Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Assessment of physical functioning and handling of tiotropium/olodaterol Respimat® in patients with COPD in a real-world clinical setting
Authors Steinmetz KO, Abenhardt B, Pabst S, Hänsel M , Kondla A , Bayer V, Buhl R
Received 24 November 2018
Accepted for publication 17 May 2019
Published 4 July 2019 Volume 2019:14 Pages 1441—1453
DOI https://doi.org/10.2147/COPD.S195852
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Karl-Otto Steinmetz,1 Birgit Abenhardt,2 Stefan Pabst,3 Michaela Hänsel,4 Anke Kondla,5 Valentina Bayer,6 Roland Buhl7
1LungCenter Darmstadt, Darmstadt, Germany; 2Pulmonary Practice, Heidelberg, Germany; 3Lung Center, Bonn-Duisdorf, Germany; 4TA Respiratory/Biosimilars, Boehringer Ingelheim International GmbH, Ingelheim am Rhein, Germany; 5Boehringer Ingelheim Pharma GmbH & Co. KG, Ingelheim am Rhein, Germany; 6Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT, USA; 7Pulmonary Department, Johannes Gutenberg University Hospital Mainz, Mainz, Germany
Background: Patients with chronic obstructive pulmonary disease (COPD) show signs of reduced physical activity from the early stages of the disease, impacting morbidity and mortality. Data suggest treatment with tiotropium, a long-acting muscarinic antagonist, and olodaterol, a long-acting ß2-agonist (LABA), as monotherapies and in combination, increases exercise capacity. This study assessed the effects of fixed-dose tiotropium/olodaterol (delivered via Respimat®,) on physical function in Global Initiative for Chronic Obstructive Lung Disease A–D patients requiring long-acting dual bronchodilation treatment in a real-world setting.
Methods: This open-label, single arm, noninterventional study measured changes in physical function in COPD patients treated with tiotropium/olodaterol 5/5 μg for approximately 6 weeks (between Visit 1 [baseline] and Visit 2). Primary end point was therapeutic success, defined as a minimum 10-point increase in Physical Functioning Questionnaire (PF-10) score. Secondary end points included change in PF-10 from Visit 1 to Visit 2, the patient’s general condition (measured by Physician’s Global Evaluation score) at Visit 1 and Visit 2, and patient satisfaction with treatment delivered via the Respimat®, device (assessed by Patient Satisfaction Questionnaire) at study end.
Results: Therapeutic success was observed in 51.5% of 1578 patients (95% confidence interval [CI] 49.0, 54.0) after approximately 6 weeks of treatment with tiotropium/olodaterol. Mean change in PF-10 score between Visit 1 and Visit 2 was 11.6 points (95% CI 10.7, 12.6). Patient general condition improved as indicated by a general improvement in scores between visits. Most patients were very satisfied or satisfied with tiotropium/olodaterol treatment (82.5%), inhalation (87.5%), and handling of Respimat®, (85.2%). One percent of patients reported an investigator-defined drug-related adverse events (AE).
Conclusion: Tiotropium/olodaterol treatment improved physical functioning in COPD patients. An associated increase in patient general condition was observed. Most patients were very satisfied or satisfied with tiotropium/olodaterol treatment, inhaling, and handling of the Respimat®, device. No unexpected drug-related AE occurred.
Keywords: tiotropium, olodaterol, chronic obstructive pulmonary disease, physical activity, noninterventional study
Plain language summary
Patients with COPD are less active. This can lead to poor quality of life and increased hospital visits. Treatment with drugs that open the airways can help patients to be more physically active. We tested if using two of these drugs together could help increase patients’ activity levels. We also looked at the patient’s general condition and whether they were happy with the inhaler device. We found that just over half of the patients improved their activity levels. Patients’ general condition also improved and most were satisfied with the treatment and inhaler. These results may mean that these drugs could help keep patients active and help them to stay well for longer.
Introduction
Studies of patients with chronic obstructive pulmonary disease (COPD) show that physical activity is already reduced in early disease stages (from Global Initiative for Chronic Obstructive Lung Disease [GOLD] Stage II).1,2 Exertional dyspnea in COPD can lead to limitation of daily activities, such as walking and standing; however, muscle deconditioning associated with physical inactivity also contributes to further inactivity in these patients.3,4 Reduced physical activity in patients with COPD is also associated with reduced quality of life, increased risk of hospital admission, and mortality.5,6
Bronchodilation with long-acting muscarinic antagonists (LAMAs) and long-acting ß2-agonists (LABAs) is the mainstay of COPD treatment.7 Treatment with the LAMA tiotropium is well established in patients with COPD and has been shown to improve disease outcomes, including exercise capacity and dynamic hyperinflation, two of the main factors contributing to reduced physical endurance and increased exertional dyspnea.8,9 Treatment with the LABA olodaterol has also demonstrated significant improvements in exercise endurance times.10 Guidelines recommend that combining different drug classes may improve efficacy with equivalent or lesser side effects.7
As improvement in physical activity is an important goal for any therapeutic approach to COPD, it has been the focus of recent clinical studies using combined therapies. A study of the dual bronchodilator QVA149 containing the LABA indacaterol and the LAMA glycopyrronium (110/50 µg metered dose, 85/43 μg delivered dose) in patients with moderate-to-severe COPD reported significant improvements in exercise endurance time compared with placebo, but found no difference between tiotropium alone and QVA149 treatment on exercise endurance.11 A Phase IV, multicenter, randomized, double-blind, placebo-controlled study (ACTIVATE) investigated treatment with a fixed-dose combination of the LAMA, aclidinium, and the LABA, formoterol fumarate (AB/FF; 400/12 μg twice daily, delivered via a multidose dry powder inhaler) in patients with moderate-to-severe COPD. Although the effects on the primary end point (trough functional residual capacity [FRC]) did not reach significance, AB/FF improved exercise endurance time and inspiratory capacity; furthermore, step counts were higher and there were fewer inactive patients after 4 weeks versus placebo.12 Following behavioral intervention at week 5, AB/FF maintained improvements in lung hyperinflation, exercise capacity, and physical activity versus placebo.12 The 12-week, randomized, part double-blind, placebo-controlled PHYSACTO® trial confirmed that LAMA/LABA (tiotropium/olodaterol, 5/5 μg) combined therapy delivered by the Respimat® inhaler, either alone or with exercise training, reduced physical activity-related dyspnea; in combination with a self-motivated behavior-modification program, exercise endurance time increased.13–15
Although evidence from randomized clinical trials is important, due to specific inclusion and exclusion criteria and close monitoring of study participants, observations do not always reflect daily practice. Interestingly, a real-world study investigating treatment with tiotropium and olodaterol (administered with separate inhalers) also showed significant improvements in physical functioning in GOLD B–D COPD patients.16 Real-world data on the effects of a fixed-dose combination therapy with tiotropium/olodaterol, administered in a single device (Respimat®), in patients with COPD who need treatment with two long-acting bronchodilators are not yet available.
The principal aim of this noninterventional study was therefore to measure the impact of a fixed-dose combination of tiotropium/olodaterol on physical functioning in patients with GOLD A–D COPD in a real-world setting. Further objectives were to gather insights into patient satisfaction with tiotropium/olodaterol treatment and the Respimat® device, as well as to evaluate the patients’ general condition and treatment safety.
Methods
Study design
This was an open-label, single-arm, noninterventional study of patients with COPD in whom combination treatment with two long-acting bronchodilators (LAMA/LABA) was indicated in accordance with marketing authorization, Summary of Product Characteristics (SmPCs), and 2015 GOLD COPD guidelines.17 The study was conducted at 258 German investigational sites, by mainly office-based pneumologists and general practitioners, between February 2016 and February 2017.
The study was approved by the Landesärztekammer Baden-Würtenberg (F-2015-081) according to national and international regulations and each participating physician was advised by their ethics committee.
Patients
This study enrolled male and female patients aged ≥40 years, with a diagnosis of COPD requiring long-acting dual bronchodilation (LAMA/LABA) treatment. Patients were required to provide written informed consent prior to participation.
Patients were excluded from study participation if they had contraindications according to the tiotropium/olodaterol SmPC or had been treated with a LAMA/LABA combination (free- or fixed-dose) in the previous 6 months. Patients continuing LABA + inhaled corticosteroid (ICS) treatment were excluded to avoid a double dosing of a LABA. Study exclusion extended to patients who were currently listed for lung transplantation or who were pregnant or breastfeeding. Patients unable to attend follow-up at the enrolling site within the planned study period of approximately 6 weeks or currently participating in a clinical trial or another noninterventional study of a drug or device were also excluded from study participation.
Study treatments
All included patients received treatment with the combination of tiotropium/olodaterol (5/5 μg) given as two puffs once daily for approximately 6 weeks, which is the average time between two medical consultations. Since this study was performed under routine conditions, there were no restrictions regarding concomitant medications other than those stated in the SmPC.
Assessments
Patients were assessed at baseline (Visit 1) and after 6 weeks (Visit 2). The initial evaluation at baseline included patient demographics; smoking history; medical history relating to year of COPD diagnosis, spirometric classification/patient group based on 2015 GOLD guidelines;17 exacerbations; hospitalizations; previous tiotropium/olodaterol administration; other respiratory therapeutics used within 6 months of starting tiotropium/olodaterol; and severity of breathlessness based on the modified Medical Research Council Questionnaire. At both baseline (Visit 1) and Week 6 (Visit 2), treatment with other respiratory agents, concomitant diseases, and concomitant medication was reported.
Study end points
The primary efficacy end point was “therapeutic success” at Visit 2 (approximately 6 weeks after starting treatment), as defined by a 10-point increase in the self-reported Physical Functioning Questionnaire (PF-10) score (a component of the 36-item Short Form Health Survey questionnaire, see supplementary analysis section for further detail)18 between Visit 1 and Visit 2. Secondary end points were changes in PF-10 score from Visit 1 to Visit 2; the patients’ general condition as evaluated by the Physician’s Global Evaluation (PGE) score at Visit 1 and Visit 2; and patient satisfaction with the treatment, inhalation, and handling of the Respimat® device was assessed at the end of the study via a patient satisfaction questionnaire on a 7-point ordinal scale, with divisions from “very satisfied” to “very dissatisfied” (Visit 2).
All adverse events (AEs), serious and nonserious, related to tiotropium/olodaterol delivered via the Respimat® device and all serious AEs irrespective of the relation to any drug were reported.
Statistical analysis
Any patient who received at least one dose of tiotropium/olodaterol was included in the treated set (TS). Patients in the TS who had a PF-10 score at Visit 1 and Visit 2 comprised the full analysis set (FAS). All analyses were descriptive. Primary and secondary end points were analyzed on the FAS. All subgroup analyses were performed on FAS. Safety end points and demographic/baseline data were analyzed on the TS.
For the primary end point, the percentage of patients with therapeutic success was presented together with the 95% CI. Summary statistics were provided for changes in PF-10 score, and the number and percentage of patients within each category was calculated for general patient condition and patients’ satisfaction with the tiotropium/olodaterol delivered via the Respimat® device.
Several subgroups were defined, including GOLD ABCD group classification according to the 2015 criteria17 (based on spirometry, symptoms and exacerbation history) and the 2017 criteria (based on symptoms and exacerbation history).7 Other subgroups of interest included treatment-naïve versus pretreated (any pretreatment with COPD therapies before the start of tiotropium/olodaterol), maintenance-naïve (neither LABA only, nor LAMA only, nor LABA + ICS) versus pre-treated patients (LABA only, LAMA only, or LABA + ICS), cardiac comorbidities, smoking status, and exacerbations during the last 12 months (results for smoking status and exacerbations during the last 12 months are presented in the supplementary analysis section).
In subgroups, the primary end point of therapeutic success was compared by chi-squared test or Fisher’s exact test as appropriate, and presented with the 95% CI. The secondary end point of change from baseline in PF-10 was compared by Wilcoxon Rank-Sum test (Mann–Whitney U test) or Kruskal–Wallis test in subgroups; 95% CIs were also calculated.
All reported P-values are nominal.
Results
In total, 1781 patients from 258 sites in Germany were screened (Figure 1). Of these, 1737 patients received at least one dose of treatment and could be included in the TS (97.5%); the FAS included 1578 patients (88.6%). Patients were followed over an observational period of approximately 6 weeks. Median duration between start of treatment and end of observation or discontinuation, whichever came first, was 5.3 weeks (range 0.0–34.1 weeks).
 |
Figure 1 Patient disposition. |
Baseline demographics and disease characteristics
Patient mean age was 66.5 years, 57.0% were male, 40.0% were smokers, and 39.8% had smoked in the past; 20.2% had never smoked (Table 1). 74.1% of patients had concomitant diseases, mostly affecting the cardiac and metabolic/endocrine systems, and 59.0% of patients received concomitant medication other than respiratory therapeutic agents.
 |
Table 1 Baseline characteristics and demographics |
Based on spirometry, symptoms, and exacerbations (2015 GOLD classification), most patients were classified as A (29.0%), followed by C and D (approximately 27.0% each), and B (17.5%). Based on symptoms and exacerbations (2017 GOLD classification), the majority of patients were classified as A (41.7%), followed by B (34.3%), C (13.9%), and D (10.1%). Approximately half of the patients had at least one exacerbation within the last 12 months, 11.5% of patients had at least one exacerbation-related hospitalization. The most frequent respiratory therapies received by patients in the previous 6 months were LAMAs (27.4%) or short-acting ß2-agonists (18.3%). Forty-eight people taking LAMA/LABA either free or as a fixed-dose combination were mistakenly included in the study population and identified after database lock. However, as it was prespecified not to exclude patients with such protocol violations from analysis sets, they stayed in the full analysis set as well as in the treated set.
Efficacy
The primary end point – therapeutic success at Visit 2 using the PF-10 questionnaire (an increase in self-reported physical function as measured by a 10-point increase in PF-10 score) – was achieved by 51.5% (95% CI 49.0%, 54.0%) of patients. Mean PF-10 score was 48.7 (95% CI 47.5, 50.0) at Visit 1 and 60.4 (95% CI 59.1, 61.6) at Visit 2.
Mean change in PF-10 score between baseline and approximately 6 weeks after start of treatment (secondary end point) was 11.6 points (95% CI 10.7, 12.6).
Analysis by GOLD classification
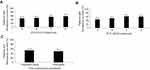
GOLD ABCD classification according to the 2015 criteria (based on spirometry, symptoms, and exacerbation history) indicated a therapeutic success rate after 6 weeks in 45.8% (95% CI 41.2%, 50.4%) of patients in GOLD subgroup A, 48.4% (95% CI 42.5%, 54.4%) in subgroup B, 54.1% (95% CI 49.2%, 58.9%) in subgroup C, and 57.5% (95% CI 52.5%, 62.3%) in subgroup D (Figure 2A). The difference in proportion of patients with therapeutic success by GOLD 2015 group was statistically significant (P=0.0027, chi-squared test).
Change in baseline PF-10 score stratified by GOLD ABCD classification according to the 2015 criteria is presented in Table 2. Change in PF-10 score between Visit 1 and Visit 2 was GOLD 2015 classification-related, with the greatest change seen in patients in GOLD C and D.
 |
Table 2 Change in baseline PF-10 score in all patients stratified by the 2015 GOLD criteria based on spirometry, symptoms, and exacerbations |
Using GOLD ABCD classification according to the 2017 criteria (based on symptoms and exacerbation history), therapeutic success rate after 6 weeks was 45.8% (95% CI 41.9%, 49.7%) in GOLD subgroup A, 51.7% (95% CI 47.4%, 56.0%) in subgroup B, 61.9% (95% CI 55.0%, 68.4%) in subgroup C, and 61.0% (95% CI 53.0%, 68.6%) in subgroup D (Figure 2B). The difference in proportion of patients with therapeutic success by GOLD 2017 group was statistically significant (P<0.0001, chi-squared test).
Change in PF-10 score between Visit 1 and Visit 2 was GOLD 2017 classification-related, with the greatest changes seen in patients in either GOLD C or D (Table 3).
 |
Table 3 Change in baseline PF-10 score in all patients stratified by the 2017 GOLD criteria based on symptoms and exacerbations |
Treatment-naïve patients versus pretreated
Among the FAS, there were more pretreated patients than treatment-naïve patients (54.4% vs 45.6%). Therapeutic success was observed in 49.6% (95% CI 46.2%, 53.0%) of pretreated patients compared with 53.8% (95% CI 50.1%, 57.5%) of treatment-naïve patients (Figure 2C); however, the difference between groups was not significant (P=0.0939, chi-squared test). Mean values of PF-10 score were higher for treatment-naïve than for pretreated patients at both visits (Visit 1: 50.6 vs 47.1; Visit 2: 63.5 vs 57.8). This was also true for mean change of PF-10 score between Visit 1 and Visit 2 (12.9 vs 10.6). The change between Visit 1 and Visit 2 was statistically significant (P=0.0129, Wilcoxon Rank-Sum test [Mann–Whitney U test]).
Maintenance-naïve patients versus pretreated
Regarding previous treatment with LABA only, LAMA only, or LABA + ICS, a greater proportion of maintenance-naïve patients (52.3%; 95% CI 49.5%, 55.2%) than patients already treated (48.9%; 95% CI 43.7%, 54.1%) achieved therapeutic success, although this was not statistically significant (P=0.2524, chi-squared test).
At Visit 1, those already treated with maintenance therapy had a higher mean PF-10 score compared with maintenance-naïve patients; mean PF-10 score at Visit 2 was 59.9 for maintenance-naïve patients and 61.9 for patients already treated. The mean PF-10 score increase between visits was slightly higher for maintenance-naïve than pretreated patients, but the change between Visit 1 and Visit 2 was not statistically significant (P=0.1943, Wilcoxon Rank-Sum test [Mann–Whitney U test]).
Patients with cardiac comorbidities
Therapeutic success was observed in 53.3% of patients with cardiac comorbidities (95% CI 49.8%, 56.8%) versus 49.7% (95% CI 46.1%, 53.3%) of patients without cardiac comorbidities (P=0.1526, chi-squared test).
At Visit 1 and Visit 2, patients without cardiac comorbidities (52.44 [95% CI 50.66, 54.22] and 63.17 [95% CI 61.43, 64.91]) had a higher mean PF-10 score than patients with cardiac comorbidities (45.21 [95% CI 43.45, 46.96] and 57.69 [95% CI 55.99, 59.39]). However, the mean change of PF-10 score between both visits was higher for patients with cardiac comorbidities (12.48 [95% CI 11.11, 13.86] versus 10.73 [95% CI 9.45, 12.01]).
General condition of the patient
The general condition of the patient, as evaluated by the physician, improved with treatment, and a shift in PGE scores from lower to higher scores was observed between Visit 1 and Visit 2 (Figure 3). At Visit 1, a large proportion of patients (52.7%) gained scores of 3 and 4, corresponding to a mediocre general condition; however, at Visit 2, the majority of patients (53.3%) achieved scores of 5 and 6, corresponding to a good general condition. The highest value, representing an excellent general condition (8), was given to 0.4% (n=7) of patients at Visit 1 and to 1.7% (n=27) of patients at Visit 2. This shift toward a higher PGE score was also observed in patients when categorized by GOLD groups. At Visit 1, 45.4% of GOLD A/B patients and 59.2% of GOLD C/D patients had PGE scores of 3 or 4. At Visit 2, both patients groups reported an improvement in PGE score with most patients scoring 5 and 6 (55.1% GOLD A/B and 51.7% GOLD C/D).
 |
Figure 3 Change from baseline PGE score in all patients.Note: Visit 2=after 6 weeks of treatment with tiotropium/olodaterol. Abbreviation: PGE, Physician’s Global Evaluation. |
Patient satisfaction with tiotropium/olodaterol Respimat®
Only one patient did not complete the satisfaction questionnaire, therefore, the response rate was 99.94%. High patient overall satisfaction rates (satisfied/very satisfied) were observed with tiotropium/olodaterol therapy (82.5%), with the great majority of patients very satisfied or satisfied with both inhaling from (87.5%) and handling of (85.2%) the Respimat® (Figure 4).
 |
Figure 4 Patient satisfaction with (A) tiotropium/olodaterol treatment, (B) inhaling from the Respimat® device, and (C) handling the Respimat® inhalation device. |
Safety
Overall, 18 (1.0%) patients reported at least one investigator-defined drug-related AE (Table 4). A total of 21 drug-related AEs were reported. These were mostly palpitations (14.3% of all events), cough (14.3%), and dry mouth (9.5%). Almost all (15 of the 18, 83.3%) patients recovered from drug-related AE events, while three patients had an ongoing AE at the end of the observation period. Five patients (0.3% of all patients) had at least one event for which a therapy was required. Treatment was discontinued in 13 patients (0.7% of all patients) due to drug-related treatment-emergent AEs, the most common reasons were palpitations (3 patients), cough (3 patients), and dry mouth (2 patients).
 |
Table 4 Summary of AEs |
Three serious AEs – cardiac arrest, cardiac failure, and cough – were documented for three patients (0.2%), two of which (cardiac arrest and cardiac failure) were fatal. The remaining event – cough – was serious for other medical reasons and resolved but led to treatment discontinuation. Only cough was considered drug-related.
Discussion
A clinically meaningful improvement in physical functioning in a COPD patient population typical of that encountered in the clinical practice setting occurred after 6 weeks of treatment with tiotropium/olodaterol. At least 50% of the patients (51.5%) achieved therapeutic success after 6 weeks, ie, a 10-point increase in the PF-10 score. This beneficial effect translated into an improvement in the patients’ general condition, as demonstrated by an improvement from baseline PGE scores. Therapeutic success was observed in more than half of the patients in the subgroups of treatment naïve, maintenance naïve, all smoking status, those with one or more exacerbations in the previous 12 months and in those with cardiac comorbidities, respectively. Interestingly, physical functioning was improved in more than 50% of patients by improving pulmonary function, without specific incentives to motivate patients to exercise. Physical functioning is affected by multiple factors and this increase could be as a result of renewed interest and motivation due to study participation.
Using the latest GOLD classification system according to the 2017 criteria (based on symptoms and exacerbation history), a higher proportion of patients in GOLD groups B, C, and D achieved therapeutic success compared with the 2015 criteria. Using either sets of criteria (spirometric vs non-spirometric), the proportions of patients with therapeutic success increased with increasing risk and disease severity. However, as with the majority of respiratory interventions, some patients did improve but did not achieve the threshold of 10 points required for “therapeutic success”, indicating room for improvement, but which may have been achieved over a longer duration of treatment.
The study demonstrated that a higher proportion of patients who were naïve to treatment and received tiotropium/olodaterol had therapeutic success than those who had received any previous COPD medication (the difference was not significant); this is likely to reflect the need for earlier treatment and subsequent increased scope for improvement in treatment-naïve patients. Therapeutic success rates were also higher in maintenance-naïve patients than pretreated with maintenance therapy (LABA only, LAMA only, or LABA + ICS) patients, although this difference was also not significant. Based on these data, tiotropium/olodaterol treatment appears suitable both as a first-line long-term therapy and as an alternate or switch option away from other therapies. Interestingly, smoking status and the presence of cardiac comorbidities did not impact on therapeutic success, again confirming that bronchodilators in COPD are effective first-line treatments largely irrespective of patient baseline characteristics. Recent findings indicate that effective bronchodilation in COPD patients can improve cardiac function mainly by reducing hyperinflation, independently supporting an early start of optimal bronchodilation in COPD.19 A clinical trial testing tiotropium/olodaterol under similar conditions is ongoing.15 Further, it is tempting to speculate that the therapeutic effects of dual bronchodilation in COPD may further improve if drug treatment is supported by behavioral measures or rehabilitation.20
Overall, at Visit 2, the majority of patients were either “very satisfied” or “satisfied” with receiving tiotropium/olodaterol treatment. Virtually identical results were observed with regard to patient satisfaction with the handling of the device. High levels of overall satisfaction with treatment, as well as satisfaction with inhaling and device handling, were reported in both treatment-naïve and pretreated patients. An observational survey found that patient satisfaction with their inhaler was associated with treatment adherence and fewer exacerbations.21 Among the most important attributes of an inhaler are ease of use and reliability,21,22 which is likely driven by the patient’s requirement to prepare and operate the device correctly in order to deliver their therapy, and because they are more likely to use an inhaler effectively if they are confident using it.23 Patients with COPD and asthma find the Respimat® device easy to use, reliable, and durable and reported high levels of satisfaction.22–24
The number of patients reporting an AE in this study was low and consistent with the safety profile of tiotropium/olodaterol.25
Potential limitations of this trial were the open-label, single-arm, noninterventional, and observational study design; hence, there was no control group and no randomization. It must be acknowledged that some patients did not complete the study due to dissatisfaction with the drug or study participation. The study was carried out over a short period of time, and so longer-lasting therapeutic effects could not be assessed; furthermore, the patients’ physical functioning and general condition were measured subjectively.
Drug administration was self-controlled and there was no treatment diary; patient use and adherence to tiotropium/olodaterol were therefore dependent on physician inquiry and could not be verified. A high proportion of patients enrolled in this study were naïve to maintenance therapy. This may suggest that many of these patients were undertreated. However, both the under-reporting of symptoms by patients and the under-treatment by physicians have been described as a serious issue worldwide26 and may be reflected here. The study was carried out in a real-world primary care setting and included a large representative sample of patients with a wide spectrum of COPD severities, background treatments, and comorbidities, thus providing valuable data and insights reflecting clinical practice.
Conclusions
These data suggest that tiotropium/olodaterol treatment helped to improve patients’ physical functioning in their daily lives, and importantly, patients were satisfied with the medication. This supports the use of tiotropium/olodaterol maintenance treatment in patients with COPD and in particular supports its use for helping patients to remain active. Further studies that will add to these data are currently ongoing (NCT02719639).27
Abbreviation list
AB/FF, aclidinium and formoterol fumarate combination; AE, adverse event; CI, confidence interval; COPD, chronic obstructive pulmonary disease; FAS, full analysis set; GERD, gastroesophageal reflux disease; GOLD, Global Initiative for Chronic Obstructive Pulmonary Disease; ICS, inhaled corticosteroid; LABA, long-acting ß2-agonist; LAMA, long-acting muscarinic antagonist; PF-10, Physical Functioning Questionnaire; PGE, Physician’s Global Evaluation; SABA, short-acting β2-agonist; SD, standard deviation; SAMA, short-acting muscarinic antagonist; SmPC, Summary of Product Characteristics; TS, treated set.
Acknowledgments
The authors received no compensation related to the development of the manuscript. This work was supported by Boehringer Ingelheim International GmbH. Medical writing assistance was provided by Victoria Kinsley, PhD, of SciMentum Ltd, who was contracted and compensated by Boehringer Ingelheim International GmbH.
Author contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
RB received grants and personal fees from Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Roche. He also received personal fees from AstraZeneca, Chiesi, Cipla and Teva, outside the submitted work. SP was an investigator in the submitted work. AK and VB are employees of Boehringer Ingelheim. MH was an employee of Boehringer Ingelheim at the time of the submitted work and is now an employee of CSL Behring GmbH. The authors report no other conflicts of interest in this work.
References
1. Troosters T, Sciurba F, Battaglia S, et al. Physical inactivity in patients with COPD, a controlled multi-center pilot-study. Respir Med. 2010;104(7):1005–1011. doi:10.1016/j.rmed.2010.01.012
2. Watz H, Waschki B, Meyer T, Magnussen H. Physical activity in patients with COPD. Eur Respir J. 2009;33(2):262–272. doi:10.1183/09031936.00024608
3. Cooper CB. Airflow obstruction and exercise. Respir Med. 2009;103(3):325–334. doi:10.1016/j.rmed.2008.10.026
4. Troosters T, van der Molen T, Polkey M, et al. Improving physical activity in COPD: towards a new paradigm. Respir Res. 2013;14:115. doi:10.1186/1465-9921-14-19
5. Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Antó JM. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61(9):772–778. doi:10.1136/thx.2006.060145
6. ZuWallack R, Esteban C. Understanding the impact of physical activity in COPD outcomes: moving forward. Eur Respir J. 2014;44(5):1107–1109. doi:10.1183/09031936.00003814
7. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Available from: http://goldcopd.org/gold-192017-global-strategy-diagnosis-management-prevention-copd/. Updated 2017.
8. Cooper CB, Celli BR, Jardim JR, et al. Treadmill endurance during 2-year treatment with tiotropium in patients with COPD: a randomized trial. Chest. 2013;144(2):490–497. doi:10.1378/chest.12-2613
9. Maltais F, Hamilton A, Marciniuk D, et al. Improvements in symptom-limited exercise performance over 8 h with once-daily tiotropium in patients with COPD. Chest. 2005;128(3):1168–1178. doi:10.1378/chest.128.3.1168
10. Maltais F, Kirsten AM, Hamilton A, De Sousa D, Voss F, Decramer M. Evaluation of the effects of olodaterol on exercise endurance in patients with chronic obstructive pulmonary disease: results from two 6-week crossover studies. Respir Res. 2016;17(1):77. doi:10.1186/s12931-016-0389-5
11. Beeh KM, Korn S, Beier J, et al. Effect of QVA149 on lung volumes and exercise tolerance in COPD patients: the BRIGHT study. Respir Med. 2014;108(4):584–592. doi:10.1016/j.rmed.2014.01.006
12. Watz H, Troosters T, Beeh KM, et al. ACTIVATE: the ffect of aclidinium/formoterol on hyperinflation, exercise capacity, and physical activity in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2017;12:2545–2558. doi:10.2147/COPD.S150887
13. Bourbeau J, Lavoie KL, Sedeno M, et al. Behaviour-change intervention in a multicentre, randomised, placebo-controlled COPD study: methodological considerations and implementation. BMJ Open. 2016;6(4):e010109. doi:10.1136/bmjopen-2015-010109
14. Troosters T, Bourbeau J, Maltais F, et al. Enhancing exercise tolerance and physical activity in COPD with combined pharmacological and non-pharmacological interventions: PHYSACTO randomised, placebo-controlled study design. BMJ Open. 2016;6(4):e010106. doi:10.1136/bmjopen-2015-010106
15. Troosters T, Maltais F, Leidy N, et al. Effect of bronchodilation, exercise training, and behavior modification on symptoms and physical activity in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(8):1021–1032. doi:10.1164/rccm.201706-1288OC
16. Sauer R, Hansel M, Buhl R, Rubin RA, Frey M, Glaab T. Impact of tiotropium + olodaterol on physical functioning in COPD: results of an open-label observational study. Int J Chron Obstruct Pulmon Dis. 2016;11:891–898.
17. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of COPD: global initiative for chronic obstructive lung disease (GOLD) 2015.
18. Brazier JE, Harper R, Jones NM, et al. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. Bmj. 1992;305(6846):160–164. doi:10.1136/bmj.305.6846.160
19. Hohlfeld JM, Vogel-Claussen J, Biller H, et al. Effect of lung deflation with indacaterol plus glycopyrronium on ventricular filling in patients with hyperinflation and COPD (CLAIM): a double-blind, randomised, crossover, placebo-controlled, single-centre trial. Lancet Respir Med. 2018;6(5):368–378. doi:10.1016/S2213-2600(18)30054-7
20. Casaburi R, Kukafka D, Cooper CB, Witek TJ
21. Chrystyn H, Small M, Milligan G, Higgins V, Gil EG, Estruch J. Impact of patients’ satisfaction with their inhalers on treatment compliance and health status in COPD. Respir Med. 2014;108(2):358–365. doi:10.1016/j.rmed.2013.09.021
22. Davis KH, Su J, Gonzalez JM, et al. Quantifying the importance of inhaler attributes corresponding to items in the patient satisfaction and preference questionnaire in patients using combivent respimat. Health Qual Life Outcomes. 2017;15(1):201. doi:10.1186/s12955-017-0780-z
23. Dekhuijzen PN, Lavorini F, Usmani OS. Patients’ perspectives and preferences in the choice of inhalers: the case for respimat(®) or HandiHaler(®). Patient Prefer Adherence. 2016;10:1561–1572. doi:10.2147/PPA.S82857
24. Schurmann W, Schmidtmann S, Moroni P, Massey D, Qidan M. Respimat soft mist inhaler versus hydrofluoroalkane metered dose inhaler: patient preference and satisfaction. Treat Respir Med. 2005;4(1):53–61.
25. Ferguson GT, Buhl R, Voss F, et al. Safety of tiotropium/olodaterol in COPD: pooled analysis of three large, 52-week randomized clinical trials. Am J Respir Crit Care Med. 2018;197:A3035.
26. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: 2018 report. Available from: https://goldcopd.org/gold-reports/.
27. ClinicalTrials.gov. Assessment of physical functioning and handling of spiolto respimat in patients with chronic obstructive pulmonary disease (COPD) requiring long-acting dual bronchodilation in routine clinical practice. Available from: https://clinicaltrials.gov/ct2/show/NCT02719639. Updated 2017.
28. Rau-Berger H, Mitfessel H, Glaab T. Tiotropium respimat® improves physical functioning in chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2010;5:367–373. doi:10.2147/COPD.S14082
Supplementary materials
PF-10 score
In this study, the PF-10 score was used, which is a component of the short form 36 health survey questionnaire (SF-36); the SF-36 is a comprehensive and sensitive measure of illness which is reliable and valid while being quick and easy to complete.1 The 10-item physical functioning questionnaire subdomain of the SF-36 can be used alone to reliably measure the effect of limited physical activity on daily living. Self-administered questions relate to restriction in daily physical activity such as vigorous (for instance, lifting heavy objects or running) and moderate activities (such as moving a table or bowling), lifting or carrying groceries, climbing several flights of stairs, climbing one flight of stairs, bending, kneeling, or stooping, walking more than one kilometer, walking several hundred meters, walking one hundred meters, and bathing or dressing yourself. Questions are answered with either “yes, limited a lot”, “yes, limited a little” or “no, not limited at all”, recorded on a 3-point Likert scale and the scores added. Scores range from 0 to 100 using the formula 100*(sum-10)/20, with higher scores indicative of better physical function.2,3
Smoking status
More than half of the patients in all smoking status stratification groups (smoker [50.8%; 95% CI 46.8%, 54.7%] vs ex-smoker [52.1%; 95% CI 48.1%, 56.0%] vs non-smoker [51.8%; 95% CI 46.1%, 57.4%]) showed therapeutic success, with no statistically significant difference between groups. At Visits 1 and 2, smokers had a higher mean PF-10 score than ex-smokers and non-smokers. The mean increase of PF-10 score between Visits 1 and 2 in all groups was between 11.3 and 12.7 points, and there was no significant difference in the increase in PF-10 score between Visit 1 and Visit 2 (P=0.8133, Wilcoxon Rank-Sum test [Mann–Whitney U test]/Kruskal–Wallis test).
Exacerbations during the last 12 months
In the group “0 exacerbations”, less than half of the patients showed therapeutic success (44.8%, 95% CI 41.3%, 48.4%). For patients in groups “1 exacerbation” and “≥2 exacerbations”, therapy was successful in 55.1% (95% CI 50.2%, 59.9%) and 61.5% (95% CI 56.4%, 66.5%) of patients, respectively (Figure S2). The difference in proportion of patients with therapeutic success was statistically significant (P<0.0001, Chi-squared test).
 |
Figure S1 Patient satisfaction questionnaire. |
 |
Figure S2 Therapeutic success at Visit 2 in all patients stratified by number of exacerbations in the previous 12 months.Note: Visit 2=after 6 weeks of treatment with tiotropium/olodaterol. |
At Visit 1, patients with 0 exacerbations and 1 exacerbation in the previous 12 months had similar mean PF-10 scores (51.41 and 50.01, respectively), which were higher than for than patients with ≥2 exacerbations (41.69). At Visit 2 mean PF-10 scores increased in all groups, the mean increase of PF-10 score was 8.28, 13.35 and 16.71 for patients with 0, 1 and ≥2 exacerbations in the previous 12 months, respectively.
Reference
1. Brazier JE, Harper R, Jones NM, et al. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. Bmj. 1992;305(6846):160–164. doi:10.1136/bmj.305.6846.160
2. Sauer R, Hansel M, Buhl R, Rubin RA, Frey M, Glaab T. Impact of tiotropium + olodaterol on physical functioning in COPD: results of an open-label observational study. Int J Chron Obstruct Pulmon Dis. 2016;11:891–898.
3. Rau-Berger H, Mitfessel H, Glaab T. Tiotropium respimat® improves physical functioning in chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2010;5:367–373. doi:10.2147/COPD.S14082
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.