Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 17
Assessment of the Effect of Rehmannia glutinosa Leaf Extract in Maintaining Skin Health: A Proof-of-Concept, Double-Blind, Randomized, Placebo-Controlled Clinical Trial
Authors Srivastava S , Huang SF, Jagtap MS
Received 8 December 2023
Accepted for publication 24 March 2024
Published 18 April 2024 Volume 2024:17 Pages 863—875
DOI https://doi.org/10.2147/CCID.S448928
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
Shalini Srivastava,1 Shu Fen Huang,2 Manjiri S Jagtap3
1Department of Clinical Development and Strategy, Vedic Lifesciences Pvt. Ltd., Andheri West, Mumbai, Maharashtra, India; 2Department of Biochemistry, NuLiv Holding Inc., Taipei City, Taiwan (Republic of China); 3Skin Cure n Care Clinic, Thane, Maharashtra, India
Correspondence: Shalini Srivastava, Department of Clinical Development and Strategy, Vedic Lifesciences Pvt. Ltd., Morya House, 118-B, Off Link Road, Andheri West, Mumbai, Maharashtra, 400053, India, Tel +91-9619477885, Email [email protected]
Purpose: A double-blind, placebo-controlled, randomized, proof-of-concept trial aimed to evaluate the efficacy and safety of VerbasnolTM [Rehmannia glutinosa Libosch leaf-based extract (RGLE)] in females, with moderate to severe acne vulgaris.
Participants and Methods: Twenty-two females aged 18 to 35 years having moderate to severe acne with Global Acne Grading System (GAGS) scores of 19 to 38 were included in the study and were randomized in a 1:1 ratio to receive either one capsule (100 mg/day) of RGLE or placebo orally after breakfast for 56 days. The primary outcome was a change in acne severity measured by the GAGS compared to the placebo on day 56. The secondary outcomes were changes in the number of inflammatory acne lesions, facial sebum secretion, quality of life, local pain and itching, skin wrinkle severity, and other skin characteristics, including radiance, luminosity, smoothness, texture, firmness, and hydration. Additionally, the percentage of responders and global tolerability and efficacy were evaluated.
Results: The mean GAGS score was reduced by 21.72% and 14.20% on day 28 in RGLE (n=10) and placebo groups (n=12), respectively, which further reduced in both groups on day 56. The RGLE group reported better improvement in other skin characteristics on day 56. No safety or tolerability concerns were reported for the extract. RGLE reduced acne and improved the skin quality in females compared to placebo as early as 28 days of supplementation.
Conclusion: RGLE supplementation at a dose of 100 mg/day has provided a clinically relevant decrease in acne severity and improved the skin hydration and quality of life of the participants with acne after 56 days of dose administration.
Keywords: acne, healthy skin, herbal extract, Rehmannia glutinosa, quality of life
Introduction
Our skin constantly remains in contact with the outside environment, acting as a barrier to protect our body from water and solute loss, disease infiltration, and chemical or physical assaults. In clinical practice, skin conditions such as acne vulgaris are generally associated with the psychological status of the affected individual.1 A recent analysis concluded that acne-affected populations had increased worldwide by 48% since 1990. The number of women with acne is 1.3 times more than men.2 Acne vulgaris is an inflammatory condition of the pilosebaceous glands that manifests as inflammatory and non-inflammatory lesions, primarily on the face (99%), followed by the upper arms, trunk, and back.3,4 The major pathogenic factor responsible for acne is increased sebum production by the sebaceous gland under the influence of circulating androgens leading to abnormal keratinization of the sebaceous duct. Additionally, the colonization of bacteria named Propionibacterium acnes results in an inflammatory response in the form of papules, pustules, nodules, and cysts.4 Large acne lesions can also leave scars that negatively impact the affected individuals’ psychosocial behavior, more so in cases of females. Research has shown that adult women with acne have higher anxiety, depression, lower self-esteem, poor self-image, and decreased quality of life.4–6 As a multifactorial condition, acne requires a holistic and individualistic management plan that simultaneously targets its several pathogenic mechanisms. Most therapies aim to reduce sebum production and inflammatory reactions, minimize scarring, and improve the individual’s appearance and psychosomatic state.7 Although isotretinoin and benzoyl peroxide have lately been the drug of choice for many dermatologists, the increasing cases of antibiotic resistance urges in developing acne management strategies that can successfully eliminate antibiotics.8,9
Rehmannia glutinosa Libosch is widely used in Traditional Chinese Medicine for its various health benefits, such as cardiovascular, neuroprotective, and nephroprotective effects.10 The R. glutinosa Libosch leaf extract (RGLE) has been used for its heat-clearing, blood circulation improvement, Qi and yin nourishing, and kidney tonifying properties. It has been officially listed in the Beijing Standard of Chinese Herbal Medicines.10,11 The main phytoconstituents comprising its leaf are iridoid glycosides (catalpol and ajugol) and phenylethanoid glycosides, including verbascosides or acteosides.12 Verbascoside is a potent anti-oxidant, anti-microbial, anti-inflammatory, and photo-protective agent; therefore, it has applications in several cosmetic products.13–15 Furthermore, this RGLE extract has also been concluded to be safe based on several histological investigation studies. One of these preclinical studies conducted by Waisundara et al indicated that R. glutinosa extract did not exert any negative impact or damage to the hepatic or renal tissues as evident by the histological analysis in streptozotocin (STZ)-induced diabetes rats and was found to be safe.16 Based on this background, the present study aimed to evaluate the safety and efficacy of RGLE in females with moderate to severe acne as per the Global Acne Grading System (GAGS) score.
Materials and Methods
Ethics Approval and Informed Consent
This study was conducted in compliance with the ethical principles of the Declaration of Helsinki, 1975, as revised in 2008; ICH-GCP, 2016; and Ethical guidelines for biomedical research on human participants, 2006, issued by the Indian Council of Medical Research (ICMR), India. It was registered on clinicaltrials.gov (NCT0937374). The study was approved and monitored by the Institutional Ethics Committee of New Healthcare Nursing Home (ECR/1388/Inst/MH/2020), Thane, India (Protocol ID: NS/201101/REHGLU/AV; Date: May 27, 2021). Only participants who provided written informed consent were recruited into the study. The study results have been reported in compliance with the Consolidated Standards of Reporting Trials (CONSORT) 2010 statement.
Study Design
This randomized, double-blind, placebo-controlled, proof-of-concept clinical study was conducted between June 2021 and October 2021 under the supervision of a qualified dermatologist. A total of 10 participants in each group were planned. Approximately 33 potential participants were screened, out of which 25 were randomized in a ratio of 1:1 to receive either RGLE 100 mg or placebo (microcrystalline cellulose). The randomization chart was generated by the block randomization method with a block size of 4 using StatsDirect software (version 3.1.17) by an independent statistician. The participants, investigator, and study researchers were blinded to the study product allocation.
Participants
The participants were recruited from the outpatient department of a dermatology clinic located in Mumbai, Maharashtra, India. Females aged 18 to 35 years with body mass index (BMI) in the range of ≥ 18.5 to ≤ 29.9 kg/m2 and having moderate to severe acne with GAGS scores of 19 to 38 were included in the study. The participants were asked to continue their usual skincare regime and abstain from other anti-acne preparations. Females with ongoing menstruation at the screening visit were rescreened, and all visits were conducted only after the end of the menstrual period.
The exclusion criteria included the consumption of any over-The-counter or prescribed oral anti-acne medications during the last 3 months, including local or systemic steroids in the previous 6 weeks, antifungals and antibiotics in the last 2 weeks before the screening, known cases of any other hormonal disorder, and consumption of >3 cups of coffee in a day.
Interventions
VerbasnolTM (RGLE) is a proprietary verbascoside [a caffeoyl phenylethanoid glycoside (C29H36O15)] extracted from R. glutinosa leaves using ethanol as a solvent through a proprietary extraction technology. It has been used in oral and topical skin care preparations. R. glutinosa leaf extract has been successfully marketed as one of the ingredients in a dietary supplement named SmarterSkin™. In the current study, participants were advised to consume one capsule of the study products (RGLE or placebo) after breakfast for 56 days. The Investigational Product (IP) and placebo capsules were matched for size, shape, color, and texture to preserve the blinding. They were packed in high-density polyethylene bottles, which were identical in size, color, and labeling. The products were manufactured in a good manufacturing practice (GMP)-compliant facility.
Study Conduct
During the screening visit, the study procedures were thoroughly explained to the research participants in a language well understood by them, following which voluntarily a duly signed and dated informed consent was obtained. The participants were screened for the eligibility criteria, and those who qualified were instructed to report on day 0 for randomization and baseline assessments. The follow-up assessments were conducted on days 28 and 56, as depicted in Figure 1. Demographic and baseline characteristics such as anthropometric parameters (height, weight and BMI), vital signs (pulse rate and blood pressure), clinical examination and medical history with prior and concomitant medication were recorded. Participants were instructed to self-administer the GAGS scale. A minimum of 90% Investigational Product (IP) compliance was required during the study.
Study Outcomes
Primary Outcome
The primary outcome of the present study was a reduction in acne severity global scores as assessed by GAGS17 on days 28 and 56 from baseline and compared to placebo.
Secondary Outcomes
The secondary outcomes were the change in the count of inflammatory acne lesions such as papules, pustules, and nodules; facial sebum secretion as assessed by Sebumeter® SM 815 (Courage + Khazaka electronic GmbH, Köln, Germany); quality of life as assessed by total scores of Acne-quality of life (QoL);18 pain and itch visual analog scale (VAS) scores of the inflammatory lesions; skin wrinkle severity as assessed by Modified Fitzpatrick Wrinkle Severity (MFWS) Scale;19 other skin characteristics including radiance, luminosity, smoothness, texture, firmness, and skin hydration as evaluated by a participant based self-assessment questionnaire using Numeric Rating Scale (NRS)20 on days 0, 28 and 56. Additionally, the percentage of responders,21 defined as the percentage of participants with improvement by one grade or more on GAGS,20 were analyzed on days 28 and 56. The global evaluation of tolerability and efficacy of the intervention was performed by the participants and the investigators, respectively, on day 56 and compared to the placebo.
The safety of the intervention was determined by monitoring vitals (pulse rate and blood pressure) and liver and renal function (aspartate transaminase, alanine aminotransferase, alkaline phosphatase, and serum creatinine) throughout the study. In addition, the occurrence of any adverse events was closely monitored.
Statistical Analysis
The sample size was determined based on previously published similar studies.22,23 The continuous variables are reported as mean (standard deviation), and categorical variables are reported as number (%). The null hypothesis was that RGLE, when consumed for 56 days, does not affect acne severity in patients suffering moderate to severe acne, as identified by no reduction in total scores of GAGS at the end of 56 days from baseline compared to placebo. The study intended to explore the trends by descriptive analysis. Hence, a significance threshold was not considered for the present study.24 All the statistical analyses were performed using MS Excel 2016.
Quality Assurance
Vedic Lifesciences monitored and audited the study to ensure compliance with the study protocol and ICH-GCP E6 (R2) guidelines.
Results
Demographic and Baseline Characteristics
Of the 28 participants screened, 25 were enrolled in the study, and 22 participants completed the study (RGLE, n=10; and placebo, n=12). The detailed participant disposition is shown in Figure 2. All efficacy and safety endpoints were evaluated using the per-protocol population. The investigational product (IP) compliance was >95% for both groups (Table 1).
 |
Table 1 Demographics and Other Characteristics at the Screening Visit |
 |
Figure 2 Participant disposition. Abbreviations: RGLE, Rehmannia glutinosa Libosch leaf-based extract; n, no of participants. |
Global Acne Grading System (GAGS)
The mean GAGS score at baseline was 24.4 (3.2) and 24.08 (1.62), which reduced to 19.1 (4.41) and 20.67 (2.93) at day 28 with a mean decrease of 5.3 (2.91) and 3.42 (3.37), in the RGLE and placebo groups, respectively. This change accounted for a 21.72% decrease in the GAGS in the RGLE group compared to 14.20% in the placebo group. (Table 2) On day 56, there was a further reduction in acne severity in both groups; however, the magnitude of reduction from baseline was greater in the RGLE group with a mean reduction of −6.7 (2.16) compared to the placebo −5.75 (3.22). The scores suggest that both the treatment groups moved to the mild from the moderate acne category by the end of the study. The reduction of the GAGS score in the placebo group may be attributed to the self-regressive nature of the Acne vulgaris (Table 2).
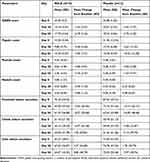 |
Table 2 Summary of GAGS Score, Inflammatory Acne Lesions, and Sebum Secretion in the per-Protocol Population |
Inflammatory Acne Lesions
The inflammatory lesions, namely papules and pustules, were reduced in both the study groups on days 28 and 56. The RGLE group was found to be more effective in reducing the pustule count than the placebo [−3.50 (3.37) vs −1.50 (4.91)]. At the same time, papule count was reduced more in the placebo group [−10.42 (12.38)] compared to the RGLE group [−5.00 (7.51)]. The inflammatory lesions in the form of nodules count were 0.8 for the RGLE group and nil in the placebo group at baseline. On day 28, in the RGLE group, the mean count of nodules decreased by 0.3 (1.34) and increased by 0.33 (0.65) in the placebo group. On day 56, in the RGLE group, the nodules completely disappeared from the face, whereas they were still present in the placebo group 0.08 (0.29). A notable reduction was observed in the pustular and nodular inflammatory lesions at day 56 in the RGLE group (Table 2).
Sebum Secretion
The sebum secretion progressively decreased by 4.20 (26.46) and 6.97 (35.38) and increased by 4.37 (24.83) on day 28 follow-up on the forehead, cheek, and chin region, respectively, in the RGLE group. While in the placebo group, the sebum secretion scores for the forehead and cheek were found to have decreased by 14.19 (56.57) and 7.08 (22.87) and increased by 1.78 (39.79) in the chin areas on day 28 (Table 2).
Acne-Quality of Life Score
The mean total score of Acne-QoL improved to 80.00 (26.41) and 80.67 (14.88) on the day 28 follow-up, with a mean increase of 22.50 (23.01) and 20.67 (22.02) in RGLE and placebo groups, respectively. The Acne QoL emotional role, social role, and symptoms showed better numerical improvements in the RGLE group on day 28 follow-up compared to the placebo. All four domains of acne QoL reported improved scores on day 28 and day 56, resulting in an improvement of total acne QoL on day 56 32.1 (29.86) in the RGLE group and 32.33 (32.86) in the placebo group (Table 3).
 |
Table 3 Summary of Acne-QoL, Pain and Itch VAS, and Wrinkle Severity |
Pain and Itch VAS
The VAS score for pain and itchiness on days 28 and 56 decreased by 3.00 (14.94) and 15.00 (27.18) respectively in the RGLE group and 12.50 (13.57) and 19.17 (25.39) respectively in the placebo group (Table 3).
Wrinkle Severity
The investigator evaluated and rated the wrinkle on the nasolabial folds visually. On day 28, the mean wrinkle severity decreased numerically by 0.10 (0.32) in the RGLE group, while no change was reported in the placebo group (Table 3).
Responder Analysis
As per the GAGS score, all the participants were in the moderately severe category at baseline in both groups. The responder analysis demonstrated that at the end of the study, 80% of participants in the RGLE group moved to the mild category from moderate. In contrast, in the placebo group, only 50% of participants moved to the mild category (Figure 3). This shows that RGLE had a better effect on the study’s primary outcome than the placebo.
 |
Figure 3 Percentage responders. Abbreviation: RGLE, Rehmannia glutinosa Libosch leaf-based extract. |
Skin Radiance, Complexion, Luminosity, Homogeneity, Smoothness, Texture, Firmness, and Skin Hydration
Participants were asked to rate a series of questions regarding radiance, complexion, luminosity, homogeneity, smoothness, texture, firmness, and skin hydration. At day 56, the mean change in the ratings in the RGLE group improved by 2.9 (2.85), 3.1 (2.77), 2.6 (2.84), 3.1 (3.03), 2.5 (3.17), 3.0 (2.54), 1.0 (2.4) and 2.8 (2.74) for radiance, complexion, luminosity, homogeneity, smoothness, texture, firmness, and skin hydration, respectively. In contrast, the placebo group reported a mean change of 1.25 (3.05), 2.42 (1.88), 2.33 (2.77), 2.33 (2.67), 2.00 (4.47), 1.67 (3.14), 0.42 (4.21) and 1.42 (2.94) for radiance, complexion, luminosity, homogeneity, smoothness, texture, firmness, and skin hydration, respectively These data demonstrated a better effect of RGLE over the placebo group on all the skin health parameters (Table 4).
 |
Table 4 Summary of NRS Rating for Various Skin Characteristics |
Global Evaluation of Efficacy and Tolerability of the Intervention
The RGLE group reported better tolerability and efficacy than the placebo group, as most of the participants and investigators in the RGLE group marked a good or excellent response during the evaluation (Table 5).
 |
Table 5 Global Evaluation of Tolerability and Efficacy of the Intervention |
Safety Outcomes
The mean pulse rate at baseline was 84.92 (5.47) and 85.15 (4.71) beats/min for the RGLE and placebo groups, respectively. No significant change from baseline values was observed in the pulse rate of the participants in both groups during the study. The mean systolic and diastolic blood pressure stayed around 115 mmHg and 75 mmHg for both groups throughout the study. The liver and renal functions were also within the normal range at the end of the study for both groups. Additionally, no adverse events were reported in this study.
Discussion
In this randomized, double-blind, placebo-controlled, proof-of-concept study, the primary objective was to clinically demonstrate the effects of RGLE on the severity of Acne vulgaris in adult women compared to the placebo. The anti-allergic and anti-inflammatory properties of the extract have been previously investigated in several animal studies.25–28 An earlier preclinical study has shown that R. glutinosa displays anti-aging effects through decreased senescence of hematopoietic stem cells.29 So far, none of the clinical studies have been conducted in humans to investigate the effect of the extract on acne vulgaris. However, many anti-acne products in the market contain R. glutinosa as one of the ingredients. The current study hypothesized that RGLE when used for 56 days, can reduce acne and improve skin quality. The study showed that the investigational product decreased acne severity by approximately 22% compared to 14% in placebo on day 28 wherein 80% of the participants moved from moderate to mild category. Being a proof-of-concept study, the change in the RGLE group on day 28 provides reasonable confidence to design a larger sample size study to assess the efficacy of RGLE. A study showing the effects of herbal polymer-based hydrogel in 24 participants reported that >50% of participants recovered from moderate-to-severe acne problems after 14 days with application three times per day.30 However, the absence of a control arm was a major limitation.
R. glutinosa leaf extract’s effect on inflammatory lesions was also proven by a decrease in the number of pustules and nodules. As pustular and nodular lesions generally manifest in bacterial infection, this noteworthy decrease in such lesions may also indicate an antibacterial effect of the RGLE.
A positive association between sebum production and inflammatory lesions has been shown in several investigations.31 As a result, the formulation’s effects on sebum discharge on the forehead, cheeks, and chin of individuals with acne-prone skin were investigated further. However, no positive results were observed in the RGLE group compared to the placebo.
On day 56, the RGLE group demonstrated a superior effect in all the skin characteristics, such as radiance, complexion, luminosity, homogeneity, smoothness, texture, firmness, and skin hydration, compared to the placebo group. These results signified a positive effect of RGLE on skin health. Females with acne are more prone to high anxiety and adverse psychological effects.32 Keeping this in mind, Acne-QoL was self-administered by the participants to evaluate their quality of life. In the present study, participants in both groups reported low scores for self-perception, emotional role, social role, and symptoms in Acne-QoL at baseline. A comparatively substantial improvement was seen in the total Acne-QoL score in the RGLE group at the end of the study. RGLE was well tolerated, as evidenced by no change in the vitals, liver and kidney function, and global tolerability evaluation by participants. Our study was the first clinical study that investigated the effect of RGLE on acne vulgaris. We included only female participants as acne prevalence is higher in females than in their male counterparts. We infer that the relatively small sample size in this study might have resulted in the lack of statistical or clinical significance of the observed effect of RGLE. The effects of RGLE on the male population with acne still need to be evaluated. Also, to confirm and generalize the results of this study, future clinical studies with a large sample size are suggested.
Conclusion
Overall, RGLE supplementation at a dose of 100 mg/day has shown a clinically relevant decrease in acne severity and, at the same time, improved the skin hydration and quality of life of the participants with acne. As already reported, R. glutinosa has potent anti-inflammatory properties, confirmed by the reduction of the inflammatory comedones in the current study by RGLE. However, no effect was observed on sebum secretion and wrinkle severity. The results of this preliminary study provide a valid proof-of-concept for designing a more extensive investigation to confirm the efficacy of RGLE supplementation in managing acne.
Abbreviations
CONSORT, Consolidated Standards Of Reporting Trials; EC, Ethics Committee; GAGS, Global Acne Grading System; GMP, Good Manufacturing Practice; IP, Investigational Product; ICH-GCP, International Conference On Harmonization - Good Clinical Practice; MFWS, Modified Fitzpatrick Wrinkle Severity Scale; NRS, Numeric Rating Scale; RGLE, Rehmannia glutinosa libosch leaf extract; TSH, Thyroid Stimulating Hormone; UPT, Urine Pregnancy Test; VAS, Visual Analog Scale.
Data Sharing Statement
The data presented in the study is available at a reasonable request from the corresponding author.
Acknowledgments
The authors thank all the participants who volunteered for the study. The authors express their gratitude to NuLiv Science USA Inc. for providing the study products and funds for executing the study. The authors acknowledge Vedic Lifesciences for facilitating the study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
NuLiv Science USA Inc. provided the study products and sponsored the study.
Disclosure
Shalini Srivastava is affiliated with Vedic Lifesciences Pvt. Ltd. Shu Fen Huang is an employee of NuLiv Holding Inc. Manjiri S Jagtap is affiliated with Skin Cure n Care Clinic. The authors report no other conflicts of interest in this work.
References
1. Gieler U, Gieler T, Peters EMJ, Linder D. Skin and psychosomatics – psychodermatology today. J Dtsch Dermatol Ges. 2020;18(11):1280–1298. doi:10.1111/ddg.14328
2. Chen H, Zhang T, Yin X, Man J, Yang X, Lü M. Magnitude and temporal trend of acne vulgaris burden in 204 countries and territories from 1990 to 2019: an analysis from the Global Burden of Disease Study 2019*. Br J Dermatol. 2022;186(4):673–683. doi:10.1111/bjd.20882
3. Sutaria AH. Acne vulgaris. StatPearls - NCBI Bookshelf; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459173/.
4. Tan J, Beissert S, Cook-Bolden F, et al. Impact of facial and truncal acne on quality of life: a multi-country population-based survey. JAAD Int. 2021;3:102–110. doi:10.1016/j.jdin.2021.03.002
5. Nguyen CM, Beroukhim K, Danesh MJ, Babikian A, Koo J, Leon A. The psychosocial impact of acne, vitiligo, and psoriasis: a review. Clin Cosmet Invest Dermatol. 2016;9:383–392. doi:10.2147/CCID.S76088
6. Bagatin E, Freitas THP, Rivitti-Machado MC, et al. Adult female acne: a guide to clinical practice [published correction appears in An Bras Dermatol. 2019 Mar-Apr;94(2):255. Machado MCR [corrected to Rivitti-Machado MC]]. An Bras Dermatol. 2019;94(1):62–75. doi:10.1590/abd1806-4841.20198203
7. Branisteanu DE, Toader MP, Porumb EA, et al. Adult female acne: clinical and therapeutic particularities (Review). Exp Ther Med. 2022;23(2):151. doi:10.3892/etm.2021.11074
8. Adler BL, Kornmehl H, Armstrong AW. Antibiotic resistance in acne treatment. JAMA Dermatol. 2017;153(8):810–811. doi:10.1001/jamadermatol.2017.1297
9. Walsh TR, Efthimiou J, Dréno B. Systematic review of antibiotic resistance in acne: an increasing topical and oral threat. Lancet Infect Dis. 2016;16(3):e23–e33. doi:10.1016/S1473-3099(15)00527-7
10. Dai X, Su S, Cai H, et al. Protective effects of total glycoside from Rehmannia glutinosa leaves on diabetic nephropathy rats via regulating the metabolic profiling and modulating the TGF-β1 and Wnt/β-catenin signaling pathway. Front Pharmacol. 2018;9:1012. doi:10.3389/fphar.2018.01012
11. Wang Y, Liao D, Qin M, Li X. Simultaneous determination of catalpol, aucubin, and geniposidic acid in different developmental stages of Rehmannia glutinosa leaves by high performance liquid chromatography. J Anal Methods Chem. 2016;2016:4956589. doi:10.1155/2016/4956589
12. Wu L, Georgiev MI, Cao H, et al. Therapeutic potential of phenylethanoid glycosides: a systematic review. Med Res Rev. 2020;40(6):2605–2649. doi:10.1002/med.21717
13. Kim SS, Son YO, Chun JC, et al. Antioxidant property of an active component purified from the leaves of paraquat-tolerant Rehmannia glutinosa. Redox Rep. 2005;10(6):311–318. doi:10.1179/135100005X83734
14. Alipieva K, Korkina L, Orhan IE, Georgiev MI. Verbascoside--a review of its occurrence, (bio)synthesis and pharmacological significance. Biotechnol Adv. 2014;32(6):1065–1076. doi:10.1016/j.biotechadv.2014.07.001
15. Vertuani S, Beghelli E, Scalambra E, et al. Activity and stability studies of verbascoside, a novel antioxidant, in dermo-cosmetic and pharmaceutical topical formulations. Molecules. 2011;16(8):7068–7080. doi:10.3390/molecules16087068
16. Waisundara VY, Huang M, Hsu A, Huang D, Tan BK. Characterization of the anti-diabetic and antioxidant effects of rehmannia glutinosa in streptozotocin-induced diabetic Wistar rats. Am J Chin Med. 2008;36(6):1083–1104. doi:10.1142/S0192415X08006594
17. Doshi A, Zaheer A, Stiller MJ. A comparison of current acne grading systems and proposal of a novel system. Int J Dermatol. 1997;36(6):416–418. doi:10.1046/j.1365-4362.1997.00099.x
18. Fehnel SE, McLeod LD, Brandman J, et al. Responsiveness of the Acne-Specific Quality of Life Questionnaire (Acne-QoL) to treatment for acne vulgaris in placebo-controlled clinical trials. Qual Life Res. 2002;11(8):809–816. doi:10.1023/a:1020880005846
19. Shoshani D, Markovitz E, Monstrey SJ, Narins DJ. The modified Fitzpatrick Wrinkle Scale: a clinical validated measurement tool for nasolabial wrinkle severity assessment. Dermatol Surg. 2008;34(Suppl 1):S85–S91. doi:10.1111/j.1524-4725.2008.34248.x
20. Dumoulin M, Gaudout D, Lemaire B. Clinical effects of an oral supplement rich in antioxidants on skin radiance in women. Clin Cosmet Invest Dermatol. 2016;9:315–324. doi:10.2147/CCID.S118920
21. Snapinn SM, Jiang Q. Responder analyses and the assessment of a clinically relevant treatment effect. Trials. 2007;8:31. doi:10.1186/1745-6215-8-31
22. Lone AH, Habib S, Ahmad T, Anwar M. Effect of a Polyherbal Unani formulation in acne vulgaris: a preliminary study. J Ayurveda Integr Med. 2012;3(4):180–183. doi:10.4103/0975-9476.104430
23. Fabbrocini G, Staibano S, De Rosa G, et al. Resveratrol-containing gel for the treatment of acne vulgaris: a single-blind, vehicle-controlled, pilot study. Am J Clin Dermatol. 2011;12(2):133–141. doi:10.2165/11530630-000000000-00000
24. Lee EC, Whitehead AL, Jacques RM, Julious SA. The statistical interpretation of pilot trials: should significance thresholds be reconsidered? BMC Med Res Methodol. 2014;14:41. doi:10.1186/1471-2288-14-41
25. Sung YY, Yoon T, Jang JY, Park SJ, Kim HK. Topical application of Rehmannia glutinosa extract inhibits mite allergen-induced atopic dermatitis in NC/Nga mice. J Ethnopharmacol. 2011;134(1):37–44. doi:10.1016/j.jep.2010.11.050
26. Kim H, Lee E, Lee S, Shin T, Kim Y, Kim J. Effect of Rehmannia glutinosa on immediate type allergic reaction. Int J Immunopharmacol. 1998;20(4–5):231–240. doi:10.1016/s0192-0561(98)00037-x
27. Ferguson EA, Littlewood JD, Carlotti DN, Grover R, Nuttall T. Management of canine atopic dermatitis using the plant extract PYM00217: a randomized, double-blind, placebo-controlled clinical study. Vet Dermatol. 2006;17(4):236–243. doi:10.1111/j.1365-3164.2006.00523.x
28. Mahajan SG, Mehta AA. Suppression of ovalbumin-induced Th2-driven airway inflammation by β-sitosterol in a Guinea pig model of asthma. Eur J Pharmacol. 2011;650(1):458–464. doi:10.1016/j.ejphar.2010.09.075
29. Bai L, Shi GY, Yang YJ, Chen W, Zhang LF, Qin C. Rehmannia glutinosa exhibits anti-aging effect through maintaining the quiescence and decreasing the senescence of hematopoietic stem cells. Animal Model Exp Med. 2018;1(3):194–202. doi:10.1002/ame2.12034
30. Lin YY, Lu SH, Gao R, et al. A novel biocompatible herbal extract-loaded hydrogel for acne treatment and repair. Oxid Med Cell Longev. 2021;2021:5598291. doi:10.1155/2021/5598291
31. Dréno B. What is new in the pathophysiology of acne, an overview. J Eur Acad Dermatol Venereol. 2017;31(Suppl 5):8–12. doi:10.1111/jdv.14374
32. Aktan S, Ozmen E, Sanli B. Anxiety, depression, and nature of acne vulgaris in adolescents. Int J Dermatol. 2000;39(5):354–357. doi:10.1046/j.1365-4362.2000.00907.x
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

