Back to Journals » Journal of Pain Research » Volume 8
Changes in saccharin preference behavior as a primary outcome to evaluate pain and analgesia in acetic acid-induced visceral pain in mice
Authors de la Puente B, Romero-Alejo E, Vela JM, Merlos M, Zamanillo D, Portillo-Salido E
Received 26 June 2015
Accepted for publication 29 July 2015
Published 6 October 2015 Volume 2015:8 Pages 663—673
DOI https://doi.org/10.2147/JPR.S91230
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Michael Schatman
Beatriz de la Puente, Elizabeth Romero-Alejo, José Miguel Vela, Manuel Merlos, Daniel Zamanillo, Enrique Portillo-Salido
Department of Pharmacology, Drug Discovery and Preclinical Development, ESTEVE, Barcelona, Spain
Abstract: Reflex-based procedures are important measures in preclinical pain studies that evaluate stimulated behaviors. These procedures, however, are insufficient to capture the complexity of the pain experience, which is often associated with the depression of several innate behaviors. While recent studies have made efforts to evidence the suppression of some positively motivated behaviors in certain pain models, they are still far from being routinely used as readouts for analgesic screening. Here, we characterized and compared the effect of the analgesic ibuprofen (Ibu) and the stimulant, caffeine, in assays of acute pain-stimulated and pain-depressed behavior. Intraperitoneal injection of acetic acid (AA) served as a noxious stimulus to stimulate a writhing response or depress saccharin preference and locomotor activity (LMA) in mice. AA injection caused the maximum number of writhes between 5 and 20 minutes after administration, and writhing almost disappeared 1 hour later. AA-treated mice showed signs of depression-like behaviors after writhing resolution, as evidenced by reduced locomotion and saccharin preference for at least 4 and 6 hours, respectively. Depression-like behaviors resolved within 24 hours after AA administration. A dose of Ibu (40 mg/kg) – inactive to reduce AA-induced abdominal writhing – administered before or after AA injection significantly reverted pain-induced saccharin preference deficit. The same dose of Ibu also significantly reverted the AA-depressed LMA, but only when it was administered after AA injection. Caffeine restored locomotion – but not saccharin preference – in AA-treated mice, thus suggesting that the reduction in saccharin preference – but not in locomotion – was specifically sensitive to analgesics. In conclusion, AA-induced acute pain attenuated saccharin preference and LMA beyond the resolution of writhing behavior, and the changes in the expression of hedonic behavior, such as sweet taste preference, can be used as a more sensitive and translational model to evaluate analgesics.
Keywords: saccharin preference, locomotor activity, pain, writhing, analgesia, ibuprofen, caffeine
Introduction
Most studies on pain and analgesia use reflex-based procedures (eg, tail flick, licking, and guarding) induced by aversive stimulation through the application of particular mechanical, thermal, electrical, and chemical stimuli to identify analgesics. This approach has been evaluated critically because it overfocuses on reflex behaviors and consequently neglects the key affective component of pain phenomena.1–4 Consequently, the development of relevant new dependent variables to increase the validity of animal models of pain is increasingly pursued.4–8 Among them, the evaluation of innate behaviors suppressed – instead of enhanced – by pain has been highlighted.6,7,9–12 A claimed advantage of selecting these behaviors as endpoints is that those drugs with analgesic properties will be associated with increased behavior rates, and, as a result, analgesic effects would be readily dissociable from motor impairment. In addition, the study of pain-suppressed behaviors should allow outlining the role of behavioral depression, which is normally associated with pain syndromes13,14 and with others aspects related to the mechanisms and determinants of the affective component of pain.15
From this perspective, any behavior spontaneously performed by an animal can be selected as target behavior to evaluate whether pain is or is not able to depress it. However, hedonically oriented behaviors, which are behaviors that have the ability to ensure a positive emotional state experienced as pleasure,16,17 are expected to be rapidly expressed and maintained by the animals at relatively high rates, which would help reduce methodological problems such as using food or water deprivation during the behavioral tasks. Furthermore, decreases in rate, frequency, duration, or intensity of highly preferred behaviors (“hedonic behaviors”) caused by pain (or other insults) can be suggestive of a deterioration of the animal global welfare and/or quality of life, which makes hedonically oriented behaviors interesting in the testing of beneficial effects of analgesics – which should restore the normal hedonic behavior of the animals.
In this study, two positively motivated behaviors, such as the natural rodent preference for sweet taste and rodent locomotor activity (LMA) in a novel environment, were selected as the main dependent variables to measure the presence of pain or analgesia. Preference for sweet taste maintains a high rate in mice and requires an intact cognitive function as well as appetitive motivation.18 A precise measurement of sweet taste preference is easy to conduct and can be determined in home cages without animal handling. This behavior has been shown to be sensitive to different pharmacological and environmental manipulations. It has been used to model anhedonia – the lack of interest or pleasure in response to hedonic stimuli or experiences – in the chronic mild stress animal model of depression.19,20 LMA measures spontaneous, instinctive behaviors of rodents that are largely motivated by the exploration of a novel environment for means of escape. Decrease in locomotion as a consequence of pain has been consistently reported in both humans and rodents,6,11,21–23 and psychomotor retardation – which includes motor impairment affecting gross locomotor skills – is also a central feature of depression.24,25
The classical preclinical pain test of acetic acid (AA)-induced abdominal constriction to induce pain was used. In this test, AA injection causes inflammation of the abdominal cavity wall and evokes sustained writhing behavior and reduced motor activity. The occurrence of this writhing behavior (abdominal cramps or stretching) per unit of time is commonly evaluated. These behaviors are considered to be reflexes and to be evidence of visceral pain,26 but the frequency of writhing decreases spontaneously with time.
The goal of the present study was to compare the analgesic sensitivity of two pain-suppressed behaviors with the AA-induced standard reflexive outcome (writhing behavior). For that purpose, the time course of AA-induced behavior (writhing) and AA-depressed behavior (saccharin preference and LMA) was first studied. Secondly, the restorative effects on both LMA and saccharin preference behavior of a dose of ibuprofen (Ibu) devoid of efficacy on AA-induced writhing were evaluated. This was performed by administering the drug before (development protocol) and after (expression protocol) the induction of pain by AA. Finally, the effects of caffeine-induced behavioral activation to assess the specificity of the different tests were evaluated.
Methods
Animals
Female CD1 mice weighing 25–30 g were used in all experiments (Charles River, L’Arbresle, France). The study protocol was approved by the local Committee of Animal Use and Care of our institution (ESTEVE) and was in accordance with the guidelines for the Care and Use of Laboratory Animals of the European Community (European Directive 2010/63/EU) and with the International Association for the Study of Pain guidelines on ethical standards for investigation in animals.27 Light/dark cycle (reverted 12/12 hours, lights on at 6 pm), temperature (22°C), and humidity (40%) were controlled. Animals had free access to food and water and were used after 14 days of acclimatization to housing conditions. All experiments were performed between 9 am and 6 pm.
Drugs
The drugs investigated were Ibu (40–320 mg/kg), supplied by Laboratorios Esteve (Barcelona, Spain), and caffeine (5–20 mg/kg), purchased from Sigma Chemical Co (Barcelona, Spain). Approximately 0.5% hydroxypropyl methylcellulose (HPMC) (Sigma Chemical Co) dissolved in saline was used as vehicle. The drugs (or the vehicle in the control group) were administered intraperitoneally (IP) at a volume of 10 mL/kg. The time of administration was chosen in order to evaluate the putative preventive or restorative effect of Ibu on target behaviors. To evaluate the preventive effect, the drug was administered 30 minutes before AA challenge (“development protocol”). To evaluate a purely restorative effect, the drug was administered 120 or 150 minutes after AA challenge for saccharin preference and LMA, respectively (“expression protocol”).
Assay of acetic acid-induced writhing
For the time course study, mice were injected 10 mL/kg of AA (0.6%) or vehicle (distilled water) by IP route. Each mouse was then placed in an individual, clear plastic observation chamber and the total number of writhes was counted for 1 hour after administration.
Based on the results of this protocol, the interval ranging between 5 and 15 minutes after AA injection was selected to evaluate the effects of Ibu and caffeine on the number of writhes. Separate groups of mice were administered vehicle (HPMC 0.5%), Ibu, or caffeine, IP, 30 minutes before 0.6% AA injection.
For scoring purposes, a “writhe” was defined as a contraction of the abdominal muscles accompanied by body elongation and hind limb extension. Data are expressed as the mean number of writhes over the 10-minute observation period.
Saccharin preference test
Mice were habituated to saccharin (0.1%, Sigma Aldrich Co, St Louis, MO, USA) consumption by means of saccharin solution diluted in tap water as sole drinking fluid for 48 hours. After habituation, the baseline saccharin preference was measured for 6 hours 1 day before the test. During the saccharin preference test, fluid consumption was measured for 24 hours with a two-bottle protocol, whereby mice were exposed to a bottle each of tap water and 0.1% saccharin solution. Water and saccharin solution intake was estimated simultaneously in control and experimental groups by weighing the bottles at 2, 4, 6, 8, and 24 hours. The animals were not previously deprived of water and food, but had no access to food during the first 6-hour preference tests. For each mouse on each day, the ratio of solution preference was calculated according to the formula below:

Novelty induced LMA evaluation
LMA was scored automatically in independent experiments. Eight standard actimeters (Linton Instrumentation Inc., Norfolk, UK) equipped with infrared beam motion detectors were used. On the day of the experiment, mice were evaluated in a dark environment. Mice were marked and weighed at the beginning of each experimental session. After administering AA, the compounds, or their vehicles, the animals were returned to their home cages and then placed in the LMA cages at the scheduled time. In the time course experiment, LMA was evaluated in separate groups of mice exposed to the chamber only once at the scheduled post-AA time (1, 2, 3, 4 or 5 hours post-AA). Moving time (seconds) was measured for 60 minutes in each separate group, with readings performed every 5 minutes.
Data analysis
Data are expressed as mean ± standard error of mean. For studies of LMA and saccharin preference, data were analyzed with two-way repeated measures analysis of variance (ANOVA), with pain and treatment drug as factors. One-way ANOVA was used for area under the curve (AUC, from 0 to 24 hours) comparison. One-way repeated measures ANOVA was used to analyze writhing test data, and one-way ANOVA with Bonferroni’s multiple comparison test as post hoc analysis was used to analyze drug treatment data. P<0.05 was considered statistically significant. Statistical analyses were carried out with the GraphPad Prism 5.00 program (GraphPad Software, San Diego, CA, USA).
Results
Acetic acid-induced stimulation of writhing and depressed sweet preference behavior
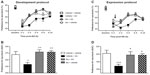
IP injection of 0.6% AA robustly induced the appearance of abdominal constrictions (writhing) in mice (Figure 1, left axis). The number of writhes peaked 5–20 minutes after AA administration (P<0.001). Then, a progressive decrease in this behavior was observed, and the effects of AA were no longer apparent after 60 minutes (P>0.05). The right axis of Figure 1 shows the preference for a saccharin solution (0.1%) in animals pretreated with AA (pain group) or its vehicle (control group). The baseline values for saccharin preference measured 1 day earlier did not vary significantly between the pain and the control groups (80.1±2.1 and 77.3±3.3%, respectively). Mice treated with the vehicle of AA demonstrated a preference for saccharin solution over water of 65%–75% at different times. Variations between baseline and test day values were observed across all experiments. These variations were attributed to the different baseline recording times – 6 hours on a continuous basis – and to mice handling on the test day, which included IP injection. Pretreatment with 0.6% but not 0.3% (data not shown) AA significantly decreased the expression of saccharin preference behavior as compared to control mice. Two-way repeated measures ANOVA (time × pain) showed a significant effect of time [F(4,48) =28.24, P<0.001] and pain [F(1,12) =15.46, P<0.01] and a significant interaction between these two factors [F(4,48) =2.76, P<0.05]. AA-induced deficit in the expression of this hedonic behavior was observed for at least 6 hours, and normal preference behavior was restored 24 hours after AA administration (Figure 1, right axis). Post hoc testing showed significantly reduced saccharin preference rates in AA-induced pain in mice at 2, 4, and 6 hours (P<0.01, P<0.001, and P<0.05, respectively), but not at 24 hours.
Acetic acid-induced decrease of LMA
LMA as a function of pretreatment interval at the same concentration of AA tested in the saccharin preference experiment is shown in Figure 2. Control mice showed peak activity during the first 5 minutes. After that, mice became habituated to the environment and their locomotion behavior progressively declined. The administration of 0.3% AA did not change LMA as compared to vehicle-treated mice (data not shown). However, mice treated with 0.6% of AA showed a strongly shortened motion time as compared to control mice (vehicle). A repeated measures two-way ANOVA showed significant effects of pain at 60 minutes [F(1,28) =20.69, P<0.001], 120 minutes [F(1,28) =16.40, P<0.001], 180 minutes [F(1,28) =9.23, P<0.01], and 240 minutes [F(1,28) =18.22, P<0.001], but not at 300 minutes [F(1,28) =0.17, P>0.05]. A significant interaction between pain and time was detected at 60 minutes [F(11,308) =6.35, P<0.001], 120 minutes [F(11,308) =7.23, P<0.001], and 180 minutes [F(11,308) =2.14, P<0.05], but not at 240 minutes [F(11,308) =0.92, P>0.05] or 300 minutes [F(11,308) =0.62, P>0.05].
Effects of ibuprofen and caffeine on acetic acid-induced writhing
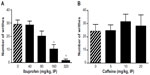
Ibu administration 30 minutes before AA led to a significant, dose-related inhibition of AA-induced writhing in mice. Doses of 160 and 320, but not 40 or 80 mg/kg, significantly inhibited AA-induced writhing behavior (P<0.01, Figure 3A). Caffeine administration, however, failed to significantly inhibit AA-induced writhing in mice at the doses of 5, 10, and 20 mg/kg IP (Figure 3B, NS).
Effects of ibuprofen on AA-induced deficit in saccharin preference behavior
Next, we aimed at determining whether an analgesic was able to revert AA-induced deficit in the saccharin preference behavior of mice in two different administration protocols, the “development” and the “expression” protocols.
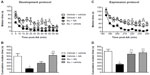
In the development protocol, 40 mg/kg of Ibu – a dose that failed to produce any analgesic effect evaluated by AA-induced writhing – or vehicle were administered 30 minutes before AA challenge. Mice receiving vehicle (vehicle + AA group) before AA injection showed a significantly depressed saccharin preference behavior as compared to control mice (vehicle + vehicle group). Repeated measures two-way ANOVA (time × pain) showed a significant effect of time [F(5,80) =27.17, P<0.001] and pain [F(1,80) =4.81, P<0.05], and a significant time × pain interaction between these two factors [F(5,80) =2.45, P<0.05]. Ibu did not affect the normal saccharin preference of vehicle-injected mice (Ibu + vehicle group) and did not prevent decreased saccharin preference in AA-treated mice (Ibu + AA group) before 2 hours, but it was able to revert the AA-induced deficit in the preference for saccharin from 2 to 6 hours (Figure 4A). Repeated measures two-way ANOVA (time × treatment) showed a significant effect of time [F(5,80) =39.29, P<0.001], treatment [F(1,80) =11.91, P<0.05], and interaction between these two factors [F(5,80) =3.49, P<0.01]. One-way ANOVA followed by Bonferroni’s post hoc test of the AUC (from 0 to 24 hours) globally suggested total restoration of saccharin preference behavior in AA-treated mice (Figure 4; P<0.01).
We took advantage of the long-term duration of the AA-induced decrease in saccharin preference to evaluate whether Ibu was able to revert the deficit once established (“expression protocol”). Thus, Ibu or vehicle was administered 2 hours after AA or vehicle challenge (arrow in Figure 4C). AA-injected mice treated with vehicle (vehicle + AA group) showed a significant decrease in saccharin preference behavior as compared to those injected with vehicle (vehicle + vehicle group). Two-way repeated measures ANOVA (time × pain) showed a significant effect of time [F(5,70) =31.34, P<0.001], pain [F(1,70) =9.13, P<0.01], and interaction between these two factors [F(5,70) =3.58, P<0.01]. Similar to the development protocol, Ibu did not change the preference for saccharin of the vehicle-injected mice, but was able to revert the deficit of the AA-injected mice (Figure 4C). Repeated measures ANOVA (time × treatment) showed a significant effect of time [F(5,70) =34.52, P<0.001], treatment [F(1,70) =5.09, P<0.05], and interaction between these two factors [F(5,70) =4.63, P<0.001]. Similarly, one-way ANOVA followed by Bonferroni’s post hoc test of the AUC was also suggestive of a restored saccharin preference behavior in AA-treated mice (Figure 4D, P<0.01).
Effects of ibuprofen on the AA-induced deficit in LMA
We next aimed to determinate whether Ibu was able to revert the AA-induced deficit in the exploratory behavior of mice, also using the two administration protocols (“development” and “expression” protocols).
In order to prevent deficit, Ibu was again administered at 40 mg/kg (inactive dose evaluated by writhing behaviors) 30 minutes before AA challenge (“development protocol”). Vehicle-treated mice injected with AA (vehicle + AA group) showed a significantly depressed LMA behavior as compared to control mice (vehicle + vehicle group). Repeated measures two-way ANOVA (time × pain) showed a significant effect of time [F(11,154) =9.81, P<0.001], pain [F(1,154) =20.10, P<0.001], and no interaction between these two factors [F(11,154) =1.68, P>0.05]. While Ibu administration failed to significantly affect the expression of the LMA behavior in the animals injected with AA vehicle (Ibu + vehicle group), it did slightly increase the LMA of AA-injected mice (Ibu + AA group), particularly over the first 10 minutes (Figure 5A). Repeated measures ANOVA (time × treatment) showed a significant effect of time [F(11,154) =31.02, P<0.0001], treatment [F(1,154) =10.72, P<0.001], and interaction between factors [F(11,154) =2.43, P<0.01]. While AUC values calculated for each experimental group indicated a partially restored saccharin preference behavior caused by Ibu in AA-injected mice (Figure 5B), one-way ANOVA followed by Bonferroni’s post hoc test of the AUC showed no statistically significant differences.
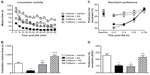
The effects of Ibu 2 hours after AA challenge (“expression protocol”) are shown in Figure 5C. Vehicle-treated mice after AA injection (vehicle + AA group) showed a significant decrease in exploratory behavior as compared to control mice (vehicle + vehicle group). Repeated measures two-way ANOVA (time × pain) showed a significant effect of time [F(11,198) =15.85, P<0.001], pain [F(1,198) =33.35, P<0.001], and interaction between these two factors [F(11,198) =1.05, P<0.05]. Ibu did not change the LMA of the mice injected with AA vehicle (Ibu + vehicle group). However, Ibu was able to fully revert LMA decrease in the AA-injected mice (Ibu + AA group). Repeated measures ANOVA (time × treatment) showed a significant effect of time [F(11,198) =30.98, P<0.001] and treatment [F(1,198) =25.14, P<0.001], but no interaction between these two factors [F(11,198) =0.27, P>0.05]. One-way ANOVA followed by Bonferroni’s post hoc test of the AUC was also suggestive of fully restored LMA behavior in AA-treated mice (Figure 5D, P<0.01).
Effects of caffeine on the AA-induced depression in saccharin preference and LMA behavior
In order to study the specificity of the endpoints, we tested the effects of caffeine, a nonanalgesic stimulant producing behavioral increases, on AA-induced depression in both LMA and saccharin preference behaviors using the development protocol. The effects on LMA are shown in Figure 6A. Caffeine was administered at 10 mg/kg (IP) 30 minutes before AA challenge. As expected, AA-injected mice treated with vehicle (vehicle + AA group) showed a significantly depressed LMA behavior as compared to control mice (vehicle + vehicle group). Repeated measures two-way ANOVA (time × pain) showed a significant effect of time [F(11,275) =16.59, P<0.001], pain [F(1,275) =46.21, P<0.001], and interaction between these two factors [F(11,275) =1.50, P>0.05]. Caffeine administration caused the expected increase in vehicle-injected mice (caffeine + vehicle group), but also increased the LMA of AA-injected mice (caffeine + AA group) as compared to control mice (vehicle + vehicle group) (Figure 6A). Repeated measures two-way (time × treatment) ANOVA showed a significant effect of time [F(11,187) =59.63, P<0.001], treatment [F(1,187) =22.32, P<0.001], and interaction between these two factors [F(11,187) =10.55, P<0.001]. One-way ANOVA followed by Bonferroni’s post hoc test of the AUC confirmed the caffeine-induced increase in LMA of both AA-treated and vehicle-treated mice (Figure 6B).
The effects on saccharin preference behavior are shown in Figure 6C. Caffeine was administered at the same dose of 10 mg/kg (IP) 30 minutes before AA challenge. AA-injected mice treated with vehicle (AA + vehicle group) showed a significant decrease in saccharin preference behavior as compared to control mice (vehicle + vehicle group). Two-way repeated measures ANOVA (time × pain) showed a significant effect of time [F(4,64) =21.36, P<0.001], pain [F(1,64) =21.33, P<0.001], and interaction between these two factors [F(4,64) =6.18, P<0,001]. Caffeine administration did not significantly affect the expression of the saccharin preference behavior of the animals injected with the vehicle of AA (caffeine + vehicle group) as compared to control mice (vehicle + vehicle group). Interestingly, caffeine administration to AA-injected mice (caffeine + AA group) did not revert the AA-induced decrease in saccharin preference. Two-way repeated measures ANOVA (time × treatment) showed a significant effect of time [F(4,68) =59.75, P<0.001], but not treatment [F(1,68) =0.12, P>0.05] or interaction between these two factors [F(4,68) =0.72, P>0.05]. AUC calculation clearly indicates the absence of caffeine effect on saccharin preference behavior deficit in AA-treated mice (Figure 6D).
Discussion
Efforts have recently been made to investigate pain and analgesia using novel paradigms that do not rely solely on reflex-based withdrawal responses.4 Decreases in burrowing,28 nesting,29 feeding,10 intracranial self-stimulation,30 wheel running,31,32 and food-maintained operant responding7 to evaluate the presence of pain and analgesia have also been reported. Also, decreased LMA as a consequence of pain has been consistently reported in both humans and rodents.6,11,21–23 The present study provides evidence that the hedonic behavior of sweet taste preference using saccharin in mice was strongly depressed by AA and that it can be used to detect the analgesic effects of drugs. The characteristic pain writhing behavior induced by AA, which lasted less than 1 hour, was followed by a substantially longer “behavioral depression” manifested by a strongly decreased expression of both saccharin preference and LMA for at least 4 hours. Pain-suppressed behaviors long after AA-induced writhing behavior is consistent with the results of a previous study showing a similarly decreased LMA for 5 hours after treatment with 0.56% AA in male ICR mice.11 However, to our knowledge, this is the first time that such a sustained depression (for at least 6 hours) of sweet taste preference after AA administration is described. Previously, the time of feeding suppression using a Liquid Ensure™ protein drink was determined 1 hour after 0.56% AA administration.10
In the present study, a visceral noxious stimulus was selected to induce pain. Visceral pain presents with important differences as compared to cutaneous somatic pain. Somatic and visceral pain are mediated, at least in part, through different neural pathways at spinal and supraspinal sites, and evoke different emotional responses.33–38 Cutaneous somatic pain is escapable, can be controlled, and characteristically evokes active emotional coping responses such as agitation, hyperactivity, fight–flight, and hypertension. In contrast, visceral pain is inescapable, cannot be controlled by the subjects themselves, and usually evokes passive coping or “conservation-withdrawal” strategies, characterized by “disengagement from” the environment, ie, behavioral quiescence and immobility, decreased reactivity to the environment, hypotension, and bradycardia.36,38 The behavioral inhibition observed after the visceral noxious stimulation in our study is consistent with this view. Recent data from our laboratory – where formalin administration to the paw, a somatic pain model, was unable to alter saccharin preference behavior in mice – further support this view (unpublished data).
Ibu started to produce significant effect in the attenuation of the number of writhes at the dose of 160 mg/kg, but the pharmacological effect of 40 mg/kg of Ibu in the saccharin preference paradigm was already consistent with analgesia, considering the whole 0–24 hours measurement period. This dose, however, was ineffective to prevent AA-depressed saccharin preference in the first 2 hours of the saccharin preference test (development protocol). A logical explanation for this is that the saccharin preference behavior reductions observed during the first 2 hours may be caused by AA-induced writhing behavior, and are not inhibited by Ibu at 40 mg/kg. These two behaviors (writhing and saccharin preference) seem incompatible because mice cannot drink and writhe at the same time. Interestingly, Ibu clearly prevented sweet preference behavior deficit after 2 hours.
The fact that 40 mg/kg of Ibu – ineffective to block pain-induced writhing – was actually effective on the pain-depressed behaviors of saccharin preference raises the possibility that the analgesic effects of drugs can be better observed with a pain-depressed endpoint than with a pain-stimulated endpoint. This conclusion agrees with those of several previous studies showing that some analgesics such as Ibu, morphine, pregabalin, or acetaminophen attenuate the affective component of pain more potently than its sensory component.8,39–41 In the present study, where decreased saccharin preference behavior reflects the affective component and increased writhing behavior reflects the sensory component of AA-induced pain, Ibu was better against the affective component than against the sensory component of pain. Furthermore, this could be indicating that a drug can have analgesic properties without inhibiting the writhing behavior. This may be of particular importance in a drug discovery context because possible analgesics may be currently being discarded based on a lack of efficacy on sensory-based pain screening experimental models.
The “expression protocol” allowed us to test the effect of the drug using a within-subject design in animals where the AA-induced deficit had already been established and once the AA-induced writhing behavior had disappeared. Before Ibu administration, AA-treated mice showed the expected depression in saccharin preference shown by the decrease observed during the first 2 hours as compared to control animals. When these animals were treated with Ibu, the preference for saccharin returned to that of vehicle-treated animals. This approach allowed avoiding the potential effect of AA-stimulated behaviors likely to compete with the target depressed behavior, as we have previously hypothesized to explain the lack of effect in the development protocol of the saccharin intake in the first 2 hours, ie, when writhing is occurring at a relatively high rate, mice cannot perform the intake of liquid.
In the present study, Ibu administration 30 minutes before AA (development protocol) was able to only partially prevent AA-induced decrease in LMA. The results obtained during the entire hour period suggest that the dose of 40 mg/kg was not sufficient to completely restore pain-depressed LMA behavior. The partial efficacy of Ibu on AA-induced deficit in LMA is consistent with the lack of efficacy observed in the AA-induced writhing test and during the first 2 hours of the saccharin preference test.
The administration of Ibu after AA (expression protocol) restored LMA, with the activity of AA-injected animals returning to that of vehicle-injected animals. In this protocol, mice received Ibu or its vehicle 150 minutes after AA injection (30 minutes before the behavioral test). Animals pretreated with Ibu – but not animals treated with the vehicle – showed LMA restoration, which is consistent with the results observed in the expression protocol of the saccharin test.
Finally, caffeine was used as a nonanalgesic stimulant to evaluate the specificity in relation with pain of the two target behaviors. Caffeine prevented LMA decrease in AA-injected mice, but also induced a strong LMA increase in vehicle-injected animals. In contrast, caffeine was unable to change the depressed saccharin preference behavior in AA-treated mice. Therefore, despite the fact that caffeine induced LMA normalization in AA-injected mice to the level of control animals, the deficit in saccharin preference behavior was not sensitive to this behavioral arousal induced by caffeine. In a previous study, Stevenson et al11 did not find such effect of caffeine on AA-depressed LMA. The reasons for this discrepancy are not clear. Similar to this study, Stevenson et al11 found that caffeine significantly increased LMA in nondepressed mice. However, they only found a nonsignificant tendency of caffeine to revert acid-depressed LMA. Discrepancy may be sex related because we used female mice and the Stevenson et al11 study used male mice. However, no sex-related differences in caffeine-induced LMA increase have been found.42 Discrepancy might also arise from the different light/dark cycles in which the two behavioral experiments were performed. In order to favor the higher levels of LMA associated with the dark (active) phase of the animal’s activity cycle, our experiments were conducted under dark conditions, while the Stevenson et al’s11 study was conducted under light conditions. While the effects of caffeine on LMA did not seem to be altered by ambient lighting,43,44 circadian fluctuations in visceral sensory functions have been reported.45 Finally, despite standardization, systematic differences in behavior across laboratories have been well documented.46
In summary, saccharin preference and LMA behaviors were altered by a visceral noxious stimulus. AA-treated mice showed signs of depression-like behaviors after writhing resolution, as evidenced by reduced saccharin preference and locomotion for at least 6 and 4 hours, respectively. The decrease observed after AA administration in sweet taste preference was probably due to ongoing pain because it was specifically reverted by an analgesic drug such as Ibu but not by the stimulant drug caffeine. The decrease observed in novelty induced locomotion after AA injection was probably also due to ongoing pain because it was reverted by Ibu. However, the AA-depressed LMA was also reverted by the stimulant caffeine, thus suggesting that this behavioral endpoint is not robust enough to evaluate analgesic drugs and should be complemented with another pain-depressed behavior endpoint. The affective and sensory components of pain were selectively affected by Ibu because the same dose of Ibu was ineffective to block writhing behavior but effective to improve pain-depressed behaviors (saccharin preference).
Consequently, hedonic behaviors are more sensitive, and translational readouts to evaluate analgesics and changes in the expression of hedonic behavior – such as sweet taste preference described in this study – can be used as a primary outcome measure to evaluate pain in mice and may complement the more traditional procedures used to assess candidate analgesics.
Disclosure
The authors report no conflicts of interest in this work.
References
Mogil JS, Crager SE. What should we be measuring in behavioral studies of chronic pain in animals? Pain. 2004;112(1/2):12–15. | |
Vierck CJ, Hansson PT, Yezierski RP. Clinical and pre-clinical pain assessment: are we measuring the same thing? Pain. 2008;135(1/2):7–10. | |
Mogil JS. Animal models of pain: progress and challenges. Nat Rev Neurosci. 2009;10(4):283–294. | |
Cobos EJ, Portillo-Salido, E. “Bedside-to-bench” behavioral outcomes in animal models of pain: beyond the evaluation of reflexes. Curr Neuropharmacol. 2013;11(6):560–591. | |
Colpaert FC, Tarayre JP, Alliaga M, Bruins Slot LA, Attal N, Koek W. Opiate self-administration as a measure of chronic nociceptive pain in arthritic rats. Pain. 2001;91(1/2):33–45. | |
Martin TJ, Buechler NL, Kahn W, Crews JC, Eisenach JC. Effects of laparotomy on spontaneous exploratory activity and conditioned operant responding in the rat: a model for postoperative pain. Anesthesiology. 2004;101(1):191–203. | |
Martin TJ, Kahn WR, Eisenach JC. Abdominal surgery decreases food-reinforced operant responding in rats: relevance of incisional pain. Anesthesiology. 2005;103(3):629–637. | |
Langford DJ, Bailey AL, Chanda ML, et al. Coding of facial expressions of pain in the laboratory mouse. Nat Methods. 2010;7(6):447–449. | |
Negus SS, Vanderah TW, Brandt MR, Bilsky EJ, Becerra L, Borsook D. Preclinical assessment of candidate analgesic drugs: recent advances and future challenges. J Pharmacol Exp Ther. 2006;319(2):507–514. | |
Stevenson GW, Bilsky EJ, Negus SS. Targeting pain-suppressed behaviors in preclinical assays of pain and analgesia: effects of morphine on acetic acid-suppressed feeding in C57BL/6J mice. J Pain. 2006;7(6):408–416. | |
Stevenson GW, Cormier J, Mercer H, et al. Targeting pain-depressed behaviors in preclinical assays of pain and analgesia: drug effects on acetic acid-depressed locomotor activity in ICR mice. Life Sci. 2009;85(7/8):309–315. | |
Morgan D, Carter CS, DuPree JP, Yezierski RP, Vierck CJ Jr. Evaluation of prescription opioids using operant-based pain measures in rats. Exp Clin Psychopharmacol. 2008;16(5):367–375. | |
Kroenke K, Wu J, Bair MJ, Krebs EE, Damush TM, Tu W. Reciprocal relationship between pain and depression: a 12-month longitudinal analysis in primary care. J Pain. 2011;12(9):964–973. | |
Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: a biopsychosocial perspective. Biol Psychiatry. 2003;54(3):399–409. | |
Fernandez E, Turk DC. Sensory and affective components of pain: separation and synthesis. Psychol Bull. 1992;112(2):205–217. | |
Pfaffmann C. The pleasures of sensation. Psychol Rev. 1960;67: 253–268. | |
Cytawa J, Trojniar W. Hedonesthesia: the nervous process determining motivated ingestive behavior. Acta Neurobiol Exp (Wars). 1978;38(2/3):139–151. | |
Valenstein ES, Kakolewski JW, Cox VC. Sex differences in taste preference for glucose and saccharin solutions. Science. 1967;156(3777):942–943. | |
Willner P, Muscat R, Papp M. Chronic mild stress-induced anhedonia: a realistic animal model of depression. Neurosci Biobehav Rev. 1992;16(4):525–534. | |
Harkin A, Houlihan DD, Kelly JP. Reduction in preference for saccharin by repeated unpredictable stress in mice and its prevention by imipramine. J Psychopharmacol. 2002;16(2):115–123. | |
Lazarus H, Neumann C. Assessing undertreatment of pain: the patients’ perspectives. J Pharm Care Pain Symptom Control. 2001;9(4):5–34. | |
Zhang L, Zhang X, Westlund KN. Restoration of spontaneous exploratory behaviors with an intrathecal NMDA receptor antagonist or a PKC inhibitor in rats with acute pancreatitis. Pharmacol Biochem Behav. 2004;77(1):145–153. | |
Matson DJ, Broom DC, Cortright DN. Locomotor activity in a novel environment as a test of inflammatory pain in rats. Methods Mol Biol. 2010;617:67–78. | |
Volkers AC, Tulen JH, van den Broek WW, Bruijn JA, Passchier J, Pepplinkhuizen L. Motor activity and autonomic cardiac functioning in major depressive disorder. J Affect Disord. 2003;76(1–3):23–30. | |
Bennabi D, Vandel P, Papaxanthis C, Pozzo T, Haffen E. Psychomotor retardation in depression: a systematic review of diagnostic, pathophysiologic, and therapeutic implications. Biomed Res Int. 2013; 2013:158746. | |
Le Bars D, Gozariu M, Cadden SW. Animal models of nociception. Pharmacol Rev. 2001;53(4):597–652. | |
Zimmermann M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain. 1983;16(2):109–110. | |
Andrews N, Legg E, Lisak D, et al. Spontaneous burrowing behaviour in the rat is reduced by peripheral nerve injury or inflammation associated pain. Eur J Pain. 2012;16(4):485–495. | |
Negus SS, Neddenriep B, Altarifi AA, Carroll FI, Leitl MD, Miller LL. Effects of ketoprofen, morphine, and kappa opioids on pain-related depression of nesting in mice. Pain. 2015;156(6):1153–1160. | |
Do Carmo GP, Stevenson GW, Carlezon WA, Negus SS. Effects of pain- and analgesia-related manipulations on intracranial self-stimulation in rats: further studies on pain-depressed behavior. Pain. 2009;144(1/2):170–177. | |
Stevenson GW, Mercer H, Cormier J, et al. Monosodium iodoacetate-induced osteoarthritis produces pain-depressed wheel running in rats: implications for preclinical behavioral assessment of chronic pain. Pharmacol Biochem Behav. 2011;98(1):35–42. | |
Cobos EJ, Ghasemlou N, Araldi D, Segal D, Duong K, Woolf CJ. Woolf inflammation-induced decrease in voluntary locomotion in mice: an operant test for evaluating inflammatory pain and analgesia. Pain. 2012;153(4):876–884. | |
Tanimoto S, Nakagawa T, Yamauchi Y, Minami M, Satoh M. Differential contributions of the basolateral and central nuclei of the amygdala in the negative affective component of chemical somatic and visceral pains in rats. Eur J Neurosci. 2003;18(8):2343–2350. | |
Menétrey D, Gannon A, Levine JD, Basbaum AI. Expression of c-fos protein in interneurons and projection neurons of the rat spinal cord in response to noxious somatic, articular, and visceral stimulation. J Comp Neurol. 1989;285(2):177–195. | |
Burstein AH, Reiss WG, Kantor E, Anderson GD. Cytochrome P450 3A4 activity in premenopausal and postmenopausal women, based on 6-beta-hydroxycortisol: cortisol ratios. Pharmacotherapy. 1998;18(6):1271–1276. | |
Bandler R, Shipley MT. Columnar organization in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci. 1994;17(9):379–389. | |
Clement CI, Keay KA, Podzebenko K, Gordon BD, Bandler R. Spinal sources of noxious visceral and noxious deep somatic afferent drive onto the ventrolateral periaqueductal gray of the rat. J Comp Neurol. 2000;425(3):323–344. | |
Nakagawa T, Katsuya A, Tanimoto S, et al. Differential patterns of c-fos mRNA expression in the amygdaloid nuclei induced by chemical somatic and visceral noxious stimuli in rats. Neurosci Lett. 2003;344(3):197–200. | |
Jensen TS. Opioids in the brain: supraspinal mechanisms in pain control. Acta Anaesthesiol Scand. 1997;41(1 Pt 2):123–132. | |
Hummel M, Lu P, Cummons TA, Whiteside GT. The persistence of a long-term negative affective state following the induction of either acute or chronic pain. Pain. 2008;140(3):436–445. | |
Rutten K, De Vry J, Robens A, Tzschentke TM, van der Kam EL. Dissociation of rewarding, anti-aversive and anti-nociceptive effects of different classes of anti-nociceptives in the rat. Eur J Pain. 2011;15(3):299–305. | |
Uzbay T, Kose A, Kayir H, Ulusoy G, Celik T. Sex-related effects of agmatine on caffeine-induced locomotor activity in Swiss Webster mice. Eur J Pharmacol. 2010;630(1–3):69–73. | |
Kallman WM, Isaac W. The effects of age and illumination on the dose-response curves for three stimulants. Psychopharmacologia. 1975;40(4):313–318. | |
Garcia AM, Cardenas FP, Morato S. The effects of pentylenetetrazol, chlordiazepoxide and caffeine in rats tested in the elevated plus-maze depend on the experimental illumination. Behav Brain Res. 2011;217(1):171–177. | |
Gschossmann JM, Buenger L, Adam B, et al. Diurnal variation of abdominal motor responses to colorectal distension and plasma cortisol levels in rats. Neurogastroenterol Motil. 2001;13(6):585–589. | |
Crabbe JC, Wahlsten D, Dudek BC. Genetics of mouse behavior: interactions with laboratory environment. Science. 1999;284(5420):1670–1672. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.