Back to Journals » Clinical Interventions in Aging » Volume 16
Association Between Systemic Immune-Inflammation Index and Diabetic Depression
Authors Wang J , Zhou D, Dai Z, Li X
Received 2 October 2020
Accepted for publication 13 December 2020
Published 11 January 2021 Volume 2021:16 Pages 97—105
DOI https://doi.org/10.2147/CIA.S285000
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Zhi-Ying Wu
Jie Wang,1 Depu Zhou,1 Zhijuan Dai,2 Xiaokun Li1
1Department of Endocrinology, Yanbian University Hospital, Yanji, Jilin, People’s Republic of China; 2Department of Endocrinology, The Second Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, People’s Republic of China
Correspondence: Xiaokun Li
Department of Endocrinology, Yanbian University Hospital, Yanji, Jilin, People’s Republic of China
Email [email protected]
Background: Depression is highly prevalent in patients with diabetes mellitus (DM). Diabetic depression has been shown to be associated with low-grade systemic inflammation. In recent years, the systemic immune-inflammation (SII) index has been developed as an integrated and novel inflammatory indicator. The aims of this study were to investigate the relationship between diabetic depression and SII levels, adjusting for a wide range of potential confounding factors, to examine the potential of SII in predicting diabetic depression.
Methods: The present cross-sectional study was conducted among adults with DM in the National Health and Nutrition Examination Survey between 2009 and 2016, the SII level was calculated as the platelet counts × neutrophil counts/lymphocyte counts. Patient Health Questionnaire‐9 was used to measure depression in patients with DM. Multivariable logistic regression and propensity score-matched analysis were used to analyze the association between SII levels and depression.
Results: A total of 2566 patients with DM were included in the study, of which 370 (13.3%) were diagnosed with depression. Multivariable logistic regression showed that high SII level was an independent risk factor for diabetic depression (OR = 1.347, 95% CI: 1.031– 1.760, P = 0.02882) after adjusting for covariates. The relationship between SII and diabetic depression was further verified by propensity score-matched analysis.
Conclusion: Our data suggest that SII is a risk factor for depression in patients with DM. The SII may be an easily accessible and cost-effective strategy for identifying depression in patients with DM. More studies are warranted to further analyze the role of SII in depression in diabetic patients.
Keywords: systemic immune-inflammation index, depressive symptoms, diabetes mellitus, NHANES
Introduction
Diabetes mellitus (DM) is one of the most prevalent chronic diseases in recent decades.1 In patients with DM, 64% experience psychological distress and 8% to 35% are diagnosed with depression.2–5 Patients with DM and depression tend to be less adherent to their therapy and have a higher rate of death.6 The complications associated with diabetes can also increase the risk of depression. Approximately 51% of depression cases are not correctly diagnosed in patients with DM, and only 31% received adequate antidepressants.7 Therefore, it is both urgent and necessary to identify depression in patients with DM.
Preclinical and clinical studies have shown a causal link between sterile low-grade inflammation and depression in patients with DM.8–12 Study showed that a high-fat diet leads to an increase in inflammatory cytokine levels and to anxiety and depressive behaviors.13,14 Antidepressant administration decreased the inflammatory cytokine levels and reversed the behavioral deficits caused by a high-fat diet.15 Inflammatory biomarkers could potentially be used to predict depression in diabetic patients. Abnormal increases in inflammatory blood cell parameters including neutrophil count, neutrophil-to-lymphocyte ratio,16,17 monocyte-to-lymphocyte ratio,18 and platelet-to-lymphocyte ratio19,20 serve as simple markers of inflammation and their ability to predict depression has been assessed. But these biomarkers involve only two types of immune-inflammatory cells and might not accurately reflect the inflammation status.
The systemic immune-inflammation index (SII) is an integrated and novel inflammatory biomarker21,22 based on neutrophil, lymphocyte, and platelet counts. The SII index was initially used to assess the prognosis of patients with solid cancers22 and coronary heart disease (CHD)23 and is now considered to accurately reflect inflammation status.24 However, the role of SII in depression in patients with DM remains unclear. We hypothesized that patients with DM and higher levels of inflammation, as measured by SII, are at a higher risk of developing depression. Therefore, we performed a cross-sectional study to assess the relationship between diabetic depression and SII levels to determine the value of SII in predicting diabetic depression.
Methods
Data and Sample Sources
The study was a two-year cross-sectional, stratified, multistage probability cluster survey. Data were obtained from the National Health and Nutrition Examination Survey (NHANES),25 which is designed to collect a wide variety of information on the potential risk factors and nutrition of the non-institutionalized, civilian, US population. The protocols for the conduct of NHANES were approved by the National Center for Health Statistics institutional review board (NCHS IRB/ERB), and informed consent was obtained from all participants (NCHS IRB/ERB protocols #2011–17). The Ethics Review Board for the National Center for Health Statistic (NCHS ERB) approved the NHANES (NCHS ERB protocols #2011–17), and all participants gave written informed consent. Following an in-home interview, NHANES participants receive a health examination at mobile examination centers. The medical and physiological status of participants is assessed, and laboratory tests conducted. Four cycles of the NHANES survey were selected to assess the association between SII and diabetic depression. The exclusion criteria were: (a) patients with missing SII data and incomplete Patient Health Questionnaire-9 (PHQ-9),26 and (b) corticosteroid, and nonsteroidal anti-inflammatory drug use.
Assessment of Depression Symptoms
In NHANES, depression was assessed using the PHQ-9.26 The PHQ-9 form was completed during the face‐to‐face mobile exam center interview and was designed to evaluate any depression symptoms in the preceding 2 weeks. Each item on the form was scored on a scale of 0 to 3, and total scores ranged from 0 to 27. In this study, PHQ‐9 score ≥ 10 was considered to indicate depression, with a specificity and sensitivity of 88%.27,28
Study Variables
Lymphocyte, neutrophil, and platelet counts were evaluated using automated hematology analyzing devices and were expressed as ×103 cells/µL. The SII level was measured as platelet count x neutrophil count/lymphocyte count.21 Details of methods about blood are described in the Supporting Methods section. Demographic characteristics included age, sex, race, marital status, education level, body mass index (BMI), smoking status, and ratio of family income to poverty (PIR); DM-related characteristics glycated hemoglobin A1c (HbA1c), diabetes duration, diabetic retinopathy (DR) and insulin use; health factors included stroke, heart failure (HF), and CHD.
Statistical Analyses
Differences in baseline characteristics in the depressive and the non-depressive-symptoms groups were compared using an independent sample t-test for continuous variables and χ2 tests for categoric variables. For the current study, the optimal cutoff value for the SII level was determined using receiver operating characteristics curve analysis. We performed multivariate logistic regression analysis to examine the association between SII and diabetic depression, with 95% confidence intervals (CI) and odds ratio (OR) calculated. In model 1, any confounding factors were not adjusted for, and age, sex, race, education, marital status, body mass index, HbA1c, insulin use, poverty income ratio, smoking status, diabetes duration, chronic conditions including stroke (yes/no), CHD (yes/no), and HF (yes/no), were adjusted for in model 2. To avoid potential bias, and because of differences in baseline characteristics, the propensity score matching (PSM) was determined.29 The study was used to ensure all reported depression selection factors were included as covariates in the model to further reduce potential confounding. Final covariates were age, sex, race, education, marital status, body mass index, HbA1c, insulin use, poverty income ratio, smoking status, diabetes duration, diabetic retinopathy, chronic conditions including stroke (yes/no), CHD (yes/no), and HF (yes/no). PSM was performed at a ratio of 1:1 using a caliper width of 0.01 of the SD of the logit of the propensity score. After PSM, model 3 was analyzed. Subgroup analysis was performed to explore if the association differed for subgroups classified using different parameters including age, sex, BMI, HbA1c, and insulin use. We conducted linear regression analyses to examine the association of SII (independent variable) and high sensitive c-reactive protein (hs-CRP), neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio (dependent variable) to examine whether SII level were associated with inflammation levels.
All analyses were performed using R (version 4.00) “MatchIt” package for PSM. P < 0.05 (two-sided) indicated significant difference.
Results
Subject Characteristics
We identified 2566 patients with DM who met our inclusion criteria. The eligible participants included 1252 women and 1314 men with a mean age of 61.4 ± 13.1 years, and a mean SII of 557.4. The number of patients diagnosed with diabetic depression was 370 (14.4%). Baseline characteristics are shown in Table 1. Depression in patients was associated with higher levels of BMI, heart failure, stroke, and SII. They were also less likely to have been married, and more likely to be in the lower age group and have a lower PIR rate (p < 0.05). Education, diabetic retinopathy, and CHD did not differ between patients with and without depression.
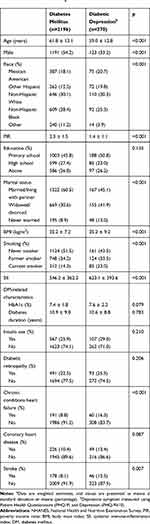 |
Table 1 Characteristics of Participants in the NHANES (2009–2016) by Depression Statusa |
SII is an Independent Risk Factor for Diabetic Depression
We constructed various models to assess the independent effects of SII on diabetic depression, after adjusting for other potential confounding factors. In univariate analysis, age, sex, race, education, BMI, PIR, smoking status, marital status, and chronic conditions were associated with a higher risk of depression (p < 0.05, Supplementary Materials Table S1). High SII levels were a risk factor for diabetic depression in univariate analysis (OR = 1.687, 95% CI: 1.351–2.107, P < 0.00001, Table 2). After adjusting for age, sex, race, education, marital status, body mass index, HbA1c, insulin use, poverty income ratio, smoking status, diabetes duration, chronic conditions including stroke (yes/no), CHD (yes/no), and HF (yes/no), high SII levels were an independent risk factor for diabetic depression (OR = 1.347, 95% CI: 1.031–1.760, P =0.02882). We excluded participants who had a diagnosis of coronary heart disease, stroke, and heart failure, a significant relationship between SII and depression still present (Supplementary Materials Table S2).
 |
Table 2 Association Between SII and Diabetic Depression |
PSM Analysis
PSM analysis was conducted to assess the relationship between SII and diabetic depression. The baseline characteristics of patients in different SII groups did not significantly differ (Table 3). Logistic regression analysis revealed that high SII levels were independently related to diabetic depression (OR = 1.452, 95% CI: 1.104–1.908, p = 0.00755).
 |
Table 3 Characteristics of Patients Before and After PSMa |
Subgroup Analysis
Subgroup analysis results are shown in Table 4. In patients with diabetic depression, there were no differences in SII levels in most pre-specified subgroups, with the exception of sex. High SII levels were independently related to depression in male patients (OR = 1.686, 95% CI: 1.162–2.447, p = 0.0059), but not in female patients.
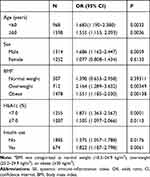 |
Table 4 Subgroup Analysis of the Associations Between SII and Diabetic Depression |
Associations Between SII and Inflammatory Markers
Correlations between SII and inflammatory markers are summarized in Table 5. The SII levels were significantly correlated with the inflammatory markers (hs-CRP, neutrophil-to-lymphocyte ratio, and platelet-to-lymphocyte ratio) in the diabetes mellitus (P<0.001), and these correlations were stronger in depressive symptoms (hs-CRP, r = 0.6073, P <0.001)
 |
Table 5 Correlations Between SII vs Different Variables in All Subjects and Depressive Symptoms |
Discussion
To the best of our knowledge, this is the first study that demonstrates the close association between SII and depression in people with DM. Our results show that patients with DM suffering from depression had significantly higher SII levels than did those without depression. Additionally, high SII levels were an independent risk factor for diabetic depression.
As a major mental illness, depression is an important chronic comorbidity of DM.30 Multiple meta-analyses show that DM is a risk factor for depression, and a bi-directional relationship has been shown between the two.6,31–33 Studies show that 20% to 40% of individuals with diabetes experienced symptoms of depression.33 Depression is associated with poor health behaviors, including smoking, physical inactivity, and caloric intake, that increase the risk of diabetes.34 Depression is associated with macrovascular complications,35 all of which cause mortality in patients with diabetes.36 It is important to identify biomarkers for early detection of depression in patients with diabetes. A potential link between chronic inflammatory states and depression has also been proposed.37
SII was determined based on the counts of three types of circulating immune cells: neutrophils, lymphocytes, and platelets. The SII level reflects the inflammatory state and could serve as a readily detectable biomarker for systemic inflammatory activity.38 Our results show that patients with diabetes suffering from depression had significantly higher SII levels than did those without depression and that high SII levels are an independent risk factor for diabetic depression. After matching the possible confounding factors, we found that SII, the neutrophil to lymphocyte ratio, and platelet to lymphocyte ratio were associated with depression, but that SII had the highest risk. The SII level provides more clinical information than do NLR and PLR. Patients with high SII levels often have thrombocytosis, neutrophilia, or lymphopenia.39 Lymphocytes and neutrophils mediate adaptive and innate immunity. Neutrophils, which constitute the largest proportion of white blood cells, are important for initiating and modulating immune processes40 and secrete neutrophil elastase to mediate chronic inflammation.41 Patients with increased neutrophil activity release reactive oxygen species, which may be involved in the development of depression. Lymphocytes are an important component of leukocytes, mediate adaptive immunity, and function in innate immunity. Lymphocytes are specific inflammatory mediators with regulatory or protective effects.
Platelets can be considered an aspecific first line inflammatory marker that can bind to leukocytes and the endothelium, influencing the function of inflammatory elements of these cells. Inflammatory elements including cytokines, epinephrine, serotonin, glutamate, dopamine, and P-selectin can activate platelets.42 Serotonin, glutamate, and other proinflammatory molecules such as IL-1, CD40L, and P-selectin originate from activated platelets and modulate platelet function in the pathophysiology of depression.43,44 The dense granules within platelets also contain glutamate42,45 and platelets are activated in patients with depression. Our results show that high SII levels are an independent risk factor for diabetic depression.
A single study that analyzed inflammation and CHD risk in patients with depression found that SII was significantly higher in patients with major depressive disorder than in the control group.46 However, further analysis was not performed, and the researchers did not adjust for potential confounding factors.
Our study had several strengths. The sample size in this study was large enough to identify a significant association between SII and depression in patients with diabetes. Moreover, the analysis of detailed covariate data allowed us to adjust for potential confounding factors that might influence the association between SII and depression. However, there are some limitations to our study. Firstly, the cross-sectional study design means that causality cannot be established. Prospective studies are needed to establish causality. Secondly, data used in this study were extracted from one blood test only. Serial testing may be more informative than a single test on admission because of the short life span of blood cells. Thirdly, SII is easy to measure in clinical practice but the loss of neutrophils, lymphocytes, and platelet counts is common and may lead to selection bias.
Conclusion
Here, we provide the first evidence that SII levels are associated with an increased risk of depression in patients with diabetes. This should be confirmed in prospective studies.
Data Sharing Statement
Publicly available datasets were analyzed in this study. The authors confirm that these data can be found here: https://www.cdc.gov/nchs/nhanes/.
Acknowledgments
We would like to thank Dr. Zhan Shaohan for editorial help.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Cannon A, Handelsman Y, Heile M, Shannon M. Burden of illness in type 2 diabetes mellitus. J Manag Care Spec Pharm. 2018;24(9a Suppl):S5–S13. doi:10.18553/jmcp.2018.24.9-a.s5
2. Matsubara M, Makino H, Washida K, et al. A prospective longitudinal study on the relationship between glucose fluctuation and cognitive function in type 2 diabetes: PROPOSAL study protocol. Diabetes Ther. 2020;11(11):2729–2737. doi:10.1007/s13300-020-00916-9
3. Riggin L. Association Between Gestational Diabetes and Mental Illness. Can J Diabetes. 2020;44(6):566–571.e563. doi:10.1016/j.jcjd.2020.06.014
4. Wong J, Mehta G. Efficacy of depression management in an integrated psychiatric-diabetes education clinic for comorbid depression and diabetes mellitus types 1 and 2. Can J Diabetes. 2020;44(6):455–460. doi:10.1016/j.jcjd.2020.03.013
5. Geraets A, Köhler S, Muzambi R, et al. The association of hyperglycaemia and insulin resistance with incident depressive symptoms over 4 years of follow-up: the Maastricht Study. Diabetologia. 2020;63(11):2315–2328. doi:10.1007/s00125-020-05247-9
6. Anderson RJ, Freedland KE, Clouse RE, Lustman PJ. The prevalence of comorbid depression in adults with diabetes: a meta-analysis. Diabetes Care. 2001;24(6):1069–1078. doi:10.2337/diacare.24.6.1069
7. Gilsanz P, Karter A, Beeri M, Quesenberry C, Whitmer R. The bidirectional association between depression and severe hypoglycemic and hyperglycemic events in type 1 diabetes. Diabetes Care. 2018;41(3):446–452. doi:10.2337/dc17-1566
8. Hall J, Ramachandran D, Roh H, et al. Obesity-linked PPARγ S273 phosphorylation promotes insulin resistance through growth differentiation factor 3. Cell Metab. 2020;32(4):665–675.e6. doi:10.1016/j.cmet.2020.08.016
9. Ridker P, MacFadyen J, Glynn R, Bradwin G, Hasan A, Rifai N. Comparison of interleukin-6, C-reactive protein, and low-density lipoprotein cholesterol as biomarkers of residual risk in contemporary practice: secondary analyses from the cardiovascular inflammation reduction trial. Eur Heart J. 2020;41(31):2952–2961. doi:10.1093/eurheartj/ehaa160
10. Liu Z, Zhang Y, Graham S, et al. Causal relationships between NAFLD, T2D and obesity have implications for disease subphenotyping. J Hepatol. 2020;73(2):263–276. doi:10.1016/j.jhep.2020.03.006
11. Slavich G, Sacher J. Stress, sex hormones, inflammation, and major depressive disorder: extending social signal transduction theory of depression to account for sex differences in mood disorders. Psychopharmacology. 2019;236(10):3063–3079. doi:10.1007/s00213-019-05326-9
12. Yuan N, Chen Y, Xia Y, Dai J, Liu C. Inflammation-related biomarkers in major psychiatric disorders: a cross-disorder assessment of reproducibility and specificity in 43 meta-analyses. Transl Psychiatry. 2019;9(1):233. doi:10.1038/s41398-019-0570-y
13. Akbaraly T, Brunner E, Ferrie J, Marmot M, Kivimaki M, Singh-Manoux A. Dietary pattern and depressive symptoms in middle age. Br J Psychiatry. 2009;195(5):408–413. doi:10.1192/bjp.bp.108.058925
14. Sharma S, Fulton S. Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. Int J Obes (Lond). 2013;37(3):382–389. doi:10.1038/ijo.2012.48
15. Finger B, Dinan T, Cryan J. High-fat diet selectively protects against the effects of chronic social stress in the mouse. Neuroscience. 2011;192:351–360. doi:10.1016/j.neuroscience.2011.06.072
16. Velasco Á, Rodríguez-Revuelta J, Olié E, et al. Neutrophil-to-lymphocyte ratio: a potential new peripheral biomarker of suicidal behavior. Eur Psychiatry. 2020;63(1):e14. doi:10.1192/j.eurpsy.2019.20
17. Sun H, Que J, Peng Y, et al. The neutrophil-lymphocyte ratio: a promising predictor of mortality in coronary care unit patients - A cohort study. Int Immunopharmacol. 2019;74:105692. doi:10.1016/j.intimp.2019.105692
18. Bilen M, Martini D, Liu Y, et al. The prognostic and predictive impact of inflammatory biomarkers in patients who have advanced-stage cancer treated with immunotherapy. Cancer. 2019;125(1):127–134. doi:10.1002/cncr.31778
19. Hu J, Zhou W, Zhou Z, Han J, Dong W. Elevated neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios predict post-stroke depression with acute ischemic stroke. Exp Ther Med. 2020;19(4):2497–2504. doi:10.3892/etm.2020.8514
20. Huang G, Chen H, Wang Q, et al. High platelet-to-lymphocyte ratio are associated with post-stroke depression. J Affect Disord. 2019;246:105–111. doi:10.1016/j.jad.2018.12.012
21. Hu B, Yang X, Xu Y, et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin Cancer Res. 2014;20(23):6212–6222. doi:10.1158/1078-0432.CCR-14-0442
22. Tong Y, Tan J, Zhou X, Song Y, Song Y. Systemic immune-inflammation index predicting chemoradiation resistance and poor outcome in patients with stage III non-small cell lung cancer. J Transl Med. 2017;15(1):221. doi:10.1186/s12967-017-1326-1
23. Yang Y, Wu C, Hsu P, et al. Systemic immune-inflammation index (SII) predicted clinical outcome in patients with coronary artery disease. Eur J Clin Invest. 2020;50(5):e13230. doi:10.1111/eci.13230
24. Hua X, Long Z, Zhang Y, et al. Prognostic value of preoperative systemic immune-inflammation index in breast cancer: a propensity score-matching study. Front Oncol. 2020;10:580. doi:10.3389/fonc.2020.00580
25. Curtin L, Mohadjer L, Dohrmann S, et al. National health and nutrition examination survey: sample design, 2007–2010. Vital Health Stat 2. 2013;(160):1–23.
26. Gilbody S, Richards D, Brealey S, Hewitt C. Screening for depression in medical settings with the Patient Health Questionnaire (PHQ): a diagnostic meta-analysis. J Gen Intern Med. 2007;22(11):1596–1602. doi:10.1007/s11606-007-0333-y
27. Fortney J, Pyne J, Edlund M, et al. A randomized trial of telemedicine-based collaborative care for depression. J Gen Intern Med. 2007;22(8):1086–1093. doi:10.1007/s11606-007-0201-9
28. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613. doi:10.1046/j.1525-1497.2001.016009606.x
29. Zhu L, She Z, Cheng X, et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31(6):1068–1077.e1063. doi:10.1016/j.cmet.2020.04.021
30. Moussavi S, Chatterji S, Verdes E, Tandon A, Patel V, Ustun B. Depression, chronic diseases, and decrements in health: results from the world health surveys. Lancet. 2007;370(9590):851–858. doi:10.1016/S0140-6736(07)61415-9
31. de Groot M, Anderson R, Freedland K, Clouse R, Lustman P. Association of depression and diabetes complications: a meta-analysis. Psychosom Med. 2001;63(4):619–630. doi:10.1097/00006842-200107000-00015
32. Mezuk B, Eaton W, Albrecht S, Golden S. Depression and type 2 diabetes over the lifespan: a meta-analysis. Diabetes Care. 2008;31(12):2383–2390. doi:10.2337/dc08-0985
33. Ali S, Stone M, Peters J, Davies M, Khunti K. The prevalence of co-morbid depression in adults with type 2 diabetes: a systematic review and meta-analysis. Diabet Med. 2006;23(11):1165–1173. doi:10.1111/j.1464-5491.2006.01943.x
34. Nusslock R, Miller G. Early-life adversity and physical and emotional health across the lifespan: a neuroimmune network hypothesis. Biol Psychiatry. 2016;80(1):23–32. doi:10.1016/j.biopsych.2015.05.017
35. Nouwen A, Adriaanse M, van Dam K, et al. Longitudinal associations between depression and diabetes complications: a systematic review and meta-analysis. Diabetic Med. 2019;36(12):1562–1572. doi:10.1111/dme.14054
36. Wu C, Hsu L, Wang SH. Association of depression and diabetes complications and mortality: a population-based cohort study. Epidemiol Psychiatr Sci. 2020;29:e96. doi:10.1017/S2045796020000049
37. Gialluisi A, Bonaccio M, Di Castelnuovo A, et al. Lifestyle and biological factors influence the relationship between mental health and low-grade inflammation. Brain Behav Immun. 2020;85:4–13. doi:10.1016/j.bbi.2019.04.041
38. Lolli C, Caffo O, Scarpi E, et al. Systemic immune-inflammation index predicts the clinical outcome in patients with mCRPC treated with abiraterone. Front Pharmacol. 2016;7:376. doi:10.3389/fphar.2016.00376
39. Geng Y, Shao Y, Zhu D, et al. Systemic immune-inflammation index predicts prognosis of patients with esophageal squamous cell carcinoma: a propensity score-matched analysis. Sci Rep. 2016;6(1):39482. doi:10.1038/srep39482
40. Chen J, Zhai E, Yuan Y, et al. Systemic immune-inflammation index for predicting prognosis of colorectal cancer. World J Gastroenterol. 2017;23(34):6261–6272. doi:10.3748/wjg.v23.i34.6261
41. Talukdar S, Oh D, Bandyopadhyay G, et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat Med. 2012;18(9):1407–1412. doi:10.1038/nm.2885
42. Herr D, Chew W, Satish R, Ong W. Pleotropic roles of autotaxin in the nervous system present opportunities for the development of novel therapeutics for neurological diseases. Mol Neurobiol. 2020;57(1):372–392. doi:10.1007/s12035-019-01719-1
43. Bayat M, Zabihi S, Karbalaei N, Haghani M. Time-dependent effects of platelet-rich plasma on the memory and hippocampal synaptic plasticity impairment in vascular dementia induced by chronic cerebral hypoperfusion. Brain Res Bull. 2020;164:299–306. doi:10.1016/j.brainresbull.2020.08.033
44. Karege F, Perret G, Bondolfi G, Schwald M, Bertschy G, Aubry J. Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res. 2002;109(2):143–148. doi:10.1016/S0165-1781(02)00005-7
45. Kalev-Zylinska ML, Green TN, Morel-Kopp M-C, et al. N-methyl-d-aspartate receptors amplify activation and aggregation of human platelets. Thromb Res. 2014;133(5):837–847. doi:10.1016/j.thromres.2014.02.011
46. Zhou L, Ma X, Wang W. Inflammation and coronary heart disease risk in patients with depression in China Mainland: a cross-sectional study. Neuropsychiatr Dis Treat. 2020;16:81–86. doi:10.2147/NDT.S216389
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
