Back to Journals » International Journal of Nanomedicine » Volume 17
Bioactive Effects of Curcumin in Human Immunodeficiency Virus Infection Along with the Most Effective Isolation Techniques and Type of Nanoformulations
Authors Butnariu M , Quispe C, Koirala N, Khadka S , Salgado-Castillo CM, Akram M, Anum R, Yeskaliyeva B, Cruz-Martins N , Martorell M , Kumar M, Vasile Bagiu R, Abdull Razis AF, Sunusi U, Muhammad Kamal R, Sharifi-Rad J
Received 1 March 2022
Accepted for publication 7 June 2022
Published 15 August 2022 Volume 2022:17 Pages 3619—3632
DOI https://doi.org/10.2147/IJN.S364501
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yan Shen
Monica Butnariu,1 Cristina Quispe,2 Niranjan Koirala,3,4 Sujan Khadka,5,6 Carla Marina Salgado-Castillo,7 Muhammad Akram,8 Rabia Anum,9 Balakyz Yeskaliyeva,10 Natália Cruz-Martins,11– 14 Miquel Martorell,15,16 Manoj Kumar,17 Radu Vasile Bagiu,18,19 Ahmad Faizal Abdull Razis,20,21 Usman Sunusi,21,22 Ramla Muhammad Kamal,21,23 Javad Sharifi-Rad7
1Chemistry & Biochemistry Discipline, University of Life Sciences “King Mihai I” from Timisoara, 300645, Calea Aradului 119, Timis, Romania; 2Facultad de Ciencias de la Salud, Universidad Arturo Prat, Iquique, 1110939, Chile; 3Department of Natural Products Research, Dr. Koirala Research Institute for Biotechnology and Biodiversity, Kathmandu, 44600, Nepal; 4Laboratory of Biotechnology, Faculty of Science and Technology, University of Macau, Macau SAR, 999078, People’s Republic of China; 5University of Chinese Academy of Sciences, Beijing, 100049, People’s Republic of China; 6State Key Laboratory of Environmental Aquatic Chemistry” with “State Key Laboratory of Environmental Aquatic Chemistry, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, Beijing, 100085, People’s Republic of China; 7Facultad de Medicina, Universidad del Azuay, Cuenca, Ecuador; 8Department of Eastern Medicine, Government College University Faisalabad, Faisalabad, Pakistan; 9SINA Health, Education and Welfare Trust, Karachi, Pakistan; 10Faculty of Chemistry and Chemical Technology, Al-Farabi Kazakh National University, Almaty, Kazakhstan; 11Faculty of Medicine, University of Porto, Porto, Portugal; 12Institute for Research and Innovation in Health (i3S), University of Porto, Porto, Portugal; 13Institute of Research and Advanced Training in Health Sciences and Technologies (CESPU), Gandra PRD, 4585-116, Portugal; 14TOXRUN-Oxicology Research Unit, University Institute of Health Sciences, CESPU, CRL, Gandra, 4585-116, Portugal; 15Department of Nutrition and Dietetics, Faculty of Pharmacy, and Centre for Healthy Living, University of Concepción, Concepción, 4070386, Chile; 16Universidad de Concepción, Unidad de Desarrollo Tecnológico, UDT, Concepción, 4070386, Chile; 17Chemical and BioChemical Processing Division, ICAR – Central Institute for Research on Cotton Technology, Mumbai, 400019, India; 18Victor Babes University of Medicine and Pharmacy of Timisoara Department of Microbiology, Timisoara, Romania; 19Preventive Medicine Study Center, Timisoara, Romania; 20Department of Food Science, Faculty of Food Science and Technology, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia; 21Natural Medicines and Products Research Laboratory, Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia; 22Department of Biochemistry, Bayero University Kano, Kano, Nigeria; 23Department of Pharmacology, Federal University Dutse, Dutse, Jigawa State, Nigeria
Correspondence: Natália Cruz-Martins; Ahmad Faizal Abdull Razis, Email [email protected]; [email protected]
Abstract: Human immunodeficiency virus (HIV) is one of the leading causes of death worldwide, with African countries being the worst affected by this deadly virus. Curcumin (CUR) is a Curcuma longa-derived polyphenol that has attracted the attention of researchers due to its antimicrobial, anti-inflammatory, antioxidant, immunomodulatory and antiviral effects. CUR also demonstrates anti-HIV effects by acting as a possible inhibitor of gp120 binding, integrase, protease, and topoisomerase II activities, besides also exerting a protective action against HIV-associated diseases. However, its effectiveness is limited due to its poor water solubility, rapid metabolism, and systemic elimination. Nanoformulations have been shown to be useful to enhance curcumin’s bioavailability and its effectiveness as an anti-HIV agent. In this sense, bioactive effects of CUR in HIV infection are carefully reviewed, along with the most effective isolation techniques and type of nanoformulations available.
Keywords: curcumin, bioactivities, HIV therapy, nanoparticles, nanoformulations
Introduction
Human immunodeficiency virus (HIV) infection is the most critical public health challenge that our society has ever faced. Since the 1980s, 75 million individuals have been infected by this deadly virus, leaving in its path a staggering burden of 32 million deaths.1 Up until 2016, it was among the top 10 leading causes of death globally.2 It remains one of the recurrent and common origins of mortality in the African region, with around 4% of the population infected; moreover, this continent alone accounts for ~70% of all infected cases.3 There are two main types of HIV: HIV-1, which is accountable for the spread of this epidemic, and HIV-2 which is mainly restricted to western regions of Africa.4,5 The harming effect of this retrovirus resides in its ability to destroy cluster of differentiation 4 (CD4) T cells, these being responsible for activating adaptive immune responses.6 This effect will usually lead to acquired immunodeficiency syndrome (AIDS), usually within the first ten years after diagnosis. This syndrome comprises a vast number of opportunistic illnesses, i.e., infections, malignancies, and specific cases of wasting and encephalopathy.7 Before introducing highly active antiretroviral therapy (HAART) in the late 90s, the life expectancy of a patient after the onset of AIDS was around 2 years. Now, with proper treatment, a patient can live for >10 years after this dangerous syndrome onset.8 Furthermore, early initiation of antiretroviral therapy (ART) following HIV diagnosis seems to be linked to a higher probability of immune reconstitution to near-normal CD4 cell levels and a dramatic decrease in transmission rates.9,10 However, currently available treatment strategies have critical challenges. Antiretroviral therapeutics require a lifelong responsibility, with poor adherence generally resulting in increased viral load and subsequent treatment failure. Also, antiretrovirals exhibit low solubility, resulting in poor bioavailability, and increasing the risk of suboptimal drug concentrations.11 Therefore, nanomedicine has been considered a potential solution to ART bioavailability issues.12 Nanoformulations can offer a wide range of possibilities in HIV treatment beyond classic ART; additionally, they can also provide the opportunity of using natural or synthetic compounds that can serve as enhancers of conventional therapeutic drugs.
Currently, there is no safe and effective treatment for HIV. Antiretroviral medication, however, can help HIV patients live longer and have fewer secondary infections. Pleiotropic chemicals, which are natural compounds, might be effective against HIV. Because of its activities such as antioxidant, anti-inflammatory, anticancer, antiviral, and antibacterial, curcumin (CUR), a yellow pigment found in the spice turmeric (Curcuma longa L.), can be used to treat a variety of disorders, including HIV-AIDS.12,14 Unlike other phytochemicals, CUR has been proved to have a wide range of functional activities, i.e. food additive, colorant, antioxidant, and as remedies .12,13 In recent years, CUR has received particular attention from the scientific community due to its antimicrobial, anti-inflammatory, antioxidant and immunomodulatory effects.14 Further, it has proven benefits in neurodegenerative,15 psychiatric,16 and cardiovascular17 diseases, as well as in diabetes mellitus, cancer,14 autoimmune18 and infectious diseases, including HIV. Briefly, CUR exerts an anti-HIV effect by acting as a possible inhibitor of gp120 binding, and of integrase, protease and topoisomerase II activities.8,19,20 However, its usefulness is somewhat finite due to its low water solubility, rapid metabolism, and rapid systemic elimination.21 Nano-particle (NP) formulations could play a key role in enhancing CUR’s bioavailability. Nanoparticles (usually 10–200 nm in diameter) can improve the loaded therapeutic molecule’s circulation duration and residency at the problematic site by enhancing penetration and retention (EPR), which is a critical feature in drug delivery technology. CUR delivery with nanotechnology not only improves solubility, quick drug metabolism, degradation, and drug stability difficulties, but it should also disseminate or target targeted tissues while reducing inadvertent harm to nearby normal cells/tissues.14,19 Several tactics have been used to increase the solubility and bioavailability of CUR as nanotechnology continues to evolve.17 The use of organic nanomaterials such as polymers, lipids, dendrimers, and polysaccharides with functionalization of targeted therapy has been proposed and explored based on specific purposes or requirements for CUR. The obtained CUR has been efficiently scaled down to nanoscale by using nano-technologies, which considerably aids in maximizing bioactivities and minimizing physical and chemical degradation of CUR.12,20,21 In this sense, this review collates the information on bioactivities of CUR in HIV infection, at same time highlighting the CUR isolation techniques along with the various types of nanoformulations available.
Curcumin: Origin and General Bioactivity in HIV
Curcumin: Origin and Chemical Structure
Turmeric has been conventionally used as a spice and for a number of remedies for centuries.22,23 CUR is the major active chemical compound present in turmeric. Belonging to the Zingiberaceae family, turmeric is composed of curcuminoids, which include cyclocurcumin and three types of CUR, present at different percentages, with 77% corresponding to CUR I, 17% to CUR II, and 3% to CUR III.24 In addition, CUR contents differ substantially depending on the different Curcuma spp., along with geographical locations that also lead to differing chemical content of CUR, probably due to hybridization of other species; so, it is very important to select the species with higher CUR contents.25 In 1815, CUR (diferuloylmethane) was obtained by Vogel and Pelletier while its chemical structure, (1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) (Figure 1) was validated by Lampe and Miłobędzka in 1913.26,27
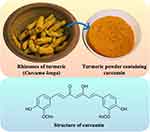 |
Figure 1 Rhizomes of turmeric and turmeric powder and chemical structure of curcumin. |
Despite phenolic compounds have gained increasing attention in the last years for their multiple health-promoting abilities, and CUR is a good example, its numerous pharmacological activities have even boosted towards a deeper knowledge on their therapeutic effects.25,28
Curcumin: Pharmacological Actions, a General Perspective
There is a plethora of research articles supporting the anti-inflammatory, antioxidant, antiseptic, analgesic, antimicrobial (such as antibacterial, antifungal, antiviral, antimalarial) and anticancer properties of C. longa, mostly attributed to the chemical compound CUR present in its chemical composition.29,30 Generally recognized as safe (GRAS) when consumed up to 12 g/day by the Food and Drug Administration (FDA) for healthy consumption without causing any side effects in humans in a clinical trial,31 there are still many turmeric features that need to be better addressed. Table 1 shows the mode of action of CUR responsible for its multiple pharmacological properties.
 |
Table 1 General Description of CUR Activities |
Curcumin Anti-HIV Bioactivity
CUR regulates the secretion of pro-inflammatory cytokine such as interleukin (IL)-4, 6, 8, and tumor necrosis factor alpha (TNF-α). Further, CUR enhances anti-inflammatory cytokines such as soluble intercellular adhesion molecule (ICAM)-1 and IL-10. CUR helps in reducing the inflammation caused by viruses and bacteria.35
Pathological Perspective
Inhibit HIV-1 Replication by Apotransferrin Nano-Particles Provide Efficient Cell Uptake
CUR encapsulation with NPs provides multiple benefits and increases drug solubility, ultimately enhancing its efficacy and stability, and improving drug degradation57 and target cells by receptor-mediated endocytosis,58 as HIV-infected cells are expressive to transferrin receptors.59 CUR-loaded with apotransferrin capsulated in NPs bind to transferring receptors, leading to cell uptake and T-cells cytotoxicity, eventually inhibiting HIV replication, at the same time also inhibiting the expression of topoisomerase II, interleukin (IL)-1β and cyclooxygenase (COX)-2 blocking HIV-induced inflammatory activities.58
HIV-1 Integrase Inhibitor
Enzyme HIV-1 integrase, integrates the HIV virus DNA to replicate further. AutoDock software has been used to run docking, giving data on structural analysis of CUR interactions, revealing that it binds to the HIV integrase, with Asp,64 His,67 Thr,66 Glu,92 Thr,93 Asp,116 Ser,119 Asn,120 and Lys159 being the binding sites for integrase. CUR also binds with the active site links the catalytic residues adjacent Asp116 and Asp,64 and close to divalent metal Mg2+ ion resulting in integrase inhibitory activity against HIV.60 For example, a study conducted in Germany testing two CUR analogs, dicaffeoylmethane and rosmarinic acid, stated that both inhibited the integrase activity, at IC50 values <10 µM. The study showed that CUR binds to lysine amino acid at active site of HIV-I integrase enzyme and inhibits its activity.61 Similarly, a study in the USA showed the inhibition of integrase with IC50 values of 40 µM for strand transfer, causing a deletion of mutant containing amino acids, that ultimately suggests anti-integrase activity.62
Inhibition of Proteases
Computational docking has also revealed that CUR inhibits proteases, through CUR binding to active sites of Asp,25 Asp,29 Asp,30 Gly,270 Asp,290 and Asp300 of HIV proteases and triggering their inhibition. CUR also inhibits proteases of HIV-1 (IC50; 100 µM) and HIV-2 (IC50; 250 µM). Increased number of hydrogen bonding promoted by the hydroxyl and/or keto–enol chemical structures are crucial for the inhibitory action of both HIV-I integrase and protease.60
Inhibition of Genome Expression
HIV-1 gene expression depends upon Tat and Rev proteins, which activate the transcription and transport mRNA that encode the viral proteins.63 CUR inhibits Tat protein, reducing HIV infection in an individual. A study reported that CUR (10–100 nM) inhibited Tat activation of HIV-1-long terminal repeats (LTR), 80% in HeLa cells. CUR also inhibits UV-activated HIV-LTR gene expression, an inhibitory effect being found in HIV-Tat protein acetylation by p300 in SupT1 cells, it was thus concluded that it acts as a lead compound in combination therapy of HIV.64
Inhibition of Kinases
Kinases have a crucial role in HIV-1 replication. IL-10 production is activated by Tat protein which is activated by protein kinase pathway.65 Studies reveal that CUR has anti-inhibitory effect of protein kinase pathway in numerous cells which ultimately is preventative with HIV and also with other chronic conditions.64,66
Inhibition and Degradation of HIV-1 Tat Protein
Tat associated with HIV-1 is an intrinsic protein that has a major role in virus replication. This Tat protein is degraded by 20S proteasome. A study showed that CUR degrades Tat protein by activating 20S proteasome, and further inhibits HIV-1 infected cells by decreasing the level of Tat-mediated LTR promoter transactivation.19 Table 2 and Figure 2 show the CUR action in HIV inhibition.
 |
Table 2 Mechanisms of Actions of Curcumin |
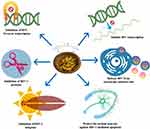 |
Figure 2 Anti-HIV activities of curcumin. |
Hence, the combination of CUR with viral coat proteins, virus-specific enzymes (RNA polymerase, integrase, protease, kinases), may affect and eliminate virus replication, infection, and damage to cells.
Activity of Curcumin in HIV-Associated Disease
CUR supplementation to the patients leads to the activation of immune components. This includes reduction in activation of allergy and inflammation and improves the innate immunity to fight against pathogens, cancer, cardiovascular diseases, and other metabolic disorders. CUR can reduce intracellular JAKs/STATs, MAPKs, NF-κB, β-catenin, and the Notch-1 pathway by regulating the gene expression of pro-inflammatory cytokines, such as IL-2, 6, 10, IL-1β, TNF-α which mediate inflammatory pathways. CUR has been widely used in the treatment of autoimmune diseases such as arthritis, psoriasis etc.38,51 Hence, CUR has been reported to show immunomodulatory effect in the treatment of various diseases. In the following sub-sections the immunomodulatory role of CUR in management of HIV-associated diseases is discussed.
Cardiovascular Disease
The HIV protease inhibitor ritonavir is responsible for causing a plethora of cardiovascular disorders, vascular dysfunction due to oxidative stress, decreased NO level and its release and increased oxygen production. In a study, CUR revealed to be able to block the ritonavir effects, triggering 71% vessel contraction, 59% endothelium-dependent relaxation and 52% endothelium-independent relaxation when compared with control group. Given these findings, CUR inhibits HIV-associated cardiovascular complications at the same time as increasing the lifespan of HIV-infected individuals.68
Neurological Disorders
Scientific study showed a positive correlation between HIV-infected individuals and neurological disorders caused by the neuroinflammation and activation of microglia cells of the central nervous system (CNS). CUR exhibited stronger protective action against neuronal damage caused by HIV-1 gp120. CUR reduces neurological disorders by inhibiting trans-activating proteins (Tat) activated HIV-1 transcription, inhibiting viral replication and also suppressed inflammatory cytokines nuclear factor (NF)-κB, tumor necrosis factor (TNF)-α, and IL-1β.69,70 CUR also protects the cortical neurons by inhibiting the HIV-1 gp120-induced elevation of the delayed rectification and transient outward K+ current.69
Gastrointestinal Disorders
Apart from the neurological disorders, cardiovascular problems and carcinogenic complications in HIV immune compromised patients, gastrointestinal disorders are another complicating situation. The prevalence of HIV-associated diarrhea is up to 14%. However, a daily dose of 1.86 g of CUR was able to resolve diarrhea in 13 ± 9 days, alongside a decrease in bloating and abdominal pain complaints as well as weight gain in a few patients.71
Carcinogenic Conditions
Epstein–Barr virus is most commonly associated with the development of B-cell lymphoma (Burkitt lymphoma, primary central nervous system lymphoma, Hodgkin and systemic non-Hodgkin lymphoma) in HIV-associated immunodeficient patients.72 CUR has been reported to act as an efficient anti-cancer and chemo-preventive agent. CUR causes inactivation of Epstein-Barr virus to cause B-cell abnormal growth and necrosis. Moreover, it has been reported that CUR enhances apoptosis of B-cell chronic lymphocytic leukemia (B-CLL) which inhibits the proliferation of Epstein-Barr Virus and thus acts as an important therapeutic agent against carcinogenic conditions.73
Tolerability of Curcumin
The current investigation on the tolerability or toxicity of the CUR showed relatively very low toxicity. Acute toxicity studies showed that 40–100 mg of high doses does not cause any lethal effect on the tested mice. Half-lethal dose for mice was more than 2 g/kg which showed that a safe dose of CUR was high. Majority of investigations showed that CUR is safe (>120 mg/m2) for clinical trials.74
Nanoformulation: Molecules, Isolation Techniques and HIV Effects
HIV being a deadly infectious agent, a substantial reduction in mortality rates related to HIV-1 infection has been recorded following application of anti-retroviral therapeutics, it being possible to manage a quickly lethal into a stable manageable illness.75 This signifies the utmost importance of bioavailability, pharmacology, cytotoxicity and dosing interval of anti-retroviral drugs for the treatment of HIV infection.75–77
For CUR, the inhibition of HIV-1 infection has been documented.78,79 CUR can inhibit HIV-1 transcription,80 mainly by Tat-activated HIV-1 transcription.80–82 In vitro, it has been observed that CUR can inhibit HIV-1 protease,83 HIV-1 integrase,84,85 and in silico binding to HIV-1 reverse transcriptase,86 protease,60 and integrase.62
On the other hand, oxidative stress has been implicated in a very long list of tissue damage in HIV/AIDS patients for which plant antioxidants can provide defense against viral replication and oxidative stress-associated cell death.87 CUR decreases the reactive oxygen species (ROS) and inflammatory mediators’ production in microglia (HIV-1-gp120-triggered), thereby protecting cortical neurons against HIV-1-mediated apoptosis.69
Nanoformulation sizes can vary from 10–100 nm in diameter and are often administrated per os or by injection as a fluid.88 Among their multiple properties, the most prominent one is their useful application in medicine for nano-drugs formulation, helping to reach the target sites promptly.17 Depending on the preparation method, nanoformulations may have a nano-spheres (drugs are uniformly dispersed) or nano-capsules (drugs are embedded inside the cavity of polymer matrix) structure. Broadly, in terms of size, nano-capsules are larger than nano-spheres and have a greater degree of polymerization; in terms of structure, nano-spheres can more easily be lyophilized in freeze-drying technology than nano-capsules.89
Nano-Formulation Techniques and Types
CUR’s role in the physico-chemical properties and biological activity of nanoformulations has been suggested by significant progress in the development of delivery systems for CUR. Among the different techniques for nano particles (NPs) elaboration, nano-precipitation is the most commonly used.89 The history of synthesizing NPs for medical purposes dates back long ago based on two principal conventionally produced ideas, one developed by Paul Ehrlich (concept of magical bullets)90 and the other by Richard Feynman (concept of miniaturization).91 Nevertheless, in 1969 it was established that epoxy resin beads can be effectively used as carrier material for the delivering of the drug under physiological pH (acidic and basic conditions).92 In an investigation by Müller et al93 working on the development of lipid NPs with wide ranges of application, the surface modified lipid NPs with poloxamine 908 and poloxamer 407 effectively acted as colloidal carriers without being phagocytosed in the system. The study established that lipid NPs can act as a suitable solid carrier for transport of drugs in the vascular system via the blood. In another study, NPs and micro particles were synthesized using a nano-precipitation process.94 In this process, nano-form polymer and drug are dissolved at ambient temperature using organic solvent with constant stirring, then stabilizer is solubilized in water at concentration of 0.5–5% w/v. Pouring the organic phase into water phase results in change in the solubility of the drug and a polymer occurs which finally develops the NPs. Finally, the solvent is removed from the system under reduced pressure which is followed by purification using centrifugation and lyophilization.17
A liposome is a spherical vesicle built from a bilayer of lipids, and is one of the most efficient drug carriers,95 and one of the commonly used systems for drugs delivery.96 Liposomes are regarded as the most successful encapsulating material to date for targeted delivery of both hydrophilic and hydrophobic drugs by entrapping in its aqueous core and lipid membrane, respectively. Their size varies from 90–150 nm in diameter and they have self-assembling capabilities in the hydrophobic or hydrophilic therapies into its empty core.96,97 There are four types of liposomes: (1) conventional type liposomes, (2) polyethylene glycol (PEG) types, (3) ligand-targeted, and (4) theranostic types. Conventional liposomes are composed of a bilayer of lipids which can develop cationic, anionic or neutral phospholipids and cholesterol, surrounding an aqueous core. In conventional liposomes aqueous space and lipid bilayer can be accommodated by water or lipid loving materials, respectively. In PEG type liposomes, the liposome surface is incorporated with the PEG to attain steric equilibrium whereas in the case of ligand targeted liposomes, ligands (antibodies, peptides, carbohydrates) are adhered to the liposome’s surface. Theranostic liposomes are a hybrid of all three previously mentioned liposomes which consists of NP along with imaging, targeting and a therapeutic element.98 Because of the longer retention periods in the bloodstream, liposomes are viewed as having an advantage in the treatment of disease, with an increasing demand being stated towards liposomal formulations application for drug delivery in medical sectors.
A nanogel (10–100 nm) is made of a hydrogel created under controlled conditions, either by physical or chemical cross-linking of polymer chains. As a result, nanogels are ideal for storing and releasing drugs. As part of this process, cells are prepared and released with active ingredients to retain their activity, improve stability, and reduce drug immunogenicity.99 To boost the bioavailability of low-molecular-weight medicines and bio macromolecules in the mouth and brain by means of salt bonds, hydrogen bonds, or hydrophobic interactions, nanogels have been designed as carriers for drug delivery.100–102 They are designed to respond to changes in pH, temperature, acidic conditions, the activity of enzymes and magnetic fields, as well as light. Changing the nanogels’ conformation may result in a “on-demand” release of any cargo that is placed into the nanogels. If the chemical composition of nanogels is altered precisely, their properties can be precisely controlled.103 Khosropanah et al observed that CUR-loaded nanogels were at least twice as powerful as free CUR, presumably due to increased absorption. There is evidence that self-assembled nanogels derived from hydrophobically modified dextrin can be effective CUR nano-carriers, according to literature. It shows that the formulation was more stable in water than in phosphate buffer saline, which was subsequently tested by dynamic light scattering and fluorescence studies.104
Role of Curcumin Nanoformulations as Anti-HIV Agents
CUR directly binds in multiple target sites related with crucial enzymes of HIV.99 An in silico study showed that CUR binds to different sites of the substrate-binding cavity of HIV protease.105 When the cells gets infected, trans-activator of transcription (Tat) is secreted which further promotes the destruction of T-cells and stimulates the formation of HIV-induced tumors. CUR provokes the destruction of Tat by proteosomal degradation and suppressed the Tat acetylation which resulted in reduced HIV proliferation.106 Similarly, Rai et al described the anti-HIV activity of Cur-AgNP via downregulation of inflammatory mediators (IL-1β, TNF-α, and IL-6), and stopping HIV replication.105 CUR combined with silver nanoparticles (AgNP) had been found to act as antiretroviral agents. HIV-1 causes neurological complications due to its ability to increase the production of pro-inflammatory cytokines.107 Furthermore, poly-proteins processing by HIV-1 protease may produce new strains of viruses. Consequently, CUR nanoformulations have been shown to be effective in reducing such difficulties because it inhibits the activity of HIV-1 proteases by binding to their active sites, such as the CCR5 (C-chemokinereceptortype5). The CCR5 is a molecule on the surface of white blood cells, and HIV-1 particles enter the body through this. Because of this, CUR molecules attach to CCR5, preventing HIV-1 from gaining access into host cells, and so protecting them from infection. Moreover, CUR’s anti-inflammatory qualities block pro-inflammatory cytokines, which contributes to a reduction in HIV-1-related problems even further.107,108
Toxic stress caused by the incompatibility of hard implants with soft brain tissue results in a neurodegenerative reactive tissue response. CUR-releasing softening polymer implants use a new concept of localized CUR delivery and also cause a minimal neuroinflammation provoked by the implant.105 Additional studies have shown that CUR nanoformulations significantly decreased lipid peroxidation and increased the enzyme and non-enzymatic antioxidants in brain.11,109–112 Poly(N-isopropyl acrylamide) and CUR nanoformulations were combined to provide a simple and direct nose-to-brain administration system for the medication.111,112 Gandapu et al used sol–oil technology to create apo-transferrin tagged CUR nanoparticles, which have a stronger anti-HIV activity than sol–CUR (IC(50) 1.75 M) because of transferrin-mediated endocytosis in T cells.58 This can also be attributable to a strong inhibition of HIV-1-induced production of Topo II, IL1, and COX-2, as well as preventing the synthesis of viral cDNA in the gag region.108 Rai et al also reported that binding of CUR with nanomaterials such as chitosan nanoparticles showed enhanced anti-HIV activity by potentiated obstruction of HIV-1 integrase, which is necessary for the replication of HIV virus.105 CUR nanoformulations had three times the anti-HIV activity over its free form, and blocked HIV-induced production of IL-1β, Topo II α, and cyclooxygenase-2 (COX-2), as well as stopping viral complementary DNA synthesis completely.113
Isolation Techniques for Curcumin
Soxhlet, ultrasonic, and microwave extraction methods are the most commonly used CUR isolation techniques.99 Heat is used to target minuscule traces of moisture in plant material in microwave aided extraction. When moisture is heated inside the plant cell by the microwave effect, it evaporates and creates great pressure on the cell wall, causing the plant cell to swell. The pressure presses the cell wall from within, stretching and eventually rupturing it, allowing the active constituents from the ruptured cells to leach out into the surrounding solvent and thereby increasing the yield of phytoconstituents. This phenomenon can be heightened even further if the plant matrix is treated with solvents that have a higher heating efficiency when exposed to microwaves.114 CUR is a thermo-labile compound that degrades after prolonged exposure to microwave radiation. A selective and rapid extraction of curcuminoids from turmeric into organic solvents using the microwave assisted extraction method has been reported, showing 60% extraction of curcuminoids with 75% purity within 1 minute, depending on the solvent used and microwave exposure time. Acetone optimized this process at a power level of 20%.115–117 Another study conducted by Najafpour (2016) showed the extraction of CUR from turmeric utilizing formic acid and microwave aided extraction. They reported 45.1 and 82.4 percentage purity of CUR, respectively.118 Although formic acid extraction was quick, its purity efficiency was lower than that of purified CUR extracted via microwave aided extraction. This was most likely due to the breakdown of CUR in formic acid. As a result, acetone has been advocated as a better extraction solvent than formic acid.117,119
Sono-capillarity and sono-poration can promote liquid penetration through channels created by bubble implosion and alter the permeability of cell membranes, respectively.119 Shirsath et al extracted 72% of CUR from Curcuma amada, a closely related species of C. longa known as “mango ginger”.120,121 A green ultrasound preparation technique was created, which proved to be more efficient than batch extraction and had a lower operating temperature than Soxhlet extraction. The acquired yield was much higher, with the added benefit of saving time. Ultrasound-assisted extraction can save energy and time, reduce extraction temperature and solvent amount, speed up energy transfer, enable selective extraction, and boost productivity.122,123 The graphic representation of extraction strategies in Figure 1 was prepared by the present reviewers. Conventional Soxhlet extraction is a traditional method for extracting lipids and other non-water-soluble compounds. Soxhlet can even store these compounds while retaining their characteristics. The disadvantages of the Soxhlet extraction technique are numerous, including the fact that it is time consuming, difficult, and requires a large number of organic solvents.124 Supercritical carbon dioxide, free of organic solvents, has been established in numerous countries for the extraction of CUR, oleoresin, and curcuminoids from Curcuma longa and Curcuma amada.125 There are also a few findings on enzyme-assisted extraction, in which pretreatment of turmeric with enzymes such as -amylase and glucoamylase resulted in a considerable increase in CUR output.126
Conclusions and Future Remarks
CUR has been used as a spice in various dishes and as traditional medicine for a long time. It has great medicinal value and is beneficial in multiple diseases. Here, its anti-HIV associated activities were discussed. Data obtained so far underline that CUR acts as an anti-HIV agent by inhibiting integrase, protease and topoisomerases, besides also exerting protective action against HIV-associated diseases, including cardiovascular, neurological and gastrointestinal disorders. The medicinal potential of CUR has just been recognized on a large scale. CUR was found to be a promising medication candidate when its physicochemical properties were modified using nanotechnology. Due to its pharmacokinetic properties, this natural component has already carved out a sizable chunk of the market. Because no substance is completely self-sufficient, there are disadvantages to be considered in this instance as well. There are still a lot of unanswered problems and issues that need to be addressed before it can be deemed an effective drug delivery mechanism. Despite the fact that we already know how nanocurcumin affects HIV, the human dosage is still at a standstill. Clinical trials in higher-order animals should cross-verify the findings of several conceptual preclinical studies. Because there are so few toxicology studies available, it is difficult to tell how effective the actual implementation is. Due to nanodrug toxicity causing DNA damage, allergic reactions, and neuroinflammation, it is necessary and recommended to do extensive and accurate investigations. As a result, the researchers must study if nano-CUR or free CUR can be employed alone or in combination formulations as supplementary medications in order to provide a conclusive report. Researchers should conduct a thorough investigation into the precise implications of nanocapsulated CUR for future therapeutic or chemotherapeutic treatments.
Acknowledgments
M.M. acknowledges ANID CENTROS BASALES ACE210012.
Disclosure
The authors report no conflicts of interest in this work.
References
1. UNAIDS. Global HIV and AIDS statistics; 2019. wwwavertorg.
2. WHO. The Top 10 Causes of Death. World Health Organization (WHO) Report; 2018.
3. World Health Organization. Global Health Observatory Data: HIV/AIDS. World Health Organization (WHO) Report; 2019.
4. Campbell-Yesufu OT, Gandhi RT. Update on human immunodeficiency virus (HIV)-2 infection. Clin Infect Dis. 2011;52(6):780–787. doi:10.1093/cid/ciq248
5. van Tienen C, van der Loeff MS. Epidemiology of HIV-2 Infection in West Africa. In: Hope TJ, Richman DD, Stevenson M, editors. Encyclopedia of AIDS. New York: Springer New York; 2018:513–523.
6. Lodi S, Phillips A, Touloumi G, et al. Time from human immunodeficiency virus seroconversion to reaching CD4+ cell count thresholds <200, <350, and <500 Cells/mm3: assessment of need following changes in treatment guidelines. Clin Infect Dis. 2011;53(8):817–825. doi:10.1093/cid/cir494
7. Hanson DL, Chu SY, Farizo KM, et al. Distribution of CD4+ T lymphocytes at diagnosis of acquired immunodeficiency syndrome-defining and other human immunodeficiency virus-related illnesses. The Adult and Adolescent Spectrum of HIV Disease Project Group. Arch Intern Med. 1995;155(14):1537–1542. doi:10.1001/archinte.1995.00430140115012
8. Poorolajal J, Hooshmand E, Mahjub H, et al. Survival rate of AIDS disease and mortality in HIV-infected patients: a meta-analysis. Public Health. 2016;139:3–12. doi:10.1016/j.puhe.2016.05.004
9. Le T, Wright EJ, Smith DM, et al. Enhanced CD4+ T-cell recovery with earlier HIV-1 antiretroviral therapy. N Engl J Med. 2013;368(3):218–230. doi:10.1056/NEJMoa1110187
10. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the international antiviral society-USA panel. JAMA. 2018;320(4):379–396. doi:10.1001/jama.2018.8431
11. Tatham LM, Rannard SP, Owen A. Nanoformulation strategies for the enhanced oral bioavailability of antiretroviral therapeutics. Ther Deliv. 2015;6(4):469–490. doi:10.4155/tde.15.4
12. Curley P, Liptrott NJ, Owen A. Advances in nanomedicine drug delivery applications for HIV therapy. Future Sci OA. 2017;4(1):FSO230–FSO30. doi:10.4155/fsoa-2017-0069
13. Farooqui T, Farooqui AA. Chapter 2 - curcumin: historical background, chemistry, pharmacological action, and potential therapeutic value. In: Farooqui T, Farooqui AA, editors. Curcumin for Neurological and Psychiatric Disorders. Academic Press; 2019:23–44.
14. Yeung AWK, Horbanczuk M, Tzvetkov NT, et al. Curcumin: total-scale analysis of the scientific literature. Molecules. 2019;24(7):1393. doi:10.3390/molecules24071393
15. Salehi B, Calina D, Docea AO, et al. Curcumin’s nanomedicine formulations for therapeutic application in neurological diseases. J Clin Med. 2020;9(2). doi:10.3390/jcm9020430
16. Trebatická J, Ďuračková Z. Psychiatric disorders and polyphenols: can they be helpful in therapy? Oxid Med Cell Longev. 2015;2015:248529. doi:10.1155/2015/248529
17. Salehi B, Del Prado-Audelo ML, Cortes H, et al. Therapeutic applications of curcumin nanomedicine formulations in cardiovascular diseases. J Clin Med. 2020;9(3):746. doi:10.3390/jcm9030746
18. Chandran B, Goel A. A randomized, pilot study to assess the efficacy and safety of curcumin in patients with active rheumatoid arthritis. Phytother Res. 2012;26(11):1719–1725. doi:10.1002/ptr.4639
19. Ali A, Banerjea AC. Curcumin inhibits HIV-1 by promoting Tat protein degradation. Sci Rep. 2016;6(1):27539–27639. doi:10.1038/srep27539
20. Jordan WC, Drew CR. Curcumin–a natural herb with anti-HIV activity. J Natl Med Assoc. 1996;88(6):333–433.
21. Siviero A, Gallo E, Maggini V, et al. Curcumin, a golden spice with a low bioavailability. J Herb Med. 2015;5(2):57–70. doi:10.1016/j.hermed.2015.03.001
22. Araújo CC, Leon LL. Biological activities of Curcuma longa L. Mem Inst Oswaldo Cruz. 2001;96(5):723–728. doi:10.1590/s0074-02762001000500026
23. Zhou Y, Xie M, Song Y, et al. Two traditional Chinese medicines curcumae radix and curcumae rhizoma: an ethnopharmacology, phytochemistry, and pharmacology review. Evid Based Complement Alternat Med. 2016;2016:4973128. doi:10.1155/2016/4973128
24. Lee W-H, Loo C-Y, Bebawy M, et al. Curcumin and its derivatives: their application in neuropharmacology and neuroscience in the 21st century. Curr Neuropharmacol. 2013;11(4):338–378. doi:10.2174/1570159X11311040002
25. Hayakawa H, Minaniya Y, Ito K, et al. Difference of curcumin content in Curcuma longa L. (Zingiberaceae) caused by Hybridization with other Curcuma species. Am J Plant Sci. 2011;02(02):111–119. doi:10.4236/ajps.2011.22013
26. Ammon HP, Wahl MA. Pharmacology of Curcuma longa. Planta Med. 1991;57(1):1–7. doi:10.1055/s-2006-960004
27. Zhou H, Beevers CS, Huang S. The targets of curcumin. Curr Drug Targets. 2011;12(3):332–347. doi:10.2174/138945011794815356
28. Maheshwari RK, Singh AK, Gaddipati J, et al. Multiple biological activities of curcumin: a short review. Life Sci. 2006;78(18):2081–2087. doi:10.1016/j.lfs.2005.12.007
29. Çıkrıkçı S, Mozio E. Biological activity of curcuminoids isolated from Curcuma longa. Rec Nat Prod. 2008;6:19–24.
30. Sharifi-Rad J, Rayess YE, Rizk AA, et al. Turmeric and its major compound curcumin on health: bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front Pharmacol. 2020;11:01021. doi:10.3389/fphar.2020.01021
31. Gupta P, Sharma A, Garg P, et al. QSAR study of curcumine derivatives as HIV-1 integrase inhibitors. Curr Comput Aided Drug Des. 2013;9(1):141–150. doi:10.2174/1573409911309010013
32. Niamsa N, Sittiwet C. Antimicrobial activity of Curcuma longa aqueous extract. J Pharmacol Toxicol. 2009;4(4):173–177. doi:10.3923/jpt.2009.173.177
33. De R, Kundu P, Swarnakar S, et al. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob Agents Chemother. 2009;53(4):1592–1597. doi:10.1128/aac.01242-08
34. Tyagi P, Singh M, Kumari H, et al. Bactericidal activity of curcumin I is associated with damaging of bacterial membrane. PLoS One. 2015;10(3):e0121313. doi:10.1371/journal.pone.0121313
35. Martins CV, da Silva DL, Neres AT, et al. Curcumin as a promising antifungal of clinical interest. J Antimicrob Chemother. 2009;63(2):337–339. doi:10.1093/jac/dkn488
36. Kumar A, Dhamgaye S, Maurya IK, et al. Curcumin targets cell wall integrity via calcineurin-mediated signaling in Candida albicans. Antimicrob Agents Chemother. 2014;58(1):167–175. doi:10.1128/AAC.01385-13
37. Peters BM, Shirtliff ME, Jabra-Rizk MA. Antimicrobial peptides: primeval molecules or future drugs? PLoS Pathog. 2010;6(10):e1001067–e67. doi:10.1371/journal.ppat.1001067
38. Mathew D, Hsu W-L. Antiviral potential of curcumin. J Funct Foods. 2018;40:692–699. doi:10.1016/j.jff.2017.12.017
39. Qin Y, Lin L, Chen Y, et al. Curcumin inhibits the replication of enterovirus 71 in vitro. Acta Pharm Sin B. 2014;4(4):284–294. doi:10.1016/j.apsb.2014.06.006
40. Flores DJ, Lee LH, Adams SD. Inhibition of curcumin-treated herpes simplex virus 1 and 2 in vero cells. Adv Microbiol. 2016;6(04):276–287. doi:10.4236/aim.2016.64027
41. Kim K, Kim KH, Kim HY, et al. Curcumin inhibits hepatitis C virus replication via suppressing the Akt-SREBP-1 pathway. FEBS Lett. 2010;584(4):707–712. doi:10.1016/j.febslet.2009.12.019
42. Lv Y, Lei N, Wang D, et al. Protective effect of curcumin against cytomegalovirus infection in Balb/c mice. Environ Toxicol Pharmacol. 2014;37(3):1140–1147. doi:10.1016/j.etap.2014.04.017
43. Mounce BC, Cesaro T, Carrau L, et al. Curcumin inhibits Zika and chikungunya virus infection by inhibiting cell binding. Antiviral Res. 2017;142:148–157. doi:10.1016/j.antiviral.2017.03.014
44. Baikerikar S. Curcumin and natural derivatives inhibit ebola viral proteins: an in silico approach. Pharmacognosy Res. 2017;9(Suppl 1):S15–S22. doi:10.4103/pr.pr_30_17
45. Chen C, Johnston TD, Jeon H, et al. An in vitro study of liposomal curcumin: stability, toxicity and biological activity in human lymphocytes and Epstein-Barr virus-transformed human B-cells. Int J Pharm. 2009;366(1–2):133–139. doi:10.1016/j.ijpharm.2008.09.009
46. Kumari N, Kulkarni AA, Lin X, et al. Inhibition of HIV-1 by curcumin A, a novel curcumin analog. Drug Des Devel Ther. 2015;9:5051–5060. doi:10.2147/dddt.S86558
47. Jin H, Qiao F, Wang Y, et al. Curcumin inhibits cell proliferation and induces apoptosis of human non-small cell lung cancer cells through the upregulation of miR-192-5p and suppression of PI3K/Akt signaling pathway. Oncol Rep. 2015;34(5):2782–2789. doi:10.3892/or.2015.4258
48. Du Q, Hu B, An HM, et al. Synergistic anticancer effects of curcumin and resveratrol in Hepa1-6 hepatocellular carcinoma cells. Oncol Rep. 2013;29(5):1851–1858. doi:10.3892/or.2013.2310
49. Marquardt JU, Gomez-Quiroz L, Arreguin Camacho LO, et al. Curcumin effectively inhibits oncogenic NF-κB signaling and restrains stemness features in liver cancer. J Hepatol. 2015;63(3):661–669. doi:10.1016/j.jhep.2015.04.018
50. Park J, Conteas CN. Anti-carcinogenic properties of curcumin on colorectal cancer. World J Gastrointest Oncol. 2010;2(4):169–176. doi:10.4251/wjgo.v2.i4.169
51. Youns M, Fathy GM. Upregulation of extrinsic apoptotic pathway in curcumin-mediated antiproliferative effect on human pancreatic carcinogenesis. J Cell Biochem. 2013;114(12):2654–2665. doi:10.1002/jcb.24612
52. Yu J, Peng Y, Wu LC, et al. Curcumin down-regulates DNA methyltransferase 1 and plays an anti-leukemic role in acute myeloid leukemia. PLoS One. 2013;8(2):e55934. doi:10.1371/journal.pone.0055934
53. Cao H, Yu H, Feng Y, et al. Curcumin inhibits prostate cancer by targeting PGK1 in the FOXD3/miR-143 axis. Cancer Chemother Pharmacol. 2017;79(5):985–994. doi:10.1007/s00280-017-3301-1
54. Liu H-T, Ho Y-S. Anticancer effect of curcumin on breast cancer and stem cells. Food Sci Hum Wellness. 2018;7(2):134–137. doi:10.1016/j.fshw.2018.06.001
55. Pourbagher-Shahri AM, Farkhondeh T, Ashrafizadeh M, et al. Curcumin and cardiovascular diseases: focus on cellular targets and cascades. Biomed Pharmacother. 2021;136:111214. doi:10.1016/j.biopha.2020.111214
56. Dong W, Yang B, Wang L, et al. Curcumin plays neuroprotective roles against traumatic brain injury partly via Nrf2 signaling. Toxicol Appl Pharmacol. 2018;346:28–36. doi:10.1016/j.taap.2018.03.020
57. Senapati S, Mahanta AK, Kumar S, et al. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3(1):7. doi:10.1038/s41392-017-0004-3
58. Gandapu U, Chaitanya RK, Kishore G, et al. Curcumin-loaded apotransferrin nanoparticles provide efficient cellular uptake and effectively inhibit HIV-1 replication in vitro. PLoS One. 2011;6(8):e23388. doi:10.1371/journal.pone.0023388
59. Qian ZM, Li H, Sun H, et al. Targeted drug delivery via the transferrin receptor-mediated endocytosis pathway. Pharmacol Rev. 2002;54(4):561–587. doi:10.1124/pr.54.4.561
60. Vajragupta O, Boonchoong P, Morris GM, et al. Active site binding modes of curcumin in HIV-1 protease and integrase. Bioorg Med Chem Lett. 2005;15(14):3364–3368. doi:10.1016/j.bmcl.2005.05.032
61. Mazumder A, Neamati N, Sunder S, et al. Curcumin analogs with altered potencies against HIV-1 integrase as probes for biochemical mechanisms of drug action. J Med Chem. 1997;40(19):3057–3063. doi:10.1021/jm970190x
62. Mazumder A, Raghavan K, Weinstein J, et al. Inhibition of human immunodeficiency virus type-1 integrase by curcumin. Biochem Pharmacol. 1995;49(8):1165–1170. doi:10.1016/0006-2952(95)98514-A
63. Karn J, Stoltzfus CM. Transcriptional and posttranscriptional regulation of HIV-1 gene expression. Cold Spring Harb Perspect Med. 2012;2(2):a006916–a16. doi:10.1101/cshperspect.a006916
64. Prasad S, Tyagi AK. Curcumin and its analogues: a potential natural compound against HIV infection and AIDS. Food Funct. 2015;6(11):3412–3419. doi:10.1039/c5fo00485c
65. Majhi A, Rahman GM, Panchal S, et al. Binding of curcumin and its long chain derivatives to the activator binding domain of novel protein kinase C. Bioorg Med Chem. 2010;18(4):1591–1598. doi:10.1016/j.bmc.2009.12.075
66. Dong HJ, Shang CZ, Peng DW, et al. Curcumin attenuates ischemia-like injury induced IL-1β elevation in brain microvascular endothelial cells via inhibiting MAPK pathways and nuclear factor-κB activation. Neurol Sci. 2014;35(9):1387–1392. doi:10.1007/s10072-014-1718-4
67. Smith JA, Daniel R. Following the path of the virus: the exploitation of host DNA repair mechanisms by retroviruses. ACS Chem Biol. 2006;1(4):217–226. doi:10.1021/cb600131q
68. Chai H, Yan S, Lin P, et al. Curcumin blocks HIV protease inhibitor ritonavir-induced vascular dysfunction in porcine coronary arteries. J Am Coll Surg. 2005;200(6):820–830. doi:10.1016/j.jamcollsurg.2005.02.030
69. Guo L, Xing Y, Pan R, et al. Curcumin protects microglia and primary rat cortical neurons against HIV-1 gp120-mediated inflammation and apoptosis. PLoS One. 2013;8(8):e70565. doi:10.1371/journal.pone.0070565
70. Yu Y, Shen Q, Lai Y, et al. Anti-inflammatory effects of curcumin in microglial cells. Front Pharmacol. 2018;9:386. doi:10.3389/fphar.2018.00386
71. Conteas CN, Panossian AM, Tran TT, et al. Treatment of HIV-associated diarrhea with curcumin. Dig Dis Sci. 2009;54(10):2188–2191. doi:10.1007/s10620-008-0597-z
72. Bibas M, Antinori A. EBV and HIV-related lymphoma. Mediterr J Hematol Infect Dis. 2009;1(2). doi:10.4084/MJHID.2009.032
73. Hayun R, Okun E, Berrebi A, et al. Rapamycin and curcumin induce apoptosis in primary resting B chronic lymphocytic leukemia cells. Leuk Lymphoma. 2009;50(4):625–632. doi:10.1080/10428190902789181
74. Fu Y-S, Chen T-H, Weng L, et al. Pharmacological properties and underlying mechanisms of curcumin and prospects in medicinal potential. Biomed Pharmacother. 2021;141:111888. doi:10.1016/j.biopha.2021.111888
75. Broder S. The development of antiretroviral therapy and its impact on the HIV-1/AIDS pandemic. Antiviral Res. 2010;85(1):1–18. doi:10.1016/j.antiviral.2009.10.002
76. Esté JA, Cihlar T. Current status and challenges of antiretroviral research and therapy. Antiviral Res. 2010;85(1):25–33. doi:10.1016/j.antiviral.2009.10.007
77. Moreno S, López Aldeguer J, Arribas JR, et al. The future of antiretroviral therapy: challenges and needs. J Antimicrob Chemother. 2010;65(5):827–835. doi:10.1093/jac/dkq061
78. Itokawa H, Shi Q, Akiyama T, et al. Recent advances in the investigation of curcuminoids. Chin Med. 2008;3:11. doi:10.1186/1749-8546-3-11
79. Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195–218. doi:10.1208/s12248-012-9432-8
80. D’Amours G, Langlois M, Mathonnet G, et al. SNP arrays: comparing diagnostic yields for four platforms in children with developmental delay. BMC Med Genomics. 2014;7(1):70. doi:10.1186/s12920-014-0070-0
81. Issuriya A, Kumarnsit E, Wattanapiromsakul C, et al. Histological studies of neuroprotective effects of Curcuma longa Linn. on neuronal loss induced by dexamethasone treatment in the rat hippocampus. Acta Histochem. 2014;116(8):1443–1453. doi:10.1016/j.acthis.2014.09.009
82. Guan WJ, Zheng XY, Zheng JP, et al. Small airway dysfunction in asymptomatic bronchial hyperresponsiveness and asthma. Allergy. 2014;69(9):1258–1259. doi:10.1111/all.12425
83. Jamil-Copley S, Bokan R, Kojodjojo P, et al. Noninvasive electrocardiographic mapping to guide ablation of outflow tract ventricular arrhythmias. Heart Rhythm. 2014;11(4):587–594. doi:10.1016/j.hrthm.2014.01.013
84. Tanaka R, Tsujii H, Yamada T, et al. Novel 3alpha-methoxyserrat-14-en-21beta-ol (PJ-1) and 3beta-methoxyserrat-14-en-21beta-ol (PJ-2)-curcumin, kojic acid, quercetin, and baicalein conjugates as HIV agents. Bioorg Med Chem. 2009;17(14):5238–5246. doi:10.1016/j.bmc.2009.05.049
85. Peters S. A second case with arrhythmogenic cardiomyopathy, provocable Brugada ECG and SCN5A mutation. Int J Cardiol. 2014;171(3):e117–8. doi:10.1016/j.ijcard.2013.12.038
86. Seal A, Aykkal R, Babu RO, et al. Docking study of HIV-1 reverse transcriptase with phytochemicals. Bioinformation. 2011;5(10):430–439. doi:10.6026/97320630005430
87. Cohly H, Asad S, Das S, et al. Effect of antioxidant (turmeric, turmerin and curcumin) on human immunodeficiency virus. Int J Mol Sci. 2003;4(2):22–33. doi:10.3390/i4020022
88. Braidot A, Hadad A. VI Latin American Congress on Biomedical Engineering CLAIB 2014, Paraná, Argentina 29, 30 & 31 october 2014. In: Biodegradable Polymeric Microparticles as Drug Delivery Devices. Cham: Springer International Publishing; 2015.
89. Rice KM, Manne NDPK, Kolli MB, et al. Curcumin nanoparticles attenuate cardiac remodeling due to pulmonary arterial hypertension. Artif Cells, Nanomed Biotechnol. 2016;44(8):1909–1916. doi:10.3109/21691401.2015.1111235
90. Kreuter J. Nanoparticles–a historical perspective. Int J Pharm. 2007;331(1):1–10. doi:10.1016/j.ijpharm.2006.10.021
91. Feynman RP. There’s plenty of room at the bottom. Resonance. 2011;16(9):890. doi:10.1007/s12045-011-0109-x
92. Khanna SC, Soliva M, Speiser P. Epoxy resin beads as a pharmaceutical dosage form. II. Dissolution studies of epoxy-amine beads and release of drug. J Pharm Sci. 1969;58(11):1385–1388. doi:10.1002/jps.2600581120
93. Müller RH, Maassen S, Weyhers H, et al. Phagocytic uptake and cytotoxicity of solid lipid nanoparticles (SLN) sterically stabilized with poloxamine 908 and poloxamer 407. J Drug Target. 1996;4(3):161–170. doi:10.3109/10611869609015973
94. Barreras-Urbina CG, Ramírez-Wong B, López-Ahumada GA, et al. Nano- and micro-particles by nanoprecipitation: possible application in the food and agricultural industries. Int J Food Prop. 2016;19(9):1912–1923. doi:10.1080/10942912.2015.1089279
95. Bozzuto G, Molinari A. Liposomes as nanomedical devices. Int J Nanomedicine. 2015;10:975–999. doi:10.2147/IJN.S68861
96. Bobo D, Robinson KJ, Islam J, et al. Nanoparticle-based medicines: a review of FDA-approved materials and clinical trials to date. Pharm Res. 2016;33(10):2373–2387. doi:10.1007/s11095-016-1958-5
97. Caster JM, Patel AN, Zhang T, et al. Investigational nanomedicines in 2016: a review of nanotherapeutics currently undergoing clinical trials. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017;9(1). doi:10.1002/wnan.1416
98. Patra JK, Das G, Fraceto LF, et al. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018;16(1):71. doi:10.1186/s12951-018-0392-8
99. Chopra H, Dey PS, Das D, et al. Curcumin nanoparticles as promising therapeutic agents for drug targets. Molecules. 2021;26(16):4998. doi:10.3390/molecules26164998
100. Wang S, Ha Y, Huang X, et al. A new strategy for intestinal drug delivery via pH-responsive and membrane-active nanogels. ACS Appl Mater Interfaces. 2018;10(43):36622–36627. doi:10.1021/acsami.8b15661
101. Cheng R, Meng F, Deng C, et al. Dual and multi-stimuli responsive polymeric nanoparticles for programmed site-specific drug delivery. Biomaterials. 2013;34(14):3647–3657. doi:10.1016/j.biomaterials.2013.01.084
102. Basniwal RK, Buttar HS, Buttar HS, et al. Curcumin nanoparticles: preparation, characterization, and antimicrobial study. J Agric Food Chem. 2011;59(5):2056–2061. doi:10.1021/jf104402t
103. Ganesh GN, Singh MK, Datri S, et al. Design and development of curcumin nanogel for squamous cell carcinoma. J Pharm Sci Res. 2019;11:1683.
104. Khosropanah MH, Dinarvand A, Nezhadhosseini A, et al. Analysis of the antiproliferative effects of curcumin and nanocurcumin in MDA-MB231 as a breast cancer cell line. Iran J Pharm Res. 2016;15:231.
105. Rai M, Ingle AP, Pandit R, et al. Curcumin and curcumin-loaded nanoparticles: antipathogenic and antiparasitic activities. Exp Rev Anti Inf Ther. 2020;18(4):367–379. doi:10.1080/14787210.2020.1730815
106. Sharma RK, Cwiklinski K, Aalinkeel R, et al. Immunomodulatory activities of curcumin-stabilized silver nanoparticles: efficacy as an antiretroviral therapeutic. Immunol Investig. 2017;46:833–846. doi:10.1080/08820139.2017.1371908
107. Keikha M, Ghazvini K. Can curcumin be used as an anti-HIV therapeutic option? Iran J Virol. 2018;12:34–37.
108. Schett G, Neurath MF. Resolution of chronic inflammatory disease: universal and tissue-specific concepts. Nat Commun. 2018;9:3261. doi:10.1038/s41467-018-05800-6
109. Potter KA, Jorfi M, Householder KT, et al. Curcumin-releasing mechanically adaptive intracortical implants improve the proximal neuronal density and blood–brain barrier stability. Acta Biomater. 2014;10(5):2209–2222. doi:10.1016/j.actbio.2014.01.018
110. Yallapu MM, Nagesh PK, Jaggi M, et al. Therapeutic applications of curcumin nanoformulations. AAPS J. 2015;17(6):1341–1356. doi:10.1208/s12248-015-9811-z
111. Sankar P, Telang AG, Kalaivanan R, et al. Oral nanoparticulate curcumin combating arsenicinduced oxidative damage in kidney and brain of rats. Toxicol Ind Health. 2013;32:410–421. doi:10.1177/0748233713498455
112. Ahmad N, Warsi MH, Iqbal Z, et al. Quantification of curcumin, demethoxycurcumin, and bisdemethoxycurcumin in rodent brain by UHPLC/ESI-Q-TOF-MS/MS after intra-nasal administration of curcuminoids loaded PNIPAM nanoparticles. Drug Test Anal. 2014;6(3):257–267. doi:10.1002/dta.1472
113. Karthikeyan A, Senthil N, Min T. Nanocurcumin: a promising candidate for therapeutic applications. Front Pharmacol. 2020;11:487. doi:10.3389/fphar.2020.00487
114. Priyadarsini KI. The chemistry of curcumin: from extraction to therapeutic agent. Molecules. 2014;19(12):20091–20112. doi:10.3390/molecules191220091
115. Li M, Ngadi MO, Ma Y. Optimisation of pulsed ultrasonic and microwave-assisted extraction for curcuminoids by response surface methodology and kinetic study. Food Chem. 2014;165:29–34. doi:10.1016/j.foodchem.2014.03.115
116. Wang L, Weller CL. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci Technol. 2006;17(6):300–312. doi:10.1016/j.tifs.2005.12.004
117. Hmar BZ, Kalita D, Srivastava B. Optimization of microwave power and curing time of turmeric rhizome (Curcuma longa L.) based on textural degradation. LWT. 2017;76:48–56. doi:10.1016/j.lwt.2016.10.044
118. Najafpour G. Formic acid and microwave assisted extraction of curcumin from turmeric (Curcuma longa L.). Int J Eng. 2016;29:145–151.
119. Chemat F, Rombaut N, Sicaire AG, et al. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason Sonochem. 2017;34:540–560. doi:10.1016/j.ultsonch.2016.06.035
120. Shirsath S, Sable S, Gaikwad S, et al. Intensification of extraction of curcumin from Curcuma amada using ultrasound assisted approach: effect of different operating parameters. Ultrason Sonochem. 2017;38:437–445. doi:10.1016/j.ultsonch.2017.03.040
121. Jatoi SA, Kikuch A, Gilani SA, et al. Phytochemical, pharmacological and ethnobotanical studies in Mango ginger (Curcuma amada Roxb.; Zingiberaceae). Phytother Res. 2007;21(6):507–516. doi:10.1002/ptr.2137
122. Mannaï A, Jableoui C, Hamrouni L, et al. DIC as a pretreatment prior to ultrasonic extraction for the improvement of rebaudioside A yield and preservation of vitamin B1 and B6. J Food Meas Charact. 2019;13(4):2764–2772. doi:10.1007/s11694-019-00197-2
123. Serpa Guerra AM, Cock JA, Vuerra AM. G yield and preservation of vitamin B1 and B6. as a pretreatment priEffect of ultra-fine friction grinding on the physical and chemical properties of curcuma (Curcuma longa L.) suspensions. J Food Sci. 2020;85:132–142. doi:10.1111/1750-3841.14973
124. Kiamahalleh MV, Najafpour-Darzi G, Rahimnejad M, et al. High performance curcumin subcritical water extraction from turmeric (Curcuma longa L.). J Chromatogr B. 2016;1022:191–198. doi:10.1016/j.jchromb.2016.04.021
125. Lv GP, Hu DJ, Zhou YQ, et al. Preparation and application of standardized typical volatile components fraction from turmeric (Curcuma longa L.) by supercritical fluid extraction and step molecular distillation. Molecules. 2018;23:1831. doi:10.3390/molecules23071831
126. Kurmudle N, Kagliwal LD, Bankar S, et al. Enzyme-assisted extraction for enhanced yields of turmeric oleoresin and its constituents. Food Biosci. 2013;3:36–41. doi:10.1016/j.fbio.2013.06.001
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
