Back to Journals » Therapeutics and Clinical Risk Management » Volume 18
Avatrombopag for the Treatment of Adult Patients with Chronic Immune Thrombocytopenia (cITP): Focus on Patient Selection and Perspectives
Authors Tsykunova G, Ghanima W
Received 3 December 2021
Accepted for publication 28 February 2022
Published 24 March 2022 Volume 2022:18 Pages 273—286
DOI https://doi.org/10.2147/TCRM.S251672
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Garry Walsh
Galina Tsykunova,1– 3 Waleed Ghanima2– 4
1Department of Hematology, Haukeland University Hospital, Bergen, Norway; 2Institute of Clinical Medicine, University of Oslo, Oslo, Norway; 3Department of Hemato-Oncology, Østfold Hospital, Grålum, Norway; 4Department of hematology, Oslo University Hospital and Institute of Clinical Medicine, University of Oslo, Oslo, Norway
Correspondence: Waleed Ghanima, Department of Hemato-Oncology, Østfold Hospital, Kalnesveien 300, Grålum, 1714, Norway, Tel +47 69860000, Email [email protected]
Abstract: Immune thrombocytopenia (ITP) is an autoimmune disorder characterized by a reduced number of circulating platelets due to immune-mediated destruction and decreased platelet production in the bone marrow. Thrombopoietin receptor agonists (TPO-RAs) are highly effective and widely used in the treatment of patients with steroid treatment failure or dependency. Avatrombopag represents a new supplement to the TPO-RAs family. It was originally approved for the treatment of thrombocytopenia in patients with chronic liver disease who are scheduled to undergo an invasive procedure. However, labeled indications for avatrombopag have been relatively recently expanded to include treatment of chronic ITP in adults with insufficient response to the previous treatments. In this article, we provide an overview of pharmacodynamics and pharmacokinetics of avatrombopag as well as results of the clinical trials related to safety and efficacy of avatrombopag with a perspective on current clinical use. Available data so far suggests that avatrombopag can be effectively used in ITP patients and has a favorable safety profile. Though further studies are needed to affirm the efficacy and safety, avatrombopag has the potential to become a TPO agonist of choice for many patients with ITP.
Keywords: ITP, thrombopoietin receptor agonists, avatrombopag
Introduction
Immune thrombocytopenia (ITP) is an autoimmune disorder characterized by immune-mediated platelet destruction combined with impaired platelet production in the bone marrow resulting in thrombocytopenia and increased risk of bleeding.1 Primary ITP is characterized by isolated thrombocytopenia (platelet counts of less than 100x109/L) in the absence of other causes. Clinical manifestations of ITP range from being asymptomatic to having mild mucocutaneous bleeding to more severe life-threatening bleeding from various organs, occasionally. Other symptoms include fatigue and various psychological manifestation.2,3 The risk of bleeding depends on many factors but most importantly on the number of platelets in the peripheral blood. Generally, the risk of life-threatening and fatal hemorrhage is highest when platelet counts drop below 10–20x109/L.4
The pathological mechanisms behind ITP are multifactorial and complex. Antiplatelet antibodies towards the different platelet receptors (such as GPIa/GPIIb/IIIa; GPIb/IX/V) seem to play a central role. In addition, T-cell mediated platelet destruction also plays a role in the pathophysiology of the disease. Antibodies facilitate the destruction of platelets by phagocytes through the Fc-dependent mechanism primary in the spleen. In addition, they lead to impaired megakaryocytic functions resulting in decreased platelet production.5
Studies have shown that thrombopoietin (TPO) levels in ITP patients are unexpectedly low for the degree of thrombocytopenia and are usually normal or only slightly elevated.6 Pathogenetic mechanisms behind remain not fully understood. TPO levels vary inversely with megakaryocyte- and platelet mass.7 It has also been suggested that platelet desialylation in the presence of platelet antibodies and the direct GPIba antibody-mediated reduction of hepatic TPO regulation may also play a role.8
The annual incidence of adult ITP is estimated to be approximately 3–6/100,000 subjects.9,10 The prevalence in adults is 9.5/100,000.11 Bleeding remains the major cause of morbidity and mortality in ITP.12 As such, the main goal of the therapy is to maintain a safe (hemostatic) platelet count to prevent clinically significant bleeding episodes. Treatment, in general, should be considered when platelet counts drop to under 20–30x109/L.13,14 Occasionally, treatment is indicated in patients with higher platelet count eg, 50x109/L in those on anticoagulation.
Corticosteroids and IVIG have remained a cornerstone of first-line treatment in ITP for many years. Unfortunately, despite a relatively high initial response rate, only about 30% of all steroid-treated patients can achieve long-term remissions. Thus, many adult ITP patients will require subsequent therapy.
Current guidelines from the American Society of Hematology recommend thrombopoietin receptor agonists (TPO-RAs) as a preferred second-line therapy for patients who fail to respond to steroids or become steroid-dependent.13 That is in line with the recommendations from the Updated international consensus report,14 which suggests rituximab or TPO-RAs as non-surgical treatment alternatives.
Thrombopoietin Receptor Agonists – Mechanism of Action, Development, and Role in ITP
Thrombopoietin (or Mpl-ligand) is a glycoprotein produced primarily on the liver and in minimal amounts in the kidney and bone marrow. Thrombopoietin acts as a regulator of platelet production through binding to thrombopoietin (TPO) receptor (c-Mpl)15 located at the megakaryocyte surface. This provides a powerful stimulus for the proliferation and differentiation of megakaryocytes by inducing intracellular mechanisms such as JAK2/STAT5, STAT 3, PI3K/Akt, ERK, MAPK/STAT1, thereby leading to increased platelet production. The process is regulated through the autoregulatory loop by binding circulating TPO to high-affinity receptors on platelets.16
Understanding of mechanisms of thrombopoiesis led to the development of the first generation thrombopoietic agents, recombinant thrombopoietin (rhTPO). Unfortunately, its further development was soon restrained due to immunogenicity resulting in the emergence of neutralizing TPO-antibodies which cross-reacted with endogenous TPO17 causing severe thrombocytopenia in healthy volunteers.
The second generation of thrombopoietic agents that were structurally different from the native TPO and thus should not trigger antibody production was subsequently developed and licensed for ITP around a decade ago. To date, there are four different thrombopoietin receptor agonists (romiplostim, eltrombopag, avatrombopag, and lusutrombopag), the first three are approved for the treatment in ITP. In addition, a fifth agent, a recombinant TPO that is only licensed in China. While primarily developed for the treatment of ITP, according to the label romiplostim and eltrombopag are licensed for several other conditions such as hematopoietic syndrome of acute radiation syndrome (romiplostim), HCV infection associated thrombocytopenia and aplastic anemia (eltrombopag).
TPO-RAs have a completely different mechanism of action compared to the other known treatment modalities in ITP. They are the only alternative that acts by directly increasing platelet production and not by inducing immunosuppression. Their introduction has dramatically changed our approach to the management of ITP.
Table 1 provides an overview of different TPO-RAs. TPO-RAs are highly effective in elevating platelet counts and prevention bleeding events in ITP. Romiplostim and eltrombopag have shown excellent short and long-term efficacy with a response rate of up to 88%, and in general, very well tolerated.18
 |
Table 1 Overview of Available Thrombopoietin Receptor Agonists and Their Key Characteristics |
Generally, TPO-RAs are considered as maintenance therapy ie patients need to take the medication continuously to maintain response. However, several recent reports indicate that up to 30% of the patients can discontinue TPO_RA without losing response. Thus, a proportion of ITP patients can discontinue TPO- RAs without losing response.19–21
Chemical Structure, Pharmacokinetics, and Dynamics of Avatrombopag
Chemistry and Administration
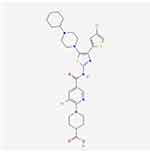
Avatrombopag is a highly lipophilic small molecule (Figure 1) and, therefore, is usually prepared as the salt, avatrombopag maleate.22 It is provided as an immediate-release tablet. Avatrombopag is produced as a 20 mg tablet.
Pharmacokinetics/Metabolism
The basis for dosage and treatment schedule in the Phase II studies of avatrombopag was provided from Phase I studies in healthy individuals, such as two double-blind, dose-rising, placebo-controlled Phase 1 studies.23 It has been shown that the increase in platelet counts is dose-dependent with a maximum increase at days 13–16 related to peak plasma concentrations. Significant platelet counts increase from baseline required avatrombopag doses ≥ 10 mg. The median time to achieve a maximum concentration and a half-life was 5–8 hours and 16–19 hours, which were dose-proportional after administration of single doses, with no difference between various ethnic groups.23
Drug absorption did not vary with food intake. Indeed, unlike eltrombopag, the administration of avatrombopag with food reduced pharmacokinetic variability. Even maximum concentration achieved with avatrombopag differed between ethnics (Figure 2) but, again, unlike eltrombopag, not the area below the concentration-time curve.24
 |
Figure 2 Mean (+SE) plasma avatrombopag concentration-time profile following single oral doses of 20, 40, and 60 mg under fed conditions in Japanese black dot line) and white (solid blue line) subjects. Semi-log scale. Reprinted from Nomoto M, Pastino G, Rege B, Aluri J, Ferry J, Han D. Pharmacokinetics, Pharmacodynamics, Pharmacogenomics, Safety, and Tolerability of Avatrombopag in Healthy Japanese and White Subjects. Clin Pharmacol Drug Dev. 2018;7:188–195. © 2017, The American College of Clinical Pharmacology.24 Abbreviation: SE, standard error. |
T1/2 for avatrombopag is independent of the dose and ranges between 18–24 hours.23
Avatrombopag is highly bound to plasma proteins (> 95%) and is metabolized mainly via hepatic enzymes CYP2C9 and CYP3A4. The studies suggest25 that CYP2C is a predominant enzyme in the metabolism of avatrombopag, and based on this knowledge, dose adjustment is advised with concomitant use of fluconazole. The drug is primarily excreted through the GI tract with 34% unmetabolized drug. Only 6% is excreted via the kidney.
Pharmacodynamics
Early in vitro studies of avatrombopag (AKR-501), similary to eltrombopag, showed a highly species-specific activity, limited to humans and chimpanzees. The drug mimicked to some degree the effect of endogenous TPO and specifically stimulated megakaryocyte colony formation from human 34+ positive hematopoietic cells. Avatrombopag acts simultaneously with TPO without inhibiting TPO binding to the receptor.26 In mice models transplanted with human fetal liver CD34+cells, daily oral administration of avatrombopag led to a dose-dependent increase in platelet counts.27 Later a randomized controlled trial in healthy Japanese and Caucasian individuals confirmed these observations showing that avatrombopag increased platelet counts in a dose-dependent fashion to maximum from baseline by day 11 and returned to baseline at around day 2724 (Figure 2). In patients with liver disease it has been shown that avatrombopag increased platelet counts but did not result in platelet activation as opposite to TPO.28
There are no studies in ITP patients directly comparing the potency of avatrombopag to romiplostim and eltrombopag. However, based on indirect comparison of all three TPO- RAs from phase 1 studies in healthy individuals, presented by Kuter,29 it seems that romiplostim is more potent after a single dose administration than a single dose administration of avatrombopag. The data on single-dose administration is not available for eltrombopag, but the latter seems less potent than avatrombopag with multiple-dose administrations.
Overview of pharmacodynamics and pharmacokinetics is provided in Table 2.
 |
Table 2 Pharmacokinetic and Pharmacodynamics of TPO-RAs Used in Treatment of ITP |
Efficacy of Avatrombopag in Adult ITP Patients
To date, there have been two randomized controlled trials of avatrombopag in the adult ITP population.
The first trial was a Phase II, double-blind, randomized dose-ranging, placebo-controlled parallel-group study.30 Those who completed this trial were eligible to continue further in the extension study (study 004, NTC00625443).
This study included 64 patients in 19 centers across the US. The main criteria were the age of 18 years or older with a diagnosis of ITP of 3 months or longer, failure to respond, or relapse after at least one line of therapy. A platelet count of <30 x 109/L was required for inclusion, whereas those on steroid therapy must have had platelet counts <50 x 109/L. Splenectomized patients were eligible for inclusion only if splenectomy was carried out over four weeks prior to the randomization. Patients randomly received either a once-daily fixed-dose of avatrombopag (2,5 mg, 5 mg, 10 mg, or 20 mg) or placebo and received treatment for 28 days.
No changes in concomitant medications were allowed during the study, and administration of rescue medication led to the withdrawal. During this study, patients received treatment on an empty stomach due to a lack of the results from the food studies at that time. The primary endpoint was response rates, defined as the proportion of subjects who achieved platelet counts ≥50 x 109/L and a minimum increase of 20 x 109/L above baseline at day 28. Response rates for the various avatrombopag groups were: 13% (2.5 mg), 53% (5 mg), 50% (10 mg) and 80% (20 mg) as compared to 0% in the placebo group. Like for the phase I studies, there was a dose-dependent response with regards to median platelet count.
Response rate for avatrombopag and placebo cohorts and median platelet count over time by treatment group in the randomized study is shown in Figure 3.
 |
Figure 3 Response rate for avatrombopag and placebo cohorts and median [Q1, Q3] PC over time by treatment group in the randomized study. Response rate (proportion of subjects who achieved PC ≥50 × 109/L and minimum PC increase of 20 × 109/L above baseline) at each time point for avatrombopag and placebo cohorts (A) and median [Q1, Q3] platelet count over time by treatment group (B) in the randomized study (LOCF). For each median (second quartile) PC presented in panel B, error bars denote the first (lower value of bar) and third (upper value of bar) quartiles. LOCF, the last observation carried forward. Reprinted from Blood, 123, Bussel JB, Kuter DJ, Aledort LM et al. A randomized trial of avatrombopag, an investigational thrombopoietin-receptor agonist, in persistent and chronic immune thrombocytopenia, 3887–3894, Copyright 2014, with permission from Elsevier.30 Abbreviation: PC, platelet count. |
Fifty-three patients completed this trial were available for the 24 weeks extension period after day 28. The extension part of the study aimed to assess participants with durable response defined as platelet counts responses at ≥75% of measurements over the last 14 weeks of the study and had a minimum of 3 platelet counts measurements. The main objective was to assess the safety and tolerability of avatrombopag. Twenty-five responders at day 28 continued in the extension study, receiving the same dose of avatrombopag /placebo for 24 weeks. Non-responders (28 patients) started an open-label treatment with avatrombopag at a starting dose of 10 mg, which could be increased to a maximum of 40 mg/ daily to maintain platelet counts of > 50 x 109/L. Overall, twenty-eight patients (53%) achieved a durable response. 40 (76%) achieved an overall response, defined as a durable or a transient platelet response. Ten out of 28 non-responders from the main study (36%) demonstrated durable responses in the extension study. Subgroups analyses showed that patients with platelet counts >15x109/l at baseline and unsplenectomized were more likely to achieve durable responses. However, the number of patients in this sub-analysis was too small to make definite conclusions.
Twenty-four participants in the extension study (45%) used steroids at the entry. Of these, 13 participants had a ≥ 50% steroid dose reduction, and 8 stopped steroids. In addition, the proportion of the subjects with bleeding episodes in both the randomized and the extension part of the study was reduced by week 14.
The second study was a 6-month multicenter, randomized double-blinded, parallel-group Phase III trial with an open-label extension phase.31 The trial took place in 27 sites in Australia and New Zealand, Africa, Asia, and Europe. The study evaluated the efficacy and safety of daily dose of avatrombopag in chronic ITP patients. The main inclusion criteria were age 18 or older, with a chronic primary ITP (at least 12 months of duration) and persistent platelet counts lower than 30x 109/L, patients could be included regardless of their splenectomy status. At least one previous treatment line for ITP therapy should have been received. In addition, the patient must have had an initial response to at least one therapy or have had a bone marrow examination within three years before inclusion to rule out other causes of thrombocytopenia. Forty-nine patients were stratified according to their platelet counts, splenectomy status, and use of concomitant medication. They were randomly assigned to receive either avatrombopag 20 mg daily or placebo treatment (double-blinded randomization 2:1). The dose could be titrated to a maximum of 40 mg to a minimum of 5 mg with the goal for platelet counts of between 50 and 150 x 109/L. In a core study, a dose reduction in concomitant medication was allowed until week 18, while the primary endpoint was evaluated during the maintenance phase (weeks 18–26). The primary endpoint, cumulative number of weeks with platelet counts of at least 50 x 109/L in the absence of rescue therapy, was significantly longer with avatrombopag (mean 12 in the avatrombopag group and 0.1 weeks in the placebo group, p<0.0001). In addition, patients in the avatrombopag group had a 66.8% platelet response on day 8 compared to no response in the placebo group. Furthermore, 33% of the patients were able to reduce their concomitant therapy compared to none in a placebo group. However, statistical significance was not reached due to the low number of participants.
The patients who discontinued the core study due to lack of response and patients who continued the core study throughout the maintenance phase were invited for enrollment into a 90- week extension period followed by the 4 weeks tapering period and 4 weeks observational period. The main goal in the extension period was to study the long-term safety and efficacy assessed by the bleeding rate, platelet counts response rate and use of rescue therapy. A short overview of both studies is provided in Table 3.
 |
Table 3 Efficacy of Avatrombopag in ITP in Phase II and III Trials |
The incidence of bleeding events was lower in the avatrombopag group when adjusted to the longer mean exposure time. Most bleeding events in both groups were grade 1 except two grade 2 and one grade 3 bleeding events observed in the avatrombopag treated group.
Quality of life (HQoL) was evaluated neither in phase II nor phase III study.
One phase III study aimed to evaluate the efficacy of avatrombopag versus eltrombopag (NTC01433978) was terminated early due to significant enrollment challenges.
There is also an ongoing phase III study (NTC04516967) is aimed to evaluate the efficacy of the drug in the pediatric population (children ≥ 6 months).
A Phase IV study to evaluate HQoL and the safety of avatrombopag after switching from other TPO-RAs (NCT04638829) is ongoing. Another real-world effectiveness study of avatrombopag in the ITP patient population (NTC04943042) is planned.
Safety of Avatrombopag in ITP Clinical Trials
Based on phase II and III trials, avatrombopag appeared to be generally safe and showed a favorable side effect profile in the ITP population.
In a phase II study,30 the most frequent adverse events (AEs) were epistaxis, headache, fatigue, and confusion. There were no differences in the rate of occurrence of AEs in avatrombopag and placebo groups during the randomized part of the study. All AEs, except increased platelet count above the desired level, were dose-independent. Increased platelet count was reported in 7 patients during the entire study resulting in permanent drug discontinuation in 5.
Recurrence of thrombocytopenia after discontinuation of avatrombopag (defined as a drop in platelet counts <10x109/L) occurred in 14% of individuals during the follow-up period, most of this occurred during week 2.
In total, five thromboembolic events occurred in 6% (4 patients) during the phase II study, with one participant experiencing two different thromboembolic events. Those events were iliac DVT, superficial thrombophlebitis, myocardial infarction, stroke, and retinal artery occlusion. Two of these events resulted in permanent study drug discontinuation. There were additional risk factors of thrombosis in three of the participants.
There was some transient liver function tests elevation with two grade 2 or 3 events in study subjects, but with no dose-dependent trends.
In total, eight subjects (13%) had temporary discontinuation of the drug across the whole study due to AE. Most were due to an increase in platelet counts (5 patients), which was the only AE resulting in permanent treatment discontinuation in > 1 patient. Permanent discontinuation of the treatment due to various reasons occurred in 10 patients (16%).
The most common and severe adverse events during the phase II trial are shown in Table 4.
 |
Table 4 Most Common and Severe (Grade 3 or 4) AEs During the Randomized and Extension Study (Total Number of Patients N=64) |
During the core phase III study,31 the most common treatment-emergent adverse events (TEAE) were headache, contusion, upper respiratory tract infection, arthralgia, epistaxis, fatigue, gingival bleeding, and petechiae, these were higher in the avatrombopag group. However, when exposure-adjusted, the incidence rates were comparable or lower than in the placebo group. In the extension study, there were some additional AEs: hypertension, pharyngitis, and nasopharyngitis.
Adverse events of special interests (AESI) were reported only in the avatrombopag-treated group. In a core study, three patients (9, 4%) experienced thromboembolic events and one additional patient in an extension phase. Three of these patients had several additional risk factors. One patient had liver tests elevation, which was considered unrelated to the study drug treatment. Only one avatrombopag patient-reported bleeding event or recurrence of thrombocytopenia during the study period. No cases of gastric atrophy or bone marrow fibrosis were reported.
An overview of treatment-emergent adverse events grade 3–4 and serious adverse events in the phase III study is shown in Table 5.
 |
Table 5 Treatment-Emergent AE Grade 3–4 and Serious AEs (Total Number of Patients N=47) in a Core Study± Extension Phase |
No deaths were reported during the study period in any of the studies.
Labeled Indications and Practical Considerations
Avatrombopag is approved in the US and Europe for use for the following indications:
- Treatment of thrombocytopenia in adult patients with chronic immune thrombocytopenia with insufficient response to a previous treatment.
Initial dosing is 20 mg once daily. Max dose is 40 mg daily. The dose must be adjusted in case of significant drug interactions
Treatments should start 10–13 days before the scheduled procedure. The procedure must be undergone 5–8 days after the last avatrombopag dose. Patients with platelet counts of 40–60x109/L should take 40 mg avatrombopag once daily for 5 consecutive days; those with platelet counts <40x109/L should take 60 mg avatrombopag for 5 consecutive days.
No dose adjustments due to drug interactions are required for this patient group.
Avatrombopag should be administered with food.
Management of Particular Populations
Patients with renal and hepatic impairment: there is no need for dose adjustments in patients with reduced renal function. Dialysis is not expected to enhance avatrombopag elimination.
Patients with hepatic impairment required no dose adjustment as hepatic impairment has no meaningful effect on pharmacokinetics.
Elderly: no dose adjustment is necessary compared to adult dosing.
Pediatric population: to date, there are no data on the safety and efficacy of avatrombopag in this population. A Phase 3 study with avatrombopag for the treatment of ITP in children ≥6 months is ongoing (NTC04516967).
Clinical Perspective in the Use of Avatrombopag
The development of TPO-RAs represents one of the major advances in the management of ITP during the last 15 years. Romiplostim and eltrombopag were first licensed for adult chronic ITP in 2008. However, their label has changed several times, and consequently, their use has gradually increased to become among the most widely used ITP treatment modalities in most countries.32 According to the updated ASH guidelines13 TPO-RA are now preferred over rituximab in patients who do not respond adequately or become dependent on corticosteroids.
In addition to their high and durable response rates, these agents have shown good safety profiles and high tolerability. Nevertheless, both romiplostim and eltrombopag have some drawbacks related to the safety profiles or mode of administration. Romiplostim is a parenteral medication with the potential of administering an incorrect dose. In addition, in the US, the drug is still administered by health care professionals, and as such, it places some restrictions on a patient’s life. Even in Europe, where the drug is licensed for self-treatment, around 2/3 of the patients may have difficulties self-administration and may require help from family members or health care professionals.33
For eltrombopag, limitations are mainly due to dietary restrictions, which may affect the efficacy and impact compliance. In addition, its use is restricted in patients with liver disease due to its hepatotoxicity, and it also carries a Black Box Warning due to the risk of decompensation in patients with chronic hepatitis C treated with ribavirin and interferon. In patients without liver disease, its use requires frequent and regular monitoring of liver function particularly during the first year after its initiation where at least monthly blood tests are required.
Avatrombopag is administered orally, once a day, requiring no dietary restrictions which obviously present an advantage over the two other preparations. In contrast to eltrombopag, hepatic events have been observed in only 3% of avatrombopag treated patients with no documented increase in bilirubin of grade 2 or higher34 representing another advantage over eltrombopag. The safety of avatrombopag has also been confirmed in patients with chronic liver disease.35 That makes avatrombopag an attractive alternative in treating ITP patients with underlying liver disorders. Based on phase II and III studies and despite a limited number of patients, it seems that avatrombopag, like both romiplostim and eltrombopag, is an effective and safe treatment alternative in the adult ITP population. As with the other two TPO RAs, avatrombopag yields high response rates with an impressive rise in platelet counts to >50x109/L already at day 8 in 66% of the patients. However, there are no studies comparing these agents’ relative efficacy and safety head-to-head.
The main limitation of avatrombopag is currently the limited safety data available. One worrying signal is the rates of thromboembolic events observed in phase II and III studies of avatrombopag, which seems higher than that reported for the two other TPO agonists. That may be due to the low number of patients, but this issue should be carefully accessed in further trials. Until more data is available, a vigilant approach has to be adopted particularly concerning those at high risk for thromboembolism, eg patients with multiple risk factors for TE.
In general, the number of patients included in both studies is relatively small and more data is required to affirm the safety of this agent. Hopefully, the ongoing studies, including phase IV study of the effectiveness and safety of avatrombopag in the ITP population, will bridge some of the current knowledge gaps with avatrombopag (NCT04943042).
Currently, avatrombopag is licensed for adult patients with chronic ITP ie ITP > 12 months since diagnosis. We do not believe that this label will be changed before more data on safety and efficacy of avatrombopag is available. Since the romiplostim and eltrombopag are currently licensed for ITP of less than one year, this means that most patients will start on these preparations first. However, if a patient fails to respond or loses response to one or both thrombopoietic agents and has chronic ITP, switching to avatrombopag is often effective and should be attempted. Several publications demonstrate the efficacy of switching between TPO-RAs when switching was conducted either due to the lack of efficacy, adverse events or platelets fluctuations.36 However, available data applies and supports first and foremost switching between romiplostim and eltrombopag. There is very limited experience regarding switching to avatrombopag from the two other TPO RAs. Al-Samkari et al reported data on 45 heavily pretreated ITP switched from romiplostim or eltrombopag to avatrombopag. The reasons for switching were inconvenience (51%), ineffectiveness (31%), or adverse events (18%). In patients switched due to ineffectiveness, the median platelet count on avatrombopag was 114 x 109/L compared to 28 x 109/L before the switch. Of 19 patients who used concomitant steroids, 12 (63%) discontinued steroids, and 32% reduced dose.37 There is also an ongoing study on safety and treatment satisfaction after switching to avatrombopag from other TPO-RAs (NCT04638829). In our practice, we switch to avatrombopag in those who have failed or lost response to any of the two other TPO-RA.
There is no reason to believe that avatrombopag would have different safety or efficacy profiles if used at an earlier stage, eg during the persistent phase of ITP or even in the newly diagnosed ITP after the failure of corticosteroids. In fact, according to the international consensus report, it is enlisted among the subsequent therapies with robust evidence, thus allowing for use at an earlier stage.14 Restrictions imposed by local regulatory authorities, reimbursement bodies, and national guidelines will be determinant in that regard.38
Generally, we recommend administration of avatrombopag according to the label. Only in an exceptional case we deviate from that eg, if no other treatment is available or if the patient prefers oral medication but cannot take eltrombopag because of drug interaction or expected difficulty because of dietary restrictions. However, when the patient can be offered any of the three available agents, our practice is to let the patient decide after presenting the three options with their pros and cons. Despite the limited evidence on safety as the main limitation, the patients frequently choose avatrombopag because of the ease of administration. Once the response is achieved, the dose should be adjusted to the lowest to maintain platelet count 50–150 x 109/L according to the schedule recommended by the manufacturer. Not infrequently, we manage to reduce the dose to every second or third day, which may substantially reduce the cost of treatment.
In contrast, for patients who do not respond to 20 mg daily within a week or two, a dose increment to 40 mg is recommended. If no response is achieved within 6 weeks, it is unlikely that the patient will respond to avatrombopag monotherapy. In this case, one should either switch to another TPO-RA or different treatment or combine avatrombopag with an immunosuppressive agent, like prednisolone or mycophenolate mofetil.39 We usually add a small dose of prednisolone (2.5–5 mg) first if satisfactory results are not achieved. This combination often results in response.38
To summarize, avatrombopag seems to be a valuable addition to the TPO treatment specter, but further studies are needed to affirm the efficacy, safety, and effect on HQoL. Considering apparent low toxicity demonstrated in studies and ease of administration, avatrombopag has the potential to become a TPO agonist of choice for many patients with ITP.
Disclosure
Waleed Ghanima reports grants from Bayer, Pfizer/BMS; ad board/honoraria from Argenix, Grifols, Amgen, MSD, Novartis, Bayer, Pfizer, Principia, Sanofi, Sobi, UCB.
Galina Tsykunova reports consultancy/honoraria from Amgen, Janssen; Ablynx, Sanofi, Sobi; research funding from Janssen. The authors report no other conflicts of interest in this work.
References
1. Cooper N, Ghanima W. Immune thrombocytopenia. N Engl J Med. 2019;381:945–955.
2. Cooper N, Kruse A, Kruse C, et al. Immune thrombocytopenia (ITP) World Impact Survey (iWISh): patient and physician perceptions of diagnosis, signs and symptoms, and treatment. Am J Hematol. 2021;96:188–198.
3. Cooper N, Kruse A, Kruse C, et al. Immune thrombocytopenia (ITP) World Impact Survey (I-WISh): impact of ITP on health-related quality of life. Am J Hematol. 2021;96:199–207.
4. Piel-Julian ML, Mahevas M, Germain J, et al. Risk factors for bleeding, including platelet count threshold, in newly diagnosed immune thrombocytopenia adults. J Thromb Haemost. 2018;16:1830–1842.
5. Zufferey A, Kapur R, Semple JW. Pathogenesis and therapeutic mechanisms in Immune Thrombocytopenia (ITP). J Clin Med. 2017;6:16.
6. Kosugi S, Kurata Y, Tomiyama Y, et al. Circulating thrombopoietin level in chronic immune thrombocytopenic purpura. Br J Haematol. 1996;93:704–706.
7. Makar RS, Zhukov OS, Sahud MA, Kuter DJ. Thrombopoietin levels in patients with disorders of platelet production: diagnostic potential and utility in predicting response to TPO receptor agonists. Am J Hematol. 2013;88:1041–1044.
8. Xu M, Li J, Neves MAD, et al. GPIbalpha is required for platelet-mediated hepatic thrombopoietin generation. Blood. 2018;132:622–634.
9. Schoonen WM, Kucera G, Coalson J, et al. Epidemiology of immune thrombocytopenic purpura in the general practice research database. Br J Haematol. 2009;145:235–244.
10. Frederiksen H, Schmidt K. The incidence of idiopathic thrombocytopenic purpura in adults increases with age. Blood. 1999;94(3):909–913.
11. Christiansen CF, Bahmanyar S, Ghanima W, et al. Chronic immune thrombocytopenia in Denmark, Sweden and Norway: the Nordic Country Patient Registry for Romiplostim. EClinicalMedicine. 2019;14:80–87.
12. Adelborg K, Kristensen NR, Norgaard M, et al. Cardiovascular and bleeding outcomes in a population-based cohort of patients with chronic immune thrombocytopenia. J Thromb Haemost. 2019;17:912–924.
13. Neunert C, Terrell DR, Arnold DM, et al. American Society of Hematology 2019 guidelines for immune thrombocytopenia. Blood Adv. 2019;3:3829–3866.
14. Provan D, Arnold DM, Bussel JB, et al. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019;3:3780–3817.
15. de Sauvage FJ, Hass PE, Spencer SD, et al. Stimulation of megakaryocytopoiesis and thrombopoiesis by the c-Mpl ligand. Nature. 1994;369:533–538.
16. Kaushansky K. The molecular mechanisms that control thrombopoiesis. J Clin Invest. 2005;115:3339–3347.
17. Li J, Yang C, Xia Y, et al. Thrombocytopenia caused by the development of antibodies to thrombopoietin. Blood. 2001;98:3241–3248.
18. Wang L, Gao Z, Chen XP, et al. Efficacy and safety of thrombopoietin receptor agonists in patients with primary immune thrombocytopenia: a systematic review and meta-analysis. Sci Rep. 2016;6:39003.
19. Gonzalez-Lopez TJ, Pascual C, Alvarez-Roman MT, et al. Successful discontinuation of eltrombopag after complete remission in patients with primary immune thrombocytopenia. Am J Hematol. 2015;90:E40–E43.
20. Cervinek L, Mayer J, Doubek M. Sustained remission of chronic immune thrombocytopenia after discontinuation of treatment with thrombopoietin-receptor agonists in adults. Int J Hematol. 2015;102:7–11.
21. Mahevas M, Fain O, Ebbo M, et al. The temporary use of thrombopoietin-receptor agonists may induce a prolonged remission in adult chronic immune thrombocytopenia. Results of a French observational study. Br J Haematol. 2014;165:865–869.
22. Al-Samkari H. Avatrombopag maleate for the treatment of periprocedural thrombocytopenia in patients with chronic liver disease. Drugs Today. 2018;54:647–655.
23. Kuter DJ, Allen LF. Avatrombopag, an oral thrombopoietin receptor agonist: results of two double-blind, dose-rising, placebo-controlled Phase 1 studies. Br J Haematol. 2018;183:466–478.
24. Nomoto M, Pastino G, Rege B, Aluri J, Ferry J, Han D. Pharmacokinetics, pharmacodynamics, pharmacogenomics, safety, and tolerability of avatrombopag in healthy Japanese and white subjects. Clin Pharmacol Drug Dev. 2018;7:188–195.
25. Nomoto M, Zamora CA, Schuck E, et al. Pharmacokinetic/pharmacodynamic drug-drug interactions of avatrombopag when coadministered with dual or selective CYP2C9 and CYP3A interacting drugs. Br J Clin Pharmacol. 2018;84:952–960.
26. Fukushima-Shintani M, Suzuki K, Iwatsuki Y, et al. AKR-501 (YM477) in combination with thrombopoietin enhances human megakaryocytopoiesis. Exp Hematol. 2008;36:1337–1342.
27. Fukushima-Shintani M, Suzuki K, Iwatsuki Y, et al. AKR-501 (YM477) a novel orally-active thrombopoietin receptor agonist. Eur J Haematol. 2009;82:247–254.
28. Michelson AD, Smolensky Koganov E, Forde EE, Carmichael SL, Frelinger AL
29. Kuter DJ. The structure, function, and clinical use of the thrombopoietin receptor agonist avatrombopag. Blood Rev. 2021:100909. doi:10.1016/j.blre.2021.100909
30. Bussel JB, Kuter DJ, Aledort LM, et al. A randomized trial of avatrombopag, an investigational thrombopoietin-receptor agonist, in persistent and chronic immune thrombocytopenia. Blood. 2014;123:3887–3894.
31. Jurczak W, Chojnowski K, Mayer J, et al. Phase 3 randomised study of avatrombopag, a novel thrombopoietin receptor agonist for the treatment of chronic immune thrombocytopenia. Br J Haematol. 2018;183:479–490.
32. Ghanima W, Cooper N, Rodeghiero F, Godeau B, Bussel JB. Thrombopoietin receptor agonists: ten years later. Haematologica. 2019;104:1112–1123.
33. Steurer M, Quittet P, Papadaki HA, et al. A large observational study of patients with primary immune thrombocytopenia receiving romiplostim in European clinical practice. Eur J Haematol. 2016;98:112–120.
34. Terrault NA, Hassanein T, Howell CD, et al. Phase II study of avatrombopag in thrombocytopenic patients with cirrhosis undergoing an elective procedure. J Hepatol. 2014;61:1253–1259.
35. Terrault N, Chen YC, Izumi N, et al. Avatrombopag before procedures reduces need for platelet transfusion in patients with chronic liver disease and thrombocytopenia. Gastroenterology. 2018;155:705–718.
36. Gonzalez-Porras JR, Godeau B, Carpenedo M. Switching thrombopoietin receptor agonist treatments in patients with primary immune thrombocytopenia. Ther Adv Hematol. 2019;10:2040620719837906.
37. Al-Samkari H, Jiang D, Gernsheimer T, et al. Adults with immune thrombocytopenia who switched to avatrombopag following prior treatment with eltrombopag or romiplostim: A multicentre US study. J Haematol. 2022. doi:10.1111/bjh.18081
38. Ghanima W, Gernsheimer T, Kuter DJ. How I treat primary ITP in adult patients who are unresponsive to or dependent on corticosteroid treatment. Blood. 2021;137:2736–2744.
39. Miltiadous O, Hou M, Bussel JB. Identifying and treating refractory ITP: difficulty in diagnosis and role of combination treatment. Blood. 2020;135:472–490.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.