Back to Journals » Therapeutics and Clinical Risk Management » Volume 12
Atrial fibrillation is a predictor of in-hospital mortality in ischemic stroke patients
Authors Ong C, Wong Y, Wu C, Su Y
Received 3 February 2016
Accepted for publication 21 April 2016
Published 29 June 2016 Volume 2016:12 Pages 1057—1064
DOI https://doi.org/10.2147/TCRM.S105703
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Cheung-Ter Ong,1,2 Yi-Sin Wong,3 Chi-Shun Wu,1 Yu-Hsiang Su1
1Department of Neurology, Chia-Yi Christian Hospital, 2Department of Nursing, Chung Jen Junior College of Nursing, Health Science and Management, Chiayi, 3Department of Family Medicine, Chia-Yi Christian Hospital, Chiayi, Taiwan, Republic of China
Background/purpose: In-hospital mortality rate of acute ischemic stroke patients remains between 3% and 18%. For improving the quality of stroke care, we investigated the factors that contribute to the risk of in-hospital mortality in acute ischemic stroke patients.
Materials and methods: Between January 1, 2007, and December 31, 2011, 2,556 acute ischemic stroke patients admitted to a stroke unit were included in this study. Factors such as demographic characteristics, clinical characteristics, comorbidities, and complications related to in-hospital mortality were assessed.
Results: Of the 2,556 ischemic stroke patients, 157 received thrombolytic therapy. Eighty of the 2,556 patients (3.1%) died during hospitalization. Of the 157 patients who received thrombolytic therapy, 14 (8.9%) died during hospitalization. History of atrial fibrillation (AF, P<0.01) and stroke severity (P<0.01) were independent risk factors of in-hospital mortality. AF, stroke severity, cardioembolism stroke, and diabetes mellitus were independent risk factors of hemorrhagic transformation. Herniation and sepsis were the most common complications of stroke that were attributed to in-hospital mortality. Approximately 70% of in-hospital mortality was related to stroke severity (total middle cerebral artery occlusion with herniation, basilar artery occlusion, and hemorrhagic transformation). The other 30% of in-hospital mortality was related to sepsis, heart disease, and other complications.
Conclusion: AF is associated with higher in-hospital mortality rate than in patients without AF. For improving outcome of stroke patients, we also need to focus to reduce serious neurological or medical complications.
Keywords: brain infarction, atrial fibrillation, outcome, risk factors, thrombolytic therapy
Introduction
Stroke is one of the most important causes of death and disability worldwide. The in-hospital mortality rates were ~6%–9% for ischemic stroke patients and 22%–45% for hemorrhagic stroke patients.1,2 However, these studies were conducted before the condition of stroke units was optimized and before intravenous recombinant-tissue plasminogen activator (rt-pa) was approved for use in acute stroke patients.
In recent years, there have been important advances in the management of acute stroke patients, such as availability of stroke units,3,4 thrombolytic therapy, and endovascular therapy performed for stroke patients who are compatible with thrombolytic therapy guidelines.5–7 Previous studies have reported that patient outcome and in-hospital mortality can be significantly improved if a patient is treated in a stroke unit.3,4 Intravenous rt-pa and endovascular therapy are known to improve outcomes in stroke patients but do not improve mortality associated with stroke.5,7 However, even with these advances in stroke management, the mortality rate of stroke patients during hospitalization remains between 3% and 18%.4,6,8–10 The predictors of in-hospital mortality were age, stroke severity, increased intracranial pressure (IICP), and pneumonia.9,11 Hemorrhagic transformation (HT) in ischemic stroke patients was reported to be associated with an increased risk for death and disability. Large area infarction, cardioembolism, and high-level blood sugar were associated with HT.12 Atrial fibrillation (AF) has been reported to be associated with poor outcome but not with in-hospital mortality.13 However, Arboix et al14 reported that AF is associated with higher in-hospital mortality rate in both ischemic and cardioembolic stroke patients. After thrombolytic therapy, patients with AF are at high risk of intracranial hemorrhage and death at discharge compared with patients without AF.15 However, one study found no association of AF with outcome in thrombolyzed patients.16 Thus, the question arises, whether AF may increase the risk of in-hospital mortality and the HT rate in stroke patients without thrombolytic therapy.
The purpose of this study was to identify the predictors of in-hospital mortality and to investigate the relationship between AF and in-hospital mortality and HT in ischemic stroke patients who were treated in stroke units, and to analyze the medical and neurological complications associated with in-hospital death.
Material and methods
Between January 1, 2007, and December 31, 2011, data of acute ischemic stroke patients admitted to the Department of Neurology of Chia-Yi Christian Hospital were collected prospectively during stroke registration. Chia-Yi Christian Hospital is an acute care, 1,000-bed teaching hospital in Southern Taiwan, Republic of China. The number of acute ischemic stroke patients treated in our hospital was >750 patients per year. All patients with acute neurological symptoms were initially attended in the emergency department. After neurological evaluation, brain computed tomography (CT) or magnetic resonance image (MRI) was performed, and stroke patients were admitted to the stroke unit, which is a part of the Department of Neurology. Blood cell count, biochemistry profile, chest radiography, and 12-lead electrocardiography were performed at the emergency department. Fasting blood sugar, cholesterol, and triglycerides were evaluated on the next day of admission. Neurological examination was performed every day. The National Institutes of Health Stroke Scale (NIHSS) was used to evaluate stroke severity. Modified Rankin Scale (mRS) was used to evaluate outcome. Good outcome was defined as mRS ≤2, whereas poor outcome was defined as mRS >2. Subtypes of stroke included large artery atherosclerosis (n=634), small vessel occlusion (n=922), cardioembolism (n=387), and undetermined etiology (n=613). Only ischemic stroke patients were included in this study. Patients with transient ischemic attack, intracerebral hemorrhage, and subarachnoid hemorrhage were excluded from the study. No other exclusion criteria were applied for patient selection in the present study.
For each patient, demographic data (age, sex, and body mass index), vascular risk factors (hypertension, diabetes mellitus, AF, hyperlipidemia, smoking, and heart disease), stroke severity score (NIHSS), neuroimaging, carotid duplex, and outcome were recorded. Variables affecting the outcome of stroke were assessed. Age was categorized as <65 years, 65–74 years, 75–84 years, or >85 years. Hypertension was defined as either systolic blood pressure ≥140 mmHg, diastolic blood pressure >90 mmHg, self-reported history of hypertension, or use of an antihypertensive agent. Diabetes mellitus was defined as pathologically elevated fasting blood sugar, self-reported patient history of diabetes mellitus, or regular use of antidiabetic medications. AF was defined as history of AF or AF observed on electrocardiogram. Hyperlipidemia was defined as serum total cholesterol level >200 mg/dL or low-density lipoprotein level >130 mg/dL (measured during acute stroke stage). Patients who had history of ischemic heart disease or congestive heart failure were defined as having heart disease. IICP was defined as evidence of brain edema, mass effect, or brain shift syndrome in brain CT or MRI associated with clinical deterioration.9 Transtentorial herniation was defined as brain CT or MRI showing brain edema associated with acute onset of unilateral or bilateral papillary dilation, loss of reactivity to light, and decline in the Glasgow coma scale ≥2 points.17 HT was defined as any sign of hemorrhage on the brain CT or MRI scans.18
The patients who had AF and regularly used anticoagulant for >3 days per week were considered as anticoagulant user, otherwise were considered as non-user. In the first week of stroke onset, anticoagulant was not given except for patients with small infarct.
Outcome variables included infection, cardiac event, vascular event, or respiration disease. Causes of death were analyzed such as transtentorial herniation, pneumonia, cardiac event, sudden death, septicemia, unknown, brain stem infarction, and other causes.19 If the patient had herniation and sepsis signs, and he (or she) died within 1 week of stroke onset, the cause of death was considered as stroke (IICP) related. If the patient had clinical evidence of systemic inflammatory response syndrome and died after 1 week of stroke onset without other causes for death, the cause of death was considered as sepsis.20 The data used in the study were collected from the Chia-Yi Christian Hospital acute stroke registry. This study has been approved by the Ethics Committee of the Chia-Yi Christian Hospital and written informed consent was obtained from each patient.
Statistical analysis
Chi-square test and independent Student’s t-test were used for univariate analysis. P<0.05 was considered statistically significant. Multivariate regression (simultaneous regression) was used to identify independent risk factors of in-hospital mortality and HT. Analysis was performed using MedCalc statistical software Version 15. (MedCalc Software, Ostend, Belgium).
Results
During the 5-year period covered by this study, 3,107 patients were admitted due to ischemic stroke, of which 451 were excluded from the study because the final diagnosis was transient ischemic attack. Thus, 2,556 patients were included in this study. CT was performed in all 2,556 patients; CT and MRI were performed in 1,362 patients. Clinical characteristics of the enrolled patients are shown in Table 1. Patients’ age ranged from 23 years to 101 years old (mean age, 69.6±11.9 years). Of the 2,556 patients, 814 patients (31.8%) were aged <65 years, 781 patients (30.5%) were aged 65–74 years, 732 patients (28.7%) were aged 75–84 years, and 229 patients (19%) were aged >85 years. Of the 2,556 patients, 1,482 (58%) were males and 1,074 (42%) were females, and 109 (4.3%) had no risk factor of stroke, 432 (16.9%) had one risk factor of stroke, 755 (29.5%) had two risk factors of stroke, 664 (26.0%) had three risk factors of stroke, and 596 (23.3%) had >3 risk factors of stroke. The most common risk factors were hypertension (80.3%), followed by hyperlipidemia (55.4%), diabetes mellitus (44%), heart disease (29.5%), stroke history (27.5%), and AF (17.1%). Types of stroke included large artery atherosclerosis (24.8% of patients), small vessel disease (36.1% of patients), cardioembolism (15.1% of patients), and undetermined etiology (24% of patients). NIHSS at admission was ≤5 in 54.5% of patients, 6–15 in 29.1% of patients, 16–25 in 10.7% of patients, and >25 in 5.7% of patients (Table 1). The median NIHSS score was 5. IICP was observed in 4.2% of patients. The prevalence of hypertension, diabetes mellitus, and AF was higher in females than in males. Of the 2,556 patients, 157 received thrombolytic therapy. At discharge, 56.8% (1,451/2,556) of total patients had a poor outcome (mRS >2), and 94.4% (1,371/1,451) of those patients were alive. The overall in-hospital mortality was 3.13% (80/2,556), which included the 8.9% (14/157) of patients receiving thrombolytic therapy who died during hospitalization. The in-hospital mortality rate was 2.4% (36/1,482) in males and 4.1% (44/1,074) in females. In-hospital mortality was higher in females than in males (P=0.02). Stroke severity was higher in females than in males. Stroke severity was strongly associated with an increase in the risk of in-hospital mortality (Table 2). In-hospital mortality was 6.28% (74/1,162) in patients with moderate-to-severe stroke (NIHSS >5) and 0.43% (6/1,394) in patients with mild stroke (NIHSS ≤5). The HT rate was higher in fatality patients than in nonfatality patients (P<0.001). Univariate analysis showed that AF, stroke subtype, stroke severity (NIHSS >15), and age ≥75 years significantly increased the risk of in-hospital mortality rate (Table 2). AF, stroke subtype, and stroke severity increased the risk of HT (Table 3). A multivariate regression analysis showed that AF, stroke severity, and stroke type were independent predictors of in-hospital mortality. AF, stroke severity, cardioembolism stroke, and diabetes mellitus were independent predictors of HT (Table 4). In comparison with large artery atherosclerosis, cardioembolic stroke increased the risk of in-hospital mortality with odds ratio 1.75 (Table 2). Of the 437 patients with AF, 62 patients used warfarin or other antithrombotic agents, 375 patients did not use any antithrombotic agent. There was no difference in the in-hospital mortality between the group that used antithrombotic agents and the group that did not use (6.45% (4/62) vs 9.33% (35/375), P=0.63). There was no significant difference in the hemorrhagic rate between the two groups (14.52% (9/62) vs 13.33% (50/375), P=0.84). Stroke-related complications such as transtentorial herniation and basilar artery occlusion were the most common causes of in-hospital mortality. Approximately 70% of in-hospital mortality was attributed to stroke severity (transtentorial herniation, basilar artery occlusion, and HT; Table 5). Middle cerebral artery occlusion-related fatality occurred mostly in the patients with large artery atherosclerosis, cardioembolism, and undetermined etiology. Multivariate logistic regression showed that only AF (P<0.001) and stroke severity (NIHSS >15; P<0.001) were independent risk factors of in-hospital mortality (Table 4). AF, stroke severity, cardioembolism stroke, and diabetes mellitus were predictors of HT (Table 4).
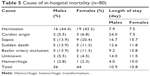  | Table 5 Cause of in-hospital mortality (n=80) |
Discussion
In-hospital mortality of acute stroke patients is affected by several factors including patient age, stroke type, stroke severity, travel time to hospital, and enrollment criteria.21–26 A German study found that the risk of in-hospital mortality was 10% in patients treated with rt-pa and 4.6% in patients not treated with rt-pa.8 A different study from Taiwan found that in-hospital mortality was 17.8% in patients treated with rt-pa.6 In our study, the total in-hospital mortality was found to be 3.13%, which included 8.9% (14/157) of patients treated with rt-pa and 2.75% (66/2,399) of patients not treated with rt-pa. This in-hospital mortality is same as that found in previous studies.1,27 Koennecke et al9 found that their overall in-hospital mortality was 5.4%; however, >50% of their patients had moderate-to-severe stroke. In our study, the in-hospital mortality rate in patients with moderate-to-severe stroke was 6.28%, which is similar to the result of Koennecke et al study.
In our study, 35 patients died due to middle cerebral artery occlusion with brain edema and herniation, and ten patients died due to basilar artery occlusion with cerebellar edema and herniation. Fatality rates in patients with large middle cerebral artery infarct as high as 78% have been reported.28 Infarct size has been reported as the major determinant of the development of life-threatening brain edema.29 Decompressive surgery in patients <60 years old had been reported to reduce mortality and severe disability in patients with malignant middle cerebral artery infarction.30 Our study found that ~60% of patients died in the hospital due to total middle cerebral artery occlusion or basilar artery occlusion with brain edema and herniation. Because most of the patients who died in hospital were older than 60 years, whether increased rate of decompressive surgery could reduce in-hospital mortality needs further investigation. This study showed that stroke severity is the most important factor of in-hospital mortality. When compared with large artery atherosclerosis, patients with cardioembolism had a high risk of death and small vessel occlusion had a lower risk of death (Table 2), which may be related to a cardioembolic occlusion of a large artery and lack of collateral circulation. Another cause for higher in-hospital mortality among patients with cardioembolic stroke may be related to recurrent embolism. Arboix et al31 found that the in-hospital mortality rate in patients with early recurrent embolization is significantly higher than in those who did not develop attacks. Small vessel occlusion had lower rate of in-hospital mortality in our study similar to the results of Arboix et al;32 they found that the mortality rate of lacunar infarct is significantly lower than other types of stroke. Our study found that the in-hospital mortality is mildly lower in anticoagulant user and there was no significant difference in the HT rate between anticoagulant user and nonuser. The result is inconsistent with that of Rosand, et al33 and Singer, et al34 studies. Their studies found that warfarin is beneficial to patients for prevention and does not significantly increase the risk of intracranial hemorrhage. This result highlights the importance of the use of anticoagulant agent for patients with AF for the prevention of ischemic stroke. Old age had been reported as a predictor of in-hospital mortality and associated with poor outcome in stroke patients.35 Our study also showed that older age of stroke patients has the tendency to increase the risk of in-hospital mortality. The study confirmed the result that old age is a predictor of in-hospital mortality and is also associated with poor outcome in stroke patients.
The in-hospital mortality rate in patient receiving thrombolysis was 8.9%, whereas the mortality rate in all the patients was 3.1% (P<0.0001). The higher mortality rate in thrombolysis patients is in contrast with the finding of ECASS II, which reported that there was no difference in 30-day and 90-day mortality in thrombolysis and nonthrombolysis patients,36 which was a randomized trial. The result is consistent with the finding in a nationwide inpatient sample study. Bateman et al analyzed the data from the US nationwide inpatient sample for the years 1999 to 2002. Their results showed that thrombolysis patient had a higher in-hospital mortality rate compared with the nonthrombolysis patients (11.4% vs 6.8%).37 The higher mortality rate in the thrombolysis group is most likely related to the baseline difference in the patient receiving thrombolysis.
Approximately 30% of in-hospital mortality was related to preventable factors. In particular, patients with septic shock (12 with pneumonia and two with urinary tract infection) showed 17.5% in-hospital fatality in this study and heart disease (cardiac event and sudden death) in about 15% of in-hospital mortality. History of angina and myocardial infarction before stroke were associated with in-hospital acute myocardial infarction and heart failure along with increased mortality at 3 months.38 Our result is compatible with the findings of this study. In order to improve the outcome of stroke patients, close monitoring of cardiac function, along with prompt diagnosis and treatment of the heart event may be necessary. A previous study showed that chest infection and urinary tract infection were frequent complications in stroke patients.39 Because chest infection may be related to aspiration, dysphagia screening is necessary in stroke patients. Whether prophylactic antibiotics should be used for the prevention of pneumonia or urinary tract infection in patients at a risk of infection needs further investigation.
It was reported that AF more likely occurs in female patients and old patients and has a higher fatality rate within 14 days.13 We found that the frequency of AF was 17.1% in our patients; the result is in accordance with 15%–21% reported by other authors.13,40–42 Wong reported that AF and ischemic heart disease were independent risk factors for in-hospital mortality.2 In our study, AF was found to be the risk factors for in-hospital mortality and HT. Heuschmann et al showed that AF, age, and stroke severity are predictors of in-hospital mortality.27 Our results also show that AF, cardioembolism type stroke, and stroke severity are predictors of in-hospital mortality. In our study, old age has the tendency to increase in-hospital mortality; however, in multivariate analysis, there is no significant difference. Our study found that AF, cardioembolism type stroke, stroke severity, and diabetes mellitus are predictors of HT. Our result is compatible with that of Wang et al,43 who found that cardioembolism stroke, severity of stroke, and diabetes mellitus are predictors of HT. Our study also found that the number of patients dying due to septic shock (17.5%, 14/80) and sudden death (12.5%, 10/80) was higher than those dying due to acute myocardial infarction (6.25%, 5/80). It can be assumed that patients who died due to sudden death may have developed ischemic heart disease; however, this was not confirmed.
A previous study showed that females aged 45–64 years had lower risk of stroke-related in-hospital mortality than males.44 However, some studies reported that female sex was associated with poor functional outcome at discharge.13,42 These studies found that females with stroke were older and more likely to have AF, they also found that females experienced cardioembolic stroke more frequently than males. Our study found that in-hospital mortality in females was higher than that in males (P=0.02), which may be attributed to more frequent AF and cardioembolic stroke in females, and stroke severity was higher in females than in males. However, after adjusting for demographic and clinical factors, there were no differences between sexes with respect to mortality.
This study had some limitations. First, this was a retrospective analysis of stroke registration data and there may have been coding error. However, the errors are unlikely to be systemic. Second, this study only included patients with ischemic stroke and did not include patients with hemorrhagic stroke. The strengths of our study are as follows: 1) all patients were managed in the stroke unit and by a neurologist, and all data were collected in a prospective and uniform manner. Data were designed for continuous monitoring of stroke. 2) All physicians in our stroke unit had experience using rt-pa in stroke patients, and all patients who were eligible for thrombolytic therapy received thrombolytic therapy.
In the study, stroke severity and AF were found to be independent predictors of in-hospital mortality in routine clinical practice of acute ischemic stroke. AF, stroke severity, cardioembolism stroke, and diabetes mellitus were predictors of HT. Our study found that AF may increase risk of in-hospital mortality and increase HT in acute stroke patients. When we treat patients with AF and acute stroke, we need to cautiously use anticoagulants, especially in patients with large infarct, previous hemorrhagic stroke, and low platelet count of high sensitive c-reactive protein (hsCRP) level.45 Approximately 30% of in-hospital mortality in acute ischemic stroke patients was found to be associated with serious neurological or medical complications. Thus, during the management of acute stroke patients, we also need focus to reduce serious complications after stroke to reduce in-hospital mortality rate. In particular, care must be targeted toward the prevention of infection and IICP.
Conclusion
From our results, we conclude that stroke in patients with AF is associated with higher in-hospital mortality rate than in patients without AF. These findings emphasize the importance of prophylaxis of stroke in patients with AF as soon as AF is diagnosed. For improving the outcome of stroke patients, we also need focus to reduce serious neurological or medical complications.
Acknowledgment
The authors would like to thank Enago (www.enago.tw) for the English language review.
Disclosure
The authors report no conflicts of interest in this work.
References
Hung TP. Changes in mortality from cerebrovascular disease and clinical pattern of stroke in Taiwan. J Formos Med Assoc. 1993;92(8):687–696. | ||
Wong KS. Risk factors for early death in acute ischemic stroke and intracerebral hemorrhage: a prospective hospital-based study in Asia. Asian Acute Stroke Advisory Panel. Stroke. 1999;30(11):2326–2330. | ||
Forti P, Maioli F, Procaccianti G, et al. Independent predictors of ischemic stroke in the elderly: prospective data from a stroke unit. Neurology. 2013;80(1):29–38. | ||
Candelise L, Gattinoni M, Bersano A, et al; PROSIT Study Group. Stroke-unit care for acute stroke patients: an observational follow-up study. Lancet. 2007;369(9558):299–305. | ||
Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995;333(24):1581–1587. | ||
Li CH, Khor GT, Chen CH, Huang P, Lin RT. Potential risk and protective factors for in-hospital mortality in hyperacute ischemic stroke patients. Kaohsiung J Med Sci. 2008;24(4):190–196. | ||
Chen CJ, Ding D, Starke RM, et al. Endovascular vs medical management of acute ischemic stroke. Neurology. 2015;85(22):1980–1990. | ||
Heuschmann PU, Kolominsky-Rabas PL, Roether J, et al; German Stroke Registers Study Group. Predictors of in-hospital mortality in patients with acute ischemic stroke treated with thrombolytic therapy. JAMA. 2004;292(15):1831–1838. | ||
Koennecke HC, Belz W, Berfelde D, et al; Berlin Stroke Register Investigators. Factors influencing in-hospital mortality and morbidity in patients treated on a stroke unit. Neurology. 2011;77(10):965–972. | ||
Lin S, Wu B, Hao ZL, et al. Characteristics, treatment and outcome of ischemic stroke with atrial fibrillation in a Chinese hospital-based stroke study. Cerebrovasc Dis. 2011;31(5):419–426. | ||
Weimar C, Ziegler A, Konig IR, Diener HC. Predicting functional outcome and survival after acute ischemic stroke. J Neurol. 2002;249(7):888–895. | ||
Paciaroni M, Agnelli G, Corea F, et al. Early hemorrhagic transformation of brain infarction: rate, predictive factors, and influence on clinical outcome: results of a prospective multicenter study. Stroke. 2008;39(8):2249–2256. | ||
Steger C, Pratter A, Martinek-Bregel M, et al. Stroke patients with atrial fibrillation have a worse prognosis than patients without: data from the Austrian Stroke registry. Eur Heart J. 2004;25(19):1734–1740. | ||
Arboix A, Garcia-Eroles L, Massons JB, Oliveres M, Pujades R, Targa C. Atrial fibrillation and stroke: clinical presentation of cardioembolic versus atherothrombotic infarction. Int J Cardiol. 2000;73(1):33–42. | ||
Saposnik G, Gladstone D, Raptis R, Zhou L, Hart RG; Investigators of the Registry of the Canadian Stroke Network (RCSN) and the Stroke Outcomes Research Canada (SORCan) Working Group. Atrial fibrillation in ischemic stroke: predicting response to thrombolysis and clinical outcomes. Stroke. 2013;44(1):99–104. | ||
Frank B, Fulton R, Weimar C, Shuaib A, Lees KR; VISTA Collaborators. Impact of atrial fibrillation on outcome in thrombolyzed patients with stroke: evidence from the Virtual International Stroke Trials Archive (VISTA). Stroke. 2012;43(7):1872–1877. | ||
Koenig MA, Bryan M, Lewin JL 3rd, Mirski MA, Geocadin RG, Stevens RD. Reversal of transtentorial herniation with hypertonic saline. Neurology. 2008;70(13):1023–1029. | ||
Ong CT, Sung SF, Wu CS, et al. Early neurological improvement after intravenous tissue plasminogen activator infusion in patients with ischemic stroke aged 80 years or older. J Chin Med Assoc. 2014;77(4):179–183. | ||
Silver FL, Norris JW, Lewis AJ, Hachinski VC. Early mortality following stroke: a prospective review. Stroke. 1984;15(3):492–496. | ||
Klouwenberg PMK, Ong DS, Bonten MJ, Cremer OL. Classification of sepsis, severe sepsis and septic shock: the impact of minor variations in data capture and definition of SIRS criteria. Intensive Care Med. 2012;38(5):811–819. | ||
Gillum LA, Johnston SC. Characteristics of academic medical centers and ischemic stroke outcomes. Stroke. 2001;32(9):2137–2142. | ||
Sacco RL. Risk factors, outcomes, and stroke subtypes for ischemic stroke. Neurology. 1997;49(5 suppl 4):S39–S44. | ||
Frankel MR, Morgenstern LB, Kwiatkowski T, et al. Predicting prognosis after stroke: a placebo group analysis from the National Institute of Neurological Disorders and Stroke rt-PA Stroke Trial. Neurology. 2000;55(7):952–959. | ||
Handschu R, Haslbeck M, Hartmann A, et al. Mortality prediction in critical care for acute stroke: severity of illness-score or coma-scale? J Neurol. 2005;252(10):1249–1254. | ||
Rordorf G, Koroshetz W, Efird JT, Cramer SC. Predictors of mortality in stroke patients admitted to an intensive care unit. Crit Care Med. 2000;28(5):1301–1305. | ||
Hsieh FI, Lien LM, Chen ST, et al. Get with the guidelines-stroke performance indicators: surveillance of stroke care in the Taiwan stroke registry: get with the guidelines-stroke in Taiwan. Circulation. 2010;122(11):1116–1123. | ||
Heuschmann PU, Kolominsky-Rabas PL, Misselwitz B, et al; German Stroke Registers Study Group. Predictors of in-hospital mortality and attributable risks of death after ischemic stroke: the German Stroke Registers Study Group. Arch Intern Med. 2004;164(16):1761–1768. | ||
Hacke W, Schwab S, Horn M, Spranger M, De Georgia M, von Kummer R. Malignant’ middle cerebral artery territory infarction: clinical course and prognostic signs. Arch Neurol. 1996;53(4):309–315. | ||
Hofmeijer J, Algra A, Kappelle LJ, van der Worp HB. Predictors of life-threatening brain edema in middle cerebral artery infarction. Cerebrovasc Dis. 2008;25(1–2):176–184. | ||
Wartenberg KE. Malignant middle cerebral artery infarction. Curr Opin Crit Care. 2012;18(2):152–163. | ||
Arboix A, Garcia-Eroles L, Massons J, Oliveres M. Predictive clinical factors of in-hospital mortality in 231 consecutive patients with cardioembolic cerebral infarction. Cerebrovasc Dis. 1998;8(1):8–13. | ||
Arboix A, Bell Y, Garcia-Eroles L, et al. Clinical study of 35 patients with dysarthria-clumsy hand syndrome. J Neurol Neurosurg Psychiatry. 2004;75(2):231–234. | ||
Rosand J, Eckman MH, Knudsen KA, Singer DE, Greenberg SM. The effect of warfarin and intensity of anticoagulation on outcome of intracerebral hemorrhage. Arch Intern Med. 2004;164(8):880–884. | ||
Singer DE, Chang Y, Fang MC, et al. The net clinical benefit of warfarin anticoagulation in atrial fibrillation. Ann Intern Med. 2009;151(5):297–305. | ||
Tian MJ, Tayal AH, Schlenk EA. Predictors of poor hospital discharge outcome in acute stroke due to atrial fibrillation. J Neurosci Nurs. 2015;47(1):20–26. | ||
Hacke W, Kaste M, Fieschi C, et al. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Second European-Australasian Acute Stroke Study Investigators. Lancet. 1998;352(9136):1245–1251. | ||
Bateman BT, Schumacher HC, Boden-Albala B, et al. Factors associated with in-hospital mortality after administration of thrombolysis in acute ischemic stroke patients: an analysis of the nationwide inpatient sample 1999 to 2002. Stroke. 2006;37(2):440–446. | ||
Micheli S, Agnelli G, Caso V, et al. Acute myocardial infarction and heart failure in acute stroke patients: frequency and influence on clinical outcome. J Neurol. 2012;259(1):106–110. | ||
Kumar S, Selim MH, Caplan LR. Medical complications after stroke. Lancet Neurol. 2010;9(1):105–118. | ||
Lamassa M, Di Carlo A, Pracucci G, et al. Characteristics, outcome, and care of stroke associated with atrial fibrillation in Europe: data from a multicenter multinational hospital-based registry (The European Community Stroke Project). Stroke. 2001;32(2):392–398. | ||
Dulli DA, Stanko H, Levine RL. Atrial fibrillation is associated with severe acute ischemic stroke. Neuroepidemiology. 2003;22(2):118–123. | ||
Jorgensen HS, Nakayama H, Reith J, Raaschou HO, Olsen TS. Acute stroke with atrial fibrillation. The Copenhagen Stroke Study. Stroke. 1996;27(10):1765–1769. | ||
Wang B-G, Yang N, Lin M, Lu B. Analysis of risk factors of hemorrhagic transformation after acute ischemic stroke: cerebral microbleeds do not correlate with hemorrhagic transformation. Cell Biochem Biophys. 2014;70(1):135–142. | ||
Towfighi A, Tai W, Markovic D, Ovbiagele B. Sex-specific temporal trends in in-hospital mortality after stroke among middle-age individuals in the United States. Stroke. 2011;42(10):2740–2745. | ||
Lee JH, Park KY, Shin JH, et al. Symptomatic hemorrhagic transformation and its predictors in acute ischemic stroke with atrial fibrillation. Eur Neurol. 2010;64(4):193–200. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




