsuoka H, et al. Topical tacrolimus therapy for antibiotic-refractory pouchitis. Dis Colon Rectum. 2013;56(10):1166–1173. doi:10.1097/DCR.0b013e31829ebd83
95. Yamamoto T, Umegae S, Matsumoto K. Mucosal healing in patients with ulcerative colitis during a course of selective leukocytapheresis therapy: a prospective cohort study. Inflamm Bowel Dis. 2010;16:1905–1911. doi:10.1002/ibd.21260
96. Passalacqua S, Ferraro P, Bresci G, et al. The Italian registry of therapeutic apheresis: granulocyte monocyte apheresis in the treatment of IBD. A multicentric study. J Clin Apher. 2011;26:332–337. doi:10.1002/jca.20315
97. Sacco R, Romano A, Mazzoni A, et al. Granulocytapheresis in steroid-dependent and steroid resistant patients with inflammatory bowel disease: a prospective observational study. J Crohns Colitis. 2013;7:e692–e697. doi:10.1016/j.crohns.2013.06.012
98. Yokoyama Y, Watanabe K, Ito H, et al. Factors associated with treatment outcome, and long-term prognosis of patients with ulcerative colitis undergoing selective depletion of myeloid lineage leucocytes: a prospective multicenter study. Cytotherapy. 2015;17:680–688. doi:10.1016/j.jcyt.2015.02.007
99. Dignass A, Akbar A, Hart A, et al. Safety and efficacy of granulocyte/monocyte apheresis in steroid dependent active ulcerative colitis with insufficient response or intolerance to immunosuppressants and/or biologics [the ART Trial]: 12-week interim results. J Crohns Colitis. 2016;10:812–820. doi:10.1093/ecco-jcc/jjw032
100. Yamamoto T, Tanaka T, Yokoyama T, et al. Efficacy of granulocyte and monocyte apheresis for antibiotic-refractory pouchitis after proctocolectomy for ulcerative colitis: an open-label, prospective, multicentre study. Therap Adv Gastroenterol. 2017;10(2):199–206. doi:10.1177/1756283X16679348
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
