Back to Journals » International Medical Case Reports Journal » Volume 8
Cardiac resynchronization therapy in a young patient with Duchenne muscular dystrophy
Authors Kono T, Ogimoto A, Nishimura K, Yorozuya T, Okura T, Higaki J
Received 28 April 2015
Accepted for publication 30 June 2015
Published 21 August 2015 Volume 2015:8 Pages 173—175
DOI https://doi.org/10.2147/IMCRJ.S87512
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ronald Prineas
Tamami Kono,1 Akiyoshi Ogimoto,1 Kazuhisa Nishimura,1 Toshihiro Yorozuya,2 Takafumi Okura,1 Jitsuo Higaki1
1Department of Cardiology, Pulmonology, Hypertension and Nephrology, 2Department of Anesthesiology and Resuscitology, Ehime University Graduate School of Medicine, Toon, Ehime, Japan
Abstract: A 32-year-old man with Duchenne muscular dystrophy (DMD) was admitted to the hospital because of worsening dyspnea and general fatigue. He had received medication therapy for cardiomyopathy with heart failure and home mechanical ventilation for respiratory failure. An electrocardiogram on admission showed intermittent third-degree atrioventricular block. Echocardiography showed global mild left ventricular systolic dysfunction with dyssynchrony (ejection fraction: 45%). He underwent implantation of a cardiac resynchronization therapy–defibrillator. His B-type natriuretic peptide level was improved after cardiac resynchronization therapy–defibrillator implantation, and he remains asymptomatic. The incidence of cardiomyopathy increases with age. By adulthood, 100% of patients have cardiac involvement.
Keywords: cardiac resynchronization therapy, Duchenne muscular dystrophy, progression of cardiomyopathy, heart failure, arrhythmia
A Letter to the Editor has been received and published for this article.
Introduction
Improvement of respiratory support and pharmacotherapy for heart failure has brought extension of life span to patients with Duchenne muscular dystrophy (DMD). However, the incidence of cardiomyopathy increases with age; 100% of patients have cardiac involvement by adulthood.1 Some kinds of arrhythmia cause fatal matters as well as worsening heart failure. Some DMD patients will need device therapy such as a permanent pacemaker2,3 or an implantable cardioverter-defibrillator (ICD).4 ICD and cardiac resynchronization therapy have not been established in DMD patients. However, it has been reported that these therapies are beneficial to cardiomyopathy patients with systolic dysfunction.5,6
Case report
A 32-year-old man with DMD was admitted to the hospital because of worsening dyspnea and general fatigue. The diagnosis of DMD was made by muscle biopsy when he was 5 years old. He had received ventilation support for respiratory failure by a volume-controlled ventilator from 20 years of age. Complete left bundle branch block (CLBBB) appeared in electrocardiogram from 3 years before this admission. At the same time, echocardiography revealed abnormal left ventricular wall motion and dyssynchrony. So, a small dose of carvedilol was started for cardiomyopathy. It was gradually increased to 10 mg/day. But he felt dizziness sometimes from 1 month before this admission. Electrocardiogram showed bradycardia with Mobitz type II second-degree atrioventricular block (Figure 1A). He was admitted for treatment of arrhythmia. On admission, his blood pressure was 90/50 mmHg, heart rate was 30 bpm, and oxygen saturation was 97% with room air. Electrocardiogram showed intermittent third-degree atrioventricular block (Figure 1B). Echocardiography showed global mild left ventricular systolic dysfunction with dyssynchrony (ejection fraction: 45%). No congenital heart disease or coronary stenosis was observed on cardiac computed tomography imaging. The results of laboratory tests showed elevation of plasma levels of creatine phosphokinase (843 U/L), troponin T (0.073 ng/mL), and B-type natriuretic peptide (BNP) (871.0 pg/mL). He underwent implantation of a cardiac resynchronization therapy–defibrillator (CRT-D) under general anesthesia because of strong systemic contracture. The device generator was implanted under the greater pectoral muscle with the assistance of a plastic surgeon because his subcutaneous tissue had very poor growth. Biventricular pacing was begun immediately after implantation of the CRT-D (Figure 2). He was discharged home without any complications. Carvedilol was gradually increased to 20 mg/day. His ventilation support was continued with the same conditions as those before CRT-D implantation. His BNP level (196.0 pg/mL) was improved 7 months after CRT-D implantation. Furthermore, his BNP level was decreased to 116.0 pg/mL after 20 months, and he remains asymptomatic. He is free from complaint and hospitalization for worsening heart failure after CRT-D implantation.
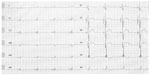  | Figure 2 An electrocardiogram shows biventricular pacing after CRT-D implantation. |
Discussion
DMD is an X-linked recessive disease that has an incidence of approximately one in 3,500 male births.1 This disease results from mutations in the dystrophin gene on chromosome Xp21. Consequently, the absence of dystrophin results in the development of skeletal muscle weakness, cardiomyopathy, and respiratory muscle weakness. The incidence of cardiomyopathy increases with age. Approximately 25% of DMD patients have cardiomyopathy by 6 years of age, and 59% are affected between 6 and 10 years of age. By adulthood, 100% of patients have cardiac involvement.1 Cardiomyopathy is due to replacement of myocardium by connective tissue or fat, and there is both cardiac dysfunction and arrhythmia. Progression of cardiomyopathy leads to heart failure and sudden death.
It has been reported that biventricular pacing is superior to conventional right ventricular pacing in patients with atrioventricular block and left ventricular systolic dysfunction.5 Our patient had CLBBB with left ventricular dyssynchrony. His cardiac function was gradually getting worse for the last 3 years. Furthermore, cardiac function was expected to deteriorate after that. Moreover, the ventricular pacing was indispensable for atrioventricular block. Only right ventricular pacing might induce left ventricular dyssynchrony and deteriorate heart failure. Therefore, cardiac resynchronization therapy was suitable for the treatment of our patient, while CRT-D decreases the risk of heart failure events compared with an ICD alone in patients with a low ejection fraction and wide QRS complex.6 However, the benefit of an ICD has not been established in DMD patients. But we could not deny the fatal ventricular arrhythmia, because this disease was a progressive disease. In addition, we thought that the reoperation for upgrade of pacemaker would not be easy for him because of his severe skeletal anomaly such as scoliosis and the risk of general anesthesia. Therefore, CRT-D was thought to be preferable for our patient as a primary prevention strategy.
Cardiac involvement occurs in some types of myopathies. Myotonic dystrophy types 1 and 2, Emery–Dreifuss muscular dystrophy, and limb–girdle muscular dystrophy (LGMD) type 1B are generally associated with conduction disease.4 These types of muscular dystrophy need to be frequently implanted with a pacemaker. It is reported that an ICD is recommended in patients with Emery–Dreifuss muscular dystrophy and LGMD type 1B because of the risk of bradycardia and ventricular arrhythmia,7 while DMD, Becker muscular dystrophy, and LGMD types 2C–2F and 2I are generally associated with a dilated cardiomyopathy.4 Progression of cardiac dysfunction increases the frequency of sudden death. Fatal ventricular arrhythmia probably relates with sudden death. CRT-D has a good possibility of the prevention of progressive heart failure and sudden death. Although device therapy must be chosen according to individual conditions, CRT-D may become one of the therapies for arrhythmia and heart failure in patients with myopathy. Ehime University institutional review board has approved this study. Written informed consent was obtained from the patient.
Disclosure
The authors report no conflicts of interest in this work.
References
Romfh A, McNally EM. Cardiac assessment in Duchenne and Becker muscular dystrophies. Curr Heart Fail Rep. 2010;7(4):212–218. | |
Fayssoil A, Orlikowski D, Nardi O, Annane D. Complete atrioventricular block in Duchenne muscular dystrophy. Europace. 2008;10(11):1351–1352. | |
Fayssoil A, Orlikowski D, Nardi O, Annane D. Pacemaker implantation for sinus node dysfunction in a young patient with Duchenne muscular dystrophy. Congest Heart Fail. 2010;16(3):127–128. | |
Groh WJ. Arrhythmias in the muscular dystrophies. Heart Rhythm. 2012;9(11):1890–1895. | |
Curtis AB, Worley SJ, Adamson PB, et al; Biventricular versus Right Ventricular Pacing in Heart Failure Patients with Atrioventricular Block (BLOCK HF) Trial Investigators. Biventricular pacing for atrioventricular block and systolic dysfunction. N Engl J Med. 2013;368(17):1585–1593. | |
Moss AJ, Hall WJ, Cannom DS, et al; MADIT-CRT Trial Investigators. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361(14):1329–1338. | |
Meune C, Van Berlo JH, Anselme F, Bonne G, Pinto YM, Duboc D. Primary prevention of sudden death in patients with lamin A/C gene mutations. N Engl J Med. 2006;354(2):209–210. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

