Back to Journals » Clinical Ophthalmology » Volume 11
Bilateral implantation of +3.0 D multifocal toric intraocular lenses: results of a US Food and Drug Administration clinical trial
Authors Lehmann R , Modi S , Fisher B, Michna M, Snyder M
Received 18 March 2017
Accepted for publication 30 May 2017
Published 20 July 2017 Volume 2017:11 Pages 1321—1331
DOI https://doi.org/10.2147/OPTH.S137413
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Robert Lehmann,1 Satish Modi,2 Bret Fisher,3 Magda Michna,4 Michael Snyder5
1Lehmann Eye Center, Nacogdoches, TX, 2Seeta Eye Center, Poughkeepsie, NY, 3The Eye Center of North Florida, Panama City, FL, 4Department of Clinical Trial Management, Alcon Laboratories, Inc., Fort Worth, TX, 5Cincinnati Eye Institute, Cincinnati, OH, USA
Purpose: The purpose of this study was to evaluate the clinical outcomes of apodized diffractive +3.0 D multifocal toric intraocular lens (IOL) implantations in subjects with preoperative corneal astigmatism.
Patients and methods: This was a prospective cohort study conducted at 21 US sites. The study population consisted of 574 subjects, aged ≥21 years, with preoperative astigmatism 0.75–2.82 D, and potential postoperative visual acuity (VA) ≥0.2 logMAR, undergoing bilateral cataract removal by phacoemulsification. The intervention was bilateral implantation of aspheric apodized diffractive +3.0 D multifocal toric or spherical multifocal nontoric IOLs. The main outcome measures were monocular uncorrected near and distance VA and safety at 12 months.
Results: A total of 373/386 and 182/188 subjects implanted with multifocal toric and nontoric IOLs, respectively, completed 12-month follow-up after the second implantation. Toric IOLs were noninferior in monocular uncorrected distance (4 m) and near (40 cm) VA but had >1 line better binocular uncorrected intermediate VA (50, 60, and 70 cm) than nontoric IOLs. Toric IOLs reduced cylinder to within 0.50 D and 1.0 D of target in 278 (74.5%) and 351 (94.1%) subjects, respectively. Mean ± standard deviation (SD) differences between intended and achieved axis orientation in the first and second implanted eyes were 5.0°±6.1° and 4.7°±4.0°, respectively. Mean ± SD 12-month IOL rotations in the first and second implanted eyes were 2.7°±5.8° and 2.2°±2.7°, respectively. No subject receiving toric IOLs required secondary surgical intervention due to optical lens properties.
Conclusion: Multifocal toric IOLs were noninferior to multifocal nontoric IOLs in uncorrected distance and near VAs in subjects with preexisting corneal astigmatism and effectively corrected astigmatism of 0.75–2.82 D.
Keywords: AcrySof® IQ ReSTOR, corneal astigmatism, phacoemulsification, target cylinder, axis orientation, IOL rotation lens, visual acuity
Introduction
Approximately 40% of subjects who undergo cataract surgery have corneal astigmatism ≥0.75 D, which can significantly limit optimal visual outcomes if left uncorrected.1,2 Toric intraocular lenses (IOLs) can correct astigmatism as well as aphakia, often resulting in freedom from spectacles at distance for astigmatic subjects. Toric IOLs are designed to eliminate the need for additional optical correction or surgical procedures to correct for preexisting corneal astigmatism. The correction of preoperative corneal astigmatism with a monofocal toric IOL has been shown to provide better visual outcomes than monofocal nontoric IOL for astigmatic patients.3–5
Multifocal IOLs are designed with refractive and/or diffractive optical properties that divide light into multiple focal points, providing subjects with simultaneous distance, intermediate, and near vision. Randomized clinical trials6–11 and the most recent Cochrane systematic review12 have concluded that multifocal IOLs improve uncorrected near vision relative to the outcomes with monofocal IOLs. In clinical practice, binocular implantation of multifocal IOLs has been shown to result in improved stereopsis and patient satisfaction scores when compared with monocular implantation after 6 months.13 Furthermore, a systematic review of the clinical outcomes of bilateral implantation of multifocal IOLs demonstrated that implantation of either refractive or diffractive multifocal IOLs resulted in good visual acuity (VA) outcomes and increased levels of spectacle independence compared with monofocal IOLs, although multifocal IOLs were also associated with higher rates of halos and glare.14
The AcrySof® IQ ReSTOR® (Alcon Research, Ltd., Fort Worth, TX, USA) apodized diffractive multifocal toric IOL has the potential to provide increased range of vision from near through intermediate to distance and to correct preoperative corneal astigmatism from 0.75 to 2.82 D. Thus, these lenses may meet astigmatic patients’ expectations of reduced need for additional refractive correction and enhanced visual function following cataract surgery.
This study presents the results of a registration trial of AcrySof IQ ReSTOR Multifocal Toric IOL Models SND1T3-SND1T6 submitted to the U.S. Food and Drug Administration (ClinicalTrials.gov identifier, NCT01424189). The objectives of this study were to compare visual and safety outcomes following bilateral implantation of AcrySof IQ ReSTOR Multifocal Toric IOL Models SND1T3-SND1T6 (multifocal toric IOLs) versus the nontoric AcrySof ReSTOR Multifocal IOL Model SN60D3 (hereafter called control multifocal nontoric IOL) in subjects with preoperative astigmatism of 0.75–2.82 D. The null hypothesis was that, at 12 months following second eye surgery, the differences in mean monocular uncorrected distance VA (UCDVA) and mean monocular uncorrected near VA (UCNVA) at fixed distance between the multifocal toric IOL and multifocal nontoric IOL would be less than a clinical performance target of 0.1 logMAR.
Methods
Study design
This prospective, nonrandomized, unmasked, parallel-group, multicenter study followed subjects bilaterally implanted with either multifocal toric or control multifocal nontoric IOLs. Follow-up continued for 12 months after implantation in the second eye. The study was conducted between July 2011 and January 2013 at 21 sites in the US, and it was managed in accordance with the ethical principles of the Declaration of Helsinki and in compliance with Good Clinical Practice, ISO 14155:2011. Before study initiation, the protocol and the informed consent form were approved by a centralized institutional review board (RCRC Institutional Review Board, Austin, TX, USA). Written informed consent was obtained from each subject (and/or legal representative, as applicable) prior to screening for this study.
Participants
Eligible subjects were included if they were scheduled for removal of bilateral cataracts by phacoemulsification; were at least age 21 years at the time of surgery; had astigmatism ≤2.82 D in both eyes, potential postoperative VA of ≥0.2 logMAR in both eyes, preoperative corrected distance VA <0.2 logMAR in each eye, and a pupil size ≥6 mm after dilation; were able to undergo the second eye surgery within 30 days of the first; and voluntarily consented to participate. Corneal astigmatism was measured using the IOLMaster® optical biometry instrument (Carl Zeiss Meditec, Inc., Dublin, CA, USA). Subjects with preoperative corneal astigmatism <0.75 D in both eyes were required to receive control multifocal nontoric IOLs, whereas subjects with preoperative corneal astigmatism ≥0.75 D and 0.75 D to 2.82 D of predicted crossed cylinder in both eyes were required to receive multifocal toric IOLs. Target cylinder was defined as the amount of anticipated residual astigmatism, as calculated with a lens-specific, web-based multifocal toric clinical calculator (www.acrysoftoriccalculator.com).
Subjects were excluded from the study if they had significant irregular corneal aberrations, as demonstrated by corneal topography; any corneal abnormality other than regular corneal astigmatism; any inflammation or edema of the cornea; or a degenerative visual disorder predicted to cause future acuity loss to <0.2 logMAR. Subjects who may have reasonably been expected to require a secondary surgical intervention (SSI) (other than YAG capsulotomy) at any time during the study as well as those who had previously undergone corneal refractive surgery were also excluded. Subjects were also excluded if they had amblyopia, clinically severe corneal dystrophy, diabetic retinopathy, extremely shallow anterior chamber not due to swollen cataract, microphthalmos, previous retinal detachment or corneal transplant, recurrent severe anterior or posterior segment inflammation of unknown etiology, rubella or traumatic cataract, iris neovascularization, glaucoma, aniridia, optic nerve atrophy, or pregnancy. During surgery, subjects were excluded from the study if any of the following exclusion criteria was met: any incision site other than temporal (±15° from the horizontal meridian); mechanical or surgical manipulation required to enlarge the pupil (pupils were required to be at least 4.5 mm in diameter just prior to IOL implantation); significant vitreous loss; significant anterior chamber hyphema; uncontrollable intraocular pressure (IOP); zonular or capsular rupture; and bag-sulcus, sulcus-sulcus, or unknown placement of the haptics.
Intraocular lenses
The multifocal toric IOL models SND1T3, SND1T4, SND1T5, and SND1T6 are apodized, diffractive, posterior chamber IOLs with ultraviolet and blue light filtering properties intended for primary implantation for the visual correction of aphakia and preexisting corneal astigmatism in adult subjects with and without presbyopia. The lens design incorporates an aspheric anterior surface to compensate for the spherical aberration of the eye and a posterior toric optical component to correct preexisting corneal astigmatism. The recommended corneal astigmatism correction range is 0.75–2.82 D, and the range of cylinder power in the IOL plane is 1.50 D–3.75 D. The near add power at the IOL plane is +3.0 D, providing a near focal distance at 40 cm.
The posterior surface of the optic of the multifocal toric IOL is marked with six indentations (three on each side), which were aligned with the postincision steep meridian of the corneal astigmatism to provide optimal astigmatism correction, as recommended by the multifocal toric calculator.
The control nontoric multifocal IOL, model SA60D3, is another type of apodized, diffractive posterior chamber IOL with ultraviolet and blue light filtering properties (Alcon Laboratories, Inc., Fort Worth, TX) that has been approved for use by the US Food and Drug Administration. It has no toric component to correct preexisting corneal astigmatism and does not incorporate asphericity to compensate for corneal spherical aberration. The ADD power at the IOL plane is +4.0 D, providing a near reading distance at 33 cm.
Preoperative evaluation
The preoperative visit included a thorough ocular and nonocular medical history and the following examinations: VA, manifest refraction, corneal topography, anterior chamber depth and axial length, IOL power (IOLMaster, Carl Zeiss Meditec, Inc. or Holladay IOL Consultant Software, Houston, TX, USA), multifocal toric IOL cylinder power and axis calculation (using the web-based multifocal toric IOL clinical calculator with a fixed surgically induced astigmatism of 0.0 D and a temporal incision location), automated keratometry, IOP, and slit-lamp and dilated fundus evaluations.
Surgical technique
Cataract surgeries were performed by experienced surgeons using a standard sutureless phacoemulsification. The incision for phacoemulsification and insertion of the lens was temporal for all eyes: 0° for the left eye and 180° for the right eye (±15° or ~1 mm). Surgery proceeded using either longitudinal (traditional) or torsional phacoemulsification. The capsulorrhexis was 5.0–5.5 mm to attain adequate IOL stabilization by overlapping the lens optic by 360°. Surgeons used only the Monarch® IOL delivery system (Alcon Laboratories), selecting an injector–cartridge combination that was qualified for the lens model and lens power being implanted.
Manual preoperative marks were made to identify the IOL axis orientation required for the implantation of multifocal toric IOL. Once the multifocal toric IOL was implanted and aligned with the intended axis, the ophthalmic viscosurgical device was carefully removed, such that the lens was positioned correctly, and the wound was closed. No axis alignment was necessary for the control multifocal nontoric IOL. Both toric and control nontoric multifocal IOLs were centered on the optical axis of the eye at the end of surgery.
Postoperative evaluations
Subjects were evaluated on the day of both surgeries, and ~1 day, 1 week, and 1 month after implantation in the first eye and 1 day, 1 week, 1 month, 6 months, and 12 months after implantation in the second eye. Lens implantation in the second eye typically occurred 7–30 days after implantation in the first eye. The primary effectiveness objective was to demonstrate that the difference in VA outcomes between the multifocal toric and control multifocal nontoric IOLs was less than a prespecified clinical performance target of 0.1 logMAR units. The VA outcome measures included mean monocular photopic UCDVA (4 m) and mean monocular photopic UCNVA at fixed distance (40 cm for the multifocal toric and 33 cm for the multifocal nontoric IOLs) in the first operative eye 12 months after the second eye implantation. UCDVA and UCNVA were tested using 100% contrast Early Treatment of Diabetic Retinopathy Study (ETDRS) charts (Precision Vision, La Salle, IL, USA).
Supportive effectiveness outcomes included intermediate VA, correction of corneal astigmatism represented as the mean percent reduction in cylinder relative to target (multifocal toric IOLs only), orientation of lens axis (multifocal toric IOLs only), and contrast sensitivity using the Vector Vision CSV-1000E contrast sensitivity test (Vector Vision, Greenville, OH, USA).
Lens axis orientation was assessed using a clinically validated custom system, Photographic Assessment of Lens Orientation, which allowed the operator to select the toric lens markers and anatomical landmarks on the eye and use their coordinate locations to quantitatively measure the axis of orientation of the implanted IOL. The cylindrical component of the multifocal toric IOL required its precise placement within the capsular bag. The flat meridian (identified with indentations on the posterior surface of the optic) of the IOL was aligned with the postincision steep meridian of the corneal astigmatism to provide optimal vision correction. The orientation of the lens axis for the multifocal toric IOL was measured at the operative visit and at each postoperative visit. Operative visit results were compared with the intended axis orientation, which was calculated using preoperative biometry measurements and the study-specific web-based multifocal toric clinical calculator. In addition, the results at each postoperative visit were compared with the operative visit results to determine rotational stability.
The primary safety outcome was the rate of actual and potential SSIs related to the optical properties of the IOLs, reported separately for the first and second operated eyes at 12 months after the second eye implantation. The rationale for assessing both types of SSIs was that factors unrelated to the criteria of interest could contribute to whether a subject actually chose, or was able, to undergo additional surgery. The secondary safety objective was to estimate the rate of severe visual disturbances/distortions, as reported by subjects using a patient-reported outcomes questionnaire (Assessment of Photic Phenomena and Lens Effects) at 12 months.15
The other safety evaluations included adverse events (including SSIs), slit-lamp and dilated fundus examination findings, subjective posterior capsular opacification, posterior capsulotomy, IOL position changes, device deficiencies, and IOP. In addition, IOL observations were evaluated, including glistenings, debris on the lens surface, forceps marks and the lens surface, and other postoperative observations.
Statistical methods
The primary driver of sample size for this study was the precision of the confidence interval (CI) on the rate of actual or potential SSIs related to the optical properties of the IOL. The study was designed such that the event rate in the toric IOL group could be estimated to as low as ~1% with 95% CI. If 0 of 300 subjects reported any event, the bound of a one-sided 95% CI would be ~1%. A minimum of 340 subjects was required to undergo bilateral implantation of toric IOLs to ensure that at least 300 eligible subjects completed the study. This assumed a dropout rate of 10%. In the control IOL group, a minimum of 170 subjects was required to undergo bilateral implantation.
Effectiveness analyses were performed on the All Implanted and Best Case datasets. All subjects with successful IOL implantation in at least one eye were considered evaluable for the All Implanted analyses. All eyes successfully implanted with at least one postoperative visit and no preoperative ocular pathology or macular degeneration at any time were evaluable for Best Case analyses. The Best Case dataset was the primary dataset of analysis for contrast sensitivity and binocular defocus. All eyes with attempted IOL implantation (whether successful or aborted after contact with the eye) were considered evaluable for safety.
The difference in means between multifocal toric and multifocal nontoric IOLs and the corresponding one-sided upper 95% confidence limit for the difference were estimated and the upper limit was compared to a clinical performance target of 0.1 logMAR unit. The effectiveness null hypothesis was rejected if the one-sided upper 95% confidence limit between the two IOLs was <0.1 logMAR unit. Age and spherical equivalent lens power (in diopters) were included as covariates in the statistical model. The primary comparison included all models of the multifocal toric IOLs combined. Analysis of the secondary effectiveness outcomes included descriptive statistics by lens group (except for reduction of cylinder, intended lens placement, and change in axis orientation, which are presented only for the multifocal toric IOL).
The evaluation of contrast sensitivity assumed that at least 90% of all subjects undergoing implantation would qualify for the “Best Case” dataset. With 276 and 138 Best Case subjects in the toric and nontoric multifocal IOL groups, respectively, and a clinical performance target of −0.15 log units, there was 89% power to demonstrate meeting the clinical performance target for contrast sensitivity at month 12, assuming no difference between groups in contrast sensitivity. Reducing the number of Best Case subjects in the toric and nontoric multifocal IOL groups to 210 and 105, respectively, would have resulted in an ~80% likelihood of meeting the clinical performance target, assuming no difference between groups in contrast sensitivity. These estimates were based on an assumed standard deviation for contrast sensitivity of 0.50 log units and a one-sided, α=0.05 test. The difference used is the mean contrast sensitivity for the toric minus the nontoric multifocal group.
Results
Subject disposition
Of the 677 subjects enrolled in the study, 103 did not undergo implantation as they failed screening, 386 were implanted with multifocal toric IOLs and 188 with control multifocal nontoric IOL (Figure 1). All but four of these subjects were implanted bilaterally: one was lost to follow-up, one became pregnant, one withdrew consent, and one had a capsulorrhexis tear during the second eye surgery, aborting implantation of the IOL. Two subjects, both implanted with the control nontoric IOL, underwent bilateral IOL explantation following the 6-month visit due to intolerance of visual disturbances/distortions. Effectiveness data recorded for these two subjects after explantation were excluded from analyses, but they were followed for safety. Altogether, 13 subjects implanted with multifocal toric IOLs and six implanted with multifocal nontoric IOLs discontinued. At 12 months, 373/386 (96.6%) subjects implanted with multifocal toric IOLs and 182/188 (96.8%) implanted with the control multifocal nontoric IOLs had completed the study.
  | Figure 1 Subject disposition flowchart (first implanted eye). |
Most subjects were white (93.7%), female (65.5%), and at least 60 years of age (Table 1). Preoperative corrected distance VA, axial length, and anterior chamber depth were similar in the two groups (Table 2).
  | Table 1 Demographic characteristics |
  | Table 2 Baseline clinical characteristics |
Visual acuity
The multifocal toric IOLs compared favorably with the control multifocal nontoric IOLs on both of the coprimary effectiveness outcomes, mean monocular UCDVA (4 m) and mean monocular UCNVA at fixed distance (40 cm for the multifocal toric IOL and 33 cm for the multifocal nontoric IOL) for the first operative eye at 12 months. For both UCDVA and UCNVA, the 95% upper confidence limit for the difference in means was less than the prespecified clinical performance target margin of 0.1 logMAR (Table 3).
Mean monocular and binocular distance-corrected near visual acuities (DCNVAs), at fixed distances of 40 cm for the multifocal toric IOL and 33 cm for the multifocal nontoric IOL, were similar in the two groups at all study visits (data not shown). The mean best distances for binocular DCNVA were ~1 cm greater than the mean best distances for binocular UCNVA at 12 months in both the multifocal toric (38 and 37 cm, respectively) and multifocal nontoric (32 and 31 cm, respectively) IOLs, indicating that any residual refractive error did not affect the mean best distances for both types of lens.
If any data were missing for the coprimary effectiveness outcomes, a sensitivity analysis was performed using two imputation methods (imputing the missing data point[s] with the median value of all postoperative visits for the subject, and imputing the missing data point[s] for a test group subject with the worst postoperative value from all test group subjects and the missing data point[s] for a control group subject with the best postoperative value from all control group subjects). The results of these analyses indicated that the conclusions for the primary effectiveness outcomes were not strongly affected by missing data.
Clinically relevant differences favoring the multifocal toric IOL were observed for mean uncorrected intermediate VA and for distance-corrected intermediate VA at all testing distances (50, 60, and 70 cm), due to the difference in near ADD power between the multifocal toric and multifocal nontoric IOLs (Table 4). The multifocal toric and control multifocal nontoric IOLs provided simultaneous visual performance for near and distance.
Correction of astigmatism
Subjects implanted with multifocal toric IOLs demonstrated a mean percent reduction in cylinder power relative to target cylinder power of ≥76.6% in both the first and second implanted eyes at all postoperative visits (Table 5). In addition, 278/373 (74.5%) subjects implanted with multifocal toric IOLs achieved a reduction in cylinder within 0.5 D of the target cylinder in the first implanted eye at 12 months, and 295/371 (79.5%) achieved this level of reduction in the second eye. Furthermore, 351/373 (94.1%) and 362/371 (97.6%) subjects achieved a reduction in cylinder within 1.0 D of the target cylinder in the first and second implanted eyes, respectively, at 12 months.
  | Table 5 Percent reduction in cylinder relative to target cylinder over time |
Orientation of lens axis
The orientation of the cylinder axis was measured upon completion of the surgical procedure for 362 subjects. In the first implanted eyes, the mean (standard deviation [SD]) absolute difference between intended and achieved axis orientation was 5.0°±6.1° (95% CI, 4.3°, 5.6°). In the second implanted eyes, the mean (SD) absolute difference was 4.7°±4.0° (95% CI, 4.2°, 5.1°).
The mean (SD) absolute IOL axis rotation at 12 months was 2.7°±5.8° (95% CI, 2.1°, 3.3°) in the first implanted eyes and 2.2°±2.7° (95% CI, 1.8°, 2.5°) in the second implanted eyes. Moreover, at all postoperative visits, the mean actual rotation was ≤1.0°±6.3° in the first and second operative eyes.
Between the operative visit and 12 months, 346/356 (97.2%) first eyes and 350/367 (98.0%) second eyes implanted with the multifocal toric IOL demonstrated absolute lens rotation of <10° (Figure 2).
  | Figure 2 Percentages of eyes showing ≥10 degrees of misalignment of multifocal toric intraocular lenses over time. |
Contrast sensitivity
No clinically relevant differences of ≥0.15 log units (ISO 11979-9: 2006) in binocular contrast sensitivity were observed between the multifocal toric and control multifocal nontoric IOLs, under either photopic or mesopic conditions and with or without a glare source (data not shown).
Safety
No subject implanted with the multifocal toric IOL experienced an actual SSI related to the optical properties of the IOL. Three subjects in the multifocal toric IOL group required lens repositioning surgery because their lenses were originally implanted at the wrong axis. Two subjects in the multifocal nontoric group experienced an actual SSI in both the first and second operative eyes; these IOLs were explanted following the 6-month visit due to intolerance of visual disturbances/distortions.
Potential SSIs were reported for four subjects implanted with the multifocal toric IOL (Table 6). One reported blurry vision in her right eye 4 months after lens implantation in that eye (first implantation). She underwent astigmatic keratotomy 6 months after initial surgery and laser-assisted in situ keratomileusis (LASIK) on her right eye 11 months after initial surgery. There were no clinically significant findings on any slit-lamp examination or dilated fundus examination, but the subject continued to report blurry vision at the end of the study. The second subject had residual astigmatism in the first implanted eye at 12 months (manifest refraction −0.75 +0.75×021). The investigator recommended that photorefractive keratectomy be performed if the subject was dissatisfied with his vision, but at the conclusion of the study, the subject had not yet scheduled the procedure. The third subject reported visual disturbances related to the optical properties of the IOL in the first and second operative eyes at 12 months. The investigator noted an inaccurate preoperative measurement for the axial length and recommended a LASIK procedure; however, the subject decided to postpone the treatment. The fourth subject reported potential SSIs for both the first and second eyes at 6 months because of complaints about visual disturbances.
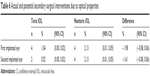  | Table 6 Actual and potential secondary surgical interventions due to optical properties |
Two subjects in the control multifocal nontoric IOL group reported potential SSIs related to visual disturbances. Reports of visual disturbances/distortions at 12 months, as measured using a patient-reported outcomes questionnaire, were similar for multifocal toric and multifocal nontoric IOLs (Table 7). The highest rates of severe visual disturbances/distortions at 12 months were for halos, reported by 28/372 (7.5%) of subjects in the multifocal toric IOL group and 20/182 (11.0%) subjects in the control multifocal nontoric IOL group.
Rates of IOL observations in the two groups were low. The cumulative rates of IOL observations in the multifocal toric group were 8.6% (32/373) in the first implanted eye and 6.7% (25/372) in the second implanted eye; in the multifocal nontoric group, these rates were 8.2% (15/182) and 7.7% (14/182), respectively. At 1 year, glistenings were observed in only 4.3% (16/373) of first implanted eyes and 4.0% (14/372) of second implanted eyes in the multifocal toric group, and in 2.7% (5/182) and 2.7% (5/182) of the first and second eyes, respectively, in the multifocal nontoric group. Moreover, none of the observed glistenings was reported to be clinically significant by the implanting surgeons.
Table 8 lists the persistent ocular serious adverse events that were present or ongoing at 12 months. The incidence of serious adverse events in each IOL group was not statistically significantly different from that reported for posterior chamber IOLs, except for SSIs. Overall, no untoward safety issues were identified for subjects implanted with the multifocal toric or nontoric IOLs. No unanticipated serious adverse device effects were reported.
  | Table 8 Serious and persistent adverse events |
Overall satisfaction
When questioned about their overall satisfaction with their vision without eyeglasses or contact lenses prior to surgery, 369/385 (95.8%) and 173/188 (92.0%) subjects implanted with multifocal toric and nontoric IOLs, respectively, reported being dissatisfied or very dissatisfied with their vision overall (Figure 3). However, 1 year after implantation, 321/371 (86.5%) and 157/180 (87.2%) subjects, respectively, reported that they were satisfied or very satisfied with their vision. When asked 1 year after implantation if they would choose the same lenses, 338/370 (91.4%) of subjects implanted with multifocal toric lenses and 163/180 (90.6%) of those implanted with multifocal nontoric lenses answered affirmatively.
  | Figure 3 Subject rating of satisfaction with overall vision without eyeglasses or contact lenses before and 1 year after intraocular lens implantation. |
Discussion
This prospective, multicenter, 12-month follow-up study showed that the multifocal toric IOL models SND1T3–SND1T6 met the clinical performance target (0.1 logMAR unit) when compared with the multifocal nontoric IOL for UCDVA (4 m) and UCNVA at fixed distance, 40 cm for the multifocal toric, and 33 cm for the multifocal nontoric IOL. Overall, implantation of multifocal toric IOLs did not introduce any new safety concerns, as determined by the rate of SSIs (actual or potential), when compared with the rate observed for current multifocal nontoric IOLs. In addition, no clinically significant glistenings were observed for either lens types.
The results of the present study confirm and extend the findings from other clinical trials of toric IOLs. A prospective, multicenter cohort study (n=49) showed significant reductions in spherical equivalent and refractive cylinder 6 months after bilateral implantation of multifocal toric IOLs (models SND1T3–SND1T5).16 The corneal cylinder remained relatively unchanged, indicating that the reduction in astigmatism can be attributed to the IOL and not to changes in the cornea. Moreover, the corneal and refractive cylinders and their spherical equivalents remained stable across postoperative visits. Reduction of cylinder in a randomized, subject-masked, 1-year study of the monofocal toric IOL model SN60T3 provided significantly better visual outcomes than monofocal nontoric IOLs.5 Similarly, other studies showed that correction of low amounts of astigmatism with the toric IOLs was associated with significantly better visual outcomes than implantation of the nontoric IOLs,3,4 indicating that correction of astigmatism by the multifocal toric IOLs may improve visual outcomes for astigmatic cataract patients who desire near, intermediate, and distance vision.
Other objective findings from a 6-month study of the multifocal toric IOL included good VA at near, intermediate, and far distances; increased reading speed; and minimal IOL rotation.16 At 6 months postoperatively, subjects reported statistically significant improvements in their ability to perform tasks at a variety of distances.17 Their rating of uncorrected vision and their overall satisfaction were also significantly better than at baseline, and most subjects (90%) were spectacle free.17
Similar results with the multifocal toric IOL were obtained in a 3-month prospective cohort study (n=19)18 and in a retrospective study with ~3 months of postoperative follow-up (44 subjects, 70 eyes).19 In the prospective study, multifocal toric IOLs provided excellent distance, intermediate, and near visual outcomes; predictability of refractive results; and rotational stability. The UCDVA was ≥0.3 logMAR (Snellen equivalent ≥20/40) in 97% of eyes, and all eyes achieved complete spectacle independence.17 Visual outcomes were even better in the retrospective study: 100% of eyes achieved UCDVA ≥0.18 logMAR (20/30 Snellen equivalent) and 100% achieved UCNVA ≥0.3 logMAR (Snellen equivalent ≥20/40).19
The evolution of toric IOL technology has allowed patients with astigmatic cataracts the possibility of achieving emmetropia without the need for additional refractive corrective procedures or vision aids. However, this is dependent on the rotational stability of the IOL, since the magnitude of the cylinder corrected is inversely proportional to the degree of axis misalignment.20 Each degree of misalignment of a toric IOL reduces the cylinder power effect by ~3.3%.21,22 Quantitative analysis of the axis of orientation of the multifocal toric IOLs showed that lens placement was accurate and that these lenses were rotationally stable in most eyes after 1-year follow-up. Similarly, a 6-month multicenter study of AT Lisa® toric 909M IOLs (909M; Carl Zeiss Meditec, Inc.) in 142 subjects found that postoperative UCDVA was always better than 0.2 logMAR, with no subject reporting having used spectacles for reading at any follow-up visit.23 At 6 months, 95.8% of IOLs had not rotated >5°. Residual astigmatism was >0.50 D in 50% of eyes, which may have been due to clinically irrelevant inaccuracies in positioning and some IOL rotation during the first postoperative day. A single-center study of the 909M-model IOL with a longer 12-month follow-up showed significant improvements in UCDVA and UCNVA and a significant decrease in refractive cylinder. In addition, optical path difference scanning showed that the IOL provided predictable astigmatic correction and was stable in the capsular bag, with limited misalignment.24
A recent case series compared 17 subjects (26 eyes) who were implanted with the 909M IOL and 42 (77 eyes) who received the AT Lisa 809M IOL, an aspheric multifocal IOL.25 Both groups experienced significant improvement in corrected distance and near VA and significant reduction in astigmatism, and they did not differ in the incidence of photic phenomena. The toric IOL group had significantly better uncorrected intermediate VA. In both groups, the postoperative astigmatic power vectors were ±0.50 D in 100% of eyes, and the spherical equivalents were within ±0.50 D in 88.5% of eyes.
A case series evaluating the Lentis Mplus toric IOL (OcuLentis GmbH, Berlin, Germany), which has a surface-embedded near section, included only eyes that had >1.50 D of preexisting corneal astigmatism (58 subjects, 89 eyes).26 After 3 months of follow-up, distance and near visual acuities, reduction in refractive cylinder, and IOL stability were excellent.
One year after implantation, 86.5% and 87.2% of subjects in this study who were implanted with multifocal toric and nontoric IOLs, respectively, reported being satisfied or very satisfied with their vision overall, and 91.4% and 90.6%, respectively, said they would choose the same lenses. Other studies have reported similar rates of dissatisfaction with implanted multifocal IOLs, with 4%–7% of these subjects requiring IOL exchange.27,28 However, most dissatisfied patients can be managed conservatively.
One limitation of this clinical study was that the vast majority of subjects were white (93.7%) and non-Hispanic (98.4%); however, these demographic characteristics are similar to those reported for subjects in the United States undergoing IOL implantation.
No additional risks were identified following bilateral implantation of toric multifocal IOL models SND1T3, SND1T4, SND1T5, and SND1T6 in subjects with corneal astigmatism of 0.75 D–2.82 D relative to the risks associated with bilateral implantation of nontoric multifocal IOL model SA60D3 in subjects with corneal astigmatism <0.75 D. The addition of a toric component to the multifocal lens offers the benefits of multifocal vision to subjects with preexisting corneal astigmatism who require cataract extraction and IOL implantation. The multifocal toric lenses can provide these subjects with a full range of vision and reduction in residual refractive cylinder.
Acknowledgments
Editorial support was provided by BelMed Professional Resources, Inc., and was funded by Alcon. This study was sponsored by Alcon Research, Ltd., Fort Worth, TX, USA.
Disclosure
Robert Lehmann is a consultant to Alcon and has received grant and research support from the sponsor. Satish Modi is a consultant to Alcon and has received grant and research support from the sponsor. Bret Fisher is a consultant to Alcon and has received grant and research support from the sponsor. Magda Michna is an employee of Alcon. Michael Snyder is a consultant to Alcon and has received grant and research support from the sponsor. The authors report no other conflicts of interest in this work.
References
Ferrer-Blasco T, Montes-Mico R, Peixoto-de-Matos SC, Gonzalez-Meijome JM, Cervino A. Prevalence of corneal astigmatism before cataract surgery. J Cataract Refract Surg. 2009;35(1):70–75. | ||
Khan MI, Muhtaseb M. Prevalence of corneal astigmatism in patients having routine cataract surgery at a teaching hospital in the United Kingdom. J Cataract Refract Surg. 2011;37(10):1751–1755. | ||
Ernest P, Potvin R. Effects of preoperative corneal astigmatism orientation on results with a low-cylinder-power toric intraocular lens. J Cataract Refract Surg. 2011;37(4):727–732. | ||
Statham M, Apel A, Stephensen D. Comparison of the AcrySof SA60 spherical intraocular lens and the AcrySof Toric SN60T3 intraocular lens outcomes in patients with low amounts of corneal astigmatism. Clin Experiment Ophthalmol. 2009;37(8):775–779. | ||
Holland E, Lane S, Horn JD, Ernest P, Arleo R, Miller KM. The AcrySof Toric intraocular lens in subjects with cataracts and corneal astigmatism: a randomized, subject-masked, parallel-group, 1-year study. Ophthalmology. 2010;117(11):2104–2111. | ||
Zhao G, Zhang J, Zhou Y, Hu L, Che C, Jiang N. Visual function after monocular implantation of apodized diffractive multifocal or single-piece monofocal intraocular lens: randomized prospective comparison. J Cataract Refract Surg. 2010;36(2):282–285. | ||
Harman FE, Maling S, Kampougeris G, et al. Comparing the 1CU accommodative, multifocal, and monofocal intraocular lenses: a randomized trial. Ophthalmology. 2008;115(6):993–1001. | ||
Hutz WW, Eckhardt HB, Rohrig B, Grolmus R. Intermediate vision and reading speed with array, Tecnis, and ReSTOR intraocular lenses. J Refract Surg. 2008;24(3):251–256. | ||
Nijkamp MD, Dolders MGT, de Brabander J, van den Borne B, Hendrikse F, Nuijts RMMA. Effectiveness of multifocal intraocular lenses to correct presbyopia after cataract surgery: a randomized controlled trial. Ophthalmology. 2004;111(10):1832–1839. | ||
Sen HN, Sarikkola AU, Uusitalo RJ, Laatikainen L. Quality of vision after AMO array multifocal intraocular lens implantation. J Cataract Refract Surg. 2004;30(12):2483–2493. | ||
Javitt JC, Steinert RF. Cataract extraction with multifocal intraocular lens implantation: a multinational clinical trial evaluating clinical, functional, and quality-of-life outcomes. Ophthalmology. 2000;107(11):2040–2048. | ||
Calladine D, Evans JR, Shah S, Leyland M. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst Rev. 2012;9:CD003169. | ||
Cionni RJ, Osher RH, Snyder ME, Nordlund ML. Visual outcome comparison of unilateral versus bilateral implantation of apodized diffractive multifocal intraocular lenses after cataract extraction: prospective 6-month study. J Cataract Refract Surg. 2009;35(6):1033–1039. | ||
de Vries NE, Nuijts RMMA. Multifocal intraocular lenses in cataract surgery: literature review of benefits and side effects. J Cataract Refract Surg. 2013;39(2):268–278. | ||
Solomon K. Patient-reported 12-month outcomes after toric multifocal IOL implantation. Presented at the ASCRS/ASOA Symposium and Congress 2014, April 25–29, 2014, Boston, Massachusetts. | ||
Alfonso JF, Knorz M, Fernandez-Vega L, et al. Clinical outcomes after bilateral implantation of an apodized +3.0 D toric diffractive multifocal intraocular lens. J Cataract Refract Surg. 2014;40(1):51–59. | ||
Knorz MC, Rincon JL, Suarez E, et al. Subjective outcomes after bilateral implantation of an apodized diffractive +3.0 D multifocal toric IOL in a prospective clinical study. J Refract Surg. 2013(11);29:762–767. | ||
Ferreira TB, Marques EF, Rodrigues A, Montes-Mico R. Visual and optical outcomes of a diffractive multifocal toric intraocular lens. J Cataract Refract Surg. 2013;39(7):1029–1035. | ||
Crema AS, Walsh A, Ventura BV, Santhiago MR. Visual outcomes of eyes implanted with a toric multifocal intraocular lens. J Refract Surg. 2014;30(7):486–491. | ||
Patel CK, Ormonde S, Rosen PH, Bron AJ. Postoperative intraocular lens rotation: a randomized comparison of plate and loop haptic implants. Ophthalmology. 1999;106(11):2190–2195. | ||
Novis C. Astigmatism and toric intraocular lenses. Curr Opin Ophthalmol. 2000;11(1):47–50. | ||
Ma JJ, Tseng SS. Simple method for accurate alignment in toric phakic and aphakic intraocular lens implantation. J Cataract Refract Surg. 2008;34(10):1631–1636. | ||
Bellucci R, Bauer NJ, Daya SM, et al; Lisa Toric Study Group. Visual acuity and refraction with a diffractive multifocal toric intraocular lens. J Cataract Refract Surg. 2013;39(10):1507–1518. | ||
Mojzis P, Pinero DP, Ctvrteckova V, Rydlova I. Analysis of internal astigmatism and higher order aberrations in eyes implanted with a new diffractive multifocal toric intraocular lens. Graefes Arch Clin Exp Ophthalmol. 2013;251(1):341–348. | ||
Frieling-Reuss EH. Comparative analysis of the visual and refractive outcomes of an aspheric diffractive intraocular lens with and without toricity. J Cataract Refract Surg. 2013;39(10):1485–1493. | ||
Venter J, Pelouskova M. Outcomes and complications of a multifocal toric intraocular lens with a surface-embedded near section. J Cataract Refract Surg. 2013;39(6):859–866. | ||
Woodward MA, Randleman B, Stulting RD. Dissatisfaction after multifocal intraocular lens implantation. J Cataract Refract Surg. 2009;35(6):992–997. | ||
de Vries NE, Webers CA, Touwslager WR, et al. Dissatisfaction after implantation of multifocal intraocular lenses. J Cataract Refract Surg. 2011;37(5):859–865. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



