Back to Journals » Neuropsychiatric Disease and Treatment » Volume 16
Change in Functional Brain Activation Patterns Induced by Olfactory Stimulation in Multiple Sclerosis
Authors OuYang Q, Wang Y, Zhang Y, Yu M, Wang X
Received 8 March 2020
Accepted for publication 1 May 2020
Published 9 June 2020 Volume 2020:16 Pages 1451—1458
DOI https://doi.org/10.2147/NDT.S252933
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jun Chen
Qingrong OuYang,1,* Yinxu Wang,2,* Yun-wei Zhang,1 Ming Yu,1 Xiaoming Wang3
1Department of Neurology, Suining Central Hospital, Sui Ning 629000, People’s Republic of China; 2Department of Rehabilitation Medicine, Affiliated Hospital of North Sichuan Medical College, Nanchong 637000, People’s Republic of China; 3Department of Neurology, Affiliated Hospital of North Sichuan Medical College, Nanchong 637000, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Ming Yu; Xiaoming Wang Email [email protected]; [email protected]
Background: Olfactory disorder is one of the commonly appearing symptoms in diseases like Parkinson’s disease (PD) and Alzheimer’s disease (AD). However, reports of olfactory changes in multiple sclerosis (MS) are scarce and usually ignored or seldom recognized by clinicians. The majority of current research is based on subjective answers obtained by smelling odorants.
Objective: To gain better insights into the central brain regions involved in the olfactory process. We measured preliminary contrast assessment characteristics of brain activation in MS patients and healthy controls using functional magnetic resonance imaging (fMRI) under an odor stimulation task.
Methods: Olfactory event-related fMRI was used to assess the olfactory network neuronal activity during passively inhaled volatile gases of lavender and rose solutions alternately in 18 MS patients and 20 healthy sex- and age-matched adults. Spearman correlation analysis was conducted between the lesions in central and olfactory-induced activated brain regions.
Results: We observed significant reductions in the number of activated brain areas compared with healthy controls in MS patients under a standard activation mode; the right insula, right amygdala, right inferior frontal gyrus, right frontal middle gyrus, and left supramarginal gyrus were activated in MS patients (t = 2.04, P < 0.05). Meanwhile, the intensity of the activated olfactory brain network revealed attenuation. The Spearman correlation analysis indicated the distribution and number of demyelination lesions, exerting a little impact on major activation of brain regions during olfactory stimulation (r = – 0.524, P = 0.054).
Conclusion: This study establishes that olfaction-related brain regions were altered in patients with MS confirmed by fMRI. The finding refreshes the awareness that olfactory disturbance involved just in structural pathology like olfactory bulbs and tracts or olfactory sulcus, which reportedly is responsible for the deficits.
Keywords: multiple sclerosis, olfactory, magnetic resonance imaging, brain
Introduction
Multiple sclerosis (MS) is a common central neural system demyelination disease in young and middle-aged people, which can cause inflammatory demyelination and neurodegeneration in the brainstem, cortex, cerebellum, spinal cord, and other neural tissues. Primary manifestation can be performed with visual, sensory, motor, and paroxysmal symptoms.1 However, some emerging evidence has indicated that the deficits in smell sense is a feature of MS.2 The current data revealed that the olfactory disturbance of MS correlates with the lesion load in olfactory brain regions in the inferior frontal and temporal lobes,3,4 olfactory brain volume,5 and the extent of disability.5–7 Reportedly, olfactory dysfunction is relevant to relapses and disease activity8 and could be an early predictor of disease progression in MS.9 The existing data suggest that olfactory disorders in MS could be used to monitor and predict disease progression in relapsing-remitting MS (RRMS) patients and even to assess the response to treatment.
To date, studies have reported psychophysical, electrophysiological, radiological, and pathological results about olfactory dysfunction of MS. The present evidence revealed that the mean incidence of olfactory function in MS patients was 11–50% (average: 35.6%) through psychophysical methods.10 Several commonly used olfactory evaluation methods include the Sniffin’ Sticks test, University of Pennsylvania Smell Identification Test, simplified odor Identification test, T/T olfactory meter test, and Connecticut Chemical sensory Clinical Research Center olfactory test among the category. Of note, the results differ in nature and extent with different methods. Changes in electrophysiology revealed that an increase in latency and a decrease in amplitude occurred in patients with olfactory impairment in MS,11 suggesting that the olfactory loop could be damaged from olfactory bulbs and tracts to the orbitofrontal and insular cortices, along with the rostrum–medial regions of the temporal lobe.12 Furthermore, radiological data supported the notion that the structure pathology related to olfactory impairment could occur in peripheral and central olfactory structure.3–5,13
Functional magnetic resonance imaging (fMRI) is a new technology based on blood oxygen-dependent level imaging, which can visualize the brain activity when subjects perform a specific task, and has become an up-and-coming technique to analyze the function of the central brain.14 In this study, the combination of event-related design with fMRI provided the advantages that high temporal and spatial consistency, high resolution, non-invasive, repeated scanning, and dynamic monitoring to investigate the sensory modality, and was independent of subjects, response bias to a certain extent. Furthermore, we assessed the characteristics of brain activation in patients with MS during olfactory stimuli. To the authors’ knowledge, this is the first study to use fMRI associated with the event-related design to investigate the neuronal activity of MS on olfactory processing.
Methods
Subjects
The diagnosis of MS in 18 patients was defined according to the McDonald Criteria, 2010 version,15 and 20 sex- and age-matched controls participated in the study. None of the significant differences in demographic variables of the two groups was confirmed using a nonparametric χ2-test, of which 4 RRMS patients with active disease were included in the MS group and the other 14 patients with clinically stable RRMS.
The exclusion criteria were as follows: (a) recent history of acute upper respiratory tract infection, toxic exposure, nasal diseases (eg, paranasal sinus and history of nasal surgery/trauma), or abnormal findings during nasal examination; (b) history of head trauma and the physical lesions have been revealed by brain MRI; (c) presence or history of other neuronal disorders that could affect olfactory function, such as Parkinson’s disease, Alzheimer’s disease, anxiety and depression, cognitive impairment, or schizophrenia; (d) history of alcohol addiction, drug dependence, allergy, long-term smoking, and/or high dose of glucocorticoid treatment.
This case–control study was conducted in accordance with the Declaration of Helsinki on ethical principle and approved by the Ethics Committee of the Affiliated Hospital of North Sichuan Medical College [approval No. 2018ER (A) 005]. All participants provided written informed consent to be included in the study.
Test Process and Design Parameters
Psychophysical Test of Olfactory Function
Argentine Hyposmia Rating Scale (AHRS)16 was used to evaluate olfactory function in all subjects before performing fMRI, all subjects were required to answer six self-administered questions related to daily experienced odors (eg, smell of fresh flowers, gasoline, decaying matter, perfume, airtight smell, and smell of soot). The total score is 25; scores ≤22 implies hyposmia and >23 denotes healthy olfactory.
Odorant and Olfactory Stimulator
The Emerging Tech Trans (ETT) olfactory stimulator (Hershey Company, USA) was operated on subjects when they accepted the fMRI scanning. It comprised a control system and odor source box. The connection between the two was attained by a polytetrafluoroethylene tube, which was then transported to the subject’s nasal terminal mask through an odor transmission tube; the operating system could sensitively control the stimulation event. Compared with traditional manual olfactory stimulation, the ETT olfactory stimulator could efficiently convert between different odors and accurately control the airflow and concentration. Moreover, the lavender and rose scents were adopted in this study; both are pleasant odors that could avoid additional stimulation to the trigeminal nerve. No significant difference in brain function activation area induced by the two odors stimuli.17 We used superthreshold concentration (0.8 %) of volatile gases with lavender and rose smells as alternating stimuli during fMRI scanning. The paradigm could minimize the impact of olfactory adaptation and desensitization because of stimulation with a single odor. All subjects remained awake with their eyes closed and maintained consistent breathing; no announcement was made ahead of olfactory stimulation to avoid autonomic imaging activity about odorant. During scanning, the smell and airflow rate were constant, and the environment was kept at a consistent temperature. The stimulation mode was as follows: 42 s of no stimulation period alternately followed by a 6-s (3 L/min) odor stimulation with either lavender or rose smell; the procedure was repeated 12 times (details in Figure 1).
 |
Figure 1 The sequence of olfactory stimulation during fMRI scanning.
Note: |
Imaging Parameters
Image acquisition was performed on a 3.0-T superconductive magnetic resonance scanner (GE Company, USA) and 32-channel head coil received fMRI data collected by the gradient echo plane imaging sequence. The scanning parameters were as follows: repetition time, 2000 ms; echo time, 30 ms; reverse angle, 90°; scanning layers, 35; layer thickness, 4 mm; matrix, 64 × 64; visual field, 24 cm × 24 cm; time points, 125.
Data Processing and Analysis
The functional images were preprocessed using statistical parametric mapping (SPM8) and MATLAB 2013a software, including the removal of the first five time-points, slice timing correction, head motion correction, normalization, and spatial smoothing. For ontoanalysis, a general linear model (GLM) matching the task block’s time vector using hemodynamic response was executed for each run. The motion correction parameters for translations and rotations in each direction were added to improve the model. Individual contrast maps were generated by the techniques mentioned above. One-sample t-tests were used to investigate both within-group activations in these contrasts, and two-sample t-tests were used to investigate the statistical inference of group effects and differences between the MS and control groups. In this study, the significance was set at P < 0.05 without correction and the cluster correction with a minimum cluster size of 10 voxels. The distribution and quantity of lesions in each brain region were counted on T2-weighted images in the MS group. Correlations between demyelination lesions and activated brain regions were analyzed by SPSS 17.0 software. The statistical threshold was set at P < 0.05.
Results
General Characteristics and Olfactory Assessment
In the MS group, 1 patient was excluded because of head movement. A total of 17 patients, including 4 men and 13 women (age: 45.14 ± 14.84 [range, 25 to 69] years), were included in this study. The disease course was 1 month to 20 years. In the control group, a total of 20 cases, 7 men and 13 women (average age: 42.63 ± 12.70 years) were enrolled. Overall, MS patients revealed significantly lower AHRS score (18.86 ± 3.7; P = 0.023) and a higher incidence of impaired identification of common odors compared with the HC group (detailed in Table 1).
 |
Table 1 Demographic and Clinical Data of Multiple Sclerosis (MS) and Healthy Control (HC) Groups |
Distribution of Activation Throughout the Brain
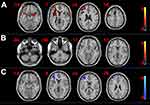
In the control group, the task-activated brain regions responding to the olfactory process were observed. The activated regions of the brain were as follows: bilateral middle frontalis, bilateral insular lobe, bilateral superior marginal gyrus, bilateral orbital frontalis, right thalamus, right anterior central gyrus, bilateral cingulate gyrus, bilateral hippocampus, bilateral almond nucleus, and bilateral superior frontalis gyrus (Figure 2A, Table 2; t = 2.11, P < 0.05). The task-activated brain regions in the MS group were as follows: right cerebellum, left insular lobe, left superior temporal gyrus, right inferior frontalis gyrus (Figure 2B, Table 3; t = 2.19, P < 0.05). Compared with the control group, the activation of the right insular lobe, right almond nucleus, right inferior forehead gyrus, right middle forehead gyrus, and left superior marginal gyrus were reduced in the MS group (Figure 2C, Table 4; t = 2.04, P < 0.05).
 |
Table 2 Montreal Neurological Institute Coordinates, Activator Voxels, and Intensities (T Values) of Activated Brain Regions in Healthy Controls |
 |
Table 3 Montreal Neurological Institute Coordinates, Activator Voxels, and Intensities (T Values) of Activated Brain Regions in Multiple Sclerosis Patients |
Correlation Between the Distribution of Demyelination Lesions and Task-Activated Brain Regions
In the MS group, the distribution and number of demyelination lesions were counted on T2-weighted images, and the frequency of lesions in each brain region was analyzed. There were 12 lateral periventricular lesions (24% frequency), 10 proximal cortical lesions (20% frequency), 10 frontotemporal lobe lesions (18% frequency), 8 brainstem lesions (16% frequency), 6 hippocampal lesions (12% frequency), and 4 lesions in other brain regions (10% frequency). The correlation analysis between the above data and olfaction-related main activated brain regions showed no significant correlation between the distribution of lesions in each brain region and the frontal lobe activated by olfactory activation in the MS group; however, there was a trend indicating that an increased number of lesions was associated with weakened olfactory activation among brain regions (Figure 3; r ≥ 0.524, P ≤ 0.054).
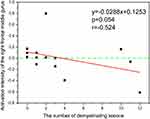 |
Figure 3 The relationship between number of demyelinating lesions and main activated regions of olfactory function in multiple sclerosis patients (n=14). |
Discussion
Olfactory disorder has become an important biomarker in neurodegenerative diseases of the early stage. In 2015, the International Association of Parkinson’s Disease and Dyskinesia included olfactory disorders in the diagnostic criteria for Parkinson’s disease.18 Emerging evidence broadened this scope of olfactory involvement, suggesting that olfactory impairment is a feature of central brain demyelinating diseases as MS. The first research reported olfactory disorder in MS in 1971.19 The available evidence support that olfactory disturbance is frequent in MS, although some studies opposed this claim.2,20 The structure pathology study revealed that the olfactory bulb and olfactory sulcus significantly atrophied in patients with MS.5,21 The pathological data provided evidence that the number of lesions in the subfrontal and temporal lobes correlate with the degree of olfactory impairment in MS patients.4 Another study on the method of rest state fMRI reported that the decrease in fractional anisotropy (FA) and mean diffusivity (MD) increase in MS central brain regions, which correlate with the olfactory formation.22 Usually, electrophysiological examination implies olfactory event-related potential (OERP), which revealed that the OERP waveform was not elicited in some patients in the MS group; meanwhile, the amplitude of OERPs declined and the incubation period prolonged.23 However, the investigation by approaches that could visualize brain activation during odor perception and olfactory information process is lacking.
In this study, AHRS scales were adopted to assess the olfactory function in MS patients. The MS group revealed significantly worse performance compared with the control group in the six commonly recognizable odors, which corroborated previous studies.8,24 Of note, cortical regions responsible for odor perception comprise primary olfactory structures, of which piriform cortex is the first cortical projection target of olfactory information; additional auxiliary regions include the olfactory tubercle, anterior olfactory nucleus, periamygdaloid cortex. Higher-ordering connected to the widely secondary olfactory cortex, namely the additional amygdala subnuclei, orbitofrontal cortex (OFC), hippocampus, parahippocampal gyrus, cingulate cortex, insula, striatum hypothalamus, and the mediodorsal thalamus.25,26 Moreover, right orbitofrontal gyrus and right piriform cortex are responsible for olfactory memory and odor recognition; the emotional correlate to odor is reportedly located in the left orbitofrontal gyrus, left insular lobe, left piriform cortex, left almond nucleus, and left suprafrontal gyrus.27 In this study, regions of activation in healthy controls were predominately localized in the secondary olfactory structures, including bilateral middle frontal lobe, bilateral insular lobe, bilateral superior marginal gyrus, bilateral orbital frontalis, right thalamus, right anterior central gyrus, bilateral cingulate gyrus, bilateral hippocampus, bilateral suprafrontal gyrus. The distinct stimuli paradigm revealed elegant activations of the central olfactory regions. However, the piriform cortex revealed no notable activation in healthy controls in this study, which could be attributed to the piriform cortex proximity to the insular and a relatively small structure in the cerebral region, which could hardly identify the activation in this region.28 Moreover, the number of activated regions and activation voxels in the right hemisphere, as well as the activation intensities, were greater than those in the left hemisphere, suggesting a possible right-sided bias in olfactory processing.
The assessment of MS patients revealed that the right cerebellum, left insular lobe, left superior temporal gyrus, and right inferior frontal gyrus were activated. The inferior frontal region has been proved to be pronounced activated during the familiarity, emotion, and intensity judgments during the odor reception.29 Previous studies revealed that insular lobe responds to the processing of the emotional aspects of smells. Accordingly, the activation of these regions indicated that the function in semantic encode and emotional perception about odors in MS patients was preserved. However, significantly fewer activated brain regions were present, and the activated voxel and activation intensities of the activated brain regions were markedly lower than those in the control group, providing relatively clear evidence for the decrease in the activity in the entire olfactory central of MS patients. Specific to the left insular lobe, right amygdala nucleus, right inferior frontal gyrus, and right middle frontal gyrus exhibited attenuated odor-related brain responses, which more belonged to the auxiliary regions in the secondary olfactory structures. A obvious hint was present for the highest-order olfactory cortex that revealed disturbance, which implied a complicated pattern of the olfactory network maladjustment in MS. From the perspective of fMRI, this study corroborated the previous landmark study, which unequivocally validated the direct proportion of the plaque activity in the olfactory-related central brain region with olfactory deficits.3,30 However, the study on olfactory structure revealed that the olfactory tract and bulb atrophied in early stages of olfactory impairment,21 suggesting the change of highest-order olfactory cortex might be secondary to the primary neuron deficit. In addition, the visualized changes were present in the olfaction-related brain network in MS, and whether differential activation of distinct brain regions exhibited a hyperactivation or compensatory olfactory process, as in Parkinson’s Disease,31 warrants further investigation.
Considering the pathological characteristics of multifocal in encephalic MS, it remains unclear whether the number of lesions affect the activation of smell-responsive brain regions. The correlation between the number of lesions and the middle frontal gyrus of the main activated brain region was investigated, but no significant association was established, and some exact causes need to be considered as follows. First, the distribution of these lesions primarily localized in the white matter in cerebral, yet the fMRI signals were weakened in white matter images and the gray matter in which distributed more blood vessels produce stronger signals during a task.32,33 Second, the study encountered methodological problems with fewer participants, which insufficiently reflect the differences in the distributions of lesions in different disease states. Specifically, a negative tendency between the number of lesions and the activation in middle frontal gyrus of MS patients appeared, which could potentially lead to the assumption that widespread demyelination and neurodegeneration in the late stages of disease, with the state of high-intensity lesions, could mediate the severe damage in olfaction-process regions. The trend implication is consistent with the findings of previous studies.8
There are some limitations to this study worth acknowledging. We were unable to group patients in terms of the pivotal element as the stage of disease, the disability extent, the severity of olfactory impairment, consequently from the small sample size. It will be necessary to expand the sample size, and the olfactory content should be combined with olfactory event-related potentials, structural imaging, and other objective means, together with biochemical immune markers in further study, which, perhaps, will provide a better understanding of the underlying pathophysiological mechanisms and the role of olfactory deficit in MS.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure
All authors report no conflicts of interest.
References
1. Good KP, Tourbier IA, Moberg P, et al. Unilateral olfactory sensitivity in multiple sclerosis. Physiol Behav. 2017;168:24–30. doi:10.1016/j.physbeh.2016.10.017
2. DeLuca GC, Joseph A, George J, et al. Olfactory pathology in central nervous system demyelinating diseases. Brain Pathol. 2015;25(5):543–551. doi:10.1111/bpa.12209
3. Zorzon M, Ukmar M, Bragadin LM, et al. Olfactory dysfunction and extent of white matter abnormalities in multiple sclerosis: a clinical and MR study. Mult Scler. 2000;6(6):386–390. doi:10.1177/135245850000600605
4. Doty RL, Cheng LI, Mannon LJ, et al. Olfactory dysfunction in multiple sclerosis: relation to plaque load in inferior frontal and temporal lobes. Ann N Y Acad Sci. 1998;855:781–786. doi:10.1111/j.1749-6632.1998.tb10658.x
5. Goektas O, Schmidt F, Bohner G, et al. Olfactory bulb volume and olfactory function in patients with multiple sclerosis. Rhinology. 2011;49(2):221–226. doi:10.4193/Rhino10.136
6. Rolet A, Magnin E, Millot J, et al. Olfactory dysfunction in multiple sclerosis: evidence of a decrease in different aspects of olfactory function. Eur Neurol. 2012;69:166–170. doi:10.1159/000345482
7. Dahlslett SB, Goektas O, Schmidt F, et al. Psychophysiological and electrophysiological testing of olfactory and gustatory function in patients with multiple sclerosis. Eur Arch Otorhinolaryngol. 2012;269(4):1163–1169. doi:10.1007/s00405-011-1812-7
8. Lutterotti A, Vedovello M, Reindl M, et al. Olfactory threshold is impaired in early, active multiple sclerosis. Mult Scler J. 2011;17(8):964–969. doi:10.1177/1352458511399798
9. Wood H. Neurophysiological and olfactory biomarkers for multiple sclerosis. Nat Rev Neurol. 2019;15(1):1. doi:10.1038/s41582-018-0119-3
10. Joseph A, DeLuca GC. Back on the scent: the olfactory system in CNS demyelinating diseases. J Neurol Neurosurg Psychiatry. 2016;87(10):1146–1154. doi:10.1136/jnnp-2015-312600
11. Hawkes CH, Shephard BC, Kobal G. Assessment of olfaction in multiple sclerosis: evidence of dysfunction by olfactory evoked response and identification tests. J Neurol Neurosurg Psychiatry. 1997;63(2):145–151. doi:10.1136/jnnp.63.2.145
12. Barresi M, Ciurleo R, Giacoppo S, et al. Evaluation of olfactory dysfunction in neurodegenerative diseases. J Neurol Sci. 2012;323(1–2):16–24. doi:10.1016/j.jns.2012.08.028
13. Holinski F, Schmidt F, Dahlslett SB, et al. MRI study: objective olfactory function and CNS pathologies in patients with multiple sclerosis. Eur Neurol. 2014;72:157–162. doi:10.1159/000362165
14. Tabert MH, Steffener J, Albers MW, et al. Validation and optimization of statistical approaches for modelling odorant-induced fMRI signal changes in olfactory-related brain areas. Neuroimage. 2007;34:1375–1390. doi:10.1016/j.neuroimage.2006.11.020
15. Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69(2):292–302. doi:10.1002/ana.22366
16. Vernetti PM, Lloret SP, Rossi M, et al. Validation of a new scale to assess olfactory dysfunction in patients with Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(4):0–361. doi:10.1016/j.parkreldis.2011.12.001
17. Toledano A, Borromeo S, Luna G, et al. Objective assessment of olfactory function using functional magnetic resonance imaging. Acta Otorrinolaringol. 2012;63:280–285. doi:10.1109/TIM.2010.2057531
18. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30(12):1591–1601. doi:10.1002/mds.26424
19. Wender M, Szmeja Z. Examination of the hearing, the vestibular system functions, taste and olfactory systems in patients with disseminated sclerosis. Neurol Neurochir Pol. 1971;5:179–184.
20. Zimmerman HM, Netsky MG. The pathology of multiple sclerosis. Res Publ Assoc Res Nerv Ment Dis. 1950;28:271–312.
21. Tanik N, Serin HI, Celikbilek A, et al. Olfactory bulb and olfactory sulcus depths are associated with disease duration and attack frequency in multiple sclerosis patients. J Neurol Sci. 2015;358(1–2):304–307. doi:10.1016/j.jns.2015.09.016
22. Erb-Eigner K, Bohner G, Goektas O, et al. Tract-based spatial statistics of the olfactory brain in patients with multiple sclerosis. J Neurol Sci. 2014;346(1–2):235–240. doi:10.1016/j.jns.2014.08.039
23. Caminiti F, De Salvo S, De Cola MC, et al. Detection of olfactory dysfunction using olfactory event related potentials in young patients with multiple sclerosis. PLoS One. 2014;9(7):e103151. doi:10.1371/journal.pone.0103151
24. Lucassen EB, Turel A, Knehans A, et al. Olfactory dysfunction in multiple sclerosis: a scoping review of the literature. Mult Scler Relat Disord. 2016;6:1–9. doi:10.1016/j.msard.2015.12.002
25. Fjældstad A, Fernandes HM, Van Hartevelt TJ, et al. Brain fingerprints of olfaction: a novel structural method for assessing olfactory cortical networks in health and disease. Sci Rep. 2017;7(1):1–13. doi:10.1038/srep42534
26. Zhou G, Lane G, Cooper SL, et al. Characterizing functional pathways of the human olfactory system. Elife. 2019;8. doi:10.7554/eLife.47177
27. Royet JP, Plailly J. Lateralization of olfactory processes. Chem Senses. 2004;29(8):731–745. doi:10.1093/chemse/bjh067
28. Cerf-Ducastel B, Murphy C. Neural substrates of cross-modal olfactory recognition memory: an fMRI study. Neuroimage. 2006;31(1):386–396. doi:10.1016/j.neuroimage.2005.11.009
29. Plailly J, Bensafi M, Pachot-Clouard M, et al. Involvement of right piriform cortex in olfactory familiarity judgments. Neuroimage. 2005;24(4):1032–1041. doi:10.1016/j.neuroimage.2004.10.028
30. Doty RL, Li C, Mannon LJ, et al. Olfactory dysfunction in multiple sclerosis. N Engl J Med. 1997;336:1918–1919. doi:10.1016/j.msard.2018.02.032
31. Moessnang C, Frank G, Bogdahn U, et al. Altered activation patterns within the olfactory network in Parkinson’s disease. Cereb Cortex. 2011;21(6):1246–1253. doi:10.1093/cercor/bhq202
32. Duvernoy HM, Delon S, Vannson JL. Cortical blood vessels of the human brain. Brain Res Bull. 1981;7(5):519–579. doi:10.1016/0361-9230(81)90007-1
33. Turner R, Bihan DL, Moonen CTW, et al. Echo‐planar time course MRI of cat brain oxygenation changes. Magn Reson Med. 1991;22(1):159–166. doi:10.1002/mrm.1910220117
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.