Back to Journals » OncoTargets and Therapy » Volume 13
Autologous Cytokine-Induced Killer Cell Immunotherapy for Patients with High-Risk Diffuse Large B Cell Lymphoma After the First Complete Remission
Authors Zhou M, Wang J , Li CP, Xu JY, Chen B
Received 19 March 2020
Accepted for publication 26 May 2020
Published 22 June 2020 Volume 2020:13 Pages 5879—5885
DOI https://doi.org/10.2147/OTT.S254291
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Federico Perche
Min Zhou,1,* Jing Wang,1,* Cui-Ping Li,2 Jing-Yan Xu,1 Bing Chen1
1Department of Hematology, The Affiliated DrumTower Hospital of Nanjing University Medical School, Nanjing, Jiangsu, People’s Republic of China; 2Department of Transfusion, BenQ Medical Center, Nanjing Medical University, Nanjing, Jiangsu, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Bing Chen Tel/ Fax +86-25-83106666-61441
Email [email protected]
Purpose: To evaluate whether autologous cytokine-induced killer (CIK) cell immunotherapy improves the prognosis of patients with high-risk diffuse large B cell lymphoma (DLBCL) after the first complete remission (CR).
Patients and Methods: Peripheral blood mononuclear cells (PBMCs) were extracted from 20 patients with high-risk DLBCL (IPI≥ 3) after the first CR. Twenty CR patients who were age- and sex-matched during the same period were selected as controls. PBMCs were cultured with IFN-γ, IL-2 and anti-CD3 mAb to generate CIK cells. These obtained cells were then transfused back into the patients; the transfusion was repeated every 3 months up to a total of four courses. Changes in peripheral blood lymphocyte subgroups and survival were assessed.
Results: Compared with the baseline proportions, the proportion of CD3+ T cells, CD3+CD8+ T cells, and NK cells in the peripheral blood were significantly higher after transfusions (p< 0.05). The 5-year DFS was improved from 45.0 ± 11.1% to 79.3 ± 9.2% in the CIK group (HR favoring CIK, 0.29; 95% CI, 0.09 to 0.92; p = 0.035), and the 5-year OS was estimated at 90 ± 6.7% for CIK versus 55 ± 11.1% for control (HR favoring CIK, 0.20; 95% CI, 0.04 to 0.93; p = 0.040). No severe side effects were observed related to CIK treatment.
Conclusion: Autologous CIK cell immunotherapy has emerged as a safe and efficacious option to improve the prognosis of patients with high-risk DLBCL after the first CR.
Keywords: diffuse large B cell lymphoma, high risk, cytokine-induced killer cells, immunotherapy
Introduction
Diffuse large B cell lymphoma (DLBCL) is the most frequent lymphoma subtype and accounts for more than 30% of all newly diagnosed lymphoma cases worldwide.1 Chemotherapy consisting of rituximab, cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) is considered standard first-line therapy for patients with DLBCL.2 However, despite improvement in first-line treatment, approximately one-third of patients will relapse or require retreatment in the first 2 years.3 The International Prognostic Index (IPI) has been the most important tool for estimating the prognosis of patients with DLBCL in recent decades.4 Patients with an IPI ≥3 are at higher risk for relapse after a complete response (CR) to first-line rituximab-based chemotherapy than patients with an IPI <3.5 Patients with a score of 3–5 had a 3-year overall survival (OS) of 63% in our previous study.6 It is essential to find a treatment to maintain CR, delay relapse, and prolong survival.
Adoptive immunotherapy is a promising novel approach for the treatment of cancer. Cytokine-induced killer (CIK) cells, a heterogeneous immune cell population induced by specific stimulation (such as rhIFN-a, rhIL-2, and anti-CD3 monoclonal antibody) of peripheral blood mononuclear cells (PBMCs), is more effective in recovering the host immunity and killing residual chemo-resistant cancer cells.7,8 CIK cells have several characters of increasing proliferation, MHC-unrestricted cytotoxic activity against a broad range of tumors, and minimal toxicity.9–11 CIK cells may be an alternative immunotherapeutic strategy for DLBCL.12 Data from a meta-analysis showing that maintenance therapy in patients with DLBCL achieving CR or PR could decrease relapse rate.13 In the present study, we aimed to examine the efficacy and safety of autologous CIK cell treatment as maintenance therapy in patients with high-risk DLBCL (IPI≥3) after the first CR.
Patients and Methods
Patient Selection
Adult patients over 18 years old reaching the first CR were eligible for this study. Patients with transformed lymphoma, evidence of central nervous system involvement, or positivity for human immunodeficiency virus were excluded. We retrospectively analyzed 20 patients receiving CIK therapy from 2010 to 2014 at Nanjing Drum Tower Hospital. Twenty CR patients who were age- and sex-matched during the same period were selected as controls. Forty patients were divided into CIK therapy versus observation. All patients received six to eight cycles of CHOP ± R therapy at 21-day intervals. CR was confirmed by (18)F-FDG PET/CT after completion of chemotherapy. CT scan was performed every 3 months after CR. This study was approved by the institutional review board of Nanjing Drum Tower Hospital, and was conducted in accordance with the Declaration of Helsinki. All CIK participants signed consent forms before recruitment.
Preparation of CIK Cells
Peripheral venous blood (60 mL) from patients was collected to extract PBMCs. The cells were incubated in a cell incubator at 37°C with 5% CO2. A total of 1000 U of rhIFN-γ was added to 1 mL of cell suspension (cell density was approximately 1–2×106/mL) on the first day. After 24 h, rhIL-2 (1000 U/mL) and anti-CD3 mAb (10 ug/mL) were added and incubated for 4 d. The cell suspensions were subcultured with fresh medium containing 1000 U/mL rhIL-2. The cells were washed and fresh medium was replaced every 3 days for 2 weeks. Culture media were removed by 3 washes with normal saline. Cells were resuspended in normal saline with human serum albumin and diverted into a transfer pack. Greater than 95% of cells were viable by the dye-exclusion test using trypan blue. CIK cells were tested twice for biological contaminants.
Treatment
Each patient received a total of 0.5–1.5×108/kg CIK cells per transfusion (one transfusion per day for 2 days). After transfusion, rhIL-2 was subcutaneously administered (100 mU/day) for 10 consecutive days. If the patient agreed, the CIK cell transfusion protocol was repeated every 3 months for 1 year (four cycles). The time taken for each component of the procedure is shown in Figure 1.
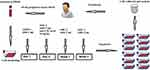 |
Figure 1 The main protocol for cytokine-induced killer (CIK) cell immunotherapy. |
Phenotypic Analysis of Peripheral Blood
The levels of T lymphocyte subsets and natural killer (NK) cells in peripheral blood before and after CIK cell transfusion were detected by flow cytometry using fluorochrome-conjugated monoclonal mouse anti-human antibodies against CD3, CD8, CD4, CD56, CD45.
Statistical Analysis
The χ2 test or Fisher’s exact test was used for discrete variables, and the Wilcoxon Mann–Whitney test was used in the analysis of continuous variables. One-way analysis of variance (ANOVA) testing was used for changes in the lymphocyte subgroup. Disease-free survival (DFS) and overall survival (OS) were estimated by the Kaplan-Meier method. Univariate and multivariate analyses were performed to evaluate factors for OS and DFS. Variables that were significant at p<0.10 were later included in the multivariate Cox regression analysis. Data were analyzed using SPSS version 21.0 software. A p-value <0.05 was considered statistically significant.
Results
Patients
The study population included 20 patients receiving CIK treatment (9 females; mean age=58±14 years) and 20 subjects as the control group (9 females; mean age = 57±14 years). The baseline characteristics of the patients are listed in Table 1. The characteristics of the patients were relatively well balanced. No significant difference was found between the two groups (p for all >0.05). The average number of CIK cells infused into patients in one cycle was 55.12±14.63×108 (range 36–84). Each patient received an average of two cycles of CIK treatments (range 1–4).
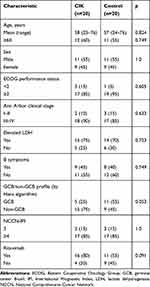 |
Table 1 Demographic Patient Characteristics by Study Arm at Diagnosis |
Changes in Lymphocyte Subgroups
Immunophenotypes of CIK are listed in Table 2. Before and two weeks after CIK cell transfusion, changes in lymphocyte subgroups in peripheral blood of CIK patients were determined by flow cytometry. As shown in Figure 2, after CIK cell transfusion, the percentages of CD3+ T cells, CD3+CD8+ T cells, and NK cells were significantly higher after treatment (P < 0.05).
 |
Table 2 Immunophenotypes of Cytokine-Induced Killer (CIK) Cells |
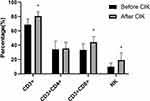 |
Figure 2 Percentages of lymphocyte subsets in the peripheral blood of 20 patients before and after cytokine-induced killer (CIK) cell transfusion (*p<0.05). |
Survival
With a median follow-up of 29.5 months (range, 6 to 105 months), the 5-year DFS was improved from 45.0 ± 11.1% to 79.3 ± 9.2% in the CIK group (HR favoring CIK, 0.29; 95% CI, 0.09 to 0.92; p = 0.035; Figure 3A). At a longer median follow-up of 35.5 months, the 5-year OS was estimated at 90 ± 6.7% for CIK versus 55 ± 11.1% for control (HR favoring CIK, 0.20; 95% CI, 0.04 to 0.93; p = 0.040; Figure 3B).
 |
Figure 3 Kaplan–Meier survival plots: (A) disease-free survival (DFS) and (B) overall survival (OS). |
According to multivariate analysis, CIK could improve DFS but not OS (Table 3). However, a significant prognostic value of B symptoms for OS and DFS was observed with wide CIs due to the small sample size.
 |
Table 3 Survival Analysis in Patients with High Risk DLBCL |
Side Effects
No severe side effects were recorded during or after CIK cell transfusions, except in one male patient, aged 47 years, who had mild flu-like symptoms, which were naturally quickly relieved.
Discussion
The goal of maintenance therapy, which requires continuous treatment administration, is to improve the quality of response, to delay disease progression and to increase long-term survival after initial therapy. Rituximab maintenance did not achieve a significant survival benefit in patients with follicular lymphoma14 or DLBCL.15 Everolimus maintenance did not improve DFS in patients already in CR.16 Lenalidomide maintenance has been shown to improve progression-free survival (PFS) in elderly patients with DLBCL after front-line therapy.17 However, no single study found a significant OS advantage. No effective maintenance treatment has been found to date.
To our knowledge, this study is the first to evaluate CIK immunotherapy as a maintenance strategy in DLBCL in CR. In our study, CIK immunotherapy was an effective treatment for maintaining CR, delaying relapse, and prolonging survival according to the univariate analysis. The survival benefit was also estimated based on analysis of the clinical characteristics of patients at diagnosis (age, sex, LDH level and B symptoms). Our analysis also found that the presence of B symptoms at diagnosis was associated with poor DFS and OS in patients after the first CR. A prognostic index that consisted of age older than 70 years and presence of B symptoms was designed, called the Oyama score, in patients with age-related EBV-associated B cell lymphoproliferative disorders. Patients with scores of zero, one or two showed median OS times of 56, 25 and 9 months, respectively.18 B symptoms were also an important predictor in refractory elderly patients diagnosed with DLBCL.19
CIK cells fall into the category of passive adoptive T cell immunotherapy against established tumors. Some studies have demonstrated that CIK cells exhibit antitumor effects against DLBCL in vivo.12,20 The cytotoxic activity of CIK cells is mediated by releasing perforin and granzyme granules and dependent on several activating receptors such as NKG2D, NKp30, NKp44, NKp46, and DNAM-1.21 CIK cells can regulate and increase cellular immune function in vivo.9 The clinical benefit of CIK cells may be due to the direct tumor killing activity in a MHC-independent way.22 CIK cells comprise CD3+CD56+, CD3+CD56−, and CD3−CD56+ cells.23 Our results suggested that the percentages of CD3+CD56+ subsets, representing the main antitumor immunoeffector cells, significantly increased after culture. CD3+CD56+ cells are able to mediate non-MHC-restricted lysis and cytokine production in the absence of TCR activation.24 CIK cell treatment can improve the immunological status of patients. In our study, the percentages of CD3+ T cells, CD3+CD8+ T cells, and NK cells in peripheral blood increased after CIK cell transfusion. NK cells are a key element of the innate immune system. A low baseline peripheral blood NK cell count was associated with impaired PFS in DLBCL.25 It has been shown that CIK cell transfusion activates CD8+ cytotoxic T cells. Activation of the immune system becomes the basis of the survival benefit derived from CIK treatment. Thus, these results together with our findings indicated that CIK cell treatment might be an optimized modality to gain improved prognosis in patients with DLBCL.
Our research has several major experimental limitations. First, this is a retrospective, nonrandomized study, and only patients from our hospital were included, which may not truly reflect patients from other centers. Moreover, few patients were included in the study to detect survival differences between CIK and observation. A second limitation of our analysis is that some patients were lost to follow-up posttreatment in both groups, which could have contributed to the shorter median follow-up time. Third, the choice of controls was subjective. Despite there being no significant difference between the two groups, we believe that some differences might have influenced survival. Thus, further prospective studies are needed to confirm our results.
In summary, autologous CIK cell immunotherapy is safe and efficacious for improving the prognosis of patients with high-risk DLBCL after the first CR. However, these findings are derived from a retrospective analysis and should be validated in larger populations through prospective studies.
Acknowledgments
This study was supported by grant from Nanjing Medical Science and Technique Development Foundation (YKK15068 and YKK17074). Min Zhou and Jing Wang are co-first authors for this study.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Li S, Young KH, Medeiros LJ. Diffuse large B-cell lymphoma. Pathology. 2018;50(1):74–87. doi:10.1016/j.pathol.2017.09.006
2. Davies A. Tailoring front-line therapy in diffuse large B-cell lymphoma: who should we treat differently? Hematology Am Soc Hematol Educ Program. 2017;2017(1):284–294. doi:10.1182/asheducation-2017.1.284
3. Maurer MJ, Ghesquières H, Jais JP, et al. Event-free survival at 24 months is a robust end point for disease-related outcome in diffuse large B-cell lymphoma treated with immunochemotherapy. J Clin Oncol. 2014;32(10):1066–1073. doi:10.1200/JCO.2013.51.5866
4. Schmitz N, Stelljes M, Bazarbachi A. Diffuse Large B-Cell Lymphoma. The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapie.
5. Sehn LH, Berry B, Chhanabhai M, et al. The revised international prognostic index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood. 2007;109(5):1857–1861. doi:10.1182/blood-2006-08-038257
6. Zhou M, Wang J, Ouyang J, et al. MYC protein expression is associated with poor prognosis in diffuse large B cell lymphoma patients treated with RCHOP chemotherapy. Tumour Biol. 2014;35(7):6757–6762. doi:10.1007/s13277-014-1907-z
7. Zhang D, Tang DG, Rycaj K. Cancer stem cells: regulation programs, immunological properties and immunotherapy. Semin Cancer Biol. 2018;52(Pt 2):94–106. doi:10.1016/j.semcancer.2018.05.001
8. Miyamoto S, Kochin V, Kanaseki T, et al. The antigen ASB4 on cancer stem cells serves as a target for CTL immunotherapy of colorectal cancer. Cancer Immunol Res. 2018;6(3):358–369. doi:10.1158/2326-6066.CIR-17-0518
9. Giraudo L, Gammaitoni L, Cangemi M, et al. Cytokine-induced killer cells as immunotherapy for solid tumors: current evidence and perspectives. Immunotherapy. 2015;7(9):999–1010. doi:10.2217/imt.15.61
10. Schmidt-Wolf IG, Negrin RS, Kiem HP, et al. Use of a SCID mouse/human lymphoma model to evaluate cytokine-induced killer cells with potent antitumor cell activity. J Exp Med. 1991;174(1):139–149. doi:10.1084/jem.174.1.139
11. Schmeel LC, Schmeel FC, Coch C, et al. Cytokine-induced killer (CIK) cells in cancer immunotherapy: report of the international registry on CIK cells (IRCC). J Cancer Res Clin Oncol. 2015;141(5):839–849. doi:10.1007/s00432-014-1864-3
12. Lu XC, Yang B, Yu RL, et al. Clinical study of autologous cytokine-induced killer cells for the treatment of elderly patients with diffuse large B-cell lymphoma. Cell Biochem Biophys. 2012;62(1):257–265. doi:10.1007/s12013-011-9273-6
13. Rozental A, Gafter-Gvili A, Vidal L, et al. The role of maintenance therapy in patients with diffuse large B cell lymphoma: A systematic review and meta-analysis. Hematol Oncol. 2019;37(1):27–34. doi:10.1002/hon.2561
14. Vitolo U, Ladetto M, Boccomini C, et al. Rituximab maintenance compared with observation after brief first-line R-FND chemoimmunotherapy with rituximab consolidation in patients age older than 60 years with advanced follicular lymphoma: a Phase III randomized study by the Fondazione Italiana Linfomi. J Clin Oncol. 2013;31(27):3351–3359. doi:10.1200/JCO.2012.44.8290
15. Gisselbrecht C, Schmitz N, Mounier N, et al. Rituximab maintenance therapy after autologous stem-cell transplantation in patients with relapsed CD20(+) diffuse large B-cell lymphoma: final analysis of the collaborative trial in relapsed aggressive lymphoma. J Clin Oncol. 2012;30(36):4462–4469. doi:10.1200/JCO.2012.41.9416
16. Witzig TE, Tobinai K, Rigacci L, et al. Adjuvant everolimus in high-risk diffuse large B-cell lymphoma: final results from the PILLAR-2 randomized phase III trial. Ann Oncol. 2018;29(3):707–714. doi:10.1093/annonc/mdx764
17. Thieblemont C, Tilly H, Gomes da Silva M, et al. Lenalidomide maintenance compared with placebo in responding elderly patients with diffuse large B-cell lymphoma treated with first-line rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Clin Oncol. 2017;35(22):2473–2481. doi:10.1200/JCO.2017.72.6984
18. Oyama T, Yamamoto K, Asano N, et al. Age-related EBV-associated B-cell lymphoproliferative disorders constitute a distinct clinicopathologic group: a study of 96 patients. Clin Cancer Res. 2007;13(17):5124–5132. doi:10.1158/1078-0432.CCR-06-2823
19. Danese MD, Griffiths RI, Gleeson ML, et al. Second-line therapy in diffuse large B-cell lymphoma (DLBCL): treatment patterns and outcomes in older patients receiving outpatient chemotherapy. Leuk Lymphoma. 2017;58(5):1094–1104. doi:10.1080/10428194.2016.1228924
20. Li SX, Zhu HL, Guo B, et al. Treatment for orbital diffuse large B-cell lymphoma in an elderly patient by autologous cytokine-induced killer cells. Ann Hematol. 2012;91(4):639–640. doi:10.1007/s00277-011-1301-5
21. Mata-Molanes JJ, Sureda González M, Valenzuela Jiménez B, et al. Cancer immunotherapy with cytokine-induced killer cells. Target Oncol. 2017;12(3):289–299. doi:10.1007/s11523-017-0489-2
22. Verneris MR, Karimi M, Baker J, et al. Role of NKG2D signaling in the cytotoxicity of activated and expanded CD8+ T cells. Blood. 2004;103(8):3065–3072. doi:10.1182/blood-2003-06-2125
23. Linn YC, Lau LC, Hui KM. Generation of cytokine-induced killer cells from leukaemic samples with in vitro cytotoxicity against autologous and allogeneic leukaemic blasts. Br J Haematol. 2002;116(1):78–86. doi:10.1046/j.1365-2141.2002.03247.x
24. Kokordelis P, Krämer B, Boesecke C, et al. CD3(+)CD56(+) natural killer-like T cells display anti-HCV activity but are functionally impaired in HIV(+) patients with acute hepatitis C. J Acquir Immune Defic Syndr. 2015;70(4):338–346. doi:10.1097/QAI.0000000000000793
25. Klanova M, Oestergaard MZ, Trněný M, et al. Prognostic impact of natural killer cell count in follicular lymphoma and diffuse large B-cell lymphoma patients treated with immunochemotherapy. Clin Cancer Res. 2019;25(15):4634–4643. doi:10.1158/1078-0432.CCR-18-3270
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
