Back to Journals » International Journal of Nanomedicine » Volume 17
Cell-Seeded Biomaterial Scaffolds: The Urgent Need for Unanswered Accelerated Angiogenesis
Authors Shokrani H, Shokrani A ![]() , Sajadi SM, Seidi F, Mashhadzadeh AH
, Sajadi SM, Seidi F, Mashhadzadeh AH ![]() , Rabiee N
, Rabiee N ![]() , Saeb MR
, Saeb MR ![]() , Aminabhavi T
, Aminabhavi T ![]() , Webster TJ
, Webster TJ ![]()
Received 14 December 2021
Accepted for publication 22 February 2022
Published 12 March 2022 Volume 2022:17 Pages 1035—1068
DOI https://doi.org/10.2147/IJN.S353062
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Lijie Grace Zhang
Hanieh Shokrani,1,* Amirhossein Shokrani,2,* S Mohammad Sajadi,3,4 Farzad Seidi,5 Amin Hamed Mashhadzadeh,6 Navid Rabiee,7,8 Mohammad Reza Saeb,9 Tejraj Aminabhavi,10,11 Thomas J Webster12,13
1Department of Chemical Engineering, Sharif University of Technology, Tehran, Iran; 2Department of Mechanical Engineering, Sharif University of Technology, Tehran, Iran; 3Department of Nutrition, Cihan University-Erbil, Erbil, 625, Iraq; 4Department of Phytochemistry, SRC, Soran University, Soran, KRG, 624, Iraq; 5Jiangsu Co–Innovation Center for Efficient Processing and Utilization of Forest Resources and International Innovation Center for Forest Chemicals and Materials, Nanjing Forestry University, Nanjing, 210037, People’s Republic of China; 6Mechanical and Aerospace Engineering, School of Engineering and Digital Sciences, Nazarbayev University, Nur-Sultan, 010000, Kazakhstan; 7Department of Physics, Sharif University of Technology, Tehran, Iran; 8School of Engineering, Macquarie University, Sydney, New South Wales, 2109, Australia; 9Department of Polymer Technology, Faculty of Chemistry, Gdańsk University of Technology, Gdańsk, Poland; 10School of Advanced Sciences, KLE Technological University, Hubballi, Karnataka, 580 031, India; 11Department of Chemistry, Karnatak University, Dharwad, 580 003, India; 12School of Health Sciences and Biomedical Engineering, Hebei University, Tianjin, People’s Republic of China; 13Center for Biomaterials, Vellore Institute of Technology, Vellore, India
*These authors contributed equally to this work
Correspondence: S Mohammad Sajadi; Navid Rabiee, Email [email protected]; [email protected]; [email protected]
Abstract: One of the most arduous challenges in tissue engineering is neovascularization, without which there is a lack of nutrients delivered to a target tissue. Angiogenesis should be completed at an optimal density and within an appropriate period of time to prevent cell necrosis. Failure to meet this challenge brings about poor functionality for the tissue in comparison with the native tissue, extensively reducing cell viability. Prior studies devoted to angiogenesis have provided researchers with some biomaterial scaffolds and cell choices for angiogenesis. For example, while most current angiogenesis approaches require a variety of stimulatory factors ranging from biomechanical to biomolecular to cellular, some other promising stimulatory factors have been underdeveloped (such as electrical, topographical, and magnetic). When it comes to choosing biomaterial scaffolds in tissue engineering for angiogenesis, key traits rush to mind including biocompatibility, appropriate physical and mechanical properties (adhesion strength, shear stress, and malleability), as well as identifying the appropriate biomaterial in terms of stability and degradation profile, all of which may leave essential trace materials behind adversely influencing angiogenesis. Nevertheless, the selection of the best biomaterial and cells still remains an area of hot dispute as such previous studies have not sufficiently classified, integrated, or compared approaches. To address the aforementioned need, this review article summarizes a variety of natural and synthetic scaffolds including hydrogels that support angiogenesis. Furthermore, we review a variety of cell sources utilized for cell seeding and influential factors used for angiogenesis with a concentrated focus on biomechanical factors, with unique stimulatory factors. Lastly, we provide a bottom-to-up overview of angiogenic biomaterials and cell selection, highlighting parameters that need to be addressed in future studies.
Keywords: tissue engineering, polymeric scaffolds, biomaterials, angiogenesis, neovascularization, biomechanical factors
Introduction
Organ transplantation is a long-term treatment for patients with end-stage diseases. Recent advances in tissue engineering have placed a focus on in vitro constructs for organ transplantation. However, most studies have been limited to simple tissues, such as skin and cardiac patches, mainly because of the need for larger amounts of vascularization in other tissues.1,2 As a consequence, most damaged tissues in the human body that cannot regenerate on their own are replaced by organs solely obtained from donors.3 However, there are several continual limitations to organ donation that have not been overcome, which have compelled scientists to identify unique biomaterial solutions that promote angiogenesis.
Limitations for organ donation include finding the right donated organ according to age, gender, geographic region, blood group, immune responses, and organ toxicity; taking various immune suppressant medications and treatments before organ donation; long recovery times; limited available; and so much more.4 It has been estimated that patients frequently wait for an organ for transplantation for around two years; while other statistics indicate that 20% of people do not survive the first year after organ transplant surgery. Unsurprisingly, over the last two years, some countries have reported a 50% reduction in organ transplantation surgeries due to the COVID-19 pandemic.5 Thus, accurately mimicking natural organ structure and function in new synthetic in vitro biomaterial scaffolds is a top priority. Mimicking natural organ structure and function is also important in terms of organ vascularization, which has proved problematic.
Numerous studies have been performed to improve angiogenesis.6 In this way, a variety of biomaterial scaffolds, implanted cells, and biomechanical stimuli strategies have been implemented to promote angiogenesis. The various biomaterial scaffolds used to enhance angiogenesis can be classified into synthetic and natural biomaterials, polymers, hydrogels, micro- and nanoparticles or even acellular scaffolds. Moreover, different types of cells can be used and seeded into such scaffolds, and further a variety of biological factors can be embedded and released from these scaffolds to maximize new vascularization.
In terms of implantable scaffolds, biomaterials must be generally chosen in a manner that has a growth rate the same as that for the tissue and scaffold degradation,7 which can be controlled principally by different fabrication methodologies.8 If scaffold deterioration happens before tissue formation, cells will lose a surface to grow onto, and they cannot survive. If scaffold degradation occurs after complete tissue formation, the cell and extracellular matrix (ECM) accumulation deprives cells of their natural function. This is exactly why choosing a suitable biomaterial for scaffold fabrication is critical for proper cell functionality.
Among the diverse kinds of biomaterials available to researchers today, utilizing natural biomaterials in comparison with synthetic biomaterials has some significant drawbacks, such as limitations in controlling physio-chemical properties, reproducibility, managing degradation rates, and pathogenic concerns during tissue extraction from its source.9–11 Among different scaffold types, injectable scaffolds, which form in the body, are far more promising for promoting angiogenesis, in terms of being less invasive, possessing faster healing times, and an opportunity to more easily fill cavities or the necrotic tissue sites. The impressive recent developments in tissue engineering and regenerative medicine haveencouraged scientists to utilize these injectable synthetic scaffolds not only for modeling diseases but also for implantation into defective sites, most notably cartilage injuries.12 Table 1 shows the biomaterials and their structures, which are covered in this manuscript as effective materials for angiogenesis. These materials belong to different types of categories, such as synthetic polymers, synthetic non-polymers, and natural biomaterials.
 |  |  |  |
Table 1 Chemical Structure of the Biomaterials Discussed in This Article |
In parallel, using acellular scaffolds not only minimizes physio-chemical concerns and degradation issues but also lets scientists focus on finding the best cell source of growth factors to use for promoting vascularization while encountering fewer immune system responses.50–52 Of note, different biological components and stimuli are necessary at the correct time and place to provide practical and complicated vascularized tissues.53,54 For example, porcine lung transplantation using a decellularized lung scaffold seeded with human endothelial and epithelial cells has been reported by Zhou et al to promote angiogenesis.55 Interestingly, a decellularized goat-lung scaffold modified with a chitosan/nanohydroxyapatite composite was utilized for other tissue investigations, such as bone regeneration.13 Although decellularized scaffolds are really encouraging, the challenges have not been fully addressed. For instance, the differences between a mouse lung scaffold with that of humans restrict researchers to use it clinically.
Ultimately, biomaterial manipulation (such as functionalization with biomolecules and/or cell/bio molecular factor loading) is necessary to optimize angiogenic signaling pathways to support vascular cell attachment, proliferation, differentiation, or migration in soft and thick tissues.56,57 Because without accurate modeling, most attempts to induce vascularization do not survive over a long time.58 Furthermore, achieving precise replacement of different cell types within an organ scaffold is very difficult.59 Unfortunately, reconstructing the decellularized tissue is another roadblock. Re-endothelialization for the prevention of thrombosis or oxygen and nutrient delivery for naked and cell re-seeded tissue is a provoking research field. Without such efforts, stable cell metabolic activities will only be maintained for a few days after cell culture. Considering all the above-mentioned obstacles, the most recent clinical trials indicate that endothelial cells and mesenchymal stem cells are the most frequent cell sources that can be cultured within the biomaterial media in order to enhance angiogenesis. Nonetheless, there exist two serious limitations. Firstly, there exist some ethical issues related to the usage of human embryos. Secondly, some immunological responses have been observed after introducing these cells to the human tissues.60,61
Immature vascularization is one of the main causes of organ transplantation failure because vessels and capillaries exchange nutrients, ions, hormones, signaling molecules62 and many other ingredients. Hence, a full vascularization network is key for tissue engineering success. To generate a full vascularization network, one must simulate the vascular niche and interactions within the scaffold to obviously affect vascular tight junctions (TJ). TJs are intercellular adhesion complexes with significant roles for the barrier function of epithelia and endothelia. They can preserve the cell polarity via limiting protein movements, controlling over the paracellular solute and regulation of water flux.63 In addition, early re-endothelialization not only decreases hyperplasia with time but also limits platelet accumulation leading to faster restoration of normal blood flow.64 Modifying chemical compositions (like surface functionalization for altering hydrophilicity/hydrophobicity),65 scaffold architecture (like tube induction, altering pore sizes, or interconnectivity),66 utilizing different growth factors (to support cell proliferation especially the differentiation of stem cells),67 combining solid scaffold biomaterials with pro-angiogenic ECM components (like collagen, hyaluronic acid or some composites of chitosan),68 and capitalizing on blood vessel-formation by stem cells cultured in an appropriate medium69 have been investigated for rapid vascularization. These methods mentioned can be utilized by themselves or together to identify the best scaffold fabrication method and cell seeding techniques. This review will highlight such attempts (Figure 1).
After trying to find a concise and succinct review article for inducing scaffold angiogenesis with all the paramount factors needed for angiogenesis thoroughly and coherently discussed, the authors of the present review did not find any. In fact, the best reference manuscript would require the collection of the three foremost roles for biomaterial angiogenesis: materials, cells and biological factors. Thus, there are several significant reasons to establish this review paper. First, the quick growth of science, particularly in tissue engineering and regenerative medicine, brings about a lack of a great classification and integration of existing research. Even if there were a few reviews about this topic, they have not been precisely arranged into a single review paper, but also there have been no reviews of the reviews. Secondly, there is inconsistency among scaffold microstructures, biomaterial properties, and ways in which in vitro and in vivo studies are conducted, as well as results that means that even a large number of reviews cannot pave the way for future studies. Thirdly, we are prone to write a comprehensive review article due to the fact that the previous ones are unanimous in terms of presenting the challenges and arduous issues of scaffold angiogenesis. Compiling such a manuscript is demanded for future researchers to finally solve such complicated issues. In the light of this review, we aim to inspect a variety of biomaterials used in scaffold fabrication (synthetic materials, natural materials, and hydrogels), discuss the diverse types of cell sources seeded in the aforementioned scaffolds/hydrogels, and review the mechanical properties of the scaffolds encompassing elasticity, stiffness and so on. This review will also provide a poignant examination of what is needed for the field to grow.
Synthetic Biomaterial Scaffolds
The field of tissue engineering always faces a complex demands on the sufficient biomaterials that meet the criteria for each specific application. The chosen biomaterials need to endorse the complicated process of tissue development and also need to serve all mechanical, chemical and biological requirements of the targeted application. These synthesized biomaterials (polymeric or non-polymeric) must have a great interaction with the seeded cells and follow an appropriate pattern of degradation, which is really critical for in vivo uses. In this section, we review how synthetic materials can meet the needs of angiogenesis and how biomimetic principles have been applied to these scaffolds to improve their performance during the angiogenesis process.
Synthetic Polymeric Biomaterial Scaffolds
Synthetic polymers have been widely studied for angiogenesis. As just one of the many examples, polycaprolactone fumarate (PCLF) scaffolds embedded with vascular endothelial growth factor (VEGF) (as the main growth factor for promoting angiogenesis as reported by many groups70), fibroblast growth factor 2 (FGF2), and/or bone morphogenetic protein 2 (BMP-2) were designed by Wagner et al showing no signs of scar formation or extensive immune response.71 Synthetic polymers have also been seeded with cells to promote angiogenesis. Specifically, poly-L-lactic acid (PLLA) scaffolds seeded with human microvascular endothelial cells (HMVECs) and human pulmonary artery smooth muscle cells (HPASMCs)36 have been studied. Synthetic polymers have also been cross-linked with a diverse range of molecules and/or coated with various proteins and biomolecules to promote angiogenesis. For example, poly(L-glutamic acid) (PLGA) cross-linked with adipic dihydrazide (ADH) and coupled with hypoxic paracrine secretion,48 polycaprolactone (PCL) scaffolds coated with collagen and VEGF,37 and poly D, L-lactide-co-glycolide (PLGA) scaffolds coated with collagen, and seeded with bone marrow mesenchymal stem cells (bmMSCs)38 are other supportive models for angiogenesis. Moreover, poly vinyl alcohol/carboxymethyl cellulose (PVA/CMC) scaffolds loaded with reduced graphene oxide (rGO) were designed to evaluate the effect of rGO on angiogenesis and have been reported to be one of the most attractive models for angiogenesis.18 Another angiogenesis template includes decorating carboxylated CNWs (CCNWs) with silver nanoparticles, which demonstrate suitable mechanical strength and antimicrobial activity. Excellent mechanical strength matched with the cancellous bone can be obtained by changing porosity between 80% and 90% and pore diameter between 150 and 500 μm, which ultimately leads to promoted angiogenesis in this model.72
Other studies have proposed synthetic vascular networks (SVNs) made of poly-3-hydroxybutyrate-co-3-hydroxy valerate (PHBV) with alginate (as a sacrificial agent), and in situ three-dimensional micro-vascular networks from PLGA as angiogenic materials.39 Not long ago, Gniesmer et al claimed that PCL scaffolds modified with chitosan (CS‐g‐PCL) and seeded with endothelial cells (ECs) as well as loaded with VEGF28 are future prospects for angiogenesis expansion. In addition, three types of polyurethane scaffolds were evaluated by another group, and the results were unanimous with previous studies demonstrating weak angiogenic responses when using polyurethane alone.44 However, Bezuidenhout et al demonstrated that porous polyurethane disks immobilized with heparin can enhance vascularization.45 Many of the above-mentioned materials have low electrical conductivity. However, bioelectricity can stimulate specific cellular phenomena such as cell growth, differentiation and proliferation, which can be applied using cyclic voltammetry.73
Many new polymers in the biomaterials’ field have also been modified to promote angiogenesis. For example, Chen et al suggested that the synthesis of a porous gelatin hydrogel modified with polyhedral oligomeric silsesquioxane (POSS) at various concentrations can sustain the release of VEGF and BMP-2 to enhance angiogenesis. The addition of POSS to gelatin is one of the reasons behind the observed accelerated protein adsorption and cell integrin α5β1 expression promoting cell adhesion to the scaffolds and vascular tube formation (Figure 2).74 Endometrial regeneration within hydrogels synthesized with heparin-poloxamer (HP) with ε-polylysine (EPL) is the reason why the aforementioned hydrogel has captured the attention of numerous scientists for angiogenesis applications.42
 |
Figure 2 In vivo applications of a Gel−POSS hybrid hydrogel primed using octafunctional POSS cages. Cell adhesion, controlled release of growth factors, repairing tissue defects, as well as the acceleration of angiogenesis are some of the uses of this platform. Reprinted with permission from Chen M, Zhang Y, Zhang W, Li J. Polyhedral oligomeric silsesquioxane-incorporated gelatin hydrogel promotes angiogenesis during vascularized bone regeneration. ACS Appl Mater Interfaces. 2020;12(20):22410–22425. Copyright 2022 American Chemical Society.74 |
In Table 2, a variety of scaffolds fabricated for angiogenesis along with the type of cultured cells studied to improve angiogenesis are listed. These systems include polymeric scaffolds (Table 2).
 |
Table 2 A Review of Templates Fabricated for Angiogenesis |
Synthetic Inorganic Biomaterial Scaffolds
Some reports have mentioned that bioactive glasses (BGs), which were introduced by Hench et al for the first time several decades ago for orthopedic/dental applications,20 can boost angiogenesis due to the emission of Si, Ca+2, B, Cu2+, Zn2+, Ag2+, and Ga81 and would be even better at promoting angiogenesis by adding Cu2+, Co2+, etc., into their structure.81 Si, as one of the first angiogenesis booster, not only promotes neovascularization82 but also stimulates collagen type I formation by cells.83 Ca+2 boosts angiogenesis by promoting epithelial cell proliferation84 and B stimulates vascularization via increasing RNA synthesis in fibroblasts necessary during wound healing.85 Ultimately, Cu and Zn have been reported as other ingredients, which86 can enhance angiogenesis between all of the mentioned ions; Cu2+ not only promotes angiogenesis but also has antibacterial properties.87 Besides, it has been demonstrated that BGs can inhibit the growth of Gram-positive and Gram-negative bacteria,88 which is relevant for reducing pH-related cell death during inflammation and increasing reactive oxygen species (ROS) generation to kill bacteria.89 For instance, poly-DL-lactic acid (PDLLA) films that contained 20 wt% BGs increased cellular VEGF expression, leading to a higher vascularization density in comparison with the control group.40 Also, increased VEGF secretion at higher BG concentrations (0.1–10 wt%) showed an undeniable dose-dependent effect on angiogenesis.41 Furthermore, the size of BG particles has been demonstrated to induce sensitivity to the angiogenesis process. Smaller particles (1–2 mm in diameter) are more effective than the larger ones (2–3.15 mm) for VEGF secretion in human CD-18CO fibroblasts, most likely due to the increased surface area and exposure of BG at smaller dimensions.27 Figure 3 shows a model of a 3D fibrous scaffold with high level of hydrophilicity and surface area that encourages ion release for angiogenesis. This platform demonstrates swift cell anchorage, spreading, and accelerated cell invasion, supporting osteogenesis as well as angiogenesis.90
 |
Figure 3 Schematic of prepared 3D fibrous scaffolds in conjunction with applied biochemical and biomechanical stimuli leading to a positive effect on angiogenesis and osteogenesis for bone repair applications. Reprinted with permission from Kim JJ, El-Fiqi A, Kim HW. Synergetic cues of bioactive nanoparticles and nanofibrous structure in bone scaffolds to stimulate osteogenesis and angiogenesis. ACS Appl Mater Interfaces. 2017;9(3):2059–2073. Copyright 2022 American Chemical Society.90 |
Recently, Vimalraj et al reported that Zn(silibinin)(phenanthroline) [Zn(sil)(phen)] and Zn(silibinin)(neocuproine) [Zn(sil)(neo)] as Zinc silibinin and mixed ligand zinc complexes with dose-dependent non-toxic traits, increased VEGF and angiopoietin-1 (Ang1) secretion in mesenchymal stem cells (MSCs) for improved angiogenesis.14 Not long ago, Chen et al reported that surface modified titanium (Ti) with a Chitosan-Catechol multilayered structure, gelatin and hydroxyapatite nanofibers increased the adhesion and proliferation of MSCs and human umbilical vein endothelial cells (HUVECs) to promote angiogenesis in bone healing after boosting paracrine effects via cell–matrix interactions.15 Furthermore, zeolites, as a member of the hydrated aluminosilicate mineral family, are really remarkable among scientists due to their ability for ion exchange and reversible dehydration. They represent an opportunity in angiogenesis tissue engineering owing to the fact that they increase epithelialization, reduce inflammation, and expand angiogenesis.91
Furthermore, materials effectively used in blood-brain-barrier (BBB) applications can be considered for some other angiogenesis applications. In this manner, in vitro BBB beads from collagen IV and fibronectin coated with induced pluripotent stem-cell (iPSC) and derived human brain microvascular endothelial cell (dhBMECs) functions were proposed by another group due to a concentration-dependent effect on hydrogen peroxide (H2O2) exertion to enhance angiogenesis.16 They further evaluated angiogenesis as a function of the concentration-dependent effect of other biomolecular factors as well as ECM components. For instance, the effect of VEGF and Wnt ligands in the presence of some pro-angiogenic ECM components (ECM components, such as collagens and proteoglycans) play critical roles in both physiological and pathological angiogenesis regulation; pro-angiogenic ECM components are the ones controlling angiogenic mechanisms enhancing the angiogenesis process for the sprouting of induced vessels.92 Their results also revealed that oxidative stress has a concentration-dependent effect on angiogenesis expansion.
One of the most visible advantages of this research is that this model has encouraged others to observe genetic mutations involved in angiogenesis in detail, which is considered a huge step forward in the field. Albeit two main challenges remain in their model: first, complicated cell–cell interactions, such as the interactions between neurons, neural progenitor cells, pericytes, and glial progenitor cells, were neglected during brain angiogenesis. If these sophisticated interactions remain neglected, there would not be enough evidence that their platform is efficient for in vivo modeling. Second, no changes were observed for capillary growth (such as varying capillary length, diameter, or branching).16,93 Clearly, such considerations need to be the focus of future studies.
Natural Biomaterials-Derived Scaffolds
Natural biomaterials are a major group of biomaterials that have been utilized in tissue engineering applications due to their nice microstructure, high interconnectivity, as well as great inherent bioactivity, which endorses them mimicking the tissue natural extracellular matrix. Playing the role of extracellular matrix provides cell adhesion, practical oxygen and nutrient delivery, as well as supporting cells for restoring the function of defected tissue. Although they enjoy lots of biological benefits, they usually suffer from weak mechanical strength and lack a fine degradation pattern (a fine degradation pattern refers to a degradation rate that is equal to the tissue regeneration rate) which must be made up via blending with synthetic materials. In this section, we will discuss different natural-based scaffolds (blended with other materials) that support the angiogenesis process.
Pro-Angiogenic Biomaterials and Anti-Angiogenic Biomaterials
When it comes to natural biomaterials, fibrin94 and hyaluronic acid (HA) have been reported as vascularization promoters during wound healing by promoting fibroblast activities.30,95 In addition, degradation products from HA increases angiogenesis, such as Ca+2.21 Furthermore, type IV collagen has a dose-dependent effect on vascular elongation. Researchers demonstrated that lower and higher doses of type IV collagen had a completely opposite effect. For instance, a moderate concentration of about 30 lg/mL promoted new vessel formation, while a concentration of about 300 lg/mL limited vessel progression.96 Collagen-based scaffolds are claimed to have a synergistic effect not only on angiogenesis but also on reducing inflammation after implantation into a diabetic rat skin wound model.75 Histopathological evaluation demonstrated promoted angiogenesis for alginate/vitamin D3 treated wounds.97 On the contrary, chitosan has been mentioned as an anti-angiogenesis agent because of its depolymerized products (low-molecular weight chitosan and chitooligosaccharides (LMWC/COs)).33,34 However, it is noteworthy to mention that some chitosan composites can accelerate angiogenesis. Other groups have worked on developing new angiogenesis biomaterials with chitosan composites; for instance, chitosan/poly–caprolactone modified by heparin and chitosan/gelatin have been proven to enhance vascularization.98 An interesting investigation by Zhang et al revealed that silk fibroin is a powerful clinical option among the natural biomaterials not only for angiogenesis but also for wound dressing and skin defect healing. A randomized single-blind parallel controlled clinical trial was done with 71 people, and their results were promising. They showed that silk fibroin film can strongly diminish the regeneration time (healing and angiogenesis of the defected skin) in comparison with other commercial dressing materials.99
Among all the existing natural biomaterials, collagen, gelatin and alginate are the most prevalent ones owing to the fact that their microstructure provides cells with a great opportunity for biological activities. They can also successfully pursue the neo-tissue formation and integration pattern by degrading within an appropriate period of time. Additionally, they endorse cell adhesion, differentiation, migration, or necrosis. Here, we have discussed some of the most interesting and up-to-date studies about angiogenesis acceleration using these three reputable natural biomaterials.
Collagen-Based Scaffolds
In one of the first models, Wong et al designed a template consisting of PLGA microfibers (similar to capillaries in terms of dimension) embedded in a type I collagen scaffold and seeded with HUVECs/VEGF to promote angiogenesis even after fiber degradation.76 Similarly, type I collagen-based scaffolds were used by Chan et al for seeding primary human microvascular endothelial cells to form CD31 positive capillary structures and clear lumens.31 Additionally, Gérard et al utilized type I collagen-based scaffolds modified with heparin and seeded them with bone marrow cells as a promising model for angiogenesis characterization.29 As a more complicated model, Haifei et al suggested heparinized porous type I collagen–chitosan scaffolds in combination with N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide (EDC) and N-hydroxysuccinimide (NHS) for promoting angiogenesis.43
To evaluate pericyte function (as the branched cells with an increasingly high effect on vascular reinforcement and blood flow regulators) which affects endothelial cell migration and stabilization,100 type I collagen hydrogels were seeded with pericytes in the presence of VEGF by Chiu et al.101 Moreover, a 3D scaffold of bacterial cellulose and bladder acellular matrix (BAM) combined with type I collagen and glycosaminoglycans (GAGs) simulated angiogenesis in the native porous nanofibrous structure of urethra after seeding with HUVECs. The results showed the expression of endothelial related proteins, which led to capillary-like tube formation.102 Like other collagen-based scaffolds, a research group formulated type I collagen hydrogels with a HUVEC/epidermal growth factor103 and added sulfhydryl groups to the porous type I and type III collagen scaffolds.77 Notably, to assess the effect of cell seeding pattern, culturing HUVECs in a cord within type I collagen/fibronectin gels was suggested by Schechner et al as a novel framework for expanding vascularization.104 The addition of 2-N,6-O-sulfated chitosan (SCS) to gelatin hydrogels for the controlled release of BMP-2 followed by VEGF is another novel synergistic angiogenesis formulation deemed successful by the upregulation of angiogenesis genes during bone reformation (Figure 4).105
 |
Figure 4 BMP-2 and VEGF release sequences from gelatin and 2-N,6-O-sulfated chitosan scaffolds. The efficient binding of VEGF to SCS is the reason behind the observed synergistic angiogenesis. Reprinted with permission from Zhang S, Chen J, Yu Y, Dai K, Wang J, Liu C. Accelerated bone regenerative efficiency by regulating sequential release of BMP-2 and VEGF and synergism with sulfated chitosan. ACS Biomater Sci Eng. 2019;5(4):1944–1955. Copyright 2022 American Chemical Society.105 |
Alginate/Gelatin-Based Scaffolds
Similar to collagen, alginate and gelatin (ie, denatured collagen), have been widely studied for angiogenesis. Alginate hydrogels loaded with alginate lyase as an enzyme for cleaving alginate polymer chains were seeded without outgrowth endothelial cells (OECs) by Campbell et al106 for improving angiogenesis. Additionally, injectable gelatin-based sponges, which were seeded with fetal rat pneumocytes, were proposed by Andrade et al78 as another template for new vascularization. Perets et al suggested that alginate scaffolds incorporate tiny poly(lactic-co-glycolic acid) microspheres to control the release of angiogenic factors, such as basic fibroblast growth factor (bFGF), and the results indicated capillary formation.35 Interestingly, vigorous angiogenic growth factors, such as VEGF, platelet-derived growth factor (PDGF), and transforming growth factor-b1 (TGF- β1) were bound to the alginate-sulfate/alginate scaffolds to boost vascularization.23 Functionalized 3D porous bacterial cellulose/gelatin (B/G) scaffolds with heparin were loaded with VEGF for the sustained release of VEGF by Wang et al to emit growth factors up to about two weeks for angiogenesis.22 Lastly, coupling gelatin with a porcine cholecyst-derived scaffold (CDS) has been demonstrated to be efficient for improving endothelial cell viability in parallel with the survival of keratinocytes (HaCaT). The conjunction of these two features is promising for angiogenesis and diabetic wound treatment. Comparison of gelatin-coupled CDS and bare CDS showed that the reinforced features resulted due to the addition of gelatin (Figure 5).107
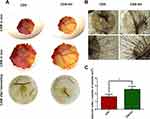 |
Figure 5 (A) First two rows show the in ovo results of blood vessel formation for cholecyst-derived scaffolds and cholecyst-derived scaffolds-gelatin and the third row, shows ex ovo results. (B) Stereomicroscopic images of vessels for both cholecyst-derived scaffolds and cholecyst-derived scaffolds-gelatin (Scale bars are in the range of 1 to 2 µm). (C) Illustration of “vascular index” which reports the angiogenesis acceleration after the addition of gelation to cholecyst-derived scaffolds. (CAM= Chick Chorioallantoic Membrane, *p value < 0.05). Reprinted with permission from Mony MP, Shenoy SJ, Raj R, et al. Gelatin-Modified Cholecyst-Derived Scaffold Promotes Angiogenesis and Faster Healing of Diabetic Wounds. ACS Appl Bio Mater. 2021;4(4):3320–3331. Copyright 2022 American Chemical Society.107 |
Alternative Natural Scaffolds
Some less commonly used natural biomaterials have been reported in this section. These biomaterials are less well-known due to their poor mechanical and biochemical properties or their high price. For instance, fibrin hydrogels with nanoparticles bound to VEGF and seeded with HUVECs were suggested by Anderson et al to support capillary formation after a short duration of time.108 He et al introduced a cyclic peptide LXW7 that binds to the integrin, αvβ3, on endothelial progenitor cells (EPCs) and EC receptors to enhance angiogenesis. Next, they designed a pro-angiogenic molecule LXW7-DS-SILY in order to functionalize the ECM scaffolds for neovascularization.49 In the next step, nano-hydroxyapatite was seeded with HUVECs to enrich vascularization due to enhanced cell viability, migration, and vascular tube formation. The group proved that reducing nitric oxide (NO) synthesis in HUVECs inhibited phosphatidylinositol 3-kinase (PI3K).109 To treat ischemic cardiomyopathy, Zarrintaj et al prepared 3D agarose-based cryogels and evaluated angiogenesis. They encapsulated cardiac stem cells using agarose. Promisingly, angiogenesis was promoted due to the enhanced secretion of cytokines. Results showed increased cell viability and proliferation as well as migration.24 It was unmistakable that plumbagin severely decreased micro-vessel formation and induced phospholipase C (PLC), extracellular-signal-regulated kinase (ERK), nuclear factor (NF), and inhibited hypoxia-inducible factor (HIF)-1 signaling pathways to stop enhanced vascularization.46
In a noteworthy manner, Momordica charantia, a kind of bitter melon, has captured the attention of researchers because of the increasing phosphorylation of ERK. Scientists claimed that it stimulated tube formation from bovine aortic endothelial cells (BAEC) to promote angiogenesis.110 As one step further, injectable platelet‐rich fibrin (PRF) with a co‐culture of OECs (sprouting endothelial cells) and primary osteoblasts (pOBs) was found to represent another unique template for angiogenesis network formation after about 1 week.111 Finally, a new framework for generating a neurovascular unit (NVU) (NVU consists of neurons, glial cells, blood vessels, and ECM proteins) composed of a fibrin-matrigel matrix seeded with BMECs/MSCs, which developed new blood vessels.112
Hydrogel Scaffolds
Hydrogels are a type of scaffolds that pose many different applications in tissue engineering. They can be utilized as space filling agents, delivery of drugs, growth factors, biomolecules or even cell encapsulation. These 3D structures can provide stimuli, which guide cells restoring the defected tissue. This class of scaffold is structurally similar to the extracellular matrix of plenty of tissues, which is an outstanding option for scaffold fabrication. They can also be processed under comparatively mild condition and can be delivered to the desired site, noninvasively. In this section, we have argued the usage of hydrogels for angiogenesis applications, which have attracted the interests of many scientists.
An injectable temperature-responsive hydrogel seeded with MSCs/stromal cell-derived factor-1 (SDF1)/VEGF was designed by scientists, and the results indicated the swift proliferation of MSCs to support angiogenesis in further steps.32 Also, Rustad et al enhanced capillary formation by using a pullulan-collagen hydrogel loaded with MSCs. They asserted that increasing the level of cytokines was the golden key for angiogenesis.25 Carrying VEGF/FGF2/keratinocyte growth factors (KGF) by micro carriers in a platelet rich plasma (PRP) hydrogel was a new solution recommended by Naderi et al.113 However, bFGF immobilization on poly ethylene glycol (PEG) hydrogels seeded with smooth muscle cells (SMCs) seemed to be more practical.47 Interestingly, epithelial cells, MSCs, and ECs showed a radical effect on angiogenesis when co-cultured and loaded on polyvinyl sponges with FGF2 growth factors by Mondrison et al.114 As with other hydrogel-based suggestions, PEG hydrogels and methacrylated gelatin (GelMA) flanked by methacrylated hyaluronic acid (HAMA) have been introduced to this field.21 A facile strategy was suggested by another team for diabetic wound treatment. For this, a high-molecular-weight hyaluronic acid (HHA) was converted to Cu-HHA/PVA using Cu2+ and physical crosslinking. Then, M2 phenotype macrophage (MΦ2) was seeded within the formed scaffold for immuno-compromization purposes and for angiogenesis. Additionally, the controlled release of Cu2+ synergistically encouraged angiogenesis (Figure 6).115 As a state-of-the-art technique for hydrogel fabrication, the idea of constructing soft hydrogels containing multi-vascular structures is extraordinary and can be accomplished by stereolithography, photoactive liquid resins, and using non-toxic natural food dyes as light blockers.116
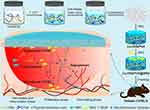 |
Figure 6 Depiction of dual cross-linked high-molecular-weight hyaluronic acid seeded hydrogels with M2 phenotype macrophages improving immunocompromization and defecting angiogenesis. Reprinted with permission from Liu S, Yu J, Zhang Q, et al. Dual cross-linked HHA hydrogel supplies and regulates MΦ2 for synergistic improvement of immunocompromise and impaired angiogenesis to enhance diabetic chronic wound healing. Biomacromolecules. 2020;21(9):3795–3806. Copyright 2022 American Chemical Society.115 |
In Table 3, a variety of scaffolds fabricated for angiogenesis along with the type of cultured cells studied to improve angiogenesis are listed. These systems include hydrogels (Table 3).
 |
Table 3 A Review of Templates Fabricated for Angiogenesis |
Fibrous Scaffolds
In modern tissue engineering, nanofibers have attracted attention due to their biomimetic ECM structure, large surface-to-volume ratio, versatility in material selection, and easy processing.118 Numerous researchers around the world have utilized electrospun nanofibrous scaffolds in their research.119 Fibrous and nano-fibrous scaffolds can mimic the extracellular matrix constituents such as collagen and this kind of simulation of natural tissue plays a key role for tissue regeneration. Among different methods of fibrous scaffold fabrication, self-assembly, electrospinning and phase separation are of great interest for scientists due to providing unique scaffolding features. In this section, we will discuss different usages of nano-fibrous scaffolds for angiogenesis acceleration. For example, one of these groups demonstrated that a fiber’s diameter affects macrophage and mast cell responses as well as VEGF secretion important for angiogenesis.120 Moreover, fiber orientation can vary with the angiogenic response. This team suggested that aligned nanofibers will improve neovascularization in comparison with other orientations.121 Furthermore, nanofibrous scaffolds made of ECM components (such as collagen) enhance EC adhesion, proliferation, and migration causing for vascularization expansion.122 To the best of our knowledge, nanofibers can control the release of loaded angiogenic small molecules (such as angiogenin) or phytochemicals (such as curcumin).123 These systems not only strongly mimic the ECM environment but are also suitable platforms to control cell fate. In fact, instead of utilizing costly growth factors, changing fiber dimensions and orientation can cause significantly favorable cell responses. Thus, state-of-the-art electrospun nanofibrous scaffolds can improve scaffold properties by improving cell differentiation and/or the release of loaded angiogenic small molecules/phytochemicals.
It is important to keep in mind that due to the swift development of nanotechnology in the 21st century, this class of materials have widely been used in tissue engineering fields due to its great physical and chemical properties. They provide a large surface-to-volume ratio, a high potential for efficient and tunable loading, great biocompatibility, as well as great potential for surface modification. Due to the aforementioned merits, many scientists have utilized these nanoscale materials (such as metal NPs and graphene-based nanomaterials) in their vascular regeneration studies. This category of materials can preserve the loaded bioactive molecules on the way to the targeted site, more efficiently.124
Other Approaches
In addition to the mentioned techniques for biomaterial manipulation, there exist some state-of-the-art methods for cell direction to the desired area, which can be a huge step toward a more efficient angiogenesis. Cell encapsulation is a new technique that provides the seeded cells a sufficiently hydrated environment before being released. Also, applying a controlled magnetic-field is a good facility to guide the cells and growth factors toward the desired areas. Additionally, utilizing the pre-vascularized scaffolds is another option for vascular tissue engineers because the existence of vascular structures in decellularized tissue can solve many problems relevant to tube formation or sprouting of formed vessels. In this section, we have argued the newer methods for angiogenesis acceleration and the assessment process of reendothelization are of great importance.
Cell Encapsulation for Vascular Tissue Engineering
Cell encapsulation within a bioactive and biodegradable hydrogel is a great option for tissue engineers, which can be utilized for in vivo cell delivery to the defected sites. It is called as an “smart procedure” owing to the fact that this class of hydrated hydrogels poses greatly tuned swelling ratio, suitable manipulated mechanical properties for cell growth and differentiation, appropriate degradation patterns (hydrolytically or enzymatically) after the tissue regeneration, as well as controlled diffusion characteristics (diffusion of oxygen or nutrients). However, the number of suitable chemistries and practical formulation methods are limited due to the fact that cells are present when gelation process happens. For instance, cell encapsulation in heparinized alginate for the sustained release of growth factors in defective areas,125 or homing of MSCs/EPCs into the scaffolds by the addition of chemokines (such as SDF1) have been suggested. After adding monomers to the unencapsulated cells, the polymerization process, on the ground of a specific mechanism, starts. Then, a kind of hydrogel forms in which cells are so-called arrested but migration and differentiation are definitely possible. In this procedure, hydrogel degradation and neo-tissue formation need to be equal as this is critical due to the fact that late or early degradation can cause failure.126,127 This new technique has been recently utilized by scientists for angiogenesis acceleration, as can be seen in (Figure 7).128
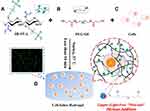 |
Figure 7 An example of in vitro 3D cell encapsulation within a hydrogel leading to cell proliferation, and migration. (A) acylated-modified sulfobetaine-derived starch (SB-ST-A), (B) dithiol-functionalized poly (ethylene glycol) (PEG-SH), (C) cells, and (D) cell-laden hydrogel. Reprinted with permission from Dong D, Li J Cui M, et al. In situ “clickable” zwitterionic starch-based hydrogel for 3D cell encapsulation. ACS Appl Mater Interfaces. 2016;8(7):4442–4455. Copyright 2022 American Chemical Society.128 |
Magnetite Scaffolds for Vascular Tissue Engineering
Magnetic scaffolding is a sort of manipulation method for cell remote controlling. The addition of magnetic nanoparticles to the fabricated scaffolds provides us with a great opportunity to control the cell functions utilizing an external magnetic field. Here, we have discussed the magnetic force-based tissue engineering (Mag-TE) as a new class of practical techniques that have recently been used in plenty of tissue engineering fields such as organ angiogenesis. Also, inducing an intrinsic microvascular network within the scaffold129 using novel magnetite tissue engineering technology130 include other promising procedures that scientists are currently pursuing. Adding some micro-channels within the scaffold can foster cell migration and/or nutrient diffusion. It is noteworthy to mention that magnetic nanoparticle-containing liposomes (MCLs) seeded with mouse iPS cell-derived Flk-11 can be shaped like sheets by a magnetic force and the results (measured by laser Doppler blood flow and capillary density analyses) revealed that this approach is helpful for therapeutic angiogenesis.131
Pre-Vascularized and Decellularized Scaffolds
The idea of using pre-vascularized scaffolds has helped researchers navigate towards decellularized tissues or multi-vascular structured hydrogels. This means that because the vascular structure already exists in decellularized tissue, issues relevant to tube formation or sprouting vessels for new cells diminish. Hence, de-epithelialized tissue with healthy and functional vascularization has captured the attention of many scientists in tissue engineering and regenerative medicine.132 Other issues, such as preventing a temperature rise and the need to find strong organic solvents, seem to be challenging133 due to protein denaturation and impairing optimal cell function. To achieve the above-mentioned aims, the use of intermittent inflation detergent-enzymatic treatment (DET) has been reported not only for removing all nuclear and cytoplasmic materials but also for preserving capillaries in more sensitive organs, such as the lung (especially, the alveolars).134 For instance, a decellularized porcine lung scaffold was reseeded with human airway epithelial progenitor cells after decellularization. Results indicated that the lung scaffold supported angiogenesis and blood circulation without coagulation for 1 hour.55 Also, small intestinal submucosa (SIS) and acellular dermal matrix (ADM) in parallel with a collagen–chondroitin sulfate–hyaluronic acid (Co–CS–HA) scaffold was seeded with the same cell types and growth factors. The results showed greater angiogenesis capacity in SIS and ADM in comparison with Co–CS–HA, which signifies the preferred use of such decellularized scaffolds.79
Re-Endothelialization Assessment
Although re-endothelialization is assessed by in vitro cell detachment under flow conditions in a bioreactor, ex vivo blood perfusion, extracorporeal systems, in vivo transplantation,135 immune histology, RNA and protein analysis, and state-of-the-art imaging techniques,136 these assays are not the end of the pathway. Unfortunately, vascularization and preventing blood clotting pose significant challenges for tissue survival.137 Thus, fully functional vascularization, which is stable over a long period of time, is seriously needed to avoid tissue necrosis. Noticeably, to pave the way for more practical angiogenesis and more accurate organ modeling, inspiration from other approaches (such as angiogenesis of severe wounds that diabetics suffer from), is promising. Moreover, following the angiogenesis platform or mechanism which can be utilized for any other tissue thicker than 400 µm, can be auxiliary.138,139 To highlight this problem, transplantation of autologous split-thickness skin grafting (STSG) with human mesenchymal stem cell sheets (HSC) flanked by pre-vascularized human mesenchymal stem cell sheets (PHCS) was introduced.140 Definitely separated from these inspirations, tissue-specific sensitivities must not be ignored. For instance, in the lung angiogenesis, particular issues like surfactant secretion after angiogenesis have been reported by Gilpin et al.137
Cell Sources
Niklason (the co-founder of Humacyte, which specializes in vascular and lung tissue engineering, was recognized as one of the top 50 most important inventions of 2010 by Time magazine) declared in 2010: “We haven’t gotten to the other side of the mountain range yet, but when we do, I hope there’s a big bus of stem cells waiting for us”.1 Although ECs and other non-stem cells have been suggested for angiogenesis growth, Niklason’s prediction is still authentic. Stem cells have been suggested by many groups to develop blood vessels. In this section, we review the different types of implanted cells within scaffolds that can achieve vascularization. To the best of our knowledge, the co-culture of primary basal-like cells with HUVECs141 has been reported to be a potent cell source for vasculature branching. The results achieved by Ren et al related that seeding a lung scaffold with endothelial and perivascular cells derived from induced pluripotent stem cells can lead to ~75% endothelial coverage. However, the dark side of these promising consequences is capillary longevity. Indeed, the formed capillaries survived in the tissue for only three days which is absolutely less than clinical expectations.142 Lung micro-vascular ECs (LMVECs) is another reliable source. Not long ago, Tsuchiya et al proved that LMVECs can bind to galactose and lack Weibel-Palade bodies, which have been identified as ultrastructurally necessary for other EC subtypes.63 In particular, plenty of researchers insist on using MSCs due to paracrine effects for angiogenesis.143 Recently, Nilforoushzadeh et al demonstrated that culturing MSCs in 3D spheroid media will improve the paracrine effect to help angiogenesis.144
MSCs and Other Most Frequent Cell Sources
Although most stem cells possess paracrine effects, mesenchymal stem cells (MSCs) are the most frequent cells used to investigate angiogenesis136 (Figure 8).145 For instance, MSCs, as the primary source of VEGF, are capable of forming tiny capillary induction in lung vascularization research.146 It is important to note that Genova et al proved that osteo-differentiation of MSCs improved when they were co-cultured with ECs, and in parallel, ECs promoted angiogenesis when near MSCs.147 Also, the delayed addition of MSCs to the culture medium was identified to accelerate capillary network formation. All of these observations confirmed Niklason’s prediction stated above.148
 |
Figure 8 The therapeutic influence of MSCs-biomaterials due to the release of trophic factors, specifically immunomodulatory or angiogenic cytokines. Reprinted with permission from Li T, Ma H, Ma H, et al. Mussel-inspired nanostructures potentiate the immunomodulatory properties and angiogenesis of mesenchymal stem cells. ACS Appl Mater Interfaces. 2019;11(19):17134–17146. Copyright 2022 American Chemical Society.145 |
Additionally, other cell sources have been used to form mature vessels. In fact, vessel maturation can be equally important for vessel formation. Lung fibroblast cells and type-II pneumocytes are pro-angiogenic and form immature capillaries.149 Recently, Panina et al reported that adipose tissue-derived stem cells (ASCs) with self-renewal properties are a prominent source of EPCs, ECs, and pericytes for vessel remodeling.150 During in vivo investigations, Yen et al hypothesized that mouse pulmonary stem/progenitor cells (mPSCs) have pro-angiogenic effects via the expression of multiple pro-angiogenic factors.151 Remarkably, although ECs are perfect cell sources, which have been used for angiogenesis time and time again, the immunogenic trait of ECs is not neglectable. Moreover, non-invasive cell harvesting is another issue in clinical trials, so allogenic cell sources are preferred. Other choices such as EPCs and iPSCs are alternatives.152 Interestingly, inflammation is said to be an integral component of angiogenesis expansion. Danese and Jackson et al declared that inflammatory cells surrounding the vasculature can promote vessel growth.153,154 Wagner et al further reported that inflammatory cells, such as monocytes/macrophages, can modulate angiogenesis in a lung model but in contrast, lymphocytes prohibit neovascularization.155 Moreover, pediatric bronchial epithelial cells and nasal epithelial cells have been infected with the influenza virus to rapidly divide after implantation.116
Some spheroids are well known as paracrine stimulators of angiogenesis and building blocks for generating new vasculature and have been investigated by scientists all around the world because three-dimensional spheroids can activate cells to secrete more of immunomodulatory paracrine factors (especially PGE2 and TNF-α-stimulated gene/protein 6 (TSG-6)). Researchers utilize this platform not only for pro-angiogenesis investigations but also for anti-angiogenesis investigations, for instance for cancer therapy. It is noteworthy to mention that anti-angiogenesis studies are important for cancer termination. For example, Maracle et al provided spheroids consisting of ECs and fibroblast-like synoviocytes. By using NF-κB signaling mediation, they demonstrated an anti-angiogenesis response for these spheroids.156,157 To address another use of spheroids in angiogenesis characterization, Lee et al revealed that the spheroids consisting of ECs and pericytes in combination with VEGF enhanced vasculature sprout formation because pericytes can cover the EC monolayer from its basal side.158 In a cell-based model of angiogenesis including co-culturing human adipose stem cells (hASCs)/ HUVECs and a growth factor cocktail (GFC), histological staining revealed a significant increase in the level of NODAGA-RGDyK binding to αvβ3 integrins in parallel with an increased tubule network density leading to neovascularization.159 Also, placental tissue-derived MSCs in serum-free medium (PlaMSCs) synthesized both angiogenic and angiostatic factors and significantly increased endothelial tube formation due to enhanced angiogenesis-related gene expression.160
Table 4 summarizes the various cell types used by different groups to foster angiogenesis.
 |
Table 4 Different Cell Sources That Can Be Seeded for Enhancing Angiogenesis |
Cell Seeding Challenges
Of course, it is worth mentioning that after choosing the right source of cells, proper placement in a 3D scaffold has a vital role for angiogenesis.137 After choosing and implanting the opted cells in their correct place, guiding cell differentiation is another serious concern.169 This cell fate guidance refers to applying diverse factors ranging from biochemical factors like growth factors to mechanical and electrical factors like scaffold stiffness, strength or even applying a gravitational force. Thus, fabrication of a suitable scaffold or hydrogel implanted with the right cell source is definitely important. One needs to model the exact physical and mechanical stimuli to uphold the seeded cells for great attachment, growth and differentiation. To end this section, it is critical to point out that controlling cell migration and proliferation, particularly of ECs, pericytes, and macrophages, are by far the most important parameters for blood vessel development in engineered tissues.170 In the hereunder section, the influence of various kinds of mechanical factors are reported as critical factors that alter cell fate or cell commitment, which is exactly why they need to be carefully characterized and correlated with angiogenesis.
Biomechanical Stimulation
Blood vessel development under the effect of mechanical factors has been under plenty of investigations. Indeed, the influence of biomaterials-induced mechanical features such as scaffold stiffness, mechanical strength, applying gravity force and other types of mechanical stimulus is the topic of many studies. Some kinds of cellular components such as growth-related factor proteins or lipids, some signaling molecules, as well as genes and mitogenic genes have been intensively reported to mediate the angiogenesis process after applying the mechanical factors. These studies demonstrate that angiogenesis acceleration must be studied under different mechanical features, which leads to the development of biomechanical engineering approaches for more efficient vascularization.
Effective Biomechanical Variables for Angiogenesis
Results indicate that scaffold stiffness,171,172 applied pressure,163,172,173 cAMP (cyclic adenosine monophosphate),174 scaffold malleability,175 adhesion strength,176 applied shear stress,80,172,177 scaffold mechanical stretch,161,162,178–180 applied mechanical tension,172 applied gravity force,181 scaffold elasticity or viscoelasticity,171,172 as well as scaffold morphological properties, such as channel induction,182 scaffold chain sizes,183 scaffold pore sizes, interconnectivity and porosity,184 are all crucial physical and mechanical properties, which affect cell migration, differentiation and angiogenesis. All the suggested values are gathered in Table 5.
 |
Table 5 Effect of Mechanical Factors on Angiogenesis |
Scaffold stiffness has been reported as an influential factor that affects angiogenesis in plenty of investigations. It needs to be approximately equal to the stiffness of the organ that has been replaced with. For instance, the stiffness of skin is about 0.86 MPa,185 bone stiffness is about 20 GPa,186 and brain stiffness is about 3KPa.187 The stiffness values need to be carefully tuned due to the fact that it has a strong influence on cell activities. For instance, Liu et al claimed that scaffold stiffness affects liver sinusoidal endothelial cell (LSECs) and hepatic stellate cell (HSCs) activation.188 Scaffold stiffness is one of the most outstanding mechanical behaviors that affect angiogenesis due to the fact that the seeded MSCs can sensitively feel and respond to the mechanical features of the host scaffold. Their differentiation, migration and orientation of cell division are highly dependent on the stiffness of the surrounding microenvironment. The researchers demonstrated that MSCs adhesion and growth are also dependent on the scaffold stiffness, which affects the osteo-differentiation and angiogenesis.186 To observe whether pressure and cAMP (as an intracellular second messenger) have any effects on angiogenesis, Linville et al designed a type I collagen gel under intermediate reverse pressure (0.75–1.5 cm H2O) and high cAMP.174 The results were unanimous with previous studies. They all concurred that pressure and cAMP strongly affects angiogenesis. Bramfeld et al demonstrated that in terms of cell adhesion strength, an optimized value between 0.4 and 1.6 MPa exists for the best response for tube formation.176 Interestingly, laminar shear stress has been reported to affect gene transcription, expression of the transformation growth factor beta (TGF- β),176 permeability of the blood vessel wall,177 and induction of changes in cell metabolism or other cellular phenomena173 which severely intervene in angiogenesis.
In the figure, all of the abovementioned biomechanical cues are gathered (Figure 9).
 |
Figure 9 Different biomechanical stimuli for angiogenesis expansion. Created with Procreate Software |
Mechanical stretching due to changes in cell death and migration161,178–180,189 and substrate malleability due to reduced expression of actin and focal adhesion plaque proteins175 have both been reported as other mechanical stimulators influencing capillary sprouting. Other reports disclose that pressure influences angiogenic resistance because it affects cell signaling factors,173 surface tension, cyclic strain, and gravitational force increasingly changing micro vascularization by capillary bleeding or preventing collapse and controlling cell differentiation,172 not only optimizing microcarrier pore size and spacing184 but also the addition of channels to a porous scaffold to promote cell growth and rapid vascularization.182
Also, the polymer chain size within the scaffold has been reported to play an effective role in angiogenesis. For instance, Perng et al changed the size of HA in collagen scaffolds (MW 6.5K and MW 220K) and their results proved that polymer chain size optimization is needed to achieve the best possible response for angiogenesis.183 Moreover, to observe an effect on cell seeding orientation, Stabler et al found that the retention of cells in the supine position is much better than in the standing position.190 Concerning the effects of bulk ECM deformation as a mechanical cue on vessel network expansion, Ruehle et al proved that load initiation time, magnitude, and mode are all key factors that can clearly affect angiogenesis via mechanotransduction signaling pathways. They believed that immediate scaffold load initiation will prevent angiogenesis and reduce sprouting; on the contrary, delayed loading can promote microvascular network formation.191
Effect of Geometric Design on Angiogenesis
To underscore the effects of interconnectivity and geometric design, porous b-tricalcium phosphate scaffolds optimized in terms of interconnectivity and geometrical properties were evaluated by computational simulation models concerning whether the platform endorsed cell activity (like proliferation and migration) or nourishment consumption by cultured cells.192 The consequences demonstrated that incorporating multiple channels within a porous b-tricalcium phosphate scaffold can significantly increase nourishment diffusion through the channels in parallel with HUVEC migration.19 Other research teamsassessed the effect of pore properties (ie, size or interconnectivity) on angiogenesis. They claimed that large pores may disturb the vascularization process in the scaffolds seeded with ECs because ECs cannot bridge pores larger than a cell diameter and this deprived them of adequate migration. The best results were obtained with pores ranging from 5 to 20 μm.193 On the contrary, hydrogels with self-assembling peptides upheld EC adhesion and capillary-network formation due to their small pores, but their flexibility caused swift cell migration, which must be controlled.194 Interestingly, the effect of cell seeding orientation on EC functions has not been neglected by scientists. It was demonstrated that pre-organizing ECs into linear vessels of parallel orientation reduced the time for vascular perfusion after transplantation.151,195
The next step was to evaluate whether the applied stimuli influenced angiogenesis. In parallel, an accurate estimation of tissue-like mechanical properties (like pressure, shear stress, or viscoelasticity) was another issue. The accuracy of these applied properties can be estimated using rheological methods, indentation and compression testing, atomic force microscopy (AFM) and macroscopic rigidity.171 Estimating properties using software, such as FEBio, can help authentic experimental results not only in terms of physiology and angiogenesis but also for fluid mechanical traits, reaction-diffusion processes, and heat transfer phenomenon.196 Such mechanical factors can be controlled by using equipment such as rolling bioreactors, bioreactors with a tubing system/pump, and hollow fiber bioreactors.80 It is worth mentioning that scientists believe that among the state of art approaches, current 3D bio printing techniques are increasingly promising in tissue engineering and can easily print numerous vascular channels, but struggle to support the geometrical complexity of tissues (like forming alveolus in lung tissue engineering due to the fact that they are extremely thin and sensitive requiring nanometer resolution), which extends to inadequate vascularization and neurogenesis formation in further steps.197
Concluding Remarks and Future Perspectives
Angiogenesis is crucial for improving tissue engineering and organ modeling. Because without efficient vasculature and capillaries, adequate nutrient delivery is blocked, and the long-term maintenance of tissue is impossible. In this review, we discussed over some advances in developing materials that can improve angiogenesis in terms of choosing more sufficient biomaterials, the proper cell source, and applied factors. In this manuscript, we presented six main sections.
- Virtually all kinds of synthetic materials (specifically, polymers) that have been studied for angiogenesis with associated seeded cell types were presented. Moreover, we addressed specific growth factors and biomolecules that have been utilized to provoke angiogenesis.
- Approximately all kinds of natural seeded scaffolds used for improved angiogenesis were addressed with seeded cell sources and growth factors. Additionally, separated from the recommended platforms, some of the single natural ingredients that show the most and least promise were compiled.
- The use of hydrogels for accelerating angiogenesis not only in terms of materials but also in terms of cells and growth factors was assembled.
- We suggested several smarter approaches for improving angiogenesis in tissue engineering and regenerative medicine; further, we just alluded to the more accessible ones.
- The most frequent cell sources used in conjunction with scaffolds, their mechanism(s) to promote angiogenesis and their sensitivities were reviewed. In addition, we provided the best cell sources for scaffold seeding.
- We stated some of the most crucial and efficient mechanical/physical traits for improving angiogenesis. Indeed, materials and their mechanical properties are inseparable.
It is abundantly clear that the ultimate effort to improve angiogenesis is ongoing as there are still numerous unsolved problems and unanswered questions including, but not limited, to:
- Providing data regarding sufficient interactions between capillaries and the epithelium surface after capillary formation. This means that even after achieving an appropriate vasculature network, interrelating vessels/capillaries with the tissue surface needs strong consideration.
- Embedding angiogenic growth factors within a scaffold in a precise pattern must be studied. Indeed, an extensive dispersion of growth factors or even cells is challenging because their respective densities in real tissue is intricate and has not yet been modeled, even if using contemporary-release methods. Another concern is dispensing the growth factors as a function of time, which helps one approach real-body simulations. Slow-release methods and/or targeted encapsulation are suggested for optimal angiogenesis.
- Moreover, the danger of an excessive immune system response against xenogeneic growth factors exists. In reality, the immune response has to be controlled not only when we implant engineered tissue but also when growth factors are added to cultured cells.
- Control over the 3D orientation of the formed vessels is another challenge that needs to be solved by utilizing native scaffolds or improving micro- and nano-fabrication technologies scaffold fabrication. However, new fabrication methods have not solved the numerous challenges relevant to angiogenesis and neurogenesis. Hence, it is clear that control over vessel and capillary density and orientation needs to be a top priority.
- Although decellularization-recellularization strategies seem to be by far the most promising pathway for obtaining robust microvascular perfusion because it consists of a native scaffold and inherent tube-like structure, as long as we find a great detergent, which does not harm protein and DNA content, such methods need to be carefully elucidated in that decellularization must not cause vascular channels or tissue disruption.
- On top of everything stated above, preventing blood coagulation within newly formed microvascular networks after cell culturing is critical.
To point out our recommendations, it seems that concentrating on discovering new biomaterials that are apt to mimic the ECM more precisely will have promising results. Moreover, using a combination of appropriate factors (biochemical, mechanical, electrical, magnetic, etc.) and delaying the addition of cells/factors may help. Also, establishing more studies to enhance acellular scaffold performance can broaden our knowledge about the exact mechanisms of angiogenesis. Additionally, various subtypes of materials are needed to reinforce existing scaffolds and can be helpful for preparing an appropriate bed for cells to grow and differentiate. It seems that presumably we have the duty to find the most analogous materials to the tissue itself without using the tissue itself, the most appropriate cell source for transferring to vessels and the most practical mechanical factors (and other stimuli) to apply to the scaffolds for angiogenesis. One item is clear from this review: we have yet to center all of the above-mentioned factors to find the golden key for accelerating angiogenesis. However, the most state-of-the-art method among scientists is utilization of decellularized tissue owing to the fact that it has an appropriate ECM and canals as well as a natural scaffold for the cells that will be seeded further. Keeping the great potential of mesenchymal stem cells in mind, this cell source is a frequent one among plenty of existing studies and should be combined with such decellularized tissue to create the perfect angiogenic environment.
Acknowledgment
The graphics are prepared using Procreate software, therefore we kindly acknowledge their professional service.
Disclosure
The authors report no conflicts of interest in this work.
References
1. De Santis MM, Alsafadi HN, Tas S, et al. Extracellular matrix reinforced bioinks for 3D bioprinting human tissue. Adv Mater. 2021;33:2005476. Wiley Online Library. doi:10.1002/adma.202005476
2. Nasr SM, Rabiee N, Hajebi S, et al. Biodegradable nanopolymers in cardiac tissue engineering: from concept towards nanomedicine. Int J Nanomedicine. 2020;15:4205. doi:10.2147/IJN.S245936
3. Matai I, Kaur G, Seyedsalehi A, McClinton A, Laurencin CT. Progress in 3D bioprinting Technology for tissue/organ regenerative engineering. Biomaterials. 2020;226:119536. Elsevier. doi:10.1016/j.biomaterials.2019.119536
4. Mahfouzi SH, Tali SHS, Amoabediny G. 3D bioprinting for lung and tracheal tissue engineering: criteria, advances, challenges, and future directions. Bioprinting. 2021;21:e00124. Elsevier. doi:10.1016/j.bprint.2020.e00124
5. Shahrezaei F, Mansouri Y, Zinatizadeh A, Akhbari A. Photocatalytic degradation of aniline using TiO2 nanoparticles in a vertical circulating photocatalytic reactor. Int J Photoenergy. 2012;2012:1–8. doi:10.1155/2012/430638
6. Ma X, Agas A, Siddiqui Z, et al. Angiogenic peptide hydrogels for treatment of traumatic brain injury. Bioact Mater. 2020;5(1):124–132. doi:10.1016/j.bioactmat.2020.01.005
7. Rabiee N, Yaraki MT, Garakani SM, et al. Recent advances in porphyrin-based nanocomposites for effective targeted imaging and therapy. Biomaterials. 2020;232:119707. doi:10.1016/j.biomaterials.2019.119707
8. Rahimnejad M, Nasrollahi Boroujeni N, Jahangiri S, et al. Prevascularized micro-/nano-sized spheroid/bead aggregates for vascular tissue engineering. Nano Micro Lett. 2021;13(1):1–24. doi:10.1007/s40820-021-00697-1
9. Mishra R, Panda AK, De Mandal S, Shakeel M, Bisht SS, Khan J. Natural anti-biofilm agents: strategies to control biofilm-forming pathogens. Front Microbiol. 2020;11:2640. Frontiers. doi:10.3389/fmicb.2020.566325
10. Maghsoudi S, Shahraki BT, Rabiee N, et al. Burgeoning polymer nano blends for improved controlled drug release: a review. Int J Nanomedicine. 2020;15:4363. doi:10.2147/IJN.S252237
11. Tavakolizadeh M, Pourjavadi A, Ansari M, et al. An environmentally friendly wound dressing based on a self-healing, extensible and compressible antibacterial hydrogel. Green Chem. 2021;23(3):1312–1329. doi:10.1039/D0GC02719G
12. Farokhi M, Mottaghitalab F, Fatahi Y, et al. Silk fibroin scaffolds for common cartilage injuries: possibilities for future clinical applications. Eur Polym J. 2019;115:251–267. Elsevier. doi:10.1016/j.eurpolymj.2019.03.035
13. Gupta SK, Dinda AK, Potdar PD, Mishra NC. Modification of decellularized goat-lung scaffold with chitosan/nanohydroxyapatite composite for bone tissue engineering applications. Biomed Res Int. 2013;2013:1–11. Hindawi. doi:10.1155/2013/651945
14. Vimalraj S, Rajalakshmi S, Saravanan S, et al. Synthesis and characterization of zinc-silibinin complexes: a potential bioactive compound with angiogenic, and antibacterial activity for bone tissue engineering. Colloids Surf B Biointerfaces. 2018;167:134–143. Elsevier. doi:10.1016/j.colsurfb.2018.04.007
15. Chen W, Xu K, Tao B, et al. Multilayered coating of titanium implants promotes coupled osteogenesis and angiogenesis in vitro and in vivo. Acta Biomater. 2018;74:489–504. Elsevier. doi:10.1016/j.actbio.2018.04.043
16. Linville RM, Arevalo D, Maressa JC, Zhao N, Searson PC. Three-dimensional induced pluripotent stem-cell models of human brain angiogenesis. Microvasc Res. 2020;132:104042. Elsevier. doi:10.1016/j.mvr.2020.104042
17. Chakraborty S, Ponrasu T, Chandel S, Dixit M, Muthuvijayan V. Reduced graphene oxide-loaded nanocomposite scaffolds for enhancing angiogenesis in tissue engineering applications. R Soc Open Sci. 2018;5:172017. The Royal Society Publishing. doi:10.1098/rsos.172017
18. Norahan MH, Pourmokhtari M, Saeb MR, Bakhshi B, Zomorrod MS, Baheiraei N. Electroactive cardiac patch containing reduced graphene oxide with potential antibacterial properties. Mater Sci Eng C. 2019;104:109921. Elsevier. doi:10.1016/j.msec.2019.109921
19. Wang X, Lin M, Kang Y. Engineering porous -tricalcium phosphate (-TCP) scaffolds with multiple channels to promote cell migration, proliferation, and angiogenesis. ACS Appl Mater Interfaces. 2019;11:9223–9232. ACS Publications. doi:10.1021/acsami.8b22041
20. Hench LL, Splinter RJ, Allen WC, Greenlee TK. Bonding mechanisms at the interface of ceramic prosthetic materials. J Biomed Mater Res. 1971;5:117–141. Wiley Online Library. doi:10.1002/jbm.820050611
21. Eke G, Mangir N, Hasirci N, MacNeil S, Hasirci V. Development of a UV crosslinked biodegradable hydrogel containing adipose derived stem cells to promote vascularization for skin wounds and tissue engineering. Biomaterials. 2017;129:188–198. Elsevier. doi:10.1016/j.biomaterials.2017.03.021
22. Wang B, Lv X, Chen S, et al. Use of heparinized bacterial cellulose based scaffold for improving angiogenesis in tissue regeneration. Carbohydr Polym. 2018;181:948–956. Elsevier. doi:10.1016/j.carbpol.2017.11.055
23. Freeman I, Cohen S. The influence of the sequential delivery of angiogenic factors from affinity-binding alginate scaffolds on vascularization. Biomaterials. 2009;30:2122–2131. Elsevier. doi:10.1016/j.biomaterials.2008.12.057
24. Zarrintaj P, Manouchehri S, Ahmadi Z, et al. Agarose-based biomaterials for tissue engineering. Carbohydr Polym. 2018;187:66–84. Elsevier. doi:10.1016/j.carbpol.2018.01.060
25. Rustad KC, Wong VW, Sorkin M, et al. Enhancement of mesenchymal stem cell angiogenic capacity and stemness by a biomimetic hydrogel scaffold. Biomaterials. 2012;33:80–90. Elsevier. doi:10.1016/j.biomaterials.2011.09.041
26. Laschke MW, Rücker M, Jensen G, et al. Improvement of vascularization of PLGA scaffolds by inosculation of in situ-preformed functional blood vessels with the host microvasculature. Ann Surg. 2008;248:939–948. LWW. doi:10.1097/SLA.0b013e31818fa52f
27. Detsch R, Stoor P, Grünewald A, Roether JA, Lindfors NC, Boccaccini AR. Increase in VEGF secretion from human fibroblast cells by bioactive glass S53P4 to stimulate angiogenesis in bone. J Biomed Mater Res A. 2014;102:4055–4061. Wiley Online Library. doi:10.1002/jbm.a.35069
28. Gniesmer S, Brehm R, Hoffmann A, et al. In vivo analysis of vascularization and biocompatibility of electrospun polycaprolactone fibre mats in the rat femur chamber. J Tissue Eng Regen Med. 2019;13:1190–1202. Wiley Online Library. doi:10.1002/term.2868
29. Gérard C, Doillon CJ. Facilitating tissue infiltration and angiogenesis in a tubular collagen scaffold. J Biomed Mater Res A. 2010;93:615–624. Wiley Online Library. doi:10.1002/jbm.a.32568
30. Lu J, Guan F, Cui F, et al. Enhanced angiogenesis by the hyaluronic acid hydrogels immobilized with a VEGF mimetic peptide in a traumatic brain injury model in rats. Regen Biomater. 2019;6:325–334. Oxford University Press. doi:10.1093/rb/rbz027
31. Chan EC, Kuo S-M, Kong AM, et al. Three dimensional collagen scaffold promotes intrinsic vascularisation for tissue engineering applications. PLoS One. 2016;11:e0149799. Public Library of Science San Francisco, CA USA. doi:10.1371/journal.pone.0149799
32. Nour S, Baheiraei N, Imani R, et al. A review of accelerated wound healing approaches: biomaterial-assisted tissue remodeling. J Mater Sci Mater Med. 2019;30(10):120. doi:10.1007/s10856-019-6319-6
33. Prashanth KVH, Tharanathan RN. Depolymerized products of chitosan as potent inhibitors of tumor-induced angiogenesis. Biochim Biophys Acta Gen Subj. 2005;1722:22–29. Elsevier. doi:10.1016/j.bbagen.2004.11.009
34. Jin H, Pi J, Yang F, et al. Ursolic acid-loaded chitosan nanoparticles induce potent anti-angiogenesis in tumor. Appl Microbiol Biotechnol. 2016;100:6643–6652. Springer. doi:10.1007/s00253-016-7360-8
35. Perets A, Baruch Y, Weisbuch F, Shoshany G, Neufeld G, Cohen S. Enhancing the vascularization of three dimensional porous alginate scaffolds by incorporating controlled release basic fibroblast growth factor microspheres. J Biomed Mater Res A. 2003;65:489–497. Wiley Online Library. doi:10.1002/jbm.a.10542
36. Hegen A, Blois A, Tiron CE, et al. Efficient in vivo vascularization of tissue engineering scaffolds. J Tissue Eng Regen Med. 2011;5:e52–e62. Wiley Online Library. doi:10.1002/term.336
37. Singh S, Wu BM, Dunn JCY. Delivery of VEGF using collagen coated polycaprolactone scaffolds stimulates angiogenesis. J Biomed Mater Res A. 2012;100:720–727. Wiley Online Library. doi:10.1002/jbm.a.34010
38. Kampmann A, Lindhorst D, Schumann P, et al. Additive effect of mesenchymal stem cells and VEGF to vascularization of PLGA scaffolds. Microvasc Res. 2013;90:71–79. Elsevier. doi:10.1016/j.mvr.2013.07.006
39. Dikici S, Claeyssens F, MacNeil S. Bioengineering vascular networks to study angiogenesis and vascularization of physiologically relevant tissue models in vitro. ACS Biomater Sci Eng. 2020;6:3513–3528. ACS Publications. doi:10.1021/acsbiomaterials.0c00191
40. Gerhardt L-C, Widdows KL, Erol MM, et al. The pro-angiogenic properties of multi-functional bioactive glass composite scaffolds. Biomaterials. 2011;32:4096–4108. Elsevier. doi:10.1016/j.biomaterials.2011.02.032
41. Day RM, Boccaccini AR, Shurey S, et al. Assessment of polyglycolic acid mesh and bioactive glass for soft-tissue engineering scaffolds. Biomaterials. 2004;25:5857–5866. Elsevier. doi:10.1016/j.biomaterials.2004.01.043
42. Zarrintaj P, Ramsey JD, Samadi A, et al. Poloxamer: a versatile tri-block copolymer for biomedical applications. Acta Biomater. 2020;110:37–67. Elsevier. doi:10.1016/j.actbio.2020.04.028
43. Haifei S, Xingang W, Shoucheng W, Zhengwei M, Chuangang Y, Chunmao H. The effect of collagen–chitosan porous scaffold thickness on dermal regeneration in a one-stage grafting procedure. J Mech Behav Biomed Mater. 2014;29:114–125. Elsevier. doi:10.1016/j.jmbbm.2013.08.031
44. Laschke MW, Strohe A, Scheuer C, et al. In vivo biocompatibility and vascularization of biodegradable porous polyurethane scaffolds for tissue engineering. Acta Biomater. 2009;5:1991–2001. Elsevier. doi:10.1016/j.actbio.2009.02.006
45. Bezuidenhout D, Davies N, Black M, Schmidt C, Oosthuysen A, Zilla P. Covalent surface heparinization potentiates porous polyurethane scaffold vascularization. J Biomater Appl. 2010;24:401–418. Sage Publications Sage UK: London, England. doi:10.1177/0885328208097565
46. Lee H-P, Chen P-C, Wang S-W, et al. Plumbagin suppresses endothelial progenitor cell-related angiogenesis in vitro and in vivo. J Funct Foods. 2019;52:537–544. Elsevier. doi:10.1016/j.jff.2018.11.040
47. DeLong SA, Moon JJ, West JL. Covalently immobilized gradients of bFGF on hydrogel scaffolds for directed cell migration. Biomaterials. 2005;26:3227–3234. Elsevier. doi:10.1016/j.biomaterials.2004.09.021
48. Zhang K, Song L, Wang J, et al. Strategy for constructing vascularized adipose units in poly (l-glutamic acid) hydrogel porous scaffold through inducing in-situ formation of ASCs spheroids. Acta Biomater. 2017;51:246–257. Elsevier. doi:10.1016/j.actbio.2017.01.043
49. He S, Walimbe T, Chen H, et al. Functionalized extracellular matrix scaffolds loaded with endothelial progenitor cells promote neovascularization and diabetic wound healing. bioRxiv. 2021. Cold Spring Harbor Laboratory. doi:10.1101/2021.02.02.429469
50. Fishman JM, Wiles K, Lowdell MW, et al. Airway tissue engineering: an update. Expert Opin Biol Ther. 2014;14:1477–1491. Taylor & Francis. doi:10.1517/14712598.2014.938631
51. Yazdi MK, Taghizadeh A, Taghizadeh M, et al. Agarose-based biomaterials for advanced drug delivery. J Control Release. 2020;326:523–543.
52. Verstegen MMA, Willemse J, Van Den Hoek S, et al. Decellularization of whole human liver grafts using controlled perfusion for transplantable organ bioscaffolds. Stem Cells Dev. 2017;26:1304–1315. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. doi:10.1089/scd.2017.0095
53. Hajebi S, Mohammadi Nasr S, Rabiee N, et al. Bioresorbable composite polymeric materials for tissue engineering applications. Int J Polym Mater Polym Biomater. 2021;1–15. doi:10.1080/00914037.2020.1765365
54. Baheiraei N, Eyni H, Bakhshi B, Najafloo R, Rabiee N. Effects of strontium ions with potential antibacterial activity on in vivo bone regeneration. Sci Rep. 2021;11(1):1–9. doi:10.1038/s41598-021-88058-1
55. Zhou H, Kitano K, Ren X, et al. Bioengineering human lung grafts on porcine matrix. Ann Surg. 2018;267:590–598. LWW. doi:10.1097/SLA.0000000000002129
56. Rabiee N, Bagherzadeh M, Ghadiri AM, et al. Multifunctional 3D hierarchical bioactive green carbon-based nanocomposites. ACS Sustain Chem Eng. 2021;9(26):8706–8720. doi:10.1021/acssuschemeng.1c00781
57. Rabiee N, Bagherzadeh M, Heidarian Haris M, et al. Polymer-coated NH2-UiO-66 for the codelivery of DOX/pCRISPR. ACS Appl Mater Interfaces. 2021;13(9):10796–10811. doi:10.1021/acsami.1c01460
58. Nichols JE, La Francesca S, Vega SP, et al. Giving new life to old lungs: methods to produce and assess whole human paediatric bioengineered lungs. J Tissue Eng Regen Med. 2017;11:2136–2152. Wiley Online Library. doi:10.1002/term.2113
59. Rabiee N, Bagherzadeh M, Jouyandeh M, et al. Natural polymers decorated MOF-MXene nanocarriers for co-delivery of doxorubicin/pCRISPR. ACS Applied Bio Mater. 2021;4(6):5106–5121. doi:10.1021/acsabm.1c00332
60. Gilpin SE, Charest JM, Ren X, et al. Regenerative potential of human airway stem cells in lung epithelial engineering. Biomaterials. 2016;108:111–119. Elsevier. doi:10.1016/j.biomaterials.2016.08.055
61. Saberianpour S, Heidarzadeh M, Geranmayeh MH, Hosseinkhani H, Rahbarghazi R, Nouri M. Tissue engineering strategies for the induction of angiogenesis using biomaterials. J Biol Eng. 2018;12:1–15. Springer. doi:10.1186/s13036-018-0133-4
62. El-Sherbiny IM, Yacoub MH. Hydrogel scaffolds for tissue engineering: progress and challenges. Glob Cardiol Sci Pract. 2013;2013:38. Hamad bin Khalifa University Press (HBKU Press). doi:10.5339/gcsp.2013.38
63. Tsuchiya T, Obata T, Hatachi G, Nagayasu T. Lung microvascular niche, repair, and engineering. Front Bioeng Biotechnol. 2020;8:105. Frontiers. doi:10.3389/fbioe.2020.00105
64. Bourke BM, Roche WR, Appleberg M. Endothelial cell harvest for seeding vascular prostheses: the influence of technique on cell function, viability, and number. J Vasc Surg. 1986;4:257–263. Elsevier. doi:10.1016/0741-5214(86)90195-3
65. Rücker M, Laschke MW, Junker D, et al. Angiogenic and inflammatory response to biodegradable scaffolds in dorsal skinfold chambers of mice. Biomaterials. 2006;27:5027–5038. Elsevier. doi:10.1016/j.biomaterials.2006.05.033
66. Druecke D, Langer S, Lamme E, et al. Neovascularization of poly (ether ester) block copolymer scaffolds in vivo: long term investigations using intravital fluorescent microscopy. J Biomed Mater Res A. 2004;68:10–18. Wiley Online Library. doi:10.1002/jbm.a.20016
67. Nillesen STM, Geutjes PJ, Wismans R, Schalkwijk J, Daamen WF, van Kuppevelt TH. Increased angiogenesis and blood vessel maturation in acellular collagen–heparin scaffolds containing both FGF2 and VEGF. Biomaterials. 2007;28:1123–1131. Elsevier. doi:10.1016/j.biomaterials.2006.10.029
68. Laschke MW, Rücker M, Jensen G, et al. Incorporation of growth factor containing Matrigel promotes vascularization of porous PLGA scaffolds. J Biomed Mater Res A. 2008;85:397–407. Wiley Online Library. doi:10.1002/jbm.a.31503
69. Shepherd BR, Enis DR, Wang F, et al. Vascularization and engraftment of a human skin substitute using circulating progenitor cell derived endothelial cells. FASEB J. 2006;20:1739–1741. Wiley Online Library. doi:10.1096/fj.05-5682fje
70. Rabiee N, Bagherzadeh M, Ghadiri AM, et al. Turning toxic nanomaterials into a safe and bioactive nanocarrier for co-delivery of DOX/pCRISPR. ACS Applied Bio Mater. 2021;4(6):5336–5351. ACS Publications. doi:10.1021/acsabm.1c00447
71. Wagner ER, Parry J, Dadsetan M, et al. VEGF-mediated angiogenesis and vascularization of a fumarate-crosslinked polycaprolactone (PCLF) scaffold. Connect Tissue Res. 2018;59:542–549. Taylor & Francis. doi:10.1080/03008207.2018.1424145
72. Seidi F, Yazdi MK, Jouyandeh M, et al. Chitosan-based blends for biomedical applications. Int J Biol Macromol. 2021;183:1818–1850. Elsevier.
73. Manouchehri S, Bagheri B, Rad SH, et al. Electroactive bio-epoxy incorporated chitosan-oligoaniline as an advanced hydrogel coating for neural interfaces. Prog Org Coat. 2019;131:389–396. Elsevier. doi:10.1016/j.porgcoat.2019.03.022
74. Chen M, Zhang Y, Zhang W, Li J. Polyhedral oligomeric silsesquioxane-incorporated gelatin hydrogel promotes angiogenesis during vascularized bone regeneration. ACS Appl Mater Interfaces. 2020;12(20):22410–22425. doi:10.1021/acsami.0c00714
75. Long G, Liu D, He X, et al. A dual functional collagen scaffold coordinates angiogenesis and inflammation for diabetic wound healing. Biomater Sci. 2020;8:6337–6349. Royal Society of Chemistry. doi:10.1039/D0BM00999G
76. Wong HK, Lam CRI, Wen F, et al. Novel method to improve vascularization of tissue engineered constructs with biodegradable fibers. Biofabrication. 2016;8:15004. IOP Publishing. doi:10.1088/1758-5090/8/1/015004
77. He Q, Zhao Y, Chen B, et al. Improved cellularization and angiogenesis using collagen scaffolds chemically conjugated with vascular endothelial growth factor. Acta Biomater. 2011;7:1084–1093. Elsevier. doi:10.1016/j.actbio.2010.10.022
78. Andrade CF, Wong AP, Waddell TK, Keshavjee S, Liu M. Cell-based tissue engineering for lung regeneration. Am J Physiol Lung Cell Mol Physiol. 2007;292:L510–L518. American Physiological Society. doi:10.1152/ajplung.00175.2006
79. Liu S, Zhang H, Zhang X, et al. Synergistic angiogenesis promoting effects of extracellular matrix scaffolds and adipose-derived stem cells during wound repair. Tissue Eng Part A. 2011;17:725–739. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. doi:10.1089/ten.tea.2010.0331
80. Hsia CCW, Hyde DM, Weibel ER. Lung structure and the intrinsic challenges of gas exchange. Compr Physiol. 2016;6:827. NIH Public Access.
81. Kargozar S, Baino F, Hamzehlou S, Hill RG, Mozafari M. Bioactive glasses: sprouting angiogenesis in tissue engineering. Trends Biotechnol. 2018;36:430–444. Elsevier. doi:10.1016/j.tibtech.2017.12.003
82. Gerhardt LC, Widdows KL, Erol MM, et al. Neocellularization and neovascularization of nanosized bioactive glass coated decellularized trabecular bone scaffolds. J Biomed Mater Res A. 2013;101:827–841. Wiley Online Library. doi:10.1002/jbm.a.34373
83. Shie M-Y, Ding S-J, Chang H-C. The role of silicon in osteoblast-like cell proliferation and apoptosis. Acta Biomater. 2011;7:2604–2614. Elsevier. doi:10.1016/j.actbio.2011.02.023
84. Walker D, Wood S, Southgate J, Holcombe M, Smallwood R. An integrated agent-mathematical model of the effect of intercellular signalling via the epidermal growth factor receptor on cell proliferation. J Theor Biol. 2006;242:774–789. Elsevier. doi:10.1016/j.jtbi.2006.04.020
85. Boyacioglu SO, Korkmaz M, Kahraman E, Yildirim H, Bora S, Ataman OY. Biological effects of tolerable level chronic boron intake on transcription factors. J Trace Elem Med Biol. 2017;39:30–35. Elsevier. doi:10.1016/j.jtemb.2016.06.009
86. Li J, Zhai D, Lv F, et al. Preparation of copper-containing bioactive glass/eggshell membrane nanocomposites for improving angiogenesis, antibacterial activity and wound healing. Acta Biomater. 2016;36:254–266. Elsevier. doi:10.1016/j.actbio.2016.03.011
87. Milan PB, Khamseh S, Zarrintaj P, et al. Copper-enriched diamond-like carbon coatings promote regeneration at the bone–implant interface. Heliyon. 2020;6:e03798. Elsevier.
88. Stoor P, Frantzen J. Influence of bioactive glass S53P4 granules and putty on osteomyelitis associated bacteria in vitro. Biomed Glas. 2017;3:79–85. De Gruyter Open. doi:10.1515/bglass-2017-0007
89. Zhang D, Leppäranta O, Munukka E, et al. Antibacterial effects and dissolution behavior of six bioactive glasses. J Biomed Mater Res A. 2010;93:475–483. Wiley Online Library. doi:10.1002/jbm.a.32564
90. Kim -J-J, El-Fiqi A, Kim H-W. Synergetic cues of bioactive nanoparticles and nanofibrous structure in bone scaffolds to stimulate osteogenesis and angiogenesis. ACS Appl Mater Interfaces. 2017;9(3):2059–2073. doi:10.1021/acsami.6b12089
91. Zarrintaj P, Mahmodi G, Manouchehri S, et al. Zeolite in tissue engineering: opportunities and challenges. MedComm. 2020;1:5–34. Wiley Online Library. doi:10.1002/mco2.5
92. Liakouli V, Cipriani P, Di Benedetto P, et al. The role of extracellular matrix components in angiogenesis and fibrosis: possible implication for systemic sclerosis. Mod Rheumatol. 2018;28(6):922–932. doi:10.1080/14397595.2018.1431004
93. Zhang J, Liu X, Ma K, et al. Collagen/heparin scaffold combined with vascular endothelial growth factor promotes the repair of neurological function in rats with traumatic brain injury. Biomater Sci. 2021;9(3):745–764. doi:10.1039/C9BM01446B
94. Zhang B, Radisic M. Organ-Level Vascularization: The Mars Mission of Bioengineering. Elsevier; 2020.
95. Liu K, Chen C, Zhang H, Chen Y, Zhou S. Adipose stem cell derived exosomes in combination with hyaluronic acid accelerate wound healing through enhancing re epithelialization and vascularization. Br J Dermatol. 2019;181:854–856. doi:10.1111/bjd.17984
96. Bahramsoltani M, Slosarek I, De Spiegelaere W, Plendl J. Angiogenesis and collagen type IV expression in different endothelial cell culture systems. Anat Histol Embryol. 2014;43(2):103–115. Wiley Online Library. doi:10.1111/ahe.12052
97. Ehterami A, Salehi M, Farzamfar S, et al. A promising wound dressing based on alginate hydrogels containing vitamin D3 cross-linked by calcium carbonate/d-glucono- -lactone. Biomed Eng Lett. 2020;10:309. Springer. doi:10.1007/s13534-020-00155-8
98. Mohebbi S, Nezhad MN, Zarrintaj P, et al. Chitosan in biomedical engineering: a critical review. Curr Stem Cell Res Ther. 2019;14:93–116. Bentham Science Publishers.
99. Zhang W, Chen L, Chen J, et al. Silk fibroin biomaterial shows safe and effective wound healing in animal models and a randomized controlled clinical trial. Adv Healthcare Mater. 2017;6(10):1700121. doi:10.1002/adhm.201700121
100. Ribatti D, Nico B, Crivellato E. The role of pericytes in angiogenesis. Int J Dev Biol. 2011;55(3):261–268. doi:10.1387/ijdb.103167dr
101. Morgan JP, Delnero PF, Zheng Y, et al. Formation of microvascular networks in vitro. Nat Protoc. 2013;8:1820–1836. Nature Publishing Group. doi:10.1038/nprot.2013.110
102. Wang B, Lv X, Li Z, et al. Urethra-inspired biomimetic scaffold: a therapeutic strategy to promote angiogenesis for urethral regeneration in a rabbit model. Acta Biomater. 2020;102:247–258. Elsevier. doi:10.1016/j.actbio.2019.11.026
103. Sweeney SM, DiLullo G, Slater SJ, et al. Angiogenesis in collagen I requires 2 1 ligation of a GFP* GER sequence and possibly p38 MAPK activation and focal adhesion disassembly. J Biol Chem. 2003;278:30516–30524. ASBMB. doi:10.1074/jbc.M304237200
104. Schechner JS, Nath AK, Zheng L, et al. In vivo formation of complex microvessels lined by human endothelial cells in an immunodeficient mouse. Proc Natl Acad Sci. 2000;97:9191–9196. National Acad Sciences. doi:10.1073/pnas.150242297
105. Zhang S, Chen J, Yu Y, Dai K, Wang J, Liu C. Accelerated bone regenerative efficiency by regulating sequential release of BMP-2 and VEGF and synergism with sulfated chitosan. ACS Biomater Sci Eng. 2019;5(4):1944–1955. doi:10.1021/acsbiomaterials.8b01490
106. Campbell KT, Stilhano RS, Silva EA. Enzymatically degradable alginate hydrogel systems to deliver endothelial progenitor cells for potential revasculature applications. Biomaterials. 2018;179:109–121. Elsevier. doi:10.1016/j.biomaterials.2018.06.038
107. Mony MP, Shenoy SJ, Raj R, et al. Gelatin-modified cholecyst-derived Scaffold promotes angiogenesis and faster healing of diabetic wounds. ACS Applied Bio Mater. 2021;4(4):3320–3331. doi:10.1021/acsabm.0c01648
108. Anderson SM, Siegman SN, Segura T. The effect of vascular endothelial growth factor (VEGF) presentation within fibrin matrices on endothelial cell branching. Biomaterials. 2011;32:7432–7443. Elsevier. doi:10.1016/j.biomaterials.2011.06.027
109. Shi X, Zhou K, Huang F, Wang C. Interaction of hydroxyapatite nanoparticles with endothelial cells: internalization and inhibition of angiogenesis in vitro through the PI3K/Akt pathway. Int J Nanomedicine. 2017;12:5781. Dove Press. doi:10.2147/IJN.S140179
110. Aljohi A, Matou-Nasri S, Liu D, Al-Khafaji N, Slevin M, Ahmed N. Momordica charantia extracts protect against inhibition of endothelial angiogenesis by advanced glycation endproducts in vitro. Food Funct. 2018;9:5728–5739. Royal Society of Chemistry. doi:10.1039/C8FO00297E
111. Dohle E, El Bagdadi K, Sader R, Choukroun J, James Kirkpatrick C, Ghanaati S. Platelet rich fibrin based matrices to improve angiogenesis in an in vitro co culture model for bone tissue engineering. J Tissue Eng Regen Med. 2018;12:598–610. Wiley Online Library. doi:10.1002/term.2475
112. Uwamori H, Higuchi T, Arai K, Sudo R. Integration of neurogenesis and angiogenesis models for constructing a neurovascular tissue. Sci Rep. 2017;7:1–11. Nature Publishing Group. doi:10.1038/s41598-017-17411-0
113. Naderi H, Matin MM, Bahrami AR. Critical issues in tissue engineering: biomaterials, cell sources, angiogenesis, and drug delivery systems. J Biomater Appl. 2011;26:383–417. Sage Publications Sage UK: London, England. doi:10.1177/0885328211408946
114. Mondrinos MJ, Koutzaki SH, Poblete HM, Crisanti MC, Lelkes PI, Finck CM. In vivo pulmonary tissue engineering: contribution of donor-derived endothelial cells to construct vascularization. Tissue Eng Part A. 2008;14:361–368. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. doi:10.1089/tea.2007.0041
115. Liu S, Yu J, Zhang Q, et al. Dual cross-linked HHA hydrogel supplies and regulates MΦ2 for synergistic improvement of immunocompromise and impaired angiogenesis to enhance diabetic chronic wound healing. Biomacromolecules. 2020;21(9):3795–3806. doi:10.1021/acs.biomac.0c00891
116. Grigoryan B, Paulsen SJ, Corbett DC, et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science. 2019;364:458–464. American Association for the Advancement of Science. doi:10.1126/science.aav9750
117. Van Hove AH, Burke K, Antonienko E, Brown E
118. Mokhames Z, Rezaie Z, Ardeshirylajimi A, Basiri A, Taheri M, Omrani MD. VEGF-incorporated PVDF/collagen nanofibrous scaffold for bladder wall regeneration and angiogenesis. Int J Polym Mater Polym Biomater. 2021;70:521–529. Taylor & Francis. doi:10.1080/00914037.2020.1740985
119. Gao W, Jin W, Li Y, et al. A highly bioactive bone extracellular matrix-biomimetic nanofibrous system with rapid angiogenesis promotes diabetic wound healing. J Mater Chem B. 2017;5:7285–7296. Royal Society of Chemistry. doi:10.1039/C7TB01484H
120. Abebayehu D, Spence AJ, McClure MJ, Haque TT, Rivera KO, Ryan JJ. Polymer scaffold architecture is a key determinant in mast cell inflammatory and angiogenic responses. J Biomed Mater Res A. 2019;107:884–892. Wiley Online Library. doi:10.1002/jbm.a.36605
121. Gugutkov D, Gustavsson J, Cantini M, Salmeron sánchez M, Altankov G. Electrospun fibrinogen–PLA nanofibres for vascular tissue engineering. J Tissue Eng Regen Med. 2017;11:2774–2784. Wiley Online Library. doi:10.1002/term.2172
122. Klumpp D, Rudisile M, Kühnle RI, et al. Three dimensional vascularization of electrospun PCL/collagen blend nanofibrous scaffolds in vivo. J Biomed Mater Res A. 2012;100:2302–2311. Wiley Online Library. doi:10.1002/jbm.a.34172
123. Nazarnezhad S, Baino F, Kim H-W, Webster TJ, Kargozar S. Electrospun nanofibers for improved angiogenesis: promises for tissue engineering applications. Nanomaterials. 2020;10:1609. Multidisciplinary Digital Publishing Institute. doi:10.3390/nano10081609
124. Cui L, Liang J, Liu H, Zhang K, Li J. Nanomaterials for angiogenesis in skin tissue engineering. Tissue Eng Part B Rev. 2020;26(3):203–216. doi:10.1089/ten.teb.2019.0337
125. Levenberg S, Rouwkema J, Macdonald M, et al. Engineering vascularized skeletal muscle tissue. Nat Biotechnol. 2005;23:879–884. Nature Publishing Group. doi:10.1038/nbt1109
126. Thevenot PT, Nair AM, Shen J, Lotfi P, Ko C-Y, Tang L. The effect of incorporation of SDF-1 into PLGA scaffolds on stem cell recruitment and the inflammatory response. Biomaterials. 2010;31:3997–4008. Elsevier. doi:10.1016/j.biomaterials.2010.01.144
127. Hoenig MR, Bianchi C, Sellke FW. Hypoxia inducible factor-1, endothelial progenitor cells, monocytes, cardiovascular risk, wound healing, cobalt and hydralazine: a unifying hypothesis. Curr Drug Targets. 2008;9:422–435. Bentham Science Publishers. doi:10.2174/138945008784221215
128. Dong D, Li J, Cui M, et al. In situ “clickable” zwitterionic starch-based hydrogel for 3D cell encapsulation. ACS Appl Mater Interfaces. 2016;8(7):4442–4455. doi:10.1021/acsami.5b12141
129. Tremblay PL, Hudon V, Berthod F, Germain L, Auger FA. Inosculation of tissue engineered capillaries with the host’s vasculature in a reconstructed skin transplanted on mice. Am J Transpl. 2005;5:1002–1010. Wiley Online Library. doi:10.1111/j.1600-6143.2005.00790.x
130. Zarrintaj P, Zangene E, Manouchehri S, et al. Conductive biomaterials as nerve conduits: recent advances and future challenges. Appl Mater Today. 2020;20:100784. Elsevier. doi:10.1016/j.apmt.2020.100784
131. Kito T, Shibata R, Ishii M, et al. iPS cell sheets created by a novel magnetite tissue engineering method for reparative angiogenesis. Sci Rep. 2013;3:1–8. Nature Publishing Group. doi:10.1038/srep01418
132. Divakar P, Caruso I, Moodie KL, Theiler RN, Hoopes PJ, Wegst UGK. Design, manufacture, and in vivo testing of a tissue scaffold for permanent female sterilization by tubal occlusion. MRS Adv. 2018;3:1685–1690. Materials Research Society. doi:10.1557/adv.2018.57
133. Gorustovich AA, Roether JA, Boccaccini AR. Effect of bioactive glasses on angiogenesis: a review of in vitro and in vivo evidences. Tissue Eng Part B Rev. 2010;16:199–207. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA. doi:10.1089/ten.teb.2009.0416
134. Maghsoudlou P, Georgiades F, Tyraskis A, et al. Preservation of micro-architecture and angiogenic potential in a pulmonary acellular matrix obtained using intermittent intra-tracheal flow of detergent enzymatic treatment. Biomaterials. 2013;34:6638–6648. Elsevier. doi:10.1016/j.biomaterials.2013.05.015
135. Hussein KH, Park K-M, Yu L, Song S-H, Woo H-M, Kwak -H-H. Vascular reconstruction: a major challenge in developing a functional whole solid organ graft from decellularized organs. Acta Biomater. 2020;103:68–80. Elsevier. doi:10.1016/j.actbio.2019.12.029
136. Merckx G, Tay H, Lo Monaco M, et al. Chorioallantoic membrane assay as model for angiogenesis in tissue engineering: focus on stem cells. Tissue Eng Part B Rev. 2020;26:519–539. Mary Ann Liebert, Inc. publishers 140 Huguenot Street, 3rd Floor New. doi:10.1089/ten.teb.2020.0048
137. Gilpin SE, Wagner DE. Acellular human lung scaffolds to model lung disease and tissue regeneration. Eur Respir Rev. 2018;27:180021. Eur Respiratory Soc. doi:10.1183/16000617.0021-2018
138. Kato Y, Iwata T, Morikawa S, Yamato M, Okano T, Uchigata Y. Allogeneic transplantation of an adipose-derived stem cell sheet combined with artificial skin accelerates wound healing in a rat wound model of type 2 diabetes and obesity. Diabetes. 2015;64:2723–2734. Am Diabetes Assoc. doi:10.2337/db14-1133
139. Auger FA, Gibot L, Lacroix D. The pivotal role of vascularization in tissue engineering. Annu Rev Biomed Eng. 2013;15:177–200. Annual Reviews. doi:10.1146/annurev-bioeng-071812-152428
140. Chen L, Xing Q, Zhai Q, et al. Pre-vascularization enhances therapeutic effects of human mesenchymal stem cell sheets in full thickness skin wound repair. Theranostics. 2017;7:117. Ivyspring International Publisher. doi:10.7150/thno.17031
141. Dickman CTD, Russo V, Thain K, et al. Functional characterization of 3D contractile smooth muscle tissues generated using a unique microfluidic 3D bioprinting Technology. FASEB J. 2020;34:1652–1664. Wiley Online Library. doi:10.1096/fj.201901063RR
142. Ren X, Moser PT, Gilpin SE, et al. Engineering pulmonary vasculature in decellularized rat and human lungs. Nat Biotechnol. 2015;33:1097–1102. Nature Publishing Group. doi:10.1038/nbt.3354
143. Zarrintaj P, Moghaddam AS, Manouchehri S, et al. Can regenerative medicine and nanotechnology combine to heal wounds? The search for the ideal wound dressing. Nanomedicine. 2017;12:2403–2422. Future Medicine. doi:10.2217/nnm-2017-0173
144. Nilforoushzadeh MA, Khodadadi Yazdi M, Baradaran Ghavami S, et al. Mesenchymal stem cell spheroids embedded in an injectable thermosensitive hydrogel: an in situ drug formation platform for accelerated wound healing. ACS Biomater Sci Eng. 2020;6:5096–5109. ACS Publications. doi:10.1021/acsbiomaterials.0c00988
145. Li T, Ma H, Ma H, et al. Mussel-inspired nanostructures potentiate the immunomodulatory properties and angiogenesis of mesenchymal stem cells. ACS Appl Mater Interfaces. 2019;11(19):17134–17146. doi:10.1021/acsami.8b22017
146. White AC, Lavine KJ, Ornitz DM. FGF9 and SHH Regulate Mesenchymal Vegfa Expression and Development of the Pulmonary Capillary Network. Oxford University Press for The Company of Biologists Limited; 2007.
147. Genova T, Petrillo S, Zicola E, et al. The crosstalk between osteodifferentiating stem cells and endothelial cells promotes angiogenesis and bone formation. Front Physiol. 2019;10:1291. Frontiers. doi:10.3389/fphys.2019.01291
148. McFadden TM, Duffy GP, Allen AB, et al. The delayed addition of human mesenchymal stem cells to pre-formed endothelial cell networks results in functional vascularization of a collagen–glycosaminoglycan scaffold in vivo. Acta Biomater. 2013;9:9303–9316. Elsevier. doi:10.1016/j.actbio.2013.08.014
149. Mura M, Binnie M, Han B, et al. Functions of type II pneumocyte-derived vascular endothelial growth factor in alveolar structure, acute inflammation, and vascular permeability. Am J Pathol. 2010;176:1725–1734. Elsevier. doi:10.2353/ajpath.2010.090209
150. Panina YA, Yakimov AS, Komleva YK, et al. Plasticity of adipose tissue-derived stem cells and regulation of angiogenesis. Front Physiol. 2018;9:1656. Frontiers. doi:10.3389/fphys.2018.01656
151. Ling T-Y, Liu Y-L, Huang Y-K, et al. Differentiation of lung stem/progenitor cells into alveolar pneumocytes and induction of angiogenesis within a 3D gelatin–microbubble scaffold. Biomaterials. 2014;35:5660–5669. Elsevier. doi:10.1016/j.biomaterials.2014.03.074
152. Spiller KL, Anfang RR, Spiller KJ, et al. The role of macrophage phenotype in vascularization of tissue engineering scaffolds. Biomaterials. 2014;35(15):4477–4488. doi:10.1016/j.biomaterials.2014.02.012
153. Danese S, Dejana E, Fiocchi C. Immune regulation by microvascular endothelial cells: directing innate and adaptive immunity, coagulation, and inflammation. J Immunol. 2007;178:6017–6022. Am Assoc Immnol. doi:10.4049/jimmunol.178.10.6017
154. Noonan DM, Barbaro ADL, Vannini N, Mortara L, Albini A. Inflammation, inflammatory cells and angiogenesis: decisions and indecisions. Cancer Metastasis Rev. 2008;27:31–40. Springer. doi:10.1007/s10555-007-9108-5
155. Wagner EM, Sánchez J, McClintock JY, Jenkins J, Moldobaeva A. Inflammation and ischemia-induced lung angiogenesis. Am J Physiol Lung Cell Mol Physiol. 2008;294:L351–L357. American Physiological Society. doi:10.1152/ajplung.00369.2007
156. Laschke MW, Menger MD. Spheroids as vascularization units: from angiogenesis research to tissue engineering applications. Biotechnol Adv. 2017;35:782–791. Elsevier. doi:10.1016/j.biotechadv.2017.07.002
157. Zimmermann JA, Mcdevitt TC. Pre-conditioning mesenchymal stromal cell spheroids for immunomodulatory paracrine factor secretion. Cytotherapy. 2014;16(3):331–345. doi:10.1016/j.jcyt.2013.09.004
158. Lee E, Takahashi H, Pauty J, et al. A 3D in vitro pericyte-supported microvessel model: visualisation and quantitative characterisation of multistep angiogenesis. J Mater Chem B. 2018;6:1085–1094. Royal Society of Chemistry. doi:10.1039/C7TB03239K
159. Grönman M, Moisio O, Li X-G, et al. Association between [68Ga] NODAGA-RGDyK uptake and dynamics of angiogenesis in a human cell-based 3D model. Mol Biol Rep. 2021;48(6):5347–5353. Springer.
160. Komaki M, Numata Y, Morioka C, et al. Exosomes of human placenta-derived mesenchymal stem cells stimulate angiogenesis. Stem Cell Res Ther. 2017;8:1–12. BioMed Central. doi:10.1186/s13287-017-0660-9
161. Heise RL, Stober V, Cheluvaraju C, Hollingsworth JW, Garantziotis S. Mechanical stretch induces epithelial-mesenchymal transition in alveolar epithelia via hyaluronan activation of innate immunity. J Biol Chem. 2011;286:17435–17444. ASBMB. doi:10.1074/jbc.M110.137273
162. Milkiewicz M, Doyle JL, Fudalewski T, Ispanovic E, Aghasi M, Haas TL. HIF 1 and HIF 2 play a central role in stretch induced but not shear stress induced angiogenesis in rat skeletal muscle. J Physiol. 2007;583:753–766. Wiley Online Library. doi:10.1113/jphysiol.2007.136325
163. Ohata K, Ott HC. Human-scale lung regeneration based on decellularized matrix scaffolds as a biologic platform. Surg Today. 2020;50:633–643. Springer. doi:10.1007/s00595-020-02000-y
164. Varley C, Hill G, Pellegrin S, et al. Autocrine regulation of human urothelial cell proliferation and migration during regenerative responses in vitro. Exp Cell Res. 2005;306:216–229. Elsevier. doi:10.1016/j.yexcr.2005.02.004
165. Nossa R, Costa J, Cacopardo L, Ahluwalia A. Breathing in vitro: designs and applications of engineered lung models. J Tissue Eng. 2021;12:20417314211008696. doi:10.1177/20417314211008696
166. Fuchs S, Motta A, Migliaresi C, Kirkpatrick CJ. Outgrowth endothelial cells isolated and expanded from human peripheral blood progenitor cells as a potential source of autologous cells for endothelialization of silk fibroin biomaterials. Biomaterials. 2006;27(31):5399–5408. doi:10.1016/j.biomaterials.2006.06.015
167. Bruzauskaite I, Raudoniute J, Denkovskij J, et al. Native matrix-based human lung alveolar tissue model in vitro: studies of the reparatory actions of mesenchymal stem cells. Cytotechnology. 2017;69(1):1–17. doi:10.1007/s10616-016-0021-z
168. Ma J, Both SK, Yang F, et al. Concise review: cell‐based strategies in bone tissue engineering and regenerative medicine. Stem Cells Transl Med. 2014;3(1):98–107. doi:10.5966/sctm.2013-0126
169. Iravani S, Varma RS. Green synthesis, biomedical and biotechnological applications of carbon and graphene quantum dots. A review. Environ Chem Lett. 2020;1–25. doi:10.1007/s10311-020-00984-0
170. Graney PL, Ben-Shaul S, Landau S, et al. Macrophages of diverse phenotypes drive vascularization of engineered tissues. Sci Adv. 2020;6:eaay6391. American Association for the Advancement of Science. doi:10.1126/sciadv.aay6391
171. de Hilster RHJ, Sharma PK, Jonker MR, et al. Human lung extracellular matrix hydrogels resemble the stiffness and viscoelasticity of native lung tissue. Am J Physiol Lung Cell Mol Physiol. 2020;318:L698–L704. American Physiological Society Bethesda, MD. doi:10.1152/ajplung.00451.2019
172. Campillo N, Jorba I, Schaedel L, et al. A novel chip for cyclic stretch and intermittent hypoxia cell exposures mimicking obstructive sleep apnea. Front Physiol. 2016;7:319. Frontiers. doi:10.3389/fphys.2016.00319
173. Da Palma RK, Nonaka PN, Campillo N, et al. Behavior of vascular resistance undergoing various pressure insufflation and perfusion on decellularized lungs. J Biomech. 2016;49:1230–1232. Elsevier. doi:10.1016/j.jbiomech.2016.02.043
174. Linville RM, Boland NF, Covarrubias G, Price GM, Tien J. Physical and chemical signals that promote vascularization of capillary-scale channels. Cell Mol Bioeng. 2016;9:73–84. Springer. doi:10.1007/s12195-016-0429-8
175. Senger D, Davis G. Angiogenesis. Cold Spring Harb Perspect Biol. 2011;3(8):a005090. doi:10.1101/cshperspect.a005090
176. Bramfeld H, Sabra G, Centis V, Vermette P. Scaffold vascularization: a challenge for three-dimensional tissue engineering. Curr Med Chem. 2010;17:3944–3967. Bentham Science Publishers. doi:10.2174/092986710793205327
177. Buchanan CF, Verbridge SS, Vlachos PP, Rylander MN. Flow shear stress regulates endothelial barrier function and expression of angiogenic factors in a 3D microfluidic tumor vascular model. Cell Adh Migr. 2014;8:517–524. Taylor & Francis. doi:10.4161/19336918.2014.970001
178. Gladman SJ, Ward RE, Michael-Titus AT, Knight MM, Priestley JV. The effect of mechanical strain or hypoxia on cell death in subpopulations of rat dorsal root ganglion neurons in vitro. Neuroscience. 2010;171:577–587. Elsevier. doi:10.1016/j.neuroscience.2010.07.009
179. Liang X, Huang X, Zhou Y, Jin R, Li Q. Mechanical stretching promotes skin tissue regeneration via enhancing mesenchymal stem cell homing and transdifferentiation. Stem Cells Transl Med. 2016;5:960–969. Wiley Online Library. doi:10.5966/sctm.2015-0274
180. Zhao J, Nishimura Y, Kimura A, et al. Chemokines protect vascular smooth muscle cells from cell death induced by cyclic mechanical stretch. Sci Rep. 2017;7(1):1–9. doi:10.1038/s41598-016-0028-x
181. Ott HC, Clippinger B, Conrad C, et al. Regeneration and orthotopic transplantation of a bioartificial lung. Nat Med. 2010;16:927–933. Nature Publishing Group. doi:10.1038/nm.2193
182. Kang Y, Chang J. Channels in a porous scaffold: a new player for vascularization. Regen Med. 2018;13:705–715. Future Medicine. doi:10.2217/rme-2018-0022
183. Perng C-K, Wang Y-J, Tsi C-H, Ma H. In vivo angiogenesis effect of porous collagen scaffold with hyaluronic acid oligosaccharides. J Surg Res. 2011;168:9–15. Elsevier. doi:10.1016/j.jss.2009.09.052
184. Mehdizadeh H, Bayrak ES, Lu C, et al. Agent-based modeling of porous scaffold degradation and vascularization: optimal scaffold design based on architecture and degradation dynamics. Acta Biomater. 2015;27:167–178. Elsevier. doi:10.1016/j.actbio.2015.09.011
185. Graham HK, McConnell JC, Limbert G, Sherratt MJ. How stiff is skin? Exp Dermatol. 2019;28:4–9. doi:10.1111/exd.13826
186. Handorf AM, Zhou Y, Halanski MA, Li W-J. Tissue stiffness dictates development, homeostasis, and disease progression. Organogenesis. 2015;11(1):1–15. doi:10.1080/15476278.2015.1019687
187. Murphy MC, Huston III, Jack J
188. Liu L, You Z, Yu H, et al. Mechanotransduction-modulated fibrotic microniches reveal the contribution of angiogenesis in liver fibrosis. Nat Mater. 2017;16:1252–1261. Nature Publishing Group. doi:10.1038/nmat5024
189. Ren Y, Zhan Q, Hu Q, Sun B, Yang C, Wang C. Static stretch induces active morphological remodeling and functional impairment of alveolar epithelial cells. Respiration. 2009;78:301–311. Karger Publishers. doi:10.1159/000207632
190. Stabler CT, Caires L
191. Ruehle MA, Eastburn EA, LaBelle SA, et al. Extracellular matrix compression temporally regulates microvascular angiogenesis. Sci Adv. 2020;6:eabb6351. American Association for the Advancement of Science. doi:10.1126/sciadv.abb6351
192. Hauser S, Jung F, Pietzsch J. Human endothelial cell models in biomaterial research. Trends Biotechnol. 2017;35(3):265–277. doi:10.1016/j.tibtech.2016.09.007
193. Narayan D, Venkatraman SS. Effect of pore size and interpore distance on endothelial cell growth on polymers. J Biomed Mater Res A. 2008;87:710–718. Wiley Online Library. doi:10.1002/jbm.a.31749
194. Narmoneva DA, Oni O, Sieminski AL, et al. Self-assembling short oligopeptides and the promotion of angiogenesis. Biomaterials. 2005;26:4837–4846. Elsevier. doi:10.1016/j.biomaterials.2005.01.005
195. Mirabella T, MacArthur JW, Cheng D, et al. 3D-printed vascular networks direct therapeutic angiogenesis in ischaemia. Nat Biomed Eng. 2017;1:1–8. Nature Publishing Group. doi:10.1038/s41551-017-0083
196. Sart S, Agathos SN, Li Y. Engineering stem cell fate with biochemical and biomechanical properties of microcarriers. Biotechnol Prog. 2013;29:1354–1366. Wiley Online Library. doi:10.1002/btpr.1825
197. Edgar L, Pu T, Porter B, et al. Regenerative medicine, organ bioengineering and transplantation. J Br Surg. 2020;107:793–800. Oxford University Press. doi:10.1002/bjs.11686
198. Narayan R, Agarwal T, Mishra D, Maiti TK, Mohanty S. Goat tendon collagen-human fibrin hydrogel for comprehensive parametric evaluation of HUVEC microtissue-based angiogenesis. Colloids Surf B Biointerfaces. 2018;163:291–300. doi:10.1016/j.colsurfb.2017.12.056
199. Mammoto A, Connor KM, Mammoto T, et al. A mechanosensitive transcriptional mechanism that controls angiogenesis. Nature. 2009;457(7233):1103–1108. doi:10.1038/nature07765
200. Xiao X, Wang W, Liu D, et al. The promotion of angiogenesis induced by three-dimensional porous beta-tricalcium phosphate scaffold with different interconnection sizes via activation of PI3K/Akt pathways. Sci Rep. 2015;5:1–11. Nature Publishing Group.
201. Lee J-H, Parthiban P, Jin G-Z, Knowles JC, Kim H-W. Materials roles for promoting angiogenesis in tissue regeneration. Prog Mater Sci. 2021;117:100732.
202. Deveci D, Marshall JM, Egginton S. Relationship between capillary angiogenesis, fiber type, and fiber size in chronic systemic hypoxia. Am J Physiol Heart Circ Physiol. 2001;281(1):H241–H252. doi:10.1152/ajpheart.2001.281.1.H241
203. Gorodnova N, Kolobov A, Mynbaev O, Simakov S. Mathematical modeling of blood flow alteration in microcirculatory network due to angiogenesis. Lobachevskii J Math. 2016;37(5):541–549. doi:10.1134/S199508021605005X
204. Ueda A, Koga M, Ikeda M, Kudo S, Tanishita K. Effect of shear stress on microvessel network formation of endothelial cells with in vitro three-dimensional model. Am J Physiol Heart Circ Physiol. 2004;287:H994–H1002. American Physiological Society. doi:10.1152/ajpheart.00400.2003
205. Nauli SM, Kawanabe Y, Kaminski JJ, Pearce WJ, Ingber DE, Zhou J. Endothelial cilia are fluid shear sensors that regulate calcium signaling and nitric oxide production through polycystin-1. Circulation. 2008;117(9):1161–1171. doi:10.1161/CIRCULATIONAHA.107.710111
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

