Back to Journals » OncoTargets and Therapy » Volume 13
Case Report: A Metabolic Complete Response to Upfront Osimertinib in a Smoker Non-Small Cell Lung Cancer Patient Harbouring EGFR G719A/V769M Complex Mutation
Authors Simionato F, Calvetti L, Cosci M, Scarparo S, Aprile G
Received 17 September 2020
Accepted for publication 29 October 2020
Published 23 November 2020 Volume 2020:13 Pages 12027—12031
DOI https://doi.org/10.2147/OTT.S280933
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Takuya Aoki
Francesca Simionato,1 Lorenzo Calvetti,1 Marco Cosci,2 Silvia Scarparo,3 Giuseppe Aprile1
1Department of Oncology, San Bortolo General Hospital, Vicenza, Italy; 2Department of Surgery, San Bortolo General Hospital, Vicenza, Italy; 3Pneumology Unit, San Bortolo General Hospital, Vicenza, Italy
Correspondence: Francesca Simionato Tel +39444753608
Email [email protected]
Abstract: Complex EGFR mutations are rare in non-small cell lung cancer (NSCLC). Limited clinical evidence is available on the efficacy of EGFR tyrosine kinase inhibitors (TKIs) in patients with NSCLC harbouring these uncommon EGFR mutations. Here, we reported the case of a complete metabolic response in a patient with advanced NSCLC carrying the uncommon EGFR G719A/V769M complex mutation treated with the first-line osimertinib.
Keywords: non-small cell lung cancer, EGFR complex mutations, osimertinib
Introduction
The treatment of NSCLC patients carrying sensitizing EGFR mutations has been revolutionized by EGFR-targeted therapies. EGFR exon 19 deletions and exon 21 L858R substitutions are the most frequent, covering about 80% of all mutations in NSCLC and resulting in a strong sensitivity to tyrosine kinase inhibitors (TKIs) as oncogenic drivers.1 Other EGFR mutations are defined uncommon and represent a heterogeneous group of molecular alterations including exon 18 point mutations, exon 20 insertions and combined complex mutations.2 Retrospective studies, reports of clinical cases and few prospective data showed inconsistent clinical activity of EGFR-TKIs in patients carrying these rare mutations.3,4
Moreover, smoking status is a well-known negative predictive clinical factor for EGFR-TKIs activity as never smokers have been found to be more sensitive to EGFR-TKIs than former or current ones.5 Intriguingly, while common mutations are mostly found in never smokers, those defined uncommon are frequent in patients with smoking history.6
Osimertinib, a third-line generation EGFR-TKI, is a novel upfront treatment option for patients affected by NSCLC with sensitizing alterations.7 However, limited clinical data of osimertinib are available to define the predictive role of uncommon EGFR mutations as the large prospective trials enrolled only NSCLC patients carrying common sensitive mutations (exon 19 deletion + exon 21 L858R).7,8 A small Phase II trial conducted on 36 NSCLC patients with uncommon EGFR mutations and treated with osimertinib demonstrated encouraging response rates with manageable toxicities.9
Here, we report a case of excellent disease response in a smoker patient with advanced NSCLC harbouring uncommon complex EGFR mutation treated with osimertinib as first-line therapy.
Case Presentation
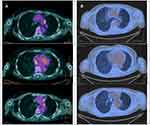
A Caucasian 69-year-old housewife with a smoking history of 40 pack-years and no significant environmental exposures underwent right upper lobectomy in February 2015 for a stage IIb lung adenocarcinoma. The patients received three cycles of adjuvant chemotherapy with cisplatin and gemcitabine and subsequent standard follow-up. She had no other relevant morbidities, nor family history of cancer. During follow-up, a Computed Tomography (CT) scan performed in November 2019 and a Positron Emission Tomography CT (PET-CT) scan in December 2019 showed disease recurrence in multiple small lung nodes and mediastinal para aortic, aorto-pulmonary and left paravertebral lymph nodes (Figures 1A and 2A). A molecular analysis looking for genetic alterations of EGFR (exon 18, 19, 20 and 21), ALK and ROS-1 genes and PDL-1 immunohistochemistry (ICH) expression were performed in the primary tumor as a new biopsy was not technically feasible. Uncommon complex EGFR mutation (exon 18 G719A and exon 20 V769M) was detected using the polymerase chain reaction (PCR)-based Sanger sequencing. No additional alterations other than EGFR mutations were found as DAKO ALK and ROS-1 D4D6 ICH were negative. DAKO PDL-1 ICH 22 C3 was also negative (Tumor Proportion Score 0%).
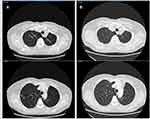 |
Figure 2 Chest CT scan. (A) Baseline disease in multiple small lung nodes. (B) Complete disease response after 12 weeks of therapy with osimertinib. |
The patient started first-line osimertinib 80 mg/day on the 2nd of February 2020. The treatment was well tolerated with no adverse events. On the 21st of April 2020, a restaging PET CT scan demonstrated a disease complete response to osimertinib (Figures 1B and 2B). The patient is still on treatment at full dose (last follow-up was on 12th October 2020).
A written informed consent was provided by the patient to have the case details and radiological images published. No institution approval was required to publish the case details.
Discussion
Nowadays, TKIs treatment is considered the gold standard in NSCLC harbouring classic EGFR mutation. In particular, TKIs demonstrated superior efficacy than standard chemotherapy in several randomized trials.7,10,11 Moreover, new clinical data suggested poor and unpredictable activity of immunotherapy in this molecular setting.12
However, limited clinical evidence is available on the efficacy of EGFR-TKIs in patients with NSCLC harbouring uncommon EGFR mutations. Intratumoral heterogeneity with the presence of different subpopulations resistant to targeted therapy is thought to be one reason of intrinsic drug resistance in some of these patients.13 In particular, tumors with high metastatic burden may be related to higher intratumoral heterogeneity and worst outcome to TKIs than oligometastatic ones.
Large retrospective analyses suggested that EGFR-TKIs have unpredictable activity in patients with tumours harbouring these uncommon mutations than the common ones.3 Complex EGFR mutations account for 3–14% of all EGFR mutations. Retrospective analyses suggest that patients carrying specific complex mutations may benefit from first and second-generation TKIs as patients with classical mutations.14,15 Moreover, a combined post hoc analysis of three prospective clinical trials of afatinib suggested a potential activity in NSCLC with certain types of uncommon EGFR mutations, including complex mutations with exon 18 and exon 21.4
In addition, smoking history has been related to a lower incidence of EGFR mutations and poor outcome to TKIs in EGFR mutant tumors in smokers. However, uncommon and complex mutations are more often in smokers than classic mutations and TKIs activity in these patients has not been clearly defined.16
At present, osimertinib is considered one first-line standard of therapy for NSCLC carrying classical mutations. However, its activity in NSCLC with these complex EGFR mutations is still unclear, although first clinical data showing promising activity are emerging. In particular, Cho et al reported four cases of complex mutations with significant response rates.9 None of these patients carried the specific complex mutation detected in our patient. As far as we know, this is the first report of remarkable EGFR-TKI antitumor activity in NSCLC carrying complex exon 18 G719A + exon 20 V769M mutation. Tumor assessment was conducted with both CT scan and PET-CT as part of an internal analysis of radiomic phenotyping of these tumors and gave us evidence of the deepresponse to osimertinib.
Certainly, this is a single report of the activity of osimertinib in these rare alterations that cannot be considered conclusive. However, we strongly believe that also single cases are clinically relevant in such a rare molecular setting where a larger series of cases are hard to collect in singular institutions.
Conclusion
Uncommon complex EGFR mutations can be present in naïve smoker NSCLC patients and may be associated with disease response to the first-line osimertinib, irrespectively to smoking status. Efforts to describe these rare molecular alterations and their predictive role for a response to EGFR-TKIs better in larger series of cases are necessary to guide clinicians to personalize therapeutic strategies for NSCLC patients.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Lee CK, Brown C, Gralla RJ, et al. Impact of EGFR inhibitor in non-small cell lung cancer on progression-free and overall survival: a meta-analysis. J Natl Cancer Inst. 2013;105(9):595–605. doi:10.1093/jnci/djt072
2. Wu JY, Yu CJ, Chang YC, Yang CH, Shih JY, Yang PC. Effectiveness of tyrosine kinase inhibitors on “uncommon” epidermal growth factor receptor mutations of unknown clinical significance in non-small cell lung cancer. Clin Cancer Res. 2011;17(11):3812–3821. doi:10.1158/1078-0432.CCR-10-3408
3. Beau-Faller M, Prim N, Ruppert AM, et al. Rare EGFR exon 18 and exon 20 mutations in non-small-cell lung cancer on 10 117 patients: a multicentre observational study by the French ERMETIC-IFCT network. Ann Oncol. 2014;25(1):126–131. doi:10.1093/annonc/mdt418
4. Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-lung 2, LUX-lung 3, and LUX-lung 6. Lancet Oncol. 2015;16(7):830–838. doi:10.1016/S1470-2045(15)00026-1
5. Kim MH, Kim HR, Cho BC, et al. Impact of cigarette smoking on response to epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitors in lung adenocarcinoma with activating EGFR mutations. Lung Cancer. 2014;84(2):196–202. doi:10.1016/j.lungcan.2014.01.022
6. Tu HY, Ke EE, Yang JJ, et al. A comprehensive review of uncommon EGFR mutations in patients with non-small cell lung cancer. Lung Cancer. 2017;114:96–102. doi:10.1016/j.lungcan.2017.11.005
7. Soria JC, Ohe Y, Vansteenkiste J, et al. Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N Engl J Med. 2018;378(2):113–125. doi:10.1056/NEJMoa1713137
8. Mok TS, Wu YL, Ahn MJ, et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. 2017;376(7):629–640. doi:10.1056/NEJMoa1612674
9. Cho JH, Lim SH, An HJ, et al. Osimertinib for patients with non-small-cell lung cancer harboring uncommon EGFR mutations: a multicenter, open-label, phase II trial (KCSG-LU15-09). J Clin Oncol. 2020;38(5):488–495. doi:10.1200/JCO.19.00931
10. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised Phase 3 trial. Lancet Oncol. 2012;13(3):239–246. doi:10.1016/S1470-2045(11)70393-X
11. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947–957. doi:10.1056/NEJMoa0810699
12. Lee CK, Man J, Lord S, et al. Checkpoint inhibitors in metastatic EGFR-mutated non-small cell lung cancer-a meta-analysis. J Thorac Oncol. 2017;12(2):403–407. doi:10.1016/j.jtho.2016.10.007
13. Lim ZF, Ma PC. Emerging insights of tumor heterogeneity and drug resistance mechanisms in lung cancer targeted therapy. J Hematol Oncol. 2019;12(1):134. doi:10.1186/s13045-019-0818-2
14. Passaro A, Prelaj A, Bonanno L, et al. Activity of EGFR TKIs in caucasian patients with NSCLC harboring potentially sensitive uncommon EGFR mutations. Clin Lung Cancer. 2019;20(2):e186–e194. doi:10.1016/j.cllc.2018.11.005
15. Yang JC, Schuler M, Popat S, et al. Afatinib for the treatment of NSCLC harboring uncommon EGFR mutations: a database of 693 cases. J Thorac Oncol. 2020;15(5):803–815. doi:10.1016/j.jtho.2019.12.126
16. Kauffmann-Guerrero D, Huber RM, Reu S, et al. NSCLC patients harbouring rare or complex EGFR mutations are more often smokers and might not benefit from first-line tyrosine kinase inhibitor therapy. Respiration. 2018;95(3):169–176. doi:10.1159/000484175
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.