Back to Journals » Neuropsychiatric Disease and Treatment » Volume 14
Biomarkers in traumatic brain injury (TBI): a review
Authors Dadas A, Washington J, Diaz-Arrastia R, Janigro D
Received 3 March 2018
Accepted for publication 20 June 2018
Published 8 November 2018 Volume 2018:14 Pages 2989—3000
DOI https://doi.org/10.2147/NDT.S125620
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Aaron Dadas,1 Jolewis Washington,1 Ramon Diaz-Arrastia,2 Damir Janigro1,3
1FloTBI Inc., Cleveland, OH, USA; 2Department of Neurology, University of Pennsylvania, Philadelphia, PA, USA; 3Department of Physiology, Case Western Reserve University, Cleveland, OH, USA
Abstract: Biomarkers can be broadly defined as qualitative or quantitative measurements that convey information on the physiopathological state of a subject at a certain time point or disease state. Biomarkers can indicate health, pathology, or response to treatment, including unwanted side effects. When used as outcomes in clinical trials, biomarkers act as surrogates or substitutes for clinically meaningful endpoints. Biomarkers of disease can be diagnostic (the identification of the nature and cause of a condition) or prognostic (predicting the likelihood of a person’s survival or outcome of a disease). In addition, genetic biomarkers can be used to quantify the risk of developing a certain disease. In the specific case of traumatic brain injury, surrogate blood biomarkers of imaging can improve the standard of care and reduce the costs of diagnosis. In addition, a prognostic role for biomarkers has been suggested in the case of post-traumatic epilepsy. Given the extensive literature on clinical biomarkers, we will focus herein on biomarkers which are present in peripheral body fluids such as saliva and blood. In particular, blood biomarkers, such as glial fibrillary acidic protein and salivary/blood S100B, will be discussed together with the use of nucleic acids (eg, DNA) collected from peripheral cells.
Keywords: peripheral markers, blood–brain barrier, post-traumatic epilepsy, fluid biomarkers, mild traumatic brain injury, neuroimaging
Introduction
According to the Food and Drug Administration (FDA), biomarkers can be broadly classified as a “characteristic that serves as an objective indicator of normal biological processes, pathogenic processes, or response to an exposure or intervention, including therapeutic interventions.”1 Biomarkers can be measured using molecular, histologic, radiographic, or physiologic tools. They can indicate health, pathology, or response to treatment, including unwanted side effects. They can act as surrogate endpoints in clinical trials or can, rarely, substitute for clinically meaningful endpoints. Biomarkers of disease can be diagnostic (identifying the nature and cause of a condition), prognostic (informing the likelihood of a person’s survival or outcome of a disease), or predictive (identifying individuals who are likely to experience a favorable or unfavorable effect from a specific treatment or exposure).2 In addition, genetic biomarkers are used to quantify the risk of developing a certain disease. For traumatic brain injury (TBI), brain-derived protein biomarkers are commonly used as discussed below.
TBIs are heterogeneous and can occur in a variety of ways. TBI can be classified by the mechanism of injury (eg, motor vehicle accidents, falls, and assaults), through clinical severity as graded by the Glasgow Coma Scale (GCS), or by the characterization of structural damage (for review, see the study by Maas et al2 and Buonora et al3). The heterogeneity of the disease makes it difficult to accurately assess the level of trauma and predict the clinical outcome for individual patients.3,4 Furthermore, the majority of initial features of injury do not translate directly into long-term consequences. These considerations have made establishing a universal and comprehensive TBI assessment protocol a challenging hurdle in the field of health care. Generally, injuries are classified as mild, moderate, or severe depending on the GCS score (which utilizes motor, eye, and verbal responses to evaluate the patient’s level of consciousness), duration of unconsciousness, and duration of post-traumatic amnesia.2,5–7 TBI is characterized by an evolving pathology: time-dependent changes almost invariably follow the initial insult.2 For this reason, biomarker-based methods for diagnosis and prognosis can only be interpreted in the context of an evolving pathology. As discussed below, the understanding of this moving target is confounded by the fact that many biomarkers have complex kinetics relating to release from brain cells, transfer across the blood–brain barrier (BBB), and clearance from the peripheral blood. These kinetic behaviors are at best indirectly related to the pathology of TBI.
Several clinical-grade imaging modalities currently exist ranging from traditional computed tomography (CT) and MRI scans to more novel imaging techniques, such as susceptibility-weighted imaging, diffusion-weighted imaging, and diffusion tensor imaging. All have been utilized in the context of TBI identification and clinical management. In addition, imaging modalities, such as functional MRI, positron emission tomography, single-photon emission computed tomography, and magnetic resonance spectroscopy, provide insight into metabolic abnormalities, which may have resulted from TBI, providing yet another level of information that could aid in diagnosis and treatment.5,8–11 Serum and cerebrospinal fluid (CSF) markers have been developed as an alternative or synergistic complement to radiological investigations of TBI and its sequelae.5,12–14 These imaging and analytic modalities, when combined, will provide a future pathway toward improved clinical outcomes and enhanced power of clinical management for TBI patients.
Uncomplicated mild TBI (mTBI) is diagnosed if the patient has no post-TBI injuries detected by a noncontrast CT head scan.15,16 A diagnosis of complicated mTBI is made if the patient has evidence of an intracranial event, such as a subdural hematoma (SDH) or epidural hematoma (EDH), subarachnoid hemorrhage, intraparenchymal hemorrhage, or brain edema.17,18 The possibility of intracranial hemorrhages is an important consideration following mTBI and is the primary motivation for performing initial CT imaging studies. In these patients, symptoms of bleeding may not be present at the time of emergency room visit and are manifest only after appropriate imaging. SDH and EDH may change the course of clinical management, as these events mandate admission to an intensive care unit and often require neurosurgical intervention.19–21 SDH is not uncommon in mTBI sequelae and is often associated with the rupture of cerebral veins, causing progressive accumulation of blood in the subdural space. Subacute and chronic SDHs are relatively common in patients with mTBI, especially in the geriatric population and when anticoagulation is present for pre-existing conditions. It is important to accurately diagnose SDH, as an expanding mass lesion may necessitate surgery. Moreover, SDH is often associated with breakdown of the BBB, disproportionate tissue inflammation, and cerebral edema, which may cause progressive neurological deterioration. These are some of the most common rationales to use surrogate biomarkers because their appearance in blood and other fluids is facilitated or caused by BBB disruption. EDHs are caused by acceleration–deceleration injuries and can be commonly ascribed to the rupture of a meningeal artery. The timely diagnosis of EDH is also of critical importance, as expanding hematomas may become life threatening, and progressive neurological deterioration, often after a lucid interval, require neurosurgical intervention. Again, this underscores the need for surrogate biomarkers when CT scans are unavailable or during transport from a remote location. Bleeding into the subarachnoid space after mTBI may also occur, but is less likely to require surgical intervention. Thus, it is critically important that acute intracranial pathology and bleeding are accurately diagnosed with a noninvasive biomarker-based test. Glial fibrillary acidic protein (GFAP), S100B, and UCHL-1 (see below) have all been used as surrogate markers of intracranial sequelae after TBI.5,12–14
Predicting patient outcome following TBI is challenging and inaccurate when based solely on clinical presentation and radiological findings because patients with seemingly comparable injuries frequently have widely variable outcomes. As underscored in the introductory paragraphs, TBI additionally evolves with time, and the injury develops into two distinct phases, a primary injury phase and secondary injury phase. Notably, the relationship between the primary phase and the secondary phase is weak and inconsistent.4 The primary phase occurs as a direct consequence of the mechanical forces experienced during head impact, which can disrupt the brain parenchyma and affect the integrity of the BBB. This is followed by a systemic and neuroinflammatory response or secondary phase, which is mediated by peripheral immune cells and the activation of immunocompetent neural cells. Molecular mediators such as cytokines, growth factors, and adhesion molecules are released, causing the activation of a complex network of pathways. The secondary injury phase can evolve over a period of hours to days and months following the primary injury. Some of these pathways are restorative in nature, while others contribute to metabolic dysregulation and hypoxic events secondary to brain swelling and edema.22–24
Nature and origin of biomarkers of TBI
Biomarkers of TBI are often measured in body fluids. Most of the data available today were obtained by measurements in CSF or blood (serum or plasma).12,17,25,26 More recently, saliva has been analyzed to show that the biomarker S100B can be measured at levels comparable with blood.27–29 In severe TBI, when access to CSF is possible, markers can be measured from the ventricles or lumbar spine.30–32 When genetic testing is the goal (see text below), DNA is extracted from cheek cells, circulating white blood cells, or isolation of free DNA “floating” in serum or (less commonly) CSF. For RNA, the favored source is the pellet of leukocytes obtained by centrifugation of whole blood, but cell-free RNA found in serum or plasma, in association with exosomes or other molecular complexes, may also provide valuable information. The cell type-specific origin of RNAs can be studied via prior separation of cells by traditional cell sorting techniques33 or by immune-affinity techniques using cell surface markers found in exosomes, which can identify their cell of origin.
A family of physiologic biomarkers, such as electroencephalogram recordings after TBI, has received notable attention.34,35 While this is not the main topic of this review, it is important to understand how the complexity of TBI has led to an equally intricate field of diagnostic and prognostic tools. In addition, recent emphasis on nonblood-derived TBI biomarkers includes genetic material and imaging findings. For the latter see the study by Zhang et al,5 where a review of imaging findings from TBI is presented and radiologic findings are discussed in the context of brain-derived blood biomarkers. The following exemplify the role of genetic biomarkers in the diagnostic and prognostic approach to TBI (reviewed in the articles by McAllister,36 Lipsky and Lin,37 Bennet et al,38 and Cotter et al;39 also see the articles by Wong and Langley40 and Meng et al41 for recent findings with genomic/genetic approaches for TBI). A discussion of gene discovery based on systems biology is also available.42
Genetic biomarkers of TBI and post-traumatic epilepsy (PTE)
The genes involved in the pathology of TBI belong to two broad categories: those that control and determine the extent of the injury (eg, inflammatory cytokines and their receptors and antagonists) and those that affect neuronal plasticity (eg, brain-derived neurotrophic factor). A new category expands these to genes involved in the cognitive process (eg, catecholamine genes).43,44 Although not well studied, the properties of dopamine (DA) may contribute to oxidative injury after TBI. In addition, chronic disruption of a number of DA-related proteins, as well as DA neurotransmission, in the striatum has been reported in experimental TBI models.45,46 Moreover, human studies using single-photon emission CT indicate that DAT expression is reduced chronically after TBI.11 In general, prognostic genetic markers are used to predict downstream pathologies and comorbidities, as in PTE.39 PTE can be characterized by a “silent” period that can last months or years. A more comprehensive understanding of predictors of epilepsy may fill a substantial knowledge gap: today, selection of treatment is still decided upon by “trial-and-error” because we lack reliable tools, such as biomarkers, to identify individuals most likely to respond to any specific intervention or individuals at risk of developing seizure disorders.47
Individuals who suffered a TBI were exposed to an increased risk of developing epilepsy. When compared with patients with no brain injury, the relative risk (RR) of epilepsy has been found to be two times higher after mTBI (RR 2.22, 95% CI 2.07–2.38) and seven times higher after severe brain injury (RR 7.40, 95% CI 6.16–8.89).48 One mechanism that has emerged as having potentially prognostic importance after TBI is systemic or brain inflammation. In the field of PTE, most genetic associations examined have been single-nucleotide polymorphisms (SNPs), a common variation within a population that can be defined as a variation of a single nucleotide, adenine (A), guanine (G), thymine (T), or cytosine (C), between individuals. A recent finding shows that an SNP of IL-1β influences the probability of PTE after TBI.49 More specifically, the rs1143634 polymorphism favored the development of PTE by affecting the CSF-to-serum ratio of this cytokine. The TT variant in rs1143634 protected from PTE. Similar dual effects of SNPs were found for GAD1 and A1AR, encoding the glutamic acid decarboxylase gene and the gene for adenosine A1 receptor, respectively.39
Pathologic significance of serum biomarkers: S100B
A common issue when dealing with biomarkers of brain pathology or health is the origin of the marker itself. Molecules, such as the glial protein S100B and neuron-specific enolase (NSE), were originally believed to directly relate to the extent of brain damage after an insult.50–52 One of the earliest documented uses of NSE and S100B was in cardiac surgery as a paradigm of iatrogenic brain damage.53,54 Only after several years was it reported that brain-derived peripheral biomarkers also act as reporters of BBB integrity even in the absence of brain injury.55,56 Thus, after an acute episode of BBB disruption, S100B is a reliable tool that can be used as a surrogate of imaging by contrast MRI5,57 or CT.5,58
It is not completely clear how these proteins leave the injured brain and enter the blood. BBB disruption25,55,57,59–63 or release independent of BBB integrity,64,65 as well as passage through the newly discovered glymphatic system,66 has been suggested as possible routes for markers appearance in blood or other biological fluids. Presumably, these proteins are first released in the brain extracellular space, a component difficult to access for repeated sampling, before being transported to the cerebral spinal fluid (CSF) where a passive diffusion from CSF to blood occurs. Some but not all these proteins are already present in the CSF and interstitial fluid, which when in communication with blood can elevate markers levels in the absence of release or ex novo synthesis.
Things are further complicated because results from different laboratories are difficult to reconcile. For example, a discrepancy between the albumin coefficient (an indicator of BBB disruption) and serum S100B (a putative reporter of BBB function) has been shown.65 Others found a concordance between serum levels of S100B and CSF:blood albumin ratios.61,67,68 A weakness in assessing BBB integrity using this albumin quotient (QA) is that patients suffering from TBI sometimes exhibit intraventricular hemorrhage. Intraventricular hemorrhage implies albumin content in the CSF, not originating from a disintegrated BBB itself but from injured vessels, a fact that might disturb the efficacy of QA to assess BBB integrity.
The hypothesis of a glymphatic system has received both support (eg, a study by Plog and Nedergaard69) and criticism.70–72 The glymphatic system has been suggested as a mediator of biomarkers extravasation after TBI.66 There are several problems with this hypothesis in the context of findings by many others. The main reason for skepticism about the interpretation of the findings by Plog et al66 is that previous studies reported that the biomarker S100B is increased within minutes after BBB disruption (carotid–jugular measures during endarterectomy,61 venous blood after osmotic disruption60,73), while the results of Plog et al were obtained at much later time points. Given also the fact that the passage in systemic circulation of S100B accurately predicts gadolinium extravasation across a leaky BBB57,74 and that S100B in serum correlates with albumin CSF:blood ratio (see discussion above), it is likely that the preferred pathway in human subjects is trans-BBB rather than the slower glymphatic pathway.
A major confounding factor for the correct interpretation of blood biomarkers related to the central nervous system (CNS) is that most of these biomarkers are expressed to varying degrees outside the CNS.75–77 Even in early work with S100B and cardiothoracic surgery, it was noted that the levels measured were inconsistent with an exclusive brain origin, but rather pointed to a peripheral source. While S100B is present in skin and fat tissue, the values measured were exorbitant even when considering a full contribution of CNS and all extracranial sources (see Table 1). In fact, by calculating the total S100B levels in a healthy individual, it was concluded that the highest value including all extracranial sources was <2 ng/mL.78 During cardiothoracic surgeries, S100B was measured at the surgical site (chest cavity) and concomitantly in serum; S100B levels were found to be almost 100-fold higher in pericardial fluid and two-fold elevated in serum compared with the ceiling value mentioned above. To address this, an effort was made to study the possibility of cross-reactivity of the test for S100B; the data demonstrated that at least with the enzyme-linked immunosorbent assay protocol used in this article, the values attributed to S100B were not due to the protein itself but other epitopes that the antibody cross-reacted with.79 Because all the spurious epitopes were found in relatively large protein, a filtration step (<50 kDa) was sufficient to remove the contribution of non-S100B antigens. The problem of cross-reactivity of tests based on immunodetection is likely to be an issue across different platforms and regardless of the antigen being measured. As also detailed above, the presence of S100B in saliva is being exploited to address this issue because saliva has a fairly low-molecular-weight cutoff, which allows S100B but not cross-reacting epitopes to be represented after capillary filtration.80
  | Table 1 Peripheral and brain S100B levels |
The contribution of fat tissue to blood levels of S100B was also studied in a large number of patients to show that even though fat expresses the protein, levels in blood are not directly affected, perhaps owing to the reduced capillary network available for protein diffusion in fat compared with other organs and tissues.26,81 Finally, the contribution of skin S100B was found to be problematic in TBI diagnosis, but with the use of a simple algorithm this could be accounted for and adjusted to show levels adjusted by skin color.78
Markers of blast vs traditional TBI
Extracranial contributions to overall biomarker levels in blood are important confounder in cases of polytrauma. Polytrauma is common in military populations after blast exposure. A rise in terrorism worldwide also fuels the polytrauma epidemic for civilian casualties. Blast-induced polytrauma poses a unique obstacle because the need for a TBI biomarker is confounded by the presence of injuries, which may increase biomarker levels in the absence of TBI.82 For the effects of polytrauma on blood biomarkers, please see below.
As in the case of impact TBI, blast injury affects the BBB.82 A recent study reports a detailed characterization of BBB opening and recovery time course in a rodent model of blast exposure.83 Early and delayed BBB “openings” were documented as well, to show preference for the extravasation of low-molecular-weight (<70 kDa) tracers in blood. BBB disruption persisted for at least 6 hours postinjury, and barrier integrity recovered by 24 hours. These findings are overall in agreement with nonblast models of TBI, in which BBB opening occurs transiently and returns to control levels. In addition, these animal model findings correlate well with the appearance of blood biomarkers related to BBB dysfunction in human studies.84 S100B was studied in a porcine model of blast injury: the early increase was not affected by the presence of systemic injuries, suggesting that as in the case of impact TBI, S100B can be used as a surrogate marker of imaging.85
Opportunities for improvements in the diagnostic characterization of TBI come from progress in the fields of blood or salivary biomarkers. These technologies are at varying stages of maturity in terms of integration into TBI clinical care: some, such as genomic stratification for therapy and outcome prognostication, are at a very early stage, whereas others, such as use of the blood biomarker S100B to stratify patients for CT imaging during the acute phase, have already been integrated into some clinical guidelines, although not widely accepted.58,86
Kinetics of biomarkers
One modern approach for identifying the presence or absence of pathophysiological changes after a head impact is the qualitative or quantitative determination of biomarkers in bodily fluids (ie, serum, saliva, CSF). Many of these biomarkers are believed to have a patient-independent kinetic behavior in the acute and chronic postinjury phases, with changes in serum biomarker levels presumed to depend mainly on source concentration and tissue of origin. It should be understood, however, that these biomarkers do not all present themselves at the same time after injury in every case, but that a biomarker’s kinetic distribution is heavily dependent on biophysical properties of the marker itself (ie, molecular weight), as well as on the anatomical and physiological characteristics of the individual (ie, glomerular filtration rate, age, gender, and skin pigmentation).78,80,83,84
A noteworthy example is seen in the temporal kinetics of S100B and GFAP shown in Figure 1A. In this example, S100B has a sharp rise within minutes after injury and returns to homeostatic levels around 1–2 hours postinjury. In contrast, abnormal serum GFAP levels persist for days after the initial event. This chart fails to emphasize the contribution of physiological variables to biomarker kinetics, however. Using a physiologically based pharmacokinetic model for biomarker distribution in blood, it was shown that this time-dependent distribution is largely based on the molecular weight of S100B and GFAP (10.7 kDa and 50.0 kDa, respectively) rather than the origin of the maker itself, as suggested in the study by Maas et al.2 We have shown that the larger molecular weight of GFAP lends to a reduced filtration by the kidneys, and thus, a longer duration of pathological serum levels after injury. These differences in postinjury serum levels are further confounded by the anatomical and physiological properties of individual patients because glomerular filtration rate is governed by a formula, which takes into account age, gender, and ethnicity. The data in Figure 1B show the results of a simulation performed according to a kinetic model for biomarkers.78 In particular, the predicted kinetic behavior of S100B and GFAP after BBB disruption is shown together with a quantitative analysis of kidney excretion of these biomarkers (Figure 1C). In this model, the barrier is “open” to 25% of free diffusion rate (to simulate an instantaneous BBB disruption event), and recovery of barrier integrity occurs 2 hours after injury. Additionally, the curve for serum GFAP is artificially increased to match the peak serum S100B, so that time-dependent properties predominate. As previously demonstrated in the study by Dadas et al,78 the half-life of GFAP in blood is considerably longer than S100B, owing to the larger molecular weight of the biomarker.
Markers of brain damage vs markers of BBB disruption
An early finding with S100B was that this blood biomarker was elevated even when brain damage was not present. It was then shown that families of soluble protein biomarkers are also indicators of BBB disruption rather than brain damage (Figure 2).55,56 This has important consequences in many clinical situations or preclinical models of systemic or brain disease. Most importantly, the usual diagnostic definitions of “positive” or “negative” predictive values need to be reformulated. In fact, in mTBI and in the absence of parenchymal lesions, the markers report the status of the BBB rather than “brain damage.” Thus, a positive predictive value should refer to contrast MRI or other sequences detecting BBB dysfunction.5 For many reasons, a comparison between blood markers and MRI has never been systemically done in TBI patients (but see the study by Thelin et al87 and Linsenmaier et al88); however, it has been studied in other pathologies.57,74,89,90 For example, the relationship between body mass index (BMI) and serum S100B values was shown not to be solely due to extracranial sources but rather reflects the clinical reality of impaired BBB integrity in morbidly obese people; factors, such as high blood pressure or metabolic disease, contribute to cerebrovascular dysfunction or BBB integrity.91–95
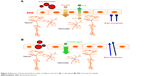  | Figure 2 Behavior of brain biomarkers under conditions of intact (A) or disrupted (B) BBB. See text for details. |
An important fact to keep in mind when analyzing blood biomarker data is the difference between a patient-specific relative increase and an increase above normal population values. For example, in one of the seminal papers showing a correlation between BMI and serum S100B,96 if one uses the normal cutoff for the test used (0.12 ng/mL), only three subjects were above the normal cutoff value, and all three were severely obese (BMI >35). The authors presented other data elsewhere but because the subjects were the same, the conclusions drawn are essentially the same.95 Another important factor to consider is the presence of comorbidities of systemic disease that may impact the BBB without directly affecting the brain. In another study, no effect of BMI on S100B was seen and only diabetic patients exhibited a high serum value.65
In polytrauma patients without TBI, the signal from biomarkers of the BBB may be elevated by conditions affecting the BBB such as hypotension (shock),93,94 sepsis,92 and pain.91 In an animal model of blast injury, a major finding was that BBB disruption, highlighted by a reduction in endothelial barrier antigen+ vessels at 7 days after injury, plays a distinct role in blast injury pathology. These effects on BBB were larger after blast with extracranial damage. Further suggestive of a role for the BBB (and not TBI) in polytrauma is the fact that similar to S100B, levels of GFAP and UCH-L1 were not able to distinguish patients with CT-negative mTBI from patients with orthopedic trauma.97,98
Biomarkers in saliva
The onset of TBI triggers an intricate sequence of physical consequences and pathophysiological responses. These sequelae can be acute (ie, intracranial lesions and BBB disruption) or chronic (ie, autoimmune response and neurodegeneration) and may occur in traumas of any severity. A critical factor in the diagnosis and management of TBI is the ability to detect these sequelae in a quick, reliable, and low-risk manner, with the ideal modality also being affordable and portable for point-of-care (POC) assessment. POC devices currently in development use blood from which platelets or red blood cells require a complicated removal process before an analysis can be completed. In addition, circumstances exist when testing of blood is problematic. For example, blood draws cannot be performed when paramedics or trained personnel are not available. Furthermore, risk of caretaker’s infection from blood products and the time and equipment necessary to separate blood components make blood diagnosis challenging. It was recently shown that saliva can act as a valid surrogate for blood for several biomarkers including S100B.27,80,99,100
The use of saliva for diagnostics has many advantages, including simple and noninvasive collection method, the fact that this body fluid does not require preprocessing, centrifugation, etc, bears minimal or no risk of contracting infections, and provides easy, low-cost storage. One of the problems, however, is that no clear-cut understanding of biomarker dynamic passage from blood to saliva was available. We have tackled the first problem as follows: we first developed a mathematical model to describe the passage of protein from systemic circulation into saliva.78,80 We used a physiologically based pharmacokinetic model to describe the distribution of brain-derived biomarkers in blood.78,101 Its main structure was expanded to include a new compartment, namely an idealized salivary gland receiving its vascular supply from the external carotid artery. The venous output can be mimicked according to the properties of jugular vein exiting the brain. Recent work102 showed that the whole saliva (WS) proteome when compared with the plasma proteome displays a larger proportion (14.5%) of low-molecular-weight proteins (<20 kDa) in contrast to only 7% for the plasma proteome. The highest fraction of proteins found in WS and blood range from 20 to 40 kDa (26%), whereas the 40 to 60 kDa range is the largest fraction for plasma (18%). This is consistent with selective permeability between blood and saliva for low-molecular-weight proteins. According to these considerations, of the commonly used protein biomarkers, only S100B and UCHL-1 are predicted to appear in saliva after an initial step of extravasation from brain to blood. The salivary “barrier” appears to be more selective than the BBB even though in reality leakage from brain to blood occurs across a disrupted barrier, while extravasation in saliva is across a healthy tissue.
Conclusion
Biomarkers are essential diagnostic and prognostic tools in a variety of neurological diseases, including TBI (eg, Figure 3). While the field of biomarker discovery has produced a plethora of molecular and genetic signatures, it still remains necessary to exactly define the nature, origin, and pathophysiological correlates of biomarkers or biomarker families. Historically, biomarkers were measured in blood or CSF; today, the use of saliva or other unconventional body fluids is gaining momentum, owing to the ease of access to secreted or filtered body fluids.
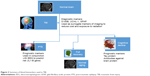  | Figure 3 Summary of blood biomarkers used in TBI. |
Disclosure
DJ and AD are employed by FloTBI, a company dealing with biomarker discovery. DJ and AD held an equity position in FloTBI. The authors report no other conflicts of interest in this work.
References
Robb MA, McInnes PM, Califf RM. Biomarkers and surrogate endpoints: developing common terminology and definitions. JAMA. 2016;315:1107–1108. | ||
Maas AIR, Menon DK, Adelson PD, et al; InTBIR Participants and Investigators. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol. 2017;16(12):987–1048. | ||
Buonora JE, Yarnell AM, Lazarus RC, et al. Multivariate analysis of traumatic brain injury: development of an assessment score. Front Neurol. 2015;6(Suppl 1):68. | ||
Pardini J, Bailes JE, Maroon JC. Mild traumatic brain injury in adults and concussion in sports. In: Winn HR, editor. Youmans Neurological Surgery. Philadelphia, PA: Elsevier; 2011:3380–3389. | ||
Zhang J, Puvenna V, Janigro D. Biomarkers of Traumatic Brain Injury and Their Relationship to Pathology. Laskowitz D, Grant G, Eds.; CRC Press: Boca Raton, FL, USA, 2016. | ||
Hawryluk GW, Manley GT. Classification of traumatic brain injury: past, present, and future. Handb Clin Neurol. 2015;127:15–21. | ||
Wilson L, Stewart W, Dams-O’Connor K, et al. The chronic and evolving neurological consequences of traumatic brain injury. Lancet Neurol. 2017;16(10):813–825. | ||
Bazarian JJ, Zhong J, Blyth B, Zhu T, Kavcic V, Peterson D. Diffusion tensor imaging detects clinically important axonal damage after mild traumatic brain injury: a pilot study. J Neurotrauma. 2007;24(9):1447–1459. | ||
Bazarian JJ, Donnelly K, Peterson DR, Warner GC, Zhu T, Zhong J. The relation between posttraumatic stress disorder and mild traumatic brain injury acquired during Operations Enduring Freedom and Iraqi Freedom: a diffusion tensor imaging study. J Head Trauma Rehabil. 2013;28:1–12. | ||
Chiara Ricciardi M, Bokkers RPH, Butman JA, et al. Trauma-specific brain abnormalities in suspected mild traumatic brain injury patients identified in the first 48 hours after injury: a blinded magnetic resonance imaging comparative study including suspected acute minor stroke patients. J Neurotrauma. 2017;34(1):23–30. | ||
Donnemiller E, Brenneis C, Wissel J, et al. Impaired dopaminergic neurotransmission in patients with traumatic brain injury: a SPET study using 123I-β-CIT and 123I-IBZM. Eur J Nucl Med. 2000;27(9):1410–1414. | ||
Kulbe JR, Geddes JW. Current status of fluid biomarkers in mild traumatic brain injury. Exp Neurol. 2016;275(Pt 3):334–352. | ||
Posti JP, Takala RS, Runtti H, et al. The levels of glial fibrillary acidic protein and ubiquitin C-terminal hydrolase-L1 during the first week after a traumatic brain injury: correlations with clinical and imaging findings. Neurosurgery. 2016;79(3):456–64. | ||
Welch RD, Ayaz SI, Lewis LM, et al. Ability of serum glial fibrillary acidic protein, ubiquitin C-terminal hydrolase-L1, and S100B to differentiate normal and abnormal head computed tomography findings in patients with suspected mild or moderate traumatic brain injury. J Neurotrauma. 2016;33(2):203– 214. | ||
Menon DK, Ercole A. Critical care management of traumatic brain injury. Handb Clin Neurol. 2017;140:239–274. | ||
Stiell IG, Wells GA, Vandemheen K, et al. The Canadian CT Head Rule for patients with minor head injury. Lancet. 2001;357(9266):1391–1396. | ||
Manley GT, Diaz-Arrastia R, Brophy M, et al. Common data elements for traumatic brain injury: recommendations from the Biospecimens and Biomarkers Working Group. Arch Phys Med Rehabil. 2010;91(11):1667–1672. | ||
Duhaime A-C, Gean AD, Haacke EM, et al. Common data elements in radiologic imaging of traumatic brain injury. Arch Phys Med Rehabil. 2010;91(11):1661–1666. | ||
Adams H, Kolias AG, Hutchinson PJ. The role of surgical intervention in traumatic brain injury. Neurosurg Clin N Am. 2016;27(4):519–528. | ||
Vega RA, Valadka AB. Natural history of acute subdural hematoma. Neurosurg Clin N Am. 2017;28(2):247–255. | ||
Smith C. Neurotrauma. Handb Clin Neurol. 2017;145:115–132. | ||
Iffland P, Grant GG, Janigro D. Mechanisms of cerebral edema leading to early seizures after traumatic brain injury. In: Lo EH, ed. Vascular Mechanisms in CNS Trauma. Springer, New York, Springer; 2014:29–36. | ||
Ivens S, Gabriel S, Greenberg G, Friedman A, Shelef I. Blood–brain barrier breakdown as a novel mechanism underlying cerebral hyperperfusion syndrome. J Neurol. 2010;257(4):615–620. | ||
Schilling L, Wahl M. Brain edema: pathogenesis and therapy. Kidney Int. 1997;59:69–75. | ||
Marchi N, Bazarian JJ, Puvenna V, et al. Consequences of repeated blood-brain barrier disruption in football players. PLoS One. 2013;8(3):e56805. | ||
Pham N, Fazio V, Cucullo L, et al. Extracranial sources of S100B do not affect serum levels. PLoS One. 2010;5(9):e12691. | ||
Lindsay A, Costello JT. Realising the potential of urine and saliva as diagnostic tools in sport and exercise medicine. Sports Med. 2017;47(1):11–31. | ||
Li Y, Sun Y, Li J, et al. Changes of ubiquitin C-terminal hydrolase-L1 levels in serum and urine of patients with white matter lesions. J Neurol Sci. 2015;357(1–2):215–221. | ||
Beharier O, Kahn J, Shusterman E, Sheiner E. S100B – a potential biomarker for early detection of neonatal brain damage following asphyxia. J Matern Fetal Neonatal Med. 2012;25(9):1523–1528. | ||
Schiavi P, Laccarino C, Servadei F. The value of the calcium binding protein S100 in the management of patients with traumatic brain injury. Acta Biomed. 2012;83:5–20. | ||
Misu T, Takano R, Fujihara K, Takahashi T, Sato S, Itoyama Y. Marked increase in cerebrospinal fluid glial fibrillar acidic protein in neuromyelitis optica: an astrocytic damage marker. J Neurol Neurosurg Psychiatry. 2009;80(5):575–577. | ||
Hayakata T, Shiozaki T, Tasaki O, et al. Changes in CSF S100B and cytokine concentrations IN early-phase severe traumatic brain injury. Shock. 2004;22(2):102–107. | ||
Zhou ZW, Li F, Zheng ZT, et al. Erythropoietin regulates immune/inflammatory reaction and improves neurological function outcomes in traumatic brain injury. Brain Behav. 2017;7(11):e00827. | ||
Reid AY, Bragin A, Giza CC, Staba RJ, Engel J. The progression of electrophysiologic abnormalities during epileptogenesis after experimental traumatic brain injury. Epilepsia. 2016;57(10):1558–1567. | ||
Klein P, Dingledine R, Aronica E, et al. Commonalities in epileptogenic processes from different acute brain insults: Do they translate? Epilepsia. 2018;59(1):37–66. | ||
McAllister TW. Genetic factors in traumatic brain injury. Handb Clin Neurol. 2015;128:723–739. | ||
Lipsky RH, Lin M. Genetic predictors of outcome following traumatic brain injury. Handb Clin Neurol. 2015;127:23–41. | ||
Bennet ER, Reuter-Rice K, Laskowitz DT. Genetic influences in traumatic brain injury. In: Laskowitz D, Grant G, editors. Translational Research in Traumatic Brain Injury. Boca Raton, FL: CRC Press/Taylor and Francis Group; 2016. Available from: https://www.ncbi.nlm.nih.gov/books/NBK326717/. Accessed October 16, 2018. | ||
Cotter D, Kelso A, Neligan A. Genetic biomarkers of posttraumatic epilepsy: a systematic review. Seizure. 2017;46:53–58. | ||
Wong VS, Langley B. Epigenetic changes following traumatic brain injury and their implications for outcome, recovery and therapy. Neurosci Lett. 2016;625:26–33. | ||
Meng Q, Zhuang Y, Ying Z, Agrawal R, Yang X, Gomez-Pinilla F. Traumatic brain injury induces genome-wide transcriptomic, methylomic, and network perturbations in brain and blood predicting neurological disorders. EBioMedicine. 2017;16:184–194. | ||
Kurowski BG, Treble-Barna A, Pitzer AJ, et al. Applying systems biology methodology to identify genetic factors possibly associated with recovery after traumatic brain injury. J Neurotrauma. 2017;34(14):2280–2290. | ||
Wagner AK, Ren D, Conley YP, et al. Sex and genetic associations with cerebrospinal fluid dopamine and metabolite production after severe traumatic brain injury. J Neurosurg. 2007;106(4):538–547. | ||
Osier ND, Dixon CE. Catecholaminergic based therapies for functional recovery after TBI. Brain Res. 2016;1640:15–35. | ||
Yan HQ, Kline AE, Ma X, Hooghe-Peters EL, Marion DW, Dixon CE. Tyrosine hydroxylase, but not dopamine beta-hydroxylase, is increased in rat frontal cortex after traumatic brain injury. Neuroreport. 2001;12(11):2323–2327. | ||
Wagner AK, Sokoloski JE, Ren D, et al. Controlled cortical impact injury affects dopaminergic transmission in the rat striatum. J Neurochem. 2005;95(2):457–465. | ||
Walker LE, Janigro D, Heinemann U, Riikonen R, Bernard C, Patel M. WONOEP appraisal: molecular and cellular biomarkers for epilepsy. Epilepsia. 2016;57(9):1354–1362. | ||
Christensen J, Pedersen MG, Pedersen CB, Sidenius P, Olsen J, Vestergaard M. Long-term risk of epilepsy after traumatic brain injury in children and young adults: a population-based cohort study. Lancet. 2009;373(9669):1105–1110. | ||
Diamond ML, Ritter AC, Failla MD, et al. IL-1beta associations with posttraumatic epilepsy development: a genetics and biomarker cohort study. Epilepsia. 2014;55(7):1109–1119. | ||
Persson L, Hardemark HG, Gustafsson J, et al. S-100 protein and neuron-specific enolase in cerebrospinal fluid and serum: markers of cell damage in human central nervous system. Stroke. 1987;18(5):911–918. | ||
Raabe A, Grolms C, Keller M, Döhnert J, Sorge O, Seifert V. Correlation of computed tomography findings and serum brain damage markers following severe head injury. Acta Neurochir. 1998;140(8):787–792. | ||
Raabe A, Grolms C, Sorge O, Zimmermann M, Seifert V. Serum S-100B protein in severe head injury. Neurosurgery. 1999;45(3):477–483. | ||
Bhattacharya K, Westaby S, Pillai R, Standing SJ, Johnsson P, Taggart DP. Serum S100B and hypothermic circulatory arrest in adults. Ann Thorac Surg. 1999;68(4):1225–1229. | ||
Usui A, Kato K, Murase M, et al. Neural tissue-related proteins (NSE, G0α, 28-kDa calbindin-D, S100b and CK-BB) in serum and cerebrospinal fluid after cardiac arrest. J Neurol Sci. 1994;123(1–2):134–139. | ||
Marchi N, Cavaglia M, Fazio V, Bhudia S, Hallene K, Janigro D. Peripheral markers of blood–brain barrier damage. Clin Chim Acta. 2004;342(1–2):1–12. | ||
Marchi N, Rasmussen P, Kapural M, et al. Peripheral markers of brain damage and blood-brain barrier dysfunction. Restor Neurol Neurosci. 2003;21(3–4):109–121. | ||
Vogelbaum MA, Masaryk T, Mazzone P, et al. S100beta as a predictor of brain metastases: brain versus cerebrovascular damage. Cancer. 2005;104:817–824. | ||
Undén J, Ingebrigtsen T, Romner B. Scandinavian guidelines for initial management of minimal, mild and moderate head injuries in adults: an evidence and consensus-based update. BMC Med. 2013;11(1):50. | ||
Járányi Z, Székely M, Bobek I, Gálfy I, Gellér L, Selmeci L. Impairment of blood-brain barrier integrity during carotid surgery as assessed by serum S-100B protein concentrations. Clin Chem Lab Med. 2003;41(10):1320–1322. | ||
Kapural M, Krizanac-Bengez L, Barnett G, et al. Serum S-100β as a possible marker of blood–brain barrier disruption. Brain Res. 2002;940(1–2):102–104. | ||
Mussack T, Hauser C, Klauss V, et al. Serum S-100B protein levels during and after successful carotid artery stenting or carotid endarterectomy. J Endovasc Ther. 2006;13(1):39–46. | ||
Raabe A, Menon DK, Gupta S, Czosnyka M, Pickard JD. Jugular venous and arterial concentrations of serum S-100B protein in patients with severe head injury: a pilot study. J Neurol Neurosurg Psychiatry. 1998;65(6):930–932. | ||
Teepker M, Munk K, Mylius V, et al. Serum concentrations of s100b and NSE in migraine. Headache. 2009;49(2):245–252. | ||
Bellander B-M, Olafsson IH, Ghatan PH, et al. Secondary insults following traumatic brain injury enhance complement activation in the human brain and release of the tissue damage marker S100B. Acta Neurochir. 2011;153(1):90–100. | ||
Kleindienst A, Schmidt C, Parsch H, Emtmann I, Xu Y, Buchfelder M. The passage of S100B from brain to blood is not specifically related to the blood-brain barrier integrity. Cardiovasc Psychiatry Neurol. 2010;2010(2):801295–801298. | ||
Plog BA, Dashnaw ML, Hitomi E, et al. Biomarkers of traumatic injury are transported from brain to blood via the glymphatic system. J Neurosci. 2015;35(2):518–526. | ||
Blyth BJ, Farhavar A, Gee C, et al. Validation of serum markers for blood-brain barrier disruption in traumatic brain injury. J Neurotrauma. 2009;26(9):1497–1507. | ||
Blyth BJ, Farahvar A, Gee CA, Hawthorn BA, Bazarian JJ. 223: serum S-100b concentrations are an accurate indicator of blood brain barrier integrity. Ann Emerg Med. 2008;51(4):538–539. | ||
Plog BA, Nedergaard M. The glymphatic system in central nervous system health and disease: past, present, and future. Annu Rev Pathol. 2018;13:379–394. | ||
Smith AJ, Yao X, Dix JA, Jin B-J, Verkman AS. Test of the ‘glymphatic’ hypothesis demonstrates diffusive and aquaporin-4-independent solute transport in rodent brain parenchyma. eLife. 2017;6:pii:e27679. | ||
Asgari M, de Zélicourt D, Kurtcuoglu V. Glymphatic solute transport does not require bulk flow. Sci Rep. 2016;6(1):38635. | ||
Jin B-J, Smith AJ, Verkman AS. Spatial model of convective solute transport in brain extracellular space does not support a “glymphatic” mechanism. J Gen Physiol. 2016;148(6):489–501. | ||
Marchi N, Angelov L, Masaryk T, et al. Seizure-promoting effect of blood–brain barrier disruption. Epilepsia. 2007;48(4):732–742. | ||
Kanner AA, Marchi N, Fazio V, et al. Serum S100beta: a noninvasive marker of blood-brain barrier function and brain lesions. Cancer. 2003;97:2806–2813. | ||
Thelin EP, Jeppsson E, Frostell A, et al. Utility of neuron-specific enolase in traumatic brain injury; relations to S100B levels, outcome, and extracranial injury severity. Crit Care. 2016;20(1):285. | ||
Savola O, Pyhtinen J, Leino TK, Siitonen S, Niemela O, Hillbom M. Effects of head and extracranial injuries on serum protein S100B levels in trauma patients. J Trauma. 2004;56(6):1229–1234. | ||
Hasselblatt M, Mooren FC, von Ahsen N, et al. Serum S100β increases in marathon runners reflect extracranial release rather than glial damage. Neurology. 2004;62(9):1634–1636. | ||
Dadas A, Washington J, Marchi N, Janigro D. Improving the clinical management of traumatic brain injury through the pharmacokinetic modeling of peripheral blood biomarkers. Fluids Barriers CNS. 2016;13(1):21. | ||
Fazio V, Bhudia SK, Marchi N, Aumayr B, Janigro D. Peripheral detection of S100β during cardiothoracic surgery: what are we really measuring? Ann Thorac Surg. 2004;78(1):46–52. | ||
Dadas A, Janigro D. The role and diagnostic significance of cellular barriers after concussive head trauma. Concussion. 2018;49(4):CNC53. | ||
Weiss L, Haydock K, Pickren JW, Lane WW. Organ vascularity and metastatic frequency. Am J Pathol. 1980;101:101–114. | ||
Hubbard WB, Greenberg S, Norris C, Eck J, Lavik E, Vandevord P. Distinguishing the unique neuropathological profile of blast polytrauma. Oxid Med Cell Longev. 2017;2017:5175249. | ||
Hue CD, Cho FS, Cao S, et al. Time course and size of blood–brain barrier opening in a mouse model of blast-induced traumatic brain injury. J Neurotrauma. 2016;33(13):1202–1211. | ||
Papa L, Brophy GM, Welch RD, et al. Time course and diagnostic accuracy of glial and neuronal blood biomarkers GFAP and UCH-L1 in a large cohort of trauma patients with and without mild traumatic brain injury. JAMA Neurol. 2016;73(5):551–560. | ||
Gyorgy A, Ling G, Wingo D, et al. Time-dependent changes in serum biomarker levels after blast traumatic brain injury. J Neurotrauma. 2011;28(6):1121–1126. | ||
Undén L, Calcagnile O, Undén J, Reinstrup P, Bazarian J. Validation of the Scandinavian guidelines for initial management of minimal, mild and moderate traumatic brain injury in adults. BMC Med. 2015;13(1):292. | ||
Thelin EP, Nelson DW, Bellander B-M. Secondary peaks of S100B in serum relate to subsequent radiological pathology in traumatic brain injury. Neurocrit Care. 2014;20(2):217–229. | ||
Linsenmaier U, Wirth S, Kanz K-G, Geyer LL. Imaging minor head injury (MHI) in emergency radiology: MRI highlights additional intracranial findings after measurement of trauma biomarker S-100B in patients with normal CCT. Br J Radiol. 2016;89(1061):20150827. | ||
Choi H, Puvenna V, Brennan C, et al. S100B and S100B autoantibody as biomarkers for early detection of brain metastases in lung cancer. Transl Lung Cancer Res. 2016;5(4):413–419. | ||
Mazzone PJ, Marchi N, Fazio V, et al. Small vessel ischemic disease of the brain and brain metastases in lung cancer patients. PLoS One. 2009;4(9):e7242. | ||
Lochhead JJ, Ronaldson PT, Davis TP. Hypoxic stress and inflammatory pain disrupt blood-brain barrier tight junctions: implications for drug delivery to the central nervous system. AAPS J. 2017;19(4):910–920. | ||
Kuperberg SJ, Wadgaonkar R. Sepsis-associated encephalopathy: the blood–brain barrier and the sphingolipid rheostat. Front Immunol. 2017;8:597. | ||
Meybohm P, Cavus E, Dörges V, et al. Release of protein S100B in haemorrhagic shock: Effects of small volume resuscitation combined with arginine vasopressin. Resuscitation. 2008;76(3):449–456. | ||
Krizbai IA, Lenzser G, Szatmari E, et al. Blood-brain barrier changes during COMPENSATED and decompensated hemorrhagic shock. Shock. 2005;24(5):428–433. | ||
Steiner J, Myint AM, Schiltz K, et al. S100B serum levels in schizophrenia are presumably related to visceral obesity and insulin resistance. Cardiovasc Psychiatry Neurol. 2010;2010(6):480707–480711. | ||
Steiner J, Schiltz K, Walter M, et al. S100B serum levels are closely correlated with body mass index: an important caveat in neuropsychiatric research. Psychoneuroendocrinology. 2010;35(2):321–324. | ||
Posti JP, Hossain I, Takala RS, et al. Glial fibrillary acidic protein and ubiquitin C-terminal hydrolase-L1 are not specific biomarkers for mild CT-negative traumatic brain injury. J Neurotrauma. 2017;34(7):1427–1438. | ||
Luoto TM, Raj R, Posti JP, Gardner AJ, Panenka WJ, Iverson GL. A systematic review of the usefulness of glial fibrillary acidic protein for predicting acute intracranial lesions following head trauma. Front Neurol. 2017;8:652. | ||
Kaczor-Urbanowicz KE, Martin Carreras-Presas C, Aro K, Tu M, Garcia-Godoy F, Wong DTW. Saliva diagnostics – current views and directions. Exp Biol Med. 2017;242(5):459–472. | ||
Michetti F, Corvino V, Geloso MC, et al. The S100B protein in biological fluids: more than a lifelong biomarker of brain distress. J Neurochem. 2012;120(5):644–659. | ||
Peters SA. Evaluation of a generic physiologically based pharmacokinetic model for lineshape analysis. Clin Pharmacokinet. 2008;47(4):261–275. | ||
Loo JA, Yan W, Ramachandran P, Wong DT. Comparative human salivary and plasma proteomes. J Dent Res. 2010;89(10):1016–1023. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

