Back to Journals » International Journal of Nanomedicine » Volume 14
Biotinylated Single-Domain Antibody-Based Blocking ELISA for Detection of Antibodies Against Swine Influenza Virus
Authors Du T, Zhu G, Wu X, Fang J, Zhou EM
Received 5 June 2019
Accepted for publication 14 November 2019
Published 29 November 2019 Volume 2019:14 Pages 9337—9349
DOI https://doi.org/10.2147/IJN.S218458
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Taofeng Du,1,2,* Guang Zhu,1,2,* Xiaoping Wu,1,2 Junyang Fang,1,2 En-Min Zhou1,2
1Department of Preventive Veterinary Medicine, College of Veterinary Medicine, Northwest A&F University, Yangling 712100, Shaanxi, People’s Republic of China; 2Scientific Observing and Experimental Station of Veterinary Pharmacology and Diagnostic Technology, Ministry of Agriculture, Yangling 712100, Shaanxi, People’s Republic of China
*These authors contributed equally to this work
Correspondence: En-Min Zhou
Department of Preventive Veterinary Medicine, College of Veterinary Medicine, Northwest A&F University, Yangling 712100, Shaanxi, People’s Republic of China
Email [email protected]
Background: Enzyme-linked immunosorbent assay (ELISA) is a common method for diagnosing swine influenza. However, the production of classical antibodies is both costly and time-consuming. As a promising alternative diagnostic tool, single-domain antibodies (sdAbs) offer the advantages of simpler and faster generation, good stability and solubility, and high affinity and specificity.
Methods: Phage display technology was used to isolate sdAbs against the SIV-NP protein from a camel VHH library. The sdAb5 was fused to the biotin acceptor peptide (BAP) and a His-Tag for its expression as monomeric and site-specific biotinylation in E.coli to develop an sdAb-based blocking ELISA (sdAb-ELISA). In the sdAb-ELISA, the anti-SIV antibodies from swine samples were used to block the binding between the biotinylated sdAb5 and SIV-NP protein coated on the ELISA plate. The specificity, sensitivity, and reproducibility of sdAb-ELISA were determined. In addition, consistency among sdAb-ELISA, commercial ELISA kit, and Western blot was evaluated.
Results: Six SIV-NP-specific sdAbs were isolated, among which sdAb5 was identified as a dominant sdAb with higher reactivity. The cut-off value of biotinylated sdAb5-based bELISA was determined to be 29.8%. Compared with the positive reference serum against five different types of swine viruses, the developed sdAb-ELISA showed 100% specificity. The detection limit of sdAb-ELISA was 1:160 in an anti-SIV positive reference serum, which is lower than that of the commercial ELISA kit (1:20). In 78 diluted anti-SIV positive serum (1:80), 21 and 42 samples were confirmed as positive by the commercial ELISA kit and sdAb-ELISA, respectively. The coefficients of variation of intra- and inter-assay were 1.79–4.57% and 5.54–9.98%, respectively. The sdAb-ELISA and commercial ELISA kit showed a consistency of 94.17% in clinical swine serum samples. Furthermore, the coincidence rate was 96.67% between the results detected by sdAb-ELISA and Western blot.
Conclusion: A specific, sensitive, and reproducible sdAb-ELISA was successfully developed, which offers a new, promising method to detect anti-SIV antibodies in swine serum.
Keywords: SIV, NP, sdAb, sdAb-ELISA
Introduction
Swine influenza virus (SIV), a detrimental pathogen that greatly challenges swine production, is composed of three influenza A virus (IAV) subtypes (H1N1, H1N2, and H3N2). Both swine and humans can be infected with IAV and exhibit similar clinical symptoms, which could manifest as an acute upper respiratory disease characterized by fever, coughing, sneezing, and anorexia.1 IAV is a single-stranded, negative-sense RNA virus that belongs to the family Orthomyxoviridae. The genome of IAV is composed of eight segments that encode for at least 10–14 proteins, whereby the highly conserved nucleoprotein (NP) is encoded by segment 5 in all IAV.2
 |
Figure 1 Schematic representation of isolating sdAbs from an immunized camel and development of sdAb-based blocking ELISA. Abbreviation: sdAb, single-domain antibody. |
The serological detection methods for influenza in swine include the hemagglutination inhibition (HI) test and enzyme-linked immunosorbent assay (ELISA).3 Although the HI test is the standard assay for detecting antibodies against SIV, it shows some limitations, such as its subjectivity, and may not be suitable for testing large numbers of samples.4 Similarly, a blocking ELISA has been developed using monoclonal antibodies against NP,5 but complex production process of monoclonal antibodies in large-scale industries is needed.
Compared to conventional antibodies composed of two heavy chains and two light chains, heavy chain only antibodies (HCAbs) found in Camelidae and sharks lack light chains and the first constant domain of the heavy chain (CH1).6 In the variable domains of heavy chain, antibodies (VHH), also known as single-domain antibodies (sdAbs) or nanobodies®, derived from a natural antibody known so far, are the smallest antigen-binding fragment. The molecular weight of the recombinant form of sdAb is about 12–15 kDa. Each sdAb contains four conserved framework regions (FR) and three complementarity-determining regions (CDR). Those molecules are attractive for biotechnological applications as they show several advantages over the conventional antibodies, such as small size, good stability and solubility, and high affinity and specificity.7–9 More importantly, sdAbs are suitable to genetic manipulation and could be obtained in different expression systems, adding to their great potential as diagnostic and therapeutic tools.10,11 Now, some sdAbs are used for clinical purposes, such as sdAb (ALX-0171) against F-protein of the respiratory syncytial virus (RSV) that targets the replication of RSV, and anti-HER2 sdAb (2Rs15d) for in vivo assessment of the HER2 status in breast cancer patients.12,13 In our previous studies, several sdAbs were successfully isolated against nonstructural protein 9 (Nsp9) of porcine reproductive and respiratory syndrome virus (PRRSV), PRRSV Nsp4, and NP of Newcastle disease virus (NDV). PRRSV Nsp9 and Nsp4-specific sdAbs demonstrated suppression of PRRSV replication in MARC-145 cells and primary porcine alveolar macrophages.14–16 Based on the NDV-NP-specific sdAb-horseradish peroxidase (HRP) fusion protein in our previous study, a competitive ELISA was successfully developed for detecting anti-NDV antibodies in chicken serum.17 In the present study, a VHH library was constructed from peripheral blood mononuclear cells (PBMCs) of an immunized Bactrian camel with SIV-NP recombinant protein, and SIV-NP specific sdAbs were isolated by phage display. Further, the lead candidate was in vivo biotinylated in E. coli and used to develop a blocking ELISA and then the specificity, sensitivity, and reproducibility were evaluated (Figure 1).
Materials and Methods
Expression and Purification of SIV-NP Recombinant Protein
The recombinant plasmid pET-32a-SIV-NP was kindly provided by Professor Chuanling Qiao, Harbin Veterinary Research Institute, Chinese Academy of Agricultural Sciences. The recombinant plasmid pET-32a-SIV-NP was transformed into E.coli Rosetta (DE3) cells (TransGen Biotech, Beijing, China). The expression of the SIV-NP recombinant protein was induced with 0.4 mM or 0.8 mM isopropyl-β-D-thiogalactopyranoside (IPTG) for 6 hrs at 25°C. The bacterial suspensions after induction were centrifuged at 8000 g for 15 mins at 4°C. Next, the pellet was harvested by centrifugation, resuspended in phosphate-buffered saline (PBS) and then lyzed by sonication. The bacterial soluble fraction was centrifuged at 10,000 g for 10 mins at 4°C, then the supernatant was loaded onto a high-affinity nickel–nitrilotriacetic acid (Ni-NTA) column (GE Healthcare, USA) to purify the SIV-NP recombinant protein. The expression and purification of SIV-NP were evaluated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The SDS-PAGE gel was stained with Coomassie Brilliant Blue.
Camel Immunization
A 2-year-old Bactrian camel was immunized with 1 mg SIV-NP recombinant protein in complete Freund Adjuvant (Sigma-Aldrich, St. Louis, MO, USA) via subcutaneous injection every 14 days. Each booster was performed under the same conditions except for the use of incomplete Freund adjuvant. Serum titration ELISA was performed over recombinant SIV-NP. Briefly, 400 ng/well of the protein was coated overnight (ON), blocked with 3% BSA in PBST (PBS containing 0.5% (v/v) Tween-20) and then serial dilutions of the serum were added into the wells and incubated at room temperature (RT) with agitation. After 1 hr, the plate was washed with PBST and the presence of SIV-NP-specific camel antibodies were detected by the use of a rabbit polyclonal serum specific for camel Immunoglobulins (prepared in our lab), followed by a goat polyclonal anti-rabbit IgG HRP conjugated (TransGen Biotech).
Library Construction
A week after the fifth immunization, 200 mL of blood was collected using tubes containing heparin. Peripheral blood mononuclear cells (PBMCs) were isolated by Leucosep tubes (Greiner Bio-One, Frickenhausen, Germany) according to the manufacturer’s instructions. Total RNA was extracted from PBMCs and used to synthesize the cDNA strand using TransScript One-Step gDNA Removal and cDNA Synthesis SuperMix (TransGen Biotech). The VHH genes were amplified by nested PCR from synthesized cDNA as described previously.18 Finally, the VHH genes were cloned into the phagemid vector pCANTAB 5E19 containing PstI and NotI restriction enzyme sites and digested with PstI and NotI restriction enzymes. The purified 5μg ligation mixture was electroporated into 800μL of E.coli TG1 cells (prepared in our lab). TG1 cells were resuspended in SOC and then plated on LB agar plates supplemented with 100 μg/mL ampicillin and 2% glucose at 37°C.18 The colonies were collected by a cell scraper and stored at −80°C in LB supplemented with 20% glycerol. A phage display VHH library was obtained after infecting bacterial cells with M13K07 helper phages.
Panning and Identification of Specific sdAbs Against SIV-NP Protein
Three rounds of panning were performed as described previously.18 Briefly, 100μL of the recombinant SIV-NP protein (10μg) was coated on 96-well plates and incubated overnight at 4°C. After washing with PBST, 200μL of blocking buffer (PBST containing 5% (w/v) skimmed milk) was added to the plates and incubated for 1 hr at room temperature (RT). The plates were washed with PBST and incubated with the phage display VHH library for 2 hrs at RT. After washing with PBST, the bound phages were eluted with 100μL of freshly prepared trimethylamine (100mM). Then, the eluted phages were neutralized with 100μL of 1M Tris–HCl buffer (pH 7.4). The neutralized phage particles were titrated by infection of TG1 cells and plated on LB agar plates supplemented with 100 μg/mL ampicillin and 2% glucose for 12 hrs at 37°C. After final rounds of panning, enrichment of the SIV-NP-specific phage particles was evaluated based on the ratios of positive phage outputs to negative phage output with plaque-forming units (pfu)/well. Then, 96 colonies were randomly picked, grown in 96-well plates in 100μL LB medium supplemented with 100 μg/mL ampicillin and 2% glucose for 6 hrs at 37°C. And, 10μL of starter culture into 24-well plates in 1 mL TB medium was inoculated. Until the OD value for 600nm reaches 0.6–0.9, the expression of soluble VHH with E-Tag was induced by adding 1 mM IPTG. Next, the cell pellet was harvested and subjected to three freeze–thaw cycles and resuspended in PBS. After centrifugation, the recombinant VHH-E-Tag protein in supernatant was obtained. The SIV-NP- specific VHHs were identified by indirect ELISA using anti-E-Tag antibody (GenScript, Piscataway, NJ, USA). Positive clones of VHH genes8 were sequenced and classified according to the CDR3 sequence. Additionally, to choose the best sdAb for establishing the following sdAb-ELISA, the specificity and reactivity of classified sdAb were detected by the indirect ELISA.
Expression and Purification of sdAb Containing Biotin Acceptor Peptide
The gene sequence of the BAP was synthesized and cloned into the multiple cloning sites (HindIII and XhoI) of the commercial prokaryotic expression vector pET-21b (Novagen, Madison, WI, USA). The modified vector was named pET-21b-BAP. The identified VHH genes were amplified by PCR and cloned into pET-21b-BAP with the XhoI and BamHI restriction enzyme sites. The recombinant plasmids were identified by sequencing. The expression of VHH-BAP fusion protein was induced with 1mM IPTG at 37°C for 4 hrs. The sdAb was purified by Ni-NTA affinity chromatography and biotinylated with a Biotin-Protein Ligase/BirA Enzyme kit (GeneCopoeia, Rockville, MD, USA) following the manufacturer’s instructions. The in vivo biotinylation of the sdAb is mediated by the biotin-protein ligase (BirA enzyme) which specifically conjugates the D-biotin to the Lys residue encoded in the BAP amino acid sequence LNDIFEAQKIEWH).20
Establishment of the Blocking ELISA
Determination of the optimal coating antigen concentration and dilution of biotinylated sdAb (10 μg/mL-1 ng/mL) were carried out by checkerboard titration in indirect ELISA. Serial dilutions of SIV-NP protein (0.5, 1, 2, and 4 μg/mL) were used to coat 96-well microtiter plates (Nunc A/S, Roskilde, Denmark) OT at 4°C. The plates were then washed three times with PBST and blocked with 200μL/well of blocking buffer (3% BSA in PBST) and incubated for 1 hr at RT. Then, it was After washed three, the biotinylated sdAb at various concentrations (10 μg/mL, 1μg/mL, 100ng/mL, 10 ng/mL, 1 ng/mL) was added to the plates at 100μL/well in blocking buffer and incubated for 1 hr at RT. After three washes with PBST, 100μL/well HRP-conjugated streptavidin (Solarbio, Beijing, China) diluted in blocking buffer at 200ng/mL was added to plates and incubated for 1 hr at RT. Following three washes, 100μL/well of 3,3ʹ,5,5ʹ-tetramethylbenzidine (TMB) was added to the plates for a colorimetric reaction for 15 mins at RT in the dark. Finally, the colorimetric reaction was stopped by adding 50 μL/well of 3 M sulfuric acid, and the OD value was read at 450 nm using the ELISA plate reader (Bio-Rad, USA). The optimal concentration range of biotinylated sdAb5 for each concentration SIV-NP was determined as follows: 100–167 ng/mL for 0.5 μg/mL SIV-NP, 25–50 ng/mL for 1 μg/mL SIV-NP and 12.5–25 ng/mL for 2 μg/mL or 4μg/mL SIV-NP. The optimal concentration of biotinylated sdAb5 (in three different dilutions) for each concentration SIV-NP was then determined using a checkboard titration. The confirmed optimal concentration of biotinylated sdAb5 for each concentration SIV-NP was as follows: 125 ng/mL for 0.5 μg/mL SIV-NP, 33 ng/mL for 1 μg/mL SIV-NP and 16ng/mL for 2 μg/mL or 4μg/mL SIV-NP. Once found, the optimal conditions for the sdAb-ELISA, the assay was performed as follows: 96-well microtiter plates (Nunc A/S, Roskilde, Denmark) were coated with 1 μg/mL SIV-NP protein (100 μL/well) diluted in PBS overnight at 4°C. Next day, the plates were washed three times with PBST and blocked for 1 hr at RT with 200 μL/well of blocking buffer (2.5% BSA in PBST). Then, to detect the presence of antibodies against SIV-NP, the reference swine positive serum was diluted in blocking buffer and 100 μL/well was added to the plates and incubated for 1 hr at RT. Following three washes with PBST, the biotinylated sdAb5 (100 μL/well at 33 ng/mL) was added into each well and incubated for 1 hr at RT and then detected using HRP- conjugated streptavidin (diluted in blocking buffer at 200ng/mL). Finally, after three washes, the HRP activity was developed for 15 mins by the addition of 100 μL/well of TMB. To conclude, the colorimetric reaction was stopped by adding 50 μL/well of 3 M sulfuric acid, and the OD450 nm was read using the ELISA plate reader (Bio-Rad).
The 110 negative swine serum samples were identified by the commercial ELISA kit (IDEXX Influenza A Ab Test, IDEXX Laboratories, Inc., Westbrook, ME, USA). As a blocking ELISA, the commercial kit is designed to detect serum antibodies to any influenza A subtype, which required serum sample is diluted 1:10. The test result is evaluated by using OD650nm of test serum samples/OD650nm of negative control (S/N). The test serum samples with S/N<0.6 and S/N 0.6 ≥ are designated positive and negative, respectively. The cut-off point of sdAb-ELISA was determined by the mean percentage inhibition (PI) value of 110 negative swine serum samples plus three standard deviations (99% confidence interval). The PI of test serum samples was calculated using the following formula: PI (%) = (1 − (OD450nm of test serum samples/OD450nm of negative serum samples)) ×100%.21–23
Validation of the Blocking ELISA
The specificity and cross-reactivity of the sdAb-ELISA were determined using the homemade swine antisera against five other swine viruses, including porcine circovirus type 2 (PCV2), porcine epidemic diarrhea virus (PEDV), porcine reproductive and respiratory syndrome virus (PRRSV), pseudorabies virus (PRV), and classical swine fever virus (CSFV).24 The sensitivity of the sdAb-ELISA was validated by the analysis of serial dilutions of an anti-SIV positive reference serum (1:10–1:640) and 78 positive serum samples which were confirmed using the commercial ELISA kit (IDEXX Influenza A Ab Test, IDEXX Laboratories, Inc., Westbrook, ME, USA) at a dilution of 1:80.24 The intra-assay and inter-assay variations were evaluated by the coefficient of variation (CV) using six positive serum samples. Each sample was replicated three times in each plate to determine the intra-assay CV. The inter-assay CV (plate-to-plate) was calculated by testing each sample using three different 96-well microtiter plates at different time points.
Evaluation of the Testing Consistency Among sdAb-ELISA, Commercial ELISA Kit, and Western Blot
To compare consistency, sdAb-ELISA, the commercial ELISA kit (IDEXX Influenza A Ab Test, IDEXX Laboratories, Inc., Westbrook, ME, USA) and Western blot were used to detect 120 clinical swine serum samples at dilution of 1:10. The coincidence rates of the test results were calculated using Microsoft Excel’s CORREL function. The Western blot analysis was performed by loading SIV-NP recombinant protein (5μg/lane) in 12% SDS-PAGE and electrotransferred onto a polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with 2.5% skin milk in PBST and then probed with swine serum (1:100). HRP-conjugated rabbit anti-pig IgG was used as the secondary antibody. Immunostained proteins were visualized using ECL Reagent (Pierce, Rockford, IL, CA, USA).25,26
Statistical Analysis
Kappa index values were calculated to evaluate the coincidence between sdAb-ELISA and the commercial ELISA kit and between sdAb-ELISA and Western blot using SPSS software (Version 20, http://www.spss.com.cn).
Results
Expression and Purification Analysis of SIV-NP
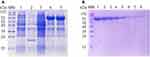
To prepare the antigen (SIV-NP) for immunizing camels, an E.coli expression system was used. As shown in Figure 2A, the soluble SIV-NP recombinant protein was expressed after induction with IPTG at 25°C for 16 hrs, and a higher-yield protein production was obtained when 0.8 mM IPTG was used as inducer. Next, the SIV-NP recombinant protein was purified through a Ni-NTA column. After buffer exchange to PBS, 60mg of SIV-NP were obtained from 1L of culture. SDS-PAGE analysis was performed and no major impurities from E.coli were seen (Figure 2B), allowing us to generate a good quality antigen for the immunization of camels.
Construction and Verification of Phage Display VHH Library
A healthy Bactrian camel was immunized with the SIV-NP protein five times. The titer of antibody against SIV-NP protein reached 1:256,000 after the last immunization as the OD450nm value from the immunized sera at highest dilution was more than three times the OD450nm value than that from the pre-immune sera (Figure 3), which indicates the camel developed strong antibody responses against SIV-NP antigen. A phage display VHH library was generated from PMBC cDNA, containing approximately 5.88 × 109 individual transformants by sequence analysis (data not shown). The results of PCR analysis from 49 randomly picked colonies showed that 46 colonies showed the presence of the VHH gene.
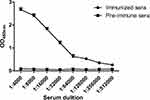 |
Figure 3 Detection of serum titration of the immunized camel against recombinant SIV-NP protein after last immunization. Abbreviations: SIV, swine influenza virus; NP, nucleoprotein. |
Selection and Characterization of SIV-NP-Specific sdAbs
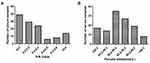
To isolate specific sdAbs against SIV-NP protein from the phage display VHH library, biopanning was performed by ELISA. The result of enrichment SIV-NP-specific phages showed that the maximal enrichment was seen after the third round, compared with values against the control panning. After the screening, 93/96 positive colonies were identified for specific binding to SIV-NP protein using ELISA. After sequencing, based on amino acid sequences of the CDR3, 93 positive colonies represented six sdAb families (sdAb1-sdAb6) (Figure 4), that occurred 2, 3, 5, 7, 75, and 1 times, respectively. The sdAb of one representative member of each family was extracted from IPTG-induced E. coli TG1 colonies. Then, the specificity of each sdAb was evaluated by indirect ELISA. The result showed that each sdAb could specifically react with the SIV-NP protein but not with the irrelevant N protein of PEDV (PEDV-N) (Figure 5A). The group of six sdAbs was compared in terms of the reactivity against SIV-NP. As shown in Figure 5B, sdAb4 and sdAb5 exhibited the highest reactivity. Based on the higher reactivity and higher frequency of the sequence of sdAb5, this sdAb was chosen for the establishment of the blocking ELISA.
Generation of Biotinylated sdAb
The sdAb5-BAP fusion protein was expressed in a prokaryotic expression system and purified by a Ni-NTA column. A 15-kDa specific band was appeared on 12% SDS-PAGE using Coomassie blue staining (Figure 6A). After purification, the yield of purified sdAb5-BAP fusion protein could be up to 80mg/L using 1L LB media. SDS-PAGE analysis showed that no major impurities from E.coli were seen (Figure 6B). After ex vivo enzymatic biotinylation, the sdAb was used to develop sdAb-ELISA.
Establishment of Blocking ELISA Based on sdAb5
The optimum concentration of biotinylated sdAb5 was identified as 33 ng/mL by indirect ELISA (Figure 7A). The results of the checkerboard titration assay showed that 1 μg/mL of SIV-NP protein was the optimal concentration for use as the coated antigen, while the optimal serum dilution was 1:10 (Figure 7B). The 110 negative swine serum samples confirmed with the commercial ELISA kit generated the cut-off value of 29.8% (23.5% + 3SD) for the sdAb-ELISA, which exhibits an average PI (x) of 23.5% and SD of 2.1%. Therefore, test serum samples with PI ≥ 29.8% and PI < 29.8% are designated positive and negative, respectively, using the sdAb-ELISA.
Specificity, Sensitivity, and Reproducibility of Blocking ELISA
To evaluate the specificity of sdAb-ELISA, antisera against PCV2, PEDV, PRRSV, PRV, and CSFV were detected by sdAb-ELISA. The results in Figure 8A show that the PI values of all antisera against viruses were lower than the cut-off value of sdAb-ELISA, except anti-SIV positive reference serum, which suggests that sdAb-ELISA is specific to antiserum against SIV.
The sensitivity of the sdAb-ELISA was determined using the anti-SIV positive reference serum with different dilutions and compared with a commercial ELISA kit. The limit of detection of sdAb-ELISA reached 1:160, which is lower than that of commercial ELISA kit (1:20) (Figure 8B). To further evaluate the sensitivity of the sdAb-ELISA, 78 commercial ELISA-anti-SIV positive serum samples (diluted 1:10) (data not shown) were diluted 1:80 to reduce the antibody level and tested again using the commercial ELISA kit and the sdAb-ELISA. The results showed that 21/78 verses 42/78 samples were SIV antibody-positive samples by the commercial ELISA and the sdAb-ELISA, respectively (Figure 8C). This finding indicates the higher sensitivity of sdAb-ELISA over the commercial ELISA kit.
The study of intra-assay and inter-assay variabilities was performed to test the reproducibility of sdAb-ELISA. The anti-SIV positive serum samples from six pigs were tested three times within a plate using sdAb-ELISA, and the intra-assay CV of PI was observed to be in the range of 1.79–4.57% with the median value at a level of 3.5%. After repeating the sdAb-ELISA experiments in three plates for six serum samples on 3 different days, the inter-assay CV of PI was found to be in the range of 5.54–9.98% with the median value at a level of 8.9% (Table 1). These results show that the sdAb-ELISA offers good reproducibility.
 |
Table 1 Estimation of Intra- and Inter-assay CV |
Comparison of the sdAb-ELISA with Commercial ELISA Kit and Western Blot
A total of 120 swine serum samples from a pig farm in Shaanxi Province were examined using both sdAb-ELISA and a commercial ELISA kit. The test results show that the positive rates of the sdAb-ELISA and commercial ELISA kit were 85.83% and 85%, respectively. When the performance of both approaches was compared, the sdAb-ELISA allowed to identify 103 positive and 17 negative serum samples, while in the commercial ELISA only 99 were positive and 4 showed to be negative. When the 17 sdAb-ELISA-negative samples were subjected to the commercial ELISA, 3 of them were positive and 14 negative (Figure 9 and Table 2). The results of both sdAb-ELISA and Western blot coincided in 116 of the 120 serum samples with an agreement rate of 96.67% (Table 2). Moreover, Kappa index values show that a high degree of coincidence exists between sdAb-ELISA and the commercial ELISA kit (Kappa = 0.76) and between sdAb-ELISA and Western blot (Kappa = 0.82). No significant differences were observed between the two groups (Kappa > 0.4). In addition, based on the results of Western blot, sdAb-ELISA has two false positive and two false negative, the commercial kit has one false positive and two false negative (Figure 10).
 |
Table 2 Comparative Analysis of bELISA with Commercial ELISA Kit and with Western Blot by Testing Swine Serum Samples |
Discussion
The diagnosis of viral infectious diseases can be determined by the presence of antigens and specific antibodies. As one of the most used methodologies, ELISA is a specific, sensitive and high-throughput detection assay that does not require an expensive experimental device. Antibodies are important for developing ELISA.27,28 For sdAb-ELISA, free antibody in the test sample could be used to block the antigen-specific antibody with plate-coated antigen. In the traditional ELISA method, polyclonal or monoclonal antibodies are commonly used, but the quality may be inconsistent among the different batch of polyclonal antibodies, and the industrialized large-scale production process of monoclonal antibodies is complicated.29 In contrast with conventional antibodies, sdAb is easily produced as recombinant proteins in bacteria and yeast expression systems, which facilitate large-scale production of sdAb and high batch-to-batch consistency.12,13 The conserved SIV-NP is an RNA-binding protein that plays a significant role in the virus life cycle and, thus, serves as a specific target protein for diagnosis of SIV infection.30 In the present study, six specific sdAbs against SIV-NP protein were isolated through phage display.
In ELISA, the biotinylation of the antigen or antibody is a pre-requisite for applying streptavidin-biotin-based sensors with detection signal amplification.31 Biotinylated molecules could be obtained by using chemical or enzymatic biotinylation. Chemical biotinylation is the process of nonspecific covalently attaching to biotin to amines, carboxylates, sulfhydryls or carbohydrates in the target molecule. The most common of enzymatic biotinylation is to genetically link BAP to the target molecule. Then, the target molecule was biotinylated by using biotin ligase (BirA) to transfer biotin to specific lysine residue BAP sequence.32,33 Enzymatic biotinylation is also performed by co-expression of biotin ligase and BAP-tagged molecule in bacterial and mammalian cells.34,35 Compared with chemical biotinylation, enzymatic biotinylation provides a technique for high selective modification of target molecules. Additionally, sdAb serves as the probe molecule in ELISA that possibly offers a higher probe density due to their small size.36 In this study, sdAb5 was biotinylated by enzymatic biotinylation, where a biotin molecule is conjugated to a specific lysine residue by the action of a biotin ligase (BirA). Moreover, the sensitivity of the developed sdAb-ELISA was improved based on this streptavidin–biotin system.
Swine influenza is the primary cause of respiratory disease among the swine industry, which has faced serious economic losses globally.37 Currently, several methods for detecting SIV include immunohistochemistry, immunofluorescence, virus isolation, ELISA, and PCR. Compared to direct virus detection, serological tests are more suitable for diagnosing acute and previous infection, surveilling disease, and evaluating immune effects of vaccines.38 For detection of antibodies against SIV, the most common serologic assay is the HI test. However, the HI test faces some limitations prior to operation, such as elimination of native agglutinins and non-specific inhibitors for viral hemagglutination from the serum samples before performing the test and preparation of virus and turkey or chicken erythrocytes.3,4 In the course of developing traditional ELISA, the production of anti-SIV antibodies is cumbersome, and antibodies require strict storage conditions. In the present study, a novel sdAb-ELISA was developed for detecting anti-SIV antibodies on the basis of biotinylated SIV-NP-specific sdAbs. Results show that the detection limit of the developed sdAb-ELISA is lower than that of the commercial ELISA kit, suggesting that this method has high sensitivity. Not only that, the developed sdAb-ELISA exhibited high consistency with both the commercial ELISA kit and Western blot, further demonstrating its great potential in the detection of anti-SIV antibodies. But, based on the results of Western blot, false-negative rates of sdAb-ELISA and the commercial kit were consistent. Compared with commercial kit, sdAb-ELISA had a higher false-positive rate, probably because sdAb-ELISA had a higher sensitivity. Potentially, the sdAb-ELISA could be used for the detection of IAV antibodies in the serum of different species, which is suggested for further research.
Conclusion
Considering the increasing use of sdAbs in the development of diagnostic tools, six specific sdAbs against SIV-NP protein were isolated from an immune phage display library in this study. The dominant sdAb was determined to be sdAb5, which exhibited a higher reactivity. Based on biotinylated sdAb5, a novel sdAb-ELISA with good specificity, sensitivity, and reproducibility was developed for detecting anti-SIV antibodies in swine serum. In comparison, the results of sdAb-ELISA are in close agreement with the measurements obtained from a commercial ELISA kit and Western blot. Overall, the developed sdAb-ELISA offers an alternative way to assess the levels of anti-SIV antibodies in swine serum, which is cost-effective, highly specific and sensitive, and has wide potential application in clinical examination.
Ethics Statement
All animal experiments were performed according to Guidance for Experimental Animal Welfare and Ethical Treatment by the Ministry of Science and Technology of China (www.most.gov.cn/fggw/zfwj/zfwj2006/zf06yw/zf06qt/200612/t20061226_39235.htm). Experimental procedures and animal use and care protocols were carried out in accordance with the guidelines of the Northwest A&F University Institutional Committee for the Care and Use of Laboratory Animals and were approved by the Committee on Ethical Use of Animals of Northwest A&F University.
Acknowledgments
This work was supported by the National Key R&D Program of China (2016YFD0500706), National Natural Science Foundation of China (31772762) and China Postdoctoral Science Foundation Grant (2018M633592). The authors would like to thank Professor Chuanling Qiao for providing the recombinant plasmid pET-32a-SIV-NP.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Vincent AL, Lager KM, Anderson TK. A brief introduction to influenza A virus in swine. Methods Mol Biol. 2014;1161:243–258.
2. Vincent AL, Perez DR, Rajao D, et al. Influenza A virus vaccines for swine. Vet Microbiol. 2017;206:35–44. doi:10.1016/j.vetmic.2016.11.026
3. Janke BH. Influenza A virus infections in swine: pathogenesis and diagnosis. Vet Pathol. 2014;51(2):410–426. doi:10.1177/0300985813513043
4. Detmer S, Gramer M, Goyal S, Torremorell M, Torrison J. Diagnostics and surveillance for Swine influenza. Curr Top Microbiol Immunol. 2013;370:85–112. doi:10.1007/82_2012_220
5. Ciacci-Zanella JR, Vincent AL, Prickett JR, Zimmerman SM, Zimmerman JJ. Detection of anti-influenza A nucleoprotein antibodies in pigs using a commercial influenza epitope-blocking enzyme-linked immunosorbent assay developed for avian species. J Vet Diagn Invest. 2010;22(1):3–9. doi:10.1177/104063871002200102
6. Hamers-Casterman C, Atarhouch T, Muyldermans S, et al. Naturally occurring antibodies devoid of light chains. Nature. 1993;363(6428):446–448. doi:10.1038/363446a0
7. Muyldermans S, Atarhouch T, Saldanha J, Barbosa JA, Hamers R. Sequence and structure of VH domain from naturally occurring camel heavy chain immunoglobulins lacking light chains. Protein Eng. 1994;7(9):1129–1135. doi:10.1093/protein/7.9.1129
8. Arbabi Ghahroudi M, Desmyter A, Wyns L, Hamers R, Muyldermans S. Selection and identification of single domain antibody fragments from camel heavy-chain antibodies. FEBS Lett. 1997;414(3):521–526. doi:10.1016/S0014-5793(97)01062-4
9. Muyldermans S. Nanobodies: natural single-domain antibodies. Annu Rev Biochem. 2013;82:775–797. doi:10.1146/annurev-biochem-063011-092449
10. Liu Y, Huang H. Expression of single-domain antibody in different systems. Appl Microbiol Biotechnol. 2018;102(2):539–551. doi:10.1007/s00253-017-8644-3
11. Salvador JP, Vilaplana L, Marco MP. Nanobody: outstanding features for diagnostic and therapeutic applications. Anal Bioanal Chem. 2019. doi:10.1007/s00216-019-01633-4
12. Steeland S, Vandenbroucke RE, Libert C. Nanobodies as therapeutics: big opportunities for small antibodies. Drug Discov Today. 2016;21(7):1076–1113. doi:10.1016/j.drudis.2016.04.003
13. Mir MA, Mehraj U, Sheikh BA, Hamdani SS. Nanobodies: the “magic bullets” in therapeutics, drug delivery and diagnostics. Hum Antibodies. 2019. doi:10.3233/HAB-190390
14. Liu H, Wang Y, Duan H, et al. An intracellularly expressed Nsp9-specific nanobody in MARC-145 cells inhibits porcine reproductive and respiratory syndrome virus replication. Vet Microbiol. 2015;181(3–4):252–260. doi:10.1016/j.vetmic.2015.10.021
15. Wang L, Zhang L, Huang B, et al. A nanobody targeting viral nonstructural protein 9 inhibits porcine reproductive and respiratory syndrome virus replication. J Virol. 2019;93:4.
16. Liu H, Liang C, Duan H, et al. Intracellularly expressed nanobodies against non-structural protein 4 of porcine reproductive and respiratory syndrome virus inhibit virus replication. Biotechnol Lett. 2016;38(7):1081–1088. doi:10.1007/s10529-016-2086-3
17. Sheng Y, Wang K, Lu Q, et al. Nanobody-horseradish peroxidase fusion protein as an ultrasensitive probe to detect antibodies against Newcastle disease virus in the immunoassay. J Nanobiotechnology. 2019;17(1):35. doi:10.1186/s12951-019-0468-0
18. Vincke C, Gutierrez C, Wernery U, Devoogdt N, Hassanzadeh-Ghassabeh G, Muyldermans S. Generation of single domain antibody fragments derived from camelids and generation of manifold constructs. Methods Mol Biol. 2012;907:145–176.
19. Kramer K, Fiedler M, Skerra A, Hock B. A generic strategy for subcloning antibody variable regions from the scFv phage display vector pCANTAB 5 E into pASK85 permits the economical production of F(ab) fragments and leads to improved recombinant immunoglobulin stability. Biosens Bioelectron. 2002;17(4):305–313. doi:10.1016/S0956-5663(01)00292-5
20. Schatz PJ. Use of peptide libraries to map the substrate specificity of a peptide-modifying enzyme: a 13 residue consensus peptide specifies biotinylation in Escherichia coli. Biotechnology (N Y). 1993;11(10):1138–1143. doi:10.1038/nbt1093-1138
21. Fevereiro M, Barros S, Fagulha T. Development of a monoclonal antibody blocking-ELISA for detection of antibodies against Maedi-Visna virus. J Virol Methods. 1999;81(1–2):101–108. doi:10.1016/S0166-0934(99)00061-0
22. Todd D, Mawhinney KA, Graham DA, Scott AN. Development of a blocking enzyme-linked immunosorbent assay for the serological diagnosis of chicken anaemia virus. J Virol Methods. 1999;82(2):177–184. doi:10.1016/S0166-0934(99)00099-3
23. Zhou D, Pei C, Yang K, et al. Development and application of a monoclonal-antibody-based blocking ELISA for detection of Japanese encephalitis virus NS1 antibodies in swine. Arch Virol. 2019;164(6):1535–1542. doi:10.1007/s00705-019-04218-9
24. Deng J, Liu Y, Jia R, et al. Development of an immunochromatographic strip for detection of antibodies against duck Tembusu virus. J Virol Methods. 2017;249:137–142. doi:10.1016/j.jviromet.2017.08.022
25. Chen Y, Zhao Q, Liu B, et al. A novel blocking ELISA for detection of antibodies against hepatitis E virus in domestic pigs. PLoS One. 2016;11(3):e0152639. doi:10.1371/journal.pone.0152639
26. Liu B, Zhao Q, Sun Y, et al. Development of a blocking ELISA for detection of antibodies against avian hepatitis E virus. J Virol Methods. 2014;204:1–5. doi:10.1016/j.jviromet.2014.03.023
27. Boonham N, Kreuze J, Winter S, et al. Methods in virus diagnostics: from ELISA to next generation sequencing. Virus Res. 2014;186:20–31. doi:10.1016/j.virusres.2013.12.007
28. Hornbeck PV. Enzyme-linked immunosorbent assays. Curr Protoc Immunol. 2015;110:21–23. doi:10.1002/0471142735.2015.110.issue-1
29. Rodrigues ME, Costa AR, Henriques M, Azeredo J, Oliveira R. Technological progresses in monoclonal antibody production systems. Biotechnol Prog. 2010;26(2):332–351. doi:10.1002/btpr.348
30. Noton SL, Simpson-Holley M, Medcalf E, et al. Studies of an influenza A virus temperature-sensitive mutant identify a late role for NP in the formation of infectious virions. J Virol. 2009;83(2):562–571. doi:10.1128/JVI.01424-08
31. Konstantinou GN. Enzyme-Linked Immunosorbent Assay (ELISA). Methods Mol Biol. 2017;1592:79–94.
32. Beckett D, Kovaleva E, Schatz PJ. A minimal peptide substrate in biotin holoenzyme synthetase-catalyzed biotinylation. Protein Sci. 1999;8(4):921–929. doi:10.1110/ps.8.4.921
33. Barat B, Wu AM. Metabolic biotinylation of recombinant antibody by biotin ligase retained in the endoplasmic reticulum. Biomol Eng. 2007;24(3):283–291. doi:10.1016/j.bioeng.2007.02.003
34. Cull MG, Schatz PJ. Biotinylation of proteins in vivo and in vitro using small peptide tags. Method Enzymol. 2000;326:430–440.
35. de Boer E, Rodriguez P, Bonte E, et al. Efficient biotinylation and single-step purification of tagged transcription factors in mammalian cells and transgenic mice. P Natl Acad Sci USA. 2003;100(13):7480–7485. doi:10.1073/pnas.1332608100
36. Huang L, Muyldermans S, Saerens D. Nanobodies (R): proficient tools in diagnostics. Expert Rev Mol Diagn. 2010;10(6):777–785. doi:10.1586/erm.10.62
37. Rajao DS, Anderson TK, Gauger PC, Vincent AL. Pathogenesis and vaccination of influenza a virus in swine. Curr Top Microbiol. 2014;385:307–326.
38. Ohst C, Saschenbrecker S, Stiba K, et al. Reliable serological testing for the diagnosis of emerging infectious diseases. Adv Exp Med Biol. 2018;1062:19–43.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.