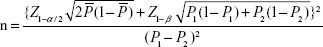Back to Journals » Clinical Ophthalmology » Volume 8
Bacterial endogenous endophthalmitis in Vietnam: a randomized controlled trial comparing vitrectomy with silicone oil versus vitrectomy alone
Authors Do T, Hon DN, Aung T, Hien N, Cowan Jr CL
Received 12 May 2014
Accepted for publication 19 June 2014
Published 28 August 2014 Volume 2014:8 Pages 1633—1640
DOI https://doi.org/10.2147/OPTH.S67589
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Tan Do,1 Do N Hon,1 Tin Aung,2 Nguyen DTN Hien,1 Claude L Cowan Jr3
1Vietnam National Institute of Ophthalmology, Hanoi, Vietnam; 2Singapore National Eye Center, Singapore; 3Georgetown University Medical Center, Washington, DC, USA
Background/aims: To compare treatment outcomes with and without silicone oil tamponade in patients undergoing pars plana vitrectomy (PPV) for severe endogenous bacterial endophthalmitis (BEE).
Methods: One hundred and eight consecutive patients with severe BEE (defined by the absence of pupil red reflex at presentation and/or dense vitreous opacity on ultrasound and no improvement after 24–36 hours of medical treatment) were randomly assigned to two treatment groups: Group 1, standard PPV with intravitreal antibiotics; and Group 2, PPV with intravitreal antibiotics and silicone tamponade. Overall success was defined as: a visual acuity ≥ count fingers at 1 meter, with an attached retina, and no intraocular oil.
Results: Fifty three patients were randomized to Group 1 and 55 patients to Group 2. The mean age of study subjects was 32 years and baseline clinical characteristics were similar in both groups. At the final follow-up visit at 9 months, the overall success rate of Group 2 (67.3%) was significantly better than Group 1 (43.4%; P=0.01). There were also fewer devastating complications (such as inoperable retinal detachment, phthisis bulbi) in Group 2 compared with Group 1 (21.8% versus 43.4%; P=0.01).
Conclusion: The outcome at 9 months of patients with BEE treated by vitrectomy with silicone oil was better than those treated by vitrectomy alone.
Keywords: bacterial endogenous endophthalmitis, vitrectomy, silicone oil endotamponade, randomized controlled trial
Background
Bacterial endogenous endophthalmitis (BEE) is a severe infection of intraocular tissues due to blood borne bacterial metastasis from other organs. Although vitrectomy, systemic antibiotics, and intraocular antibiotic injections are the mainstays of treatment, the final functional result is often poor due to diffuse tissue necrosis and retinal detachment post-vitrectomy.1,2
Silicone oil was proven to be bactericidal experimentally by Ozadamar et al in 1999.3 Implementing this finding, Azad et al and Kaynak et al have conducted two clinical trials which showed some benefit of silicone oil as an adjunct to vitrectomy in endophthalmitis treatment. Subjects treated with vitrectomy with silicone oil were found to have better functional outcome with fewer additional operations.1,2 However the trials were conducted for post-traumatic and postoperative endophthalmitis respectively and evidence for silicone oil use in BEE is lacking.
We carried out a randomized controlled trial for BEE comparing vitrectomy with silicone oil tamponade with conventional vitrectomy alone.
Materials and methods
Consecutive subjects with BEE were screened for enrollment into this prospective, randomized controlled trial conducted at a single center (Vietnam National Institute of Ophthalmology). Written, informed consent was obtained from all participants. The study had the approval of the Ethical Review Committee of Hanoi Medical University and was carried out in accordance with the tenets of the Declaration of Helsinki. All patients underwent a systemic evaluation and review of the medical history to identify any risk factors or precipitating factors for BEE.
The inclusion criteria were as follows:
- Clinically diagnosed with BEE, as evidenced by the presence of pain, blurred vision, eyelid edema, conjunctival chemosis and hyperemia, corneal edema, anterior chamber cells and hypopyon, vitritis and/or retinitis, and/or chorioretinal abscesses.
- The absence of red pupil reflex or grade 5 of vitreous opacity according to the grading system of endophthalmitis vitrectomy study (EVS) of 1995,4 with visual acuity (VA) limited to light perception, and/or dense vitreous opacity on ultrasound consistent with that level of vision.
- No response to medical treatment with intravitreal and systemic antibiotics alone and clinical picture deteriorated over first 24–36 hours whose initial VA ranged from 20/200 to hand motion.
- Bacteria found on Gram stain from anterior chamber and/or vitreous taps.
The exclusion criteria were as follows:
- History of previous ocular trauma or surgery.
- Eyes with no light perception.
- Eyes with retinal detachment and/or choroidal detachment (seen clinically and/or ultrasonographically).
- Patients <75 years old, or =5 years old.
Once the clinical diagnosis had been made, anterior chamber (AC) tap and vitreous tap were performed. The samples were first directly examined by Gram stain before culturing into appropriate media (blood agar, chocolate agar [35°C–37°C, 5%–10% CO2], and MacConkey agar [35°C–37°C for Columbia blood agar with Hemin and vitamin K for anaerobic bacteria]). If bacteria were not found on Gram stain, the subject would be excluded from the study. Blood culture was performed in cases with fever, and urine culture was performed for all patients. A standard systemic workup was done for all including infectious disease physician consultation, blood tests (including complete blood count, glucose, electrolytes, HIV, hepatitis B surface antigen), urinalysis (including pH, protein, glucose, bilirubin, cells), sinus X-ray, chest X-ray, and abdominal ultrasound. Patients were randomized into two study groups, Group 1 underwent vitrectomy alone and Group 2 underwent vitrectomy with silicone oil tamponade with randomization occurring at two time points:
- Upon diagnosis of BEE clinically and microbiologically, if the red reflex remained, patients would be first treated medically (by intravitreal and systemic antibiotics-please see the sections Materials and methods and Associated medical treatment) and closely monitored over 24 hours. If there was poor response to the medical treatment (more pain and/or more hypopyon, and/or more vitreous opacity) after 24–36 hours, the patients would be enrolled into the study and randomized to either of the two study groups.4
- For patients in whom the red reflex was poor at presentation due to vitreous cavity being densely opaque clinically and/or ultrasonographically, patients would be immediately randomized into either of the two study groups after informed consent.
The randomization list was generated automatically with help of random allocation software (developed by Saghaei M, Department of Anesthesia, Isfahan University of Medical Sciences, Isfahan, Iran).
Associated medical treatment
A standardized treatment protocol was used with all patients receiving vancomycin (1 mg/0.1 mL for intravitreal injection; 1 g/day or 40 mg/kg/day divided into four doses for intravenous injection) and ceftazidime (2 mg/0.1 mL for intravitreal injection; 2 g/day or 75 mg/kg/day divided into two doses intravenously). Patients who were allergic to penicillin were given amikacin (400 μg/0.1 mL for intravitreal injection; 6 mg/kg/12 h for intravenous injection). Intravenous antibiotics were given for at least 1 week (up to 2 weeks) depending on the clinical responses (such as the grade of AC hypopyon, the residual pus at the vitreous base) and were always followed by another week of oral antibiotics.
Steroids were prescribed 48 hours after the initialization of antibiotics if there was favorable clinical judgment (dexamethasone 0.4 mg/0.1 mL for intravitreal injection along with 1 mg/kg/day of prednisolone by mouth for 4 weeks).4
Vitrectomy procedure
A three-port 20-gauge vitrectomy was performed under the ophthalmic endoscopy system (FiberTech Co., Ltd, Tokyo, Japan). After a complete vitrectomy had been performed (with posterior vitreous detachment in all cases, and vitreous completely removed up to equator), patients randomized to Group 1 received 0.05 mL of vancomycin (1 mg/0.1 mL) and 0.05 mL of ceftazidime (2 mg/0.1 mL), and patients in Group 2 were injected with silicone oil as well as vancomycin and ceftazidime.5 If retinal tears were caused during the time of surgery, they were treated with endolaser and endotamponaded with short term gas (SF6) in Group 1 and with silicone oil in Group 2. If retinal detachment occurred after the surgery in Group 1, patients were all reoperated to reattach the retina, using silicone oil tamponade.
Postoperative follow-up
Patients were scheduled for examination at 1 week, 1 month, 3 months, 6 months, and 9 months postoperatively. Silicone oil was removed at 3 months follow-up if the eye was stable and fulfilled the following criteria: no inflammation, normal intraocular pressure (IOP), flat retina, and no active proliferative vitreoretinopathy (PVR). If there was localized retinal detachment, this had to be fully corrected before the oil removal.1,2 Cases where oil could not be removed were considered failures.
The technique of silicone oil removal
This maneuver was performed through two-port technique: one opening for infusion which created the passive pressure to egress the oil gradually through the other opening. Air–fluid exchange was done when needed to expel all small oil bubbles.6
Study outcome criteria
The study outcome criteria included functional success, anatomical success, and overall success, as well as complication rates:
- Functional success: was defined as of VA ≥ counting fingers (CF) at 1 meter, and any better than preoperative VA.
- Anatomical success: was defined as of media clarity grade 2 or better (according to grading system of EVS, 1995).4
- Overall success: was defined as a VA ≤ CF at 1 meter and better than pre-operative VA and media clarity ≥ grade 2, and an attached retina without intraocular oil.
All intraoperative and postoperative complications were noted, such as retinal tears, retinal detachment, macular anomalies, cataract, phthisis bulbi, and others (with comparison between two groups). The complications related to oil removal (such as retinal detachment, and suprachoroidal hemorrhage) were also recorded.
Sample size calculation and data analysis
The sample size for a significance level of 0.05 with a power of 1–b =0.90 for all of our comparisons was calculated using the formula for a single blinded randomized controlled trial
| (1) |
where n is sample size in each group; P1 is the estimated success rate of the control group (0.5); P2 is the estimated success rate of the study group (0.8); and P– is the mean success rate of both groups.
The calculated sample size was 52 for each group.
Results
Between August 2008 and October 2010 108 consecutive patients who fulfilled our inclusion criteria were recruited into the study and were randomized into two groups: 53 patients in Group 1 and 55 patients in Group 2. All patients were available for the 9 months postoperative visit (primary endpoint) although some patients missed some of the interim visits. Demographic and systemic characteristics of study subjects were summarized in Table 1 as well as clinical features upon presentation in Table 2.
  | Table 2 Clinical features of study subjects upon presentation |
The surgical outcomes
At 9 months postoperatively, on the eyes which has been free from oil for about 6 months (all cases in Group 2 and reoperated cases for retinal detachments in Group 1), there was a significantly better outcome in Group 2 in terms of overall success (Group 1, 43.4% versus Group 2, 67.3%; P=0.01) and functional outcome (Group 1, 50.9% versus Group 2, 74.6%, P=0.02). The rate of VA improvement ≥ over baseline tended to be better in Group 2, 40% versus 22.6% of Group 1 (P=0.0521). The anatomical result in Group 2 also had a trend of being better (Group 1, 64.2% versus Group 2, 80%; P=0.07).
Patient age had a clear effect on the final functional outcome, being better in the group of patients older than 40 years (relative risk [RR] =2.39, P=0.02). In contrast, there was no association between sex (P=0.62) or history of systemic risk factors (P=0.603) with final success rate.
In terms of preoperative VA, the group with VA of better than light perception at presentation had better overall outcomes than those with light perception only (P=0.03). Other signs of severity of BEE, namely the height of hypopyon, presence of vitreous opacity, retinal necrosis, and vitreous opacity on ultrasound, were also associated with poorer final outcome (P=0.034, 0.009, 0.01, and 0.017 respectively).
Postoperative complications
In Group 1, the rate of retinal detachment over the early study period (within the first 3 months) was higher (36/53, 67.9%) compared to Group 2 (3/55, 5.45%; P=0.0000001). In Group 1, most of the retinal detachments occurred within first postoperative week due superior retinal dialysis (27/53, 50.9%, which were not seen at the time of surgery), the remaining were due to later retinal breaks happening within the first month of follow up. All subjects with retinal detachment had to be reoperated to reattach the retina under silicone oil. In contrast, the retinal detachments within first 3 months in Group 2 were typically due to traction under the oil bubble and were normally repaired with scleral buckling.
We compared the safety of silicone oil removal in patients of Group 1 and those of Group 2 and found that the rate of retinal detachment after silicone oil removal in Group 1 was much higher than that of Group 2 (7/19, 36.8% versus 3/45, 6.6%; P=0.009. Relative risk of 4.31).
There was also a significant difference in the rate of irreversible visual loss complications (fibrosed and atrophic macula; inoperable retinal detachment; phthisis bulbi and evisceration) in Group 1 compared with Group 2 (43.4% versus 21.8% with P=0.01, Table 5).
There were nine cases of oil in the anterior chamber: four in Group 1 and five in Group 2. In four cases (two in each group), silicone oil was removed earlier than scheduled due to oil related complications leaving no sequelae. There was only one case of high IOP post-surgery, which was treated successfully with glaucoma eye drops. One case of oil emulsification was noted which required oil removal.
Discussion
In this randomized clinical trial, we found significant differences between two groups over time. At 9 months after the surgery (study endpoint), Group 2 (who underwent vitrectomy with silicone oil) had better functional outcome and overall success. This finding agrees with the conclusions from previous studies,1,2 and shows the long term advantages of endotamponade with silicone oil for endophthalmitis. The treatment of BEE in the South East Asian region, particularly with the major pathogen Klebsiella pneumoniae from hepatobiliary infections, has been generally challenging with very poor outcomes.7 More recently, thanks to advances of vitrectomy, the reported treatment outcome of BEE has improved modestly to a success rate of 33%7 to 40%.8 Most authors agreed upon the crucial role of vitrectomy in improving visual function, eyes which had been treated surgically seemed to have three times more chances of recovering useful vision and three times less chance of being eviscerated.7–10 When compared with the surgical outcome from similar studies in the region, we found our result seemed to be more favorable, our overall success rate of 74.6% in Group 2 compared with 30% by Yoon et al.9
The better outcome of silicone oil assisted vitrectomy could be explained as follows: silicone oil may help antibiotics to eradicate the microbes leading to a quicker recovery.3 Some authors in the past hesitated to use silicone oil in endophthalmitis eyes due to the fear of microbial pooling and uncontrolled antibiotic concentration behind the oil bubble.11 However, later study showed the inhibiting effect of silicone oil for microbes including anaerobes.3 Furthermore, antibiotics penetrated so well into the sub-oil space that oral ciprofloxacine (0.75 g twice a day) could easily bring about the microbial bactericidal concentration (MBC) and/or microbial inhibiting concentration (MIC).11 Silicone oil could also help prevent postoperative retinal detachment. Another advantage of using silicone oil is that the oil keeps the media clear, which helps postoperative examination and additional laser treatment when needed. Oil also has good surface tension, pushing the retina against the eye wall, and blocking retinal tears.1,2 There are some disadvantages to performing vitrectomy in severely infected BEE eyes: 1) the media opacity obscures visualization, leading to unexpected damage to the retina; 2) the infected and necrosed retina may be vulnerable during the operation to surgical traction especially at the vitreous base, where there is strong vitreo-retinal adherence; 3) the inflammation may continue after the surgery, causing late necrosis of underperfused zones like the ora serrata (retinal dialysis); and 4) ciliary body damage (necrosis, shock) may cause postoperative hypotony. All these issues may cause retinal detachment.1,2,5,12 Thus, vitrectomy in BEE could be considered as a double-edged sword, on one hand, it may help to clear the infection while on the other hand, it could increase the risk of retinal detachment.
Our study has shown notable differences in post-vitrectomy retinal detachment rates in eyes with BEE. In Group 1, 50.9% (25/53) of patients experienced a retinal detachment within the first post operative week. These detachments were associated with a superior retinal dialysis and all patients required re-operation with silicone oil tamponade to obtain re-attachment and stabilization of the eye. Among the other cases, there were nine more cases of retinal detachment that occurred later, making the total number of 36 retinal detachments (67.9%) in Group 1. In contrast, in Group 2, there were no retinal detachments during the hospitalization period and within first 3 months there were only three cases of retinal detachment (5.45%) caused by residual traction under the oil bubble.
Most of late complications in the treatment course of BEE are related to PVR. In the 27 cases of retinal detachment in Group 1 where repair with silicone oil assistance was performed, the vicious cycle of PVR was triggered.13 Although the retina was attached initially under oil, PVR was further accentuated by the second surgery, leading to later recurrent retinal detachment in nine cases. All these nine cases were considered inoperable, and oil was left permanently in the eye. Among 36 oil-filled eyes of Group 1, at 3 months after the procedure, there were only 19 cases that were considered suitable candidates for oil removal. Unfortunately, the recurrent rate of retinal detachment in this group was also high, up to 36.8% (7/19). Compared with the rate of recurrent retinal detachment after oil removal in Group 2, we saw a clear difference: 3/45, 6.6% in group 2 versus 7/19, 36.8% (P=0.008). In other words, the relative risk of oil removal in Group 1 compared to Group 2 was 4.31 (range, 1.22–15.27). The recurrent retinal detachment was almost always due to the progressive PVR, causing new breaks and/or tractions.6,14,15
All recurrent retinal detachments after surgery were complicated due to the combination of rhegmatogenous and tractional factors.13,14 The prognosis was poor and many eyes became phthisical. At the final follow-up, the number of very poor outcomes (including macular fibrosis, inoperable retinal detachment, phthisis bulbi, evisceration) in Group 1 was significantly higher than that of Group 2 (43.4% versus 21.8% with P=0.02) and this is reflected in the overall success rates.
Most of the patients in our trial from Vietnam with BEE were previously fit and healthy (75%) with no other medical conditions, and with the mean age of only 32 years. In contrast, in the 17 year review of Jackson et al8 BEE usually occurred in patients with pre-existing diabetes mellitus, with mean age of 48.9 years. In Vietnam, the most commonly seen predisposing systemic diseases were upper respiratory infections. Meanwhile another review in the South East Asia region showed that the most common risk factor for BEE was hepatobiliary infections (13/27 cases equivalent to 48%).16 However, none of recruited subjects had fever during the hospitalization time so the proposed mechanism of bacteria metastasis was bacteremia rather than septicemia. The source of many of these infections remains unclear due to the low rate of culture positivity. We subsequently ran another analysis using polymerase chain reaction (PCR) and sequencing technique to evaluate the vitreous samples from another 23 patients with BEE and we found 47.8% (11/23) positive with Streptococcus pneumoniae. Basing on that evidence we suspect that we may have needed a more sensitive assay for the tests in our initial study. In addition, the high rate of upper respiratory infections in Vietnamese patients may have contributed to episodic asymptomatic bacteremia in our patients and the widespread inappropriate use of antibiotics may have resulted in the disease remaining sub clinical.
We found that the positive rate of vitreous analysis was much higher than that of aqueous taps (on smear 81.5% versus 48.1%; on culture 13.9% versus 3.7%). This finding was similar with previous studies.17–20 The overall positive rate of bacterial cultures was relatively low, only 19/108 cases accounting for 17.6%. The most frequently seen pathogen was Staphylococcus aureus (6/19, accounted for 31.5%), followed by Streptococcus pneumoniae (3/19, 15.7%), and Pseudomonas aeruginosa (3/19, 15.7%) (Table 4). In other countries of South East Asia, the most common bacterium were Gram negative rods Klebsiella from hepatobiliary infections.7,19 In Europe and North America, the most common isolates were Gram positive cocci like Staphylococcus and Streptococcus from arthritis and endocarditis.8,19,21,22
  | Table 4 Bacteria isolated from samples in the study |
In the study, we found that markers of severity of BEE like preoperative VA, hypopyon height (in mm), vitreous opacity grade, and fundus affected areas (necrosis, abscesses) were associated with poorer final result, which is in agreement with previous studies.23–25 The EVS study has already shown the benefit of urgent vitrectomy for post cataract endophthalmitis with no pupillary red reflex and vision acuity limited to light perception.4 The difference in BEE patients was that the anterior segment was also usually severely involved. If the anterior segment was very opaque the vitrectomy indication would need to be based on the ultrasound evaluation (as shown in the inclusion criteria). Intraoperatively, we typically found retinal necrosis and chorioretinal abscesses, findings not commonly associated with endophthalmitis complicating cataract surgery, and which likely contributed to the poorer prognosis (Table 3). In cases treated initially with antibiotics the clinical evolution needed to be closely monitored for a timely surgical indication.
The main limitation of this study was the low rate of positive cultures, which may be partly explained by the prior use of antibiotics from referring centers, and the small volume of collected samples. In future, in order to elucidate the bacterial basis of this disease, we may need to use other methods such as PCR and sequencing which can bring about a higher sensitivity and specificity.
Conclusion
In this randomized control trial for BEE, we found better functional and overall outcomes in subjects treated by vitrectomy with silicone oil compared to vitrectomy alone. There was a high rate of retinal detachment in the group treated by vitrectomy alone. There was a low rate of oil related complications. We thus recommend that severe cases of BEE be treated by vitrectomy with silicone oil.
Acknowledgment
Our deepest thanks to the Vietnam National Institute of Ophthalmology, which funded this study.
Disclosure
The authors report no conflicts of interest in this work.
References
Azad R, Ravi K, Talwar D, Rajpal, Kumar N. Pars plana vitrectomy with and without silicone oil tamponade in post-traumatic endophthalmitis. Graefe’s Arch Clin Exp Ophthalmol. 2003;241(6):478–483. | ||
Kaynak S, Oner FH, Focak N, et al. Surgical management of postoperative endophthalmitis: comparision of 2 techniques. J Cataract Refract Surg. 2003;29(5):966–969. | ||
Ozadamar A, Aras C, Ozturk R, Akin E, karacorlu M, Ercikan C. In vitro antimicrobial activity of silicone oil against endophthalmitis-causing agents. Retina. 1999;19(2):122–126. | ||
Endophthalmitis Vitrectomy Study Group. Results of the Endophthalmitis Vitrectomy Study. Arch Ophthalmol. 1995;113(12):1479–1496. | ||
Kuhn F, Gini G. Ten years after... are findings of the Endophthalmitis Vitrectomy Study still relevant today? Graefe’s Arch Clin Exp Ophthalmol. 2005;243(12):1197–1199. | ||
Sharma T, Gopal L, Shanmugam P, et al. Management of recurrent retinal detachment in silicone oil-filled eyes. Retina. 2002;22(2):153–157. | ||
Wong JS, Chan TK, Lee HM, et al. Endogenous bacterial endophthalmitis. Ophthalmology. 2000;107(8):1483–1491. | ||
Jackson TL, Eykyn JS, Graham EM, Graham EM, Stanford MR. Endogenous bacterial endophthalmitis: a 17-year prospective study and review of 267 reported cases. Surv Opthalmol. 2003;48(4): 403–423. | ||
Yoon YH, Lee SU, Sohn JH, Lee SE. Result of early vitrectomy for endogenous Klebsiella pneumoniae endophthalmitis. Retina. 2003; 23(3):366–370. | ||
Yonekawa Y, Chan RV, Reddy AK, Pieroni CG, Lee TC, Lee S. Early intravitreal treatment of endogenous bacterial endophthalmitis. Clin Experiment Ophthalmol. 2011;39(8):771–778. | ||
Talwar D, Kulkarni A, Azad R, et al. Intraocular ciprofloxacin levels after oral administration in silicone oil filled eyes. Invest Ophthalmol Vis Sci. 2003;44(2):505–509. | ||
Doft BM, Kelsey SF, Wisniewski SR. Retinal detachment in the Endophthalmitis Vitrectomy Study. Arch Ophthalmol. 2000;118(12): 1661–1665. | ||
Cowley M, Conway BP, Campochiaro PA, kaiser D, Gaskin H. Clinical risk factors for proliferative vitreoretinopathy. Arch Ophthalmol. 1989;107(8):1147–1151. | ||
Raman R, Sharma T. Management of recurrent retinal detachment in silicone oil-filled eyes. Tech Ophthalmology. 2010;8(3):122–126. | ||
Jonas JB, Knorr HL, Rank RM, Budde W. Retinal redetachment after removal of intraocular silicone oil tamponade. Br J Ophthalmol. 2001; 85(10):1203–1207. | ||
Chee SP, Jap A. Endogenous endophthalmitis. Curr Opin Ophthalmol. 2001;12(6):464–470. | ||
Marelli DJ. Endogenous endophthalmitis. Clinical Eye and Vision Care. 1995;7(3):143–149. | ||
Ness T, Pelz K, Hansen LL. Endogenous endophthalmitis: microorganisms, disposition and prognosis. Acta Ophthalmol Scand. 2007;85(8): 852–856. | ||
Shankar K, Gyanendra L, Hari S, Narayan SD. Culture proven endogenous bacterial endophthalmitis in apparently healthy individuals. Ocul Immunol Inflamm. 2009;17(6):396–399. | ||
The endophthalmitis vitrectomy study group. Microbiologic factors and visual outcome in endophthalmitis vitrectomy study. Am J Ophthalmol. 1996;122(6):830–846. | ||
Binder MI, Chua J, Kaiser PK, et al. Endogenous endophthalmitis: an 18-year review of culture-posistive cases at a tertiary care center. Medicine. 2003;82(2):97–105. | ||
Leibovitch I, Lai T, Raymond G, Zadeh R, Nathan F, Selva D. Endogenous endophthalmitis: a 13-year review at a tertiary hospital in South Australia. Scand J Infect Dis. 2005;37(3):184–189. | ||
Bartz-Schmidt KU, Bermig J, Kirchhof B, Wiedemann P, Walter P, Heimann K. Prognostic factors associated with the visual outcome after vitrectomy for endophthalmitis. Graefe’s Arch Clin Exp Ophthalmol. 1996;234(1 supplement):s51–s58. | ||
Dacey MP, Valencia M, Lee MB, et al. Echographic findings in Infectious endophthalmitis. Arch Ophthalmol. 1994;112(10):1325–1333. | ||
Fan JC, Niederer RL, von Lany H, Polkinghorne PJ. Infectious endophthalmitis: clinical features, management and visual outcomes. Clin Experiment Ophthalmol. 2008;36(7):631–636. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.