Back to Journals » Therapeutics and Clinical Risk Management » Volume 16
Bandage Contact Lenses versus Deproteinized Calf Blood Extract Eye Gel for Recurrent Corneal Erosion Syndrome: A Case-Control Study
Authors Li J , Ma Y, Huang X, Xu L, Tang S
Received 20 August 2020
Accepted for publication 21 October 2020
Published 13 November 2020 Volume 2020:16 Pages 1109—1115
DOI https://doi.org/10.2147/TCRM.S277282
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Jing Li, Yu Ma, Xiaohan Huang, Lanfang Xu, Shaohua Tang
Department of Ophthalmology, Beijing Jishuitan Hospital, Beijing, People’s Republic of China
Correspondence: Jing Li; Shaohua Tang
Beijing Jishuitan Hospital, 31 Xinjiekou East St., Xicheng District, Beijing 100035, People’s Republic of China
Tel +86-10-58398208
Email [email protected]; [email protected]
Background: The clinical efficacy of eye drops in the treatment of recurrent corneal erosion syndrome (RCES) is not satisfactory. Many studies have confirmed the positive effect of the bandage contact lens (BCL) in corneal diseases, but not many in patients with RCES. The purpose of this study is to investigate the efficacy of the BCL compared with deproteinized calf blood extract eye gel in the initial treatment of RCES.
Methods: Forty-seven patients with RCES treated in our hospital from September 2010 to September 2018 were retrospectively analyzed, including 24 cases (26 eyes) in the bandage contact lenses (BCLs) group wearing bandage contact lens and 23 cases (24 eyes) in the drug group treated with deproteinized calf blood extract eye gel. The efficacy was evaluated after 3 months of treatment, with a mean follow-up time of 21.15 ± 1.71 months in the BCL group and 20.87 ± 1.89 months in the drug group. Corneal erosion resolution, pain relief, visual acuity recovery time, recurrence and complications were observed.
Results: After 3 months of treatment, 22 eyes (22/26, 84.6%) in the BCLs group achieved complete resolution, compared with 14 eyes (14/24, 58.3%) in the drug group (P < 0.05). The corneal healing time in the BCLs group was 4.77 ± 4.51 weeks, which was significantly shorter than that in the drug group (9.83 ± 5.93 weeks (P < 0.01)). At 1 and 2 months after treatment, the visual analogue score (VAS) in the BCLs group (3.28 ± 1.15 at 1 month and 1.90 ± 0.77 at 2 months) decreased more significantly than that in the drug group (4.54 ± 0.89 at 1 month and 2.43 ± 0.93 at 2months, P =0.000 at 1 month and P=0.034 at 2 months). At 3 months after treatment, the mean BCVA in the BCL group (logMAR 0.03± 0.08) improved more significantly than that in the drug group (logMAR 0.14± 0.12,P=0.001). The complete recovery time of visual acuity was 5.46 ± 4.43 weeks in the BCLs group, compared with 10.33 ± 6.12 weeks in the drug group (P =0.003). During further follow-up, recurrence was observed in 2 eyes (2/22, 9.1%) of the BCLs group and 6 eyes (6/14, 42.8%) of the drug group. No patient in both groups developed adverse side effects.
Conclusion: Bandage contact lenses are safe and effective in the initial treatment of RCES. Compared with topical deproteinized calf blood extract eye gel, the use of BCLs can provide a higher cure rate, better pain control, faster visual recovery and lower recurrence rate.
Trial Registration: Chinese Clinical Trial Registry, ChiCTR2000031241. Registered 25 March 2020- Retrospectively registered, http://www.chictr.org.cn/edit.aspx?pid=51309andhtm=4.
Keywords: bandage contact lens, recurrent corneal erosion syndrome, deproteinized calf blood extract eye gel
Plain Language Summary
There is no consensus on the treatment of recurrent corneal erosion syndrome (RCES). The initial management of RCES is generally treated with ocular lubricants. However, some patients relapse repeatedly and have to resort to more aggressive interventions such as invasive surgery. The bandage contact lens (BCL) is a less invasive treatment modality than surgical options. Many studies have confirmed the positive effect of BCLs in corneal diseases, but not many in patients with RCES. This study is by far the largest series investigating BCLs in the initial treatment of RCES. Our results show that BCLs are effective and safe for the treatment of RCES. Compared with topical deproteinized calf blood extract eye gel, the use of BCLs can provide a higher cure rate, better pain control, faster visual recovery and lower recurrence rate. We hope that our study could provide feasibility and a preliminary basis for future exploration and clinical application.
Background
Recurrent corneal erosion syndrome (RCES) is characterized by repeated episodes of pain, photophobia and blur vision due to poor epithelial adhesion. RCES is a relatively common disease affecting mainly working adults, which could lead to social and economic consequences.1 The pathogenesis of RCES is associated with poor adhesion of the corneal epithelium to its underlying stroma. Trauma is the initiating factor for most patients with RCES, however, there may be underlying primary epithelial basement membrane dystrophy (EBMD) that leads to symmetrical erosions in multiple corneal areas.2–4
The initial management of RCES is generally treated with topical eye drops, including artificial tears, ocular lubricants, as well as autologous serum,5,6 and the symptoms can be relieved in most patients. The deproteinized calf blood extract is a protein-free, standardized dialysate/ultrafiltrate derived from calf blood, which has been shown to enhance wound healing in both experimental animal studies and man. It is a cellular respiratory activator, which could enhance the uptake of oxygen and glucose by cells, enhance the synthesis of adenosine triphosphate, and promote the repair and regeneration of tissues.7,8 Deproteinized calf blood extract uses sodium carboxymethylcellulose as a carrier to make ophthalmic gel, which has both lubricating and corneal repair effects, and its efficacy is better than that of conventional lubricants alone. It has been widely used in various persistent corneal epithelial injury diseases and has achieved encouraging efficacy. It has also achieved some efficacy in the treatment of recurrent corneal erosion syndrome, but it is often difficult to prevent the recurrence of corneal erosion.9,10 Some patients relapse repeatedly and have to resort to more aggressive interventions such as invasive surgery,11–14 which inevitably has its risks. Many treatment modalities report varying degrees of success but no definitive treatment regime has been agreed.15 The bandage contact lens (BCL) is a less invasive treatment modality than surgical options. The BCL has been used for ocular surface protection and corneal disease therapy in recent years and has obtained encouraging effects.16–20 However, there are not many reports about the efficacy of BCLs on recurrent corneal erosion syndrome. The purpose of this study is to evaluate the efficacy and safety of bandage contact lenses in the initial treatment of recurrent corneal erosion syndrome and to provide suggestions for clinical practice.
Methods
Study Design and Participants
This study retrospectively collected 47 patients who were treated in the Department of Ophthalmology of our hospital due to RCES from September 2010 to September 2018, including 24 patients (26 eyes) in the BCLs group and 23 patients (24 eyes) in the drug group. (Table 1). Inclusion criteria: (1) Diagnosis of RCES; (2) No history of surgery for recurrent corneal erosion; (3) No history of wearing contact lens. Exclusion criteria:(1) With infectious keratitis; (2) Other corneal dystrophies except for epithelial basement membrane dystrophy (EMBD); (3) History of previous refractive surgery or other corneal surgery;(4) Combined with systemic diseases affecting corneal repair (such as epidermolysis bullosa, psoriasis, etc.);(5) Those with allergic constitutions that cannot tolerate the treatment. The study procedure was approved by the Ethics Committee of Beijing Jishuitan Hospital and executed under its supervision. The study adhered to the tenets of the Declaration of Helsinki and all participants signed informed consent.
 |
Table 1 Demographic and Clinical Characteristics of Patients |
Interventions
The BCLs group was treated with continuous BCL wear for 3 months, and the silicone hydrogel soft BCL (PureVision, Bausch and Lomb, NJ) used in this study had a 14-mm diameter and high oxygen transmissibility (130DK/t), which was approved for continuous wear for up to 21 days. In our study, the BCLs were replaced every 2 weeks to decrease the risk of infectious keratitis. During BCL wearing, it is necessary to immediately remove the BCL in case of any noticeable foreign body sensation or infection. Both placing and removing of the BCLs were performed by the same ophthalmologist. Any adverse events possibly related to the use of the BCL were recorded.
In the drug group, the deproteinized calf blood extract eye gel (5g, 20%, Shenyang Xingqi Pharmaceutical Co., Ltd) was used for 3 months, 4 times a day (the 4th time before going to bed). The deproteinized calf blood extract is a protein-free, standardized dialysate/ultrafiltrate derived from calf blood, which mainly contains small peptides, amino acids, nucleotides, oligosaccharides, and other bioactive substances. Before corneal healing, 0.5% levofloxacin eye drops were prescribed four times a day in both groups, while after corneal healing, the topical levofloxacin was reduced to twice a day in the bandage lens group to prevent infection and discontinued in the drug group.
In both groups, sodium hyaluronate eye drops (0.1%, Santen Pharmaceutical Co., Ltd.) were used for 3 months, 4 times a day.
Follow-Up and Outcome Measures
Patients in both groups were followed up weekly during the first month and every 2 weeks during the second and third months after treatment. After 3 months of treatment, the efficacy of the treatment was evaluated, and patients who achieved complete remission were further followed up to 18 months to observe the recurrence, while during this period, follow up visits were arranged every month for the first six months, then every two months for the next 12 months. At the initial visit, the patient’s medical and family history, causes and triggers of the disease, time to initial onset, number of attacks and previous therapy were inquired in detail and recorded. Before and after treatment, the symptoms of RCES (pain, foreign body sensation, redness, tearing or photophobia) were recorded and patients were asked about the grading of pain, and best-corrected visual acuity, slit-lamp biomicroscopy, and corneal fluorescein staining were performed, to record the size and site of loose epithelium and corneal epithelial defects, the presence of microcysts in the epithelium and EBMD.
The main outcome measures included pain evaluated using a visual analogue score (VAS: 0 = no pain at all, 10 = worst pain imaginable),21 best-corrected visual acuity (BCVA), corneal abnormality changes monitored by corneal fluorescein staining. Visual acuity recovery time, improvement rate and cure rate were further evaluated. Visual acuity recovery time refers to the time required for visual acuity to return to the best corrected visual acuity before the first onset. The BCVA was recorded by Snellen chart and analyzed by logarithm of the minimum angle of resolution (LogMAR) between two groups. At 3 months, the patients of both groups were categorized as per the following conditions: (1) cure: complete resolution of symptoms with no visible loose epithelium and corneal fluorescein staining; (2) improvement: partial resolution of symptoms with partial repairment of corneal abnormality; (3) invalid: no significant change or even aggravation of symptoms and corneal abnormalities. A recurrence was noted if the patient developed subjective symptoms or corneal erosions appeared during the follow-up period. All the treatment and follow-up were carried out by the same professionally trained doctor throughout the whole study period, thus eliminating interobserver bias.
Statistical Analysis
Statistical analysis was performed using SPSS software (version 23.0, IBM SPSS Co., Ltd.). Categorical variables were compared using the chi-square test or Fisher’s exact test, and continuous variables were compared using the t-test. A P value of 0.05 was considered significant.
Results
The demographic and clinical characteristics of the patients are shown in Table 1. There were no significant differences in age, gender, number of previous episodes, time from trauma or diagnosis of EBMD to initial episode, cause of disease and size of corneal erosions between the two groups. (Table 1).
Cure Rate and Corneal Healing Time
After 3 months of treatment, 22 eyes (22/26, 84.6%) in the BCLs group achieved complete resolution, compared with 14 eyes (14/24, 58.3%) in the drug group, with significant statistical differences. (Table 2). The time to cure for cornea in the BCLs group was (4.77±4.51) weeks, which was significantly shorter than that in the drug group (9.83±5.93) weeks. (Table 3).
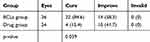 |
Table 2 Comparison of Cure Rates and Improvement Rates at After 3 Months of Treatment Between the Two Groups of Patients (Eyes, %) |
 |
Table 3 Comparison of Corneal Healing Time, Visual Recovery Time and BCVA After Treatment Between the Two Groups of Patients (Mean±SD) |
Pain Relief
After treatment, the pain of patients in both groups was gradually relieved, and the pain relief of the BCLs group was better than that of the drug group. At 1 and 2 months after treatment, the VAS pain score in the BCLs group decreased more significantly than that in the drug group, with a statistically significant difference. At 3 months after treatment, the VAS pain score in the BCLs group was still lower than that in the drug group, but the difference was not statistically significant. (Table 4).
 |
Table 4 Pain Scores at Different Time Points Before and After Treatment in Two Groups of Patients (Mean±SD, Points) |
Vision Recovery
There was no significant statistical difference in baseline BCVA between the two groups. After 3 months of treatment, the BCVA improved in both groups, with mean BCVA of logMAR 0.03±0.08(20/20 Snellen equivalent) in the BCL group and logMAR 0.14±0.12(20/25 Snellen equivalent) in the drug group, and the BCVA improved more in the BCL group(P=0.001).The complete recovery time of visual acuity in the BCLs group was (5.46 ± 4.43) weeks, which was significantly shorter than that in the drug group (10.33 ± 6.12) weeks. (Table 3).
Recurrence Observation
Patients who achieved complete cure (22 in the BCLs group and 14 in the drug group) were further followed up to 18 months to observe recurrence. During further follow-up, recurrence was observed in 2 eyes (2/22, 9.1%) of the BCLs group and 6 eyes (6/14,42.8%) of the drug group, and the difference was statistically significant (P=0.026). In addition to the low recurrence rate, patients in the BCLs group appeared to relapse later than these in the drug group. Both patients with recurrence in the BCLs group complained of ocular symptoms, but minor corneal abnormalities were visible in only one of them. The symptoms of both patients were relieved and tolerable after bedtime lubricant application. Corneal abnormalities were observed in all relapsed patients in the drug group, however, 3 of them did not respond well to bedtime lubricants and had to turn to BCL therapy, after which the symptoms were relieved. (Table 5).
 |
Table 5 Recurrence After Complete Resolution (Number of Eyes, %) |
Complications Observation
In both groups, no serious complications such as infectious keratitis were reported during the entire study. In the BCLs group, BCLs fit satisfactorily in 25 eyes (25/26,96.2%). Sliding of BCL was observed in one patient by slit-lamp examination during follow up, and the BCL was reset in time. None of the patients in the BCLs group developed any complications related to contact lenses.
Discussion
Our results showed that the use of BCLs is more effective for corneal healing promotion and pain control than using eye drops alone, which is the routine therapy in the initial management of RCES.
Although there is no consensus on RCES treatment, promoting to regain sufficient and stable adhesion between the corneal epithelial and the underlying stroma is essential for the management of RCES.17,22 We speculated that wearing BCLs would help mainly in three aspects. First, BCLs help to form a more stable tear film on the ocular surface and provide a scaffold for the migration of epithelial cells, thereby promoting corneal epithelial repair; Secondly, the mechanical barrier function of BCLs can avoid the friction of the eyelid on the cornea, thereby protecting the newly formed corneal epithelium, and allowing time for the defective anchoring system to achieve faster and more stable healing. Thirdly, the BCL wear could provide more sustained and stable pain relief, further reducing eyelid rubbing on the cornea.23,24
The therapeutic effects of wearing BCLs on RCES have been studied. Consistent with our findings, two studies confirmed the positive role of BCL in the treatment of RCES. Fraunfelder20 retrospectively studied 12 patients treated with BCL and found that 9 (75%) had no further recurrence of RCES after approximately 1 year of follow-up, while Ahad25 compared 14 patients treated with BCL and 15 patients treated with ocular lubricants (OLs) and showed that patients on BCLs achieved a complete resolution more quickly, although BCLs do not increase the likelihood of complete resolution when compared with OLs in the initial management of RCES. In contrast to our findings, Williams and Buckley26 reported that BCL using on RCES therapy is less effective than ocular lubricants, with higher rates of recurrence and complications. Further comparison showed that Our study was different from the study of William in many aspects. First, the type of BCL in our study was silicone hydrogel BCL with higher oxygen permeability, while in their study was hydrogel BCL with relatively lower oxygen permeability; Second, a more broad-spectrum prophylactic antibiotic eyedrop (levofloxacin vs chloramphenicol), and a longer wear time of the contact lens overall (3 months vs 2 months) may help to reduce the incidence of complications and improve the efficacy of the BCL.23
In our study, the patients had substantially more favorable outcomes compared to previous reports on the use of BCL for RCES.20,25,26 To the best of our knowledge, the present study reported for the first time the statistically significant improvements in VAS pain scores and visual recovery times with BCL versus ocular lubricant for RCES. The recurrence rate (2/26,7.7%) in the BCLs group was also acceptable during the 18 months follow-up after treatment. In addition to the encouraging cure rate, better pain relief, faster visual recovery and lower recurrence rate after BCL wear provide additional evidence for the effectiveness of BCL treatment. Besides, although not observed, contact lens-related complications, especially corneal infections, should be closely monitored rather than ignored during the BCL application, and the risk and signs of infection must be detailed to the patients.27,28 The efficacy of the drug treatment group in this study was similar to that of other eye drops previously reported for the treatment of RCES. The reason for the lower efficacy in the drug treatment group than in the BCls group may be considered to be related to the lack of mechanical support and barrier protection of the eye drops on corneal abnormalities. Despite these encouraging findings, this study has inherent limitations of retrospective observational studies, and further prospective studies are required to confirm our conclusions.
Conclusions
In summary, our results show that BCLs are effective and safe for the treatment of RCES. Compared with topical deproteinized calf blood extract eye gel, the use of BCLs can provide a higher cure rate, better pain control, faster visual recovery and lower recurrence rate. Therefore, our study suggests that BCL therapy can be considered in the management of RCES, and we hope that our study could provide feasibility and a preliminary basis for future exploration and clinical application.
Abbreviations
RCES, recurrent corneal erosion syndrome; BCL, bandage contact lens; BCLs, bandage contact lenses; BCVA, best-corrected visual acuity; VAS, visual analogue scale; OLs, ocular lubricants.
Data Sharing Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of the Beijing Jishuitan Hospital and completed under its supervision. And the participant provided written informed consent to take part in the study. It was in accordance with all the ethical requirements.
Consent for Publication
All authors agree to publish in the magazine.
Acknowledgments
The authors thanks for the corneal disease group of Jishuitan Hospital.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Funding
This research was funded by the “Beijing Municipal Administration of Hospitals Incubating Program”, code: PX2018019.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Moutray TN, Frazer DG, Jackson AJ. Recurrent erosion syndrome – the patient’s perspective. Contact Lens Anterior Eye. 2011;34(3):139–143. doi:10.1016/j.clae.2010.11.006
2. Nanba H, Mimura T, Mizuno Y, et al. Clinical course and risk factors of recurrent corneal erosion. Medicine. 2019;98(16):e14964.
3. Miller DD, Hasan SA, Simmons N, et al. Recurrent corneal erosion: a comprehensive review. Clin Ophthalmol. 2019;13:325–335. doi:10.2147/OPTH.S157430
4. Laibson PR. Recurrent corneal erosions and epithelial basement membrane dystrophy. Sci Clin Pract. 2010;36(5):315–317. doi:10.1097/ICL.0b013e3181f18ff7
5. Ziakas NG, Boboridis KG, Terzidou C, et al. Long-term follow up of autologous serum treatment for recurrent corneal erosions. Clin Experiment Ophthalmol. 2010;38(7):683–687. doi:10.1111/j.1442-9071.2010.02304.x
6. Del Castillo JMB, de la Casa JMM, Sardiña RC, et al. Treatment of recurrent corneal erosions using autologous serum. Cornea. 2002;21(8):781–783. doi:10.1097/00003226-200211000-00010
7. Egger SF, Huber-Spitzy V, Alzner E, et al. Corneal wound healing after superficial foreign body injury: vitamin A and dexpanthenol versus a calf blood extract. Ophthalmology. 1999;213(4):246–249. doi:10.1159/000027429
8. Huibin L, Liu Z, Xuemin L, et al. Effect of lacrimal plugs combined with deproteinized calf blood extract eye gel for filamentary keratitis[J]. J Ocul Biol Dis Infor. 2010;3(4):134–140. doi:10.1007/s12177-011-9066-6
9. Xiao-Guang N. Therapeutic effect of anterior corneal stromal puncture combined with deproteinized calf blood extract eye gel for recurrent corneal erosion. Recent Adv Ophthalmol. 2011;31(11):1077–1079.
10. Zhang C, Wei M. Clinical effect of deproteinized calf blood extract eye gel for corneal epithelial defect. Discussion Clin Cases. 2018;5(2):7–10. doi:10.14725/dcc.v5n2p7
11. Avni Zauberman N, Artornsombudh P, Elbaz U, et al. Anterior stromal puncture for the treatment of recurrent corneal erosion syndrome: patient clinical features and outcomes. Am J Ophthalmol. 2014;157(2):273–279.e1. doi:10.1016/j.ajo.2013.10.005
12. Singh RP, Raj D, Pherwani A, et al. Alcohol delamination of the corneal epithelium for recalcitrant recurrent corneal erosion syndrome: a prospective study of efficacy and safety. Br J Ophthalmol. 2007;91(7):908–911. doi:10.1136/bjo.2006.112912
13. Vo RC, Chen JL, Sanchez PJ, et al. Long-term outcomes of epithelial debridement and diamond burr polishing for corneal epithelial irregularity and recurrent corneal erosion. Cornea. 2015;34(10):1259–1265. doi:10.1097/ICO.0000000000000554
14. Dedes W, Faes L, Schipper I, et al. Phototherapeutic keratectomy (PTK) for treatment of recurrent corneal erosion: correlation between etiology and prognosis – prospective longitudinal study. Graefe’s Arch Clin Exp Ophthalmol. 2015;253(10):1745–1749. doi:10.1007/s00417-015-2990-6
15. Watson S, Lee H. Interventions for recurrent corneal erosion: a cochrane systematic review. Eye (London, England). 2013;27(11):1330–1331. doi:10.1038/eye.2013.191
16. Rashad R, Weed MC, Quinn N, et al. Extended wear bandage contact lenses decrease pain and preserve vision in patients with epidermolysis bullosa: case series and review of literature. Ocul Immunol Inflamm. 2020;28(3):379–383. doi:10.1080/09273948.2019.1587472
17. Shimazaki J, Shigeyasu C, Saijo-Ban Y, et al. Effectiveness of bandage contact lens application in corneal epithelialization and pain alleviation following corneal transplantation; prospective, randomized clinical trial. BMC Ophthalmol. 2016;16(1):174. doi:10.1186/s12886-016-0346-6
18. Blackmore SJ. The use of contact lenses in the treatment of persistent epithelial defects. Contact Lens Anterior Eye. 2010;33(5):239–244. doi:10.1016/j.clae.2010.06.004
19. Choi JA, Chung S. Combined application of autologous serum eye drops and silicone hydrogel lenses for the treatment of persistent epithelial defects. Sci Clin Pract. 2011;37(6):370–373. doi:10.1097/ICL.0b013e318233c9bb
20. Fraunfelder FW, Cabezas M. Treatment of recurrent corneal erosion by extended-wear bandage contact lens. Cornea. 2011;30(2):164–166. doi:10.1097/ICO.0b013e3181e84689
21. Bae SH, Shin YJ, Kim HK, Hyon JY, Wee WR, Park SG. Vitamin D supplementation for patients with dry eye syndrome refractory to conventional treatment. Sci Rep. 2016;6(1):33083.
22. Lin SR, Aldave AJ, Chodosh J. Recurrent corneal erosion syndrome. Br J Ophthalmol. 2019;103(9):1204–1208. doi:10.1136/bjophthalmol-2019-313835
23. Rubinstein MP. Applications of contact lens devices in the management of corneal disease. Eye (Lond). 2003;17(8):872–876. doi:10.1038/sj.eye.6700560
24. Lim L, Tan DT, Chan WK. Therapeutic use of bausch & lomb purevision contact lenses. CLAO J. 2001;27(4):179–185.
25. Ahad MA, Anandan M, Tah V, et al. Randomized controlled study of ocular lubrication versus bandage contact lens in the primary treatment of recurrent corneal erosion syndrome. Cornea. 2013;32(10):1311–1314. doi:10.1097/ICO.0b013e31829dec39
26. Williams R, Buckley RJ. Pathogenesis and treatment of recurrent erosion. Br J Ophthalmol. 1985;69(6):435–437. doi:10.1136/bjo.69.6.435
27. Alipour F, Khaheshi S, Soleimanzadeh M, Heidarzadeh S, Heydarzadeh S. Contact lens-related complications: a review. J Ophthalmic Vis Res. 2017;12(2):193–204.
28. Feizi S, Masoudi A, Hosseini SB, Kanavi MR, Javadi MA. Microbiological evaluation of bandage soft contact lenses used in management of persistent corneal epithelial defects. Cornea. 2019;38(2):146–150.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
