Back to Journals » Patient Preference and Adherence » Volume 14
Assessing Adherence to Inhaled Corticosteroids Among Adults with Asthma in Kuwait Using the Medication Adherence Report Scale for Asthma
Authors Albassam A , Alharbi A, Awaisu A
Received 6 February 2020
Accepted for publication 12 May 2020
Published 9 June 2020 Volume 2020:14 Pages 963—970
DOI https://doi.org/10.2147/PPA.S248655
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Johnny Chen
Abdullah Albassam,1 Amal Alharbi,2 Ahmed Awaisu3
1Department of Pharmacy Practice, Faculty of Pharmacy, Kuwait University, Kuwait City, Kuwait; 2Department of Pharmacy, Farwaniya Hospital, Ministry of Health, Kuwait City, Kuwait; 3Department of Clinical Pharmacy and Practice, College of Pharmacy, QU Health, Qatar University, Doha, Qatar
Correspondence: Abdullah Albassam Email [email protected]
Purpose: Inhaled corticosteroids (ICS) are the mainstay maintenance therapy for asthma management. Non-adherence to ICS, which can lead to exacerbations and poor asthma control, is commonly reported among adult patients with asthma. The level of adherence to ICS among adult patients with asthma in Kuwait has not been previously published. The aim of this study was to assess adherence to ICS among adults with asthma in Kuwait.
Patients and Methods: A cross-sectional study was conducted among adult patients with asthma using ICS therapy at ambulatory asthma clinics in Kuwait. Adherence to ICS was assessed using the Medication Adherence Report Scale for Asthma (MARS-A). Descriptive and logistic regression analyses were conducted using SPSS version 23.
Results: One hundred and forty-nine patients with a mean age of 41.42 (12.75) years participated in the study. The majority of the participants (82.6%) reported low adherence to ICS. Approximately, three-quarters of the participants reported that they used ICS either when needed or when they felt breathless. Furthermore, about half of them admitted that they tried to avoid using ICS (46.3%), or forgot to take ICS (51%), or only used ICS before performing an exercise that made them breathless (54%). In the multivariate logistic regression analysis, independent predictors (age, education, and smoking status) were not found to significantly influence the overall adherence to ICS therapy.
Conclusion: Adherence to ICS among adult patients with asthma was found to be low in ambulatory care settings in Kuwait. Future studies should aim to determine the barriers to ICS adherence among patients with asthma, with a focus on developing effective intervention strategies.
Keywords: adherence, asthma, Kuwait, inhaled corticosteroid, medication adherence report scale
Introduction
Asthma is a chronic inflammatory disease of the airways that is characterized by recurrent episodes of wheezing, breathlessness, coughing, and chest tightness.1 According to the World Health Organization (WHO), about 235 million people have asthma globally and its prevalence has increased by 50% every decade.2 The economic cost of asthma is estimated to exceed the costs associated with tuberculosis and HIV/AIDS combined.3 In 2012, the prevalence of asthma among adults in Kuwait was estimated to be 15%, with a total of 93,923 individuals diagnosed with this condition.4 The annual total direct medical cost of asthma treatment in Kuwait was estimated to be more than KD 58 million (USD 207 million).4 On the other hand, the mean mortality rate associated with asthma in Kuwait is 1.59 per 100,000 population annually.5 In the United States (US), asthma mortality rate was 0.94 per 100,000 population in 2018.6
Persistent asthma is controlled with long-term controller medications, particularly anti-inflammatory agents. As with other chronic conditions, adherence to medications in asthma is essential to ensure effective management and favorable health outcomes.7 It was previously reported by the WHO that the average rate of adherence to treatment was about 50% among patients with chronic diseases in developed countries.8 In developing countries, where access to health care and medicines is limited, this average rate of adherence is expected to be even lower.8
Adherence monitoring techniques to evaluate the use of ICS therapy are currently employed in practice. Yet, each technique has its own advantages and disadvantages. A recent study demonstrated that hair analysis can be used to monitor adherence to ICS therapy.9 Yet, the sensitivity of analytical method for each prescribed medicine was a major limitation.9 On the other hand, indirect measurement of adherence may be classified as objective or subjective. Objective methods include medication electronic monitoring or medication event monitoring systems (MEMS) or electronic devices, prescription refill monitoring, and canister weighing.10,11 Electronic monitoring is considered as the gold standard for measuring adherence to ICS.10 However, the drawbacks of this method include its high cost and limited availability in developing countries.12 Prescription refills and canister weighing are other indirect objective methods for measuring medication adherence.12 These methods can provide population-level estimates and with trajectory modelling one can identify different patterns of use;13 however, they are coupled with drawbacks, like medication dumping, sharing therapy with other patients with asthma, and the need for access to national databases. As a result, the actual amount of the administered dose cannot be easily determined.10,12 On the other hand, self-reporting is a subjective way of measuring adherence. An example of this method is the use of validated adherence scales or questionnaires. These scales are frequently used; however, they may overestimate the actual adherence. In addition, these scales are prone to recall bias and may be affected by poor memory.12,14 Careful administration of self-report adherence questionnaires may contribute in identifying potential problems associated with suboptimal adherence and in determining the prevalence of adherence rates among patients with asthma.12
Studies from different geographical areas around the world have evaluated the prevalence of non-adherence to ICS among patients with asthma in both developed and developing countries. In the US, the reported adherence rates among patients with asthma ranged from 22% to 50%.15–19 Two studies evaluated the level of adherence to ICS in the United Kingdom.20,21 In those studies, suboptimal adherence was detected, with less than 80% of ICS taken in 65% of the cases and less than 50% of ICS medication prescriptions being filled among 35% of patients with asthma attending a specialized clinic. In Denmark, among 509 adults with asthma, two-thirds admitted to changing their ICS doses without consulting their clinicians.22 In the Middle Eastern region, adherence to ICS was reported in 25% of patients attending adult pulmonary clinics in Oman.23 Poor adherence was also reported in 55% of adult patients with asthma attending three private clinics in Iran.24 In Egypt, the ICS adherence rate was 21% among patients with asthma.25 However, this rate increased to 55% in those who received asthma-related education.
More studies about adherence to ICS in patients with asthma are still warranted, especially in developing countries, where education and training on the appropriate use of ICS in asthma is still lacking or suboptimal. In Kuwait, the level of ICS adherence among patients with asthma has not been previously reported. Such study is highly warranted given the high prevalence and financial burden of asthma in Kuwait. The aim of this study was to assess the level of adherence to ICS therapy using a validated tool among adult patients with asthma in Kuwait.
Patients and Methods
Study Design and Population
This is a quantitative, cross-sectional study, designed to assess the level of adherence to ICS therapy among adult patients with asthma in Kuwait. The study was conducted in four urban ambulatory asthma clinics. Kuwait is a Middle-Eastern country with an area of 17,820 km2 and an estimated population of 3,892,000 people (2015 estimate).26
Study Sample and Data Collection
Patients were eligible to participate in the study if they were adults (21 years or older), and diagnosed with asthma. Patients who were newly diagnosed with asthma were excluded from the study. The study used convenience sampling strategy. Patients were approached for consent by the study staff who either administered the Medication Adherence Report Scale-Asthma (MARS-A) survey in face-to-face basis or handed the survey to the patient for self-administration. Asthma diagnosis was confirmed with patients or the caring physician before recruitment and completion of the survey. Participation in the study was voluntary and patients were free to refuse to participate or to withdraw at any time without any consequences. Those who agreed to participate were given a written consent form and the survey. Data were collected anonymously to ensure confidentiality. Other collected data included socio-demographics, educational level, and smoking status.
Ethical Consideration
This study was approved by the Student Research Ethical Committee in the Health Science Centre (HSC) at Kuwait University (Ref. No. WMTJ/302). The study was performed in accordance with the tenets of the Declaration of Helsinki and guidelines of the Good Clinical Practice, with respect to the rights and dignity of the participants. All patients provided written consent form.
Measurement of Adherence to Inhaled Corticosteroids
The study used a previously validated 10-item questionnaire – the Medication Adherence Report Scale for Asthma (MARS-A) – that was proven to have good internal, construct, and criterion validity in English.27 Adherence to ICS was measured using the MARS-A, a self-reported adherence tool that demonstrated good test–retest reliability (r=0.65, p-value <0.001), internal consistency reliability of 0.85, sensitivity of 0.82, and specificity of 0.69. Moreover, this scale assesses both types of non-adherence: intentional (eg, “I try to avoid using it”), and unintentional (eg, “I forget to take it”). Furthermore, it contains a generic statement (“I use it regularly”), and an asthma-related statement (‘I only use it when I feel breathless’). Social desirability bias was minimized using negative statements that portray non-adherence as a normal behavior. The responses were measured using a 5-point Likert scale (1 = Never, 2 = Rarely, 3 = Sometimes, 4 = Often, 5 = Always). Self-reported adherence is reported as the average score of the 10 items (1–5), where higher scores indicate higher levels of reported adherence. High self-reported adherence was defined as a MARS-A score of 4.5 or higher.28
Translation of MARS-A into Arabic
The survey was translated into Arabic using accredited translators who were native speakers of English and fluent in Arabic. Translation was verified for compatibility with the original version in a process of forward and backward translation, performed by persons who were native speakers of Arabic and fluent in English. Face validity of the survey was assessed among five researchers in pharmacy practice for clarity of the questions. Then, the survey was pretested for content, design, readability, and comprehension on 26 people, and modifications were made as necessary so that the survey was simple to understand and answer, yet provided accurate data. Internal consistency reliability of the items in the Arabic version of the MARS-A was determined using Cronbach’s α to be 0.79. This indicates that the Arabic version of the MARS-A had satisfactory performance. Permission to use and translate the instrument was obtained from the developer (Prof. Rob Horne).
Data Analysis
Data were entered into the Statistical Package for Social Sciences (SPSS, version 23, IBM Corp, Armonk, NY, USA). Both descriptive and inferential analyses were conducted. Reported adherence to medication scores were entered as the dependent variable, with groups of independent variables entered in the following order: gender, age, educational level, and smoking status. The patients were categorized in terms of their adherence level as having high or low self-reported adherence. The results are presented as means (standard deviations; SD), median (interquartile range), and percentage.
Univariate logistic regression was performed to determine the relationship of each independent variable with the overall adherence to ICS therapy. All variables with p ≤0.25 in the univariate analysis were included in a multiple logistic regression to determine the factors that were independently associated with overall adherence to ICS therapy. The results of the multivariate logistic analysis are reported showing the odds ratio (OR) and 95% confidence interval (CI). Statistical significance was accepted at p < 0.05. For the regression model, response options for the dependent variable were categorized as either “low adherence” or “high adherence”. The predictor variables were categorized as follows: (1) age: 18–39 years or ≥40 years; (2) educational level: low [≤12 years of formal education] or high [>12 years of formal education]; (3) gender: male or female; (4) smoking status: smoker or non-smoker.
Results
Demographic Characteristics of Adult Patients with Asthma
Of 165 adults with asthma using ICS who were approached to participate in the study, 149 consented to participate (a response rate of 90.3%). The mean ± SD (median, IQR) age of the patients was 41.42 ± 12.75 (42, 18.75) years. Of the 149 participants, 112 (75.2%) were female, and 86 (57.7%) had high level (>12 years) of education. The majority of the participants were non-smokers (85%). Table 1 presents the demographic characteristics of the participants.
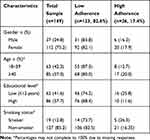 |
Table 1 Participants’ Demographic Characteristics According to Their Adherence Level |
Adherence to Inhaled Corticosteroids Among Adult Patients with Asthma
The majority of the participants (82.6%) reported low overall adherence. The overall adherence scores of the asthma patients ranged from 1 to 5, with a mean (SD) score of 3.4 (0.86) and median (IQR) of 4 (3). Approximately three-quarters of the adult patients with asthma reported that they either used the ICS when needed (72%) [median (IQR): 2 (4)], or used it when they felt breathless (72.5%) [median (IQR): 2 (3.5)]. About half of the participants admitted that they tried to avoid using the ICS (46.3%) [median (IQR): 4(2)], or forgot to take the ICS (51%) [median (IQR): 3 (2)], or stopped taking the ICS for a while (45%) [median (IQR): 4 (2)], or only used the ICS before performing an exercise that might make them breathless (54%) [median (IQR); 3 (4)]. Furthermore, about one-third of the participants reported that they altered the dose (39.6%) [median (IQR); 5 (4)], or used it as a reserve if other treatment did not work (39%) [median (IQR); 4 (2.5)], or took it less than instructed (33%) [median (IQR); 5 (2)]. The percentages of the participants who reported that they engaged in each trait of the non-adherent behavior represented by the 10 items of the scale are shown in Table 2.
 |
Table 2 The Percentage of Patients’ Engagements in Each Action of Non-Adherence and the Mean Score for Each Item* |
Predictors of Adherence to ICS Among Adult Patients with Asthma
Table 3 shows the results of the multivariate analysis for factors independently associated with overall adherence to ICS therapy. In the univariate analysis, factors with p-value ≤0.25 were age, education, and smoking status, which were included in the multivariate logistic regression model. As shown in Table 3, these factors did not significantly influence the adherence to ICS therapy (p>0.05).
 |
Table 3 Association Between Overall Adherence and Participants’ Characteristics |
Discussion
This study was performed using a previously validated instrument to determine adherence to ICS among adult patients with asthma in Kuwait. To our knowledge, there were no prior published studies evaluating adherence to ICS in Kuwait. The present findings identified the existing problems related to the use of ICS therapy in asthma. Unfortunately, only 17.4% of the participants reported high adherence to ICS. The current study revealed low adherence to ICS in Kuwait. In the Middle Eastern region, a Jordanian study reported that two-thirds of parents of children with asthma were adherent to ICS therapy.29 Moreover, a UAE study involving 200 patients with asthma reported that only 5% of the study participants were on ICS therapy and half of them were on short-acting beta-2 agonists.30 It was estimated that two-thirds of asthma patients in Saudi Arabia were poorly controlled.31 Such figures need to be investigated in relation to factors related to uncontrolled asthma, considering several aspects (eg, patients’ beliefs about asthma and medication adherence).
Although poor adherence to ICS is not uncommon in the literature, the rate of high adherence in the present study was lower than those reported in other studies.28,32-36 Moreover, the MARS-A mean score in our study (mean=3.4) was found to be lower than that reported by other studies.27,28 Consistent with previous similar research, no correlation was found between reported adherence and some of the demographic information.27,37 The findings of the present study merit attention, because low adherence to ICS was previously proven to have negative consequences on health outcomes. A UK study measured the effect of ICS adherence on asthma exacerbations (the need for oral corticosteroid use, asthma-related hospitalization or asthma-related emergency department visits).16 It was estimated that 24% of asthma exacerbations were related to ICS nonadherence.
The mean scores of individual items from the MARS-A scale indicated low adherence. In this study, about 39% of the participants admitted that they sometimes, often, or always use ICS therapy as a reserve if their other inhaler does not work. This finding was higher than that found by Horne and Weinman (2002), where only 15% of participants admitted to such practice.27 Moreover, one of the lowest mean scores was for the second item (“I only use it when I feel breathless”), in which about 73% of the participants reported that they sometimes, often, or always use their ICS only when they feel breathless. This figure was much higher than those reported in other studies in the literature.27 Moreover, Williams et al (2007) found that non-adherence to ICS was associated with lower use of rescue medications, indicating that these patients might have a lower perception of their need for ICS medication due to their milder form of the disease: this might reflect poor understanding regarding the nature of asthma.18 This is consistent with another study, in which the investigators reported that 53% of patients with asthma thought that their asthma was acute and episodic.38
Patients in the present study exhibited both intentional and unintentional non-adherence. About 35% of the participants decided to miss a dose of their ICS. A lower percentage was observed by a UK study, in which 23% of the participants admitted that they sometimes, often, or always missed a dose.27 In the same study, 22% of patients tried to avoid using their ICS.27 However, our data suggest a higher rate, with 46% of the patients trying to avoid using their ICS. In terms of dose alteration, our finding was lower than that found by Horne and Weinman (49%) but higher than that observed by Laforestt et al (18.3%).27,39 The prevalence of patients stopping their medication in the present study (45%) was higher than that observed by Horne and Weinman (28%) and Laforest et al (31.6%).27,39 It was suggested that nonadherence to ICS might be attributed to patients’ concerns related to ICS side effects.40
On the other hand, more than half of the participants were involved in unintentional non-adherence. This was seen with 51% of patients forgetting to take their ICS medication. This was higher than the findings of both Horne and Weinman (29%) and Laforest et al (25.4%). 27,39 In such situations, reminder techniques should be discussed with the patients.15,41 Previous research evaluated strategies used by older adults with asthma and found that those incorporating their use of ICS with their day-to-day activities were more likely to have good adherence than those with other strategies or no strategy at all.42
Although ICS is the mainstay therapy for patients with asthma, 39% of the participants in the present study used it as a reserve in case other treatments failed. A lower figure was observed in the literature, whereby 15% of the participants used their ICS as a reserve.27 Moreover, nearly half of the participants in the present study used ICS before involvement in activities that might make them breathless. This might be related to patients confusing the roles of controller medications (eg, ICS) and the role of reliever therapy (eg, short-acting beta-2 agonist).
In the present study, around one-third (34%) of the participants reported that they used their ICS less than instructed and about half of them (54%) used it as instructed. In line with this finding, a previous study had similar results, with one-quarter of the patients admitting that they used their ICS less than instructed.27 Nonetheless, 72% and 73% used their ICS when needed and when feeling breathless, respectively. These findings deserve attention, since more than half of the patients reported using ICS less than instructed; however, the majority of the participants did not use their ICS properly, as observed in the first and second items. This might reflect patients’ denial of being non-adherent. Gamble et al (2009) reported that about 88% of patients with difficult to treat asthma initially denied their non-adherence to their ICS.21
Implications for Future Research, Policy and Practice
Non-adherence is a direct reason for therapeutic failure in patients with chronic diseases. Effective communication with patients to uncover the potential individual barriers that prevent them from engaging in effective self-management is warranted. In addition, clinicians have an important role in enhancing medication adherence in which they can carefully consider patients’ understanding of the role of ICS therapy in asthma, and highlight the difference between preventer and reliever medications in asthma therapy. Future research should focus on investigating barriers to adherence to ICS in a larger sample of Kuwaiti asthma patients, and on testing different strategies or interventions for overcoming non-adherence through longitudinal research.
Limitations
The present study was limited by the small sample size and the use of convenience sampling method. These limitations might have introduced bias, limiting the generalizability of the findings to all adult patients with asthma in Kuwait. Moreover, the study included limited number of predictors, and other important predictors, such as patient/drug characteristics, were missed out. Additionally, social desirability bias might have contributed to high positive response rate since the survey was interviewer-administered for many participants. Finally, the sample was gender-biased as the majority were female patients.
Conclusion
The present study demonstrated high rates of non-adherence to ICS in patients with asthma. The MARS-A survey, which was proven to be a good and valid tool, was used to measure adherence to ICS therapy among asthma patients in Kuwait. The results of our study highlight the importance of asthma therapy in adults with asthma, and patients’ education regarding the role of asthma medications, most importantly ICS. Patient-clinicianconsultations can be improved by exploring and assessing triggers to non-adherence.
Acknowledgments
We would like to express our gratitude to Prof. Rob Horne for granting permission to use the MARS-A tool, and Dr. Eman Abahussain for her hard work in organizing the ethical approval of this study. We also wish to offer our thanks to Prof. Abdelmoneim Awad for his support with data analyses and interpretation. We would like to acknowledge the medical staff working in various asthma clinics for their great support.
Disclosure
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors and the authors report no conflicts of interest in this work.
References
1. Global Initiative for Asthma. Pocket Guide for Asthma Management and Prevention; 2019.
2. World Health Organization. Asthma fact sheets. Published 2017. Available from: https://www.who.int/news-room/fact-sheets/detail/asthma.
3. Braman S. The global burden of asthma. Chest. 2006;130:4S–12S. doi:10.1378/chest.130.1_suppl.4S
4. Khadadah M. The cost of asthma in Kuwait. Med Princ Pract. 2013;22(1):87–91. doi:10.1159/000341154
5. Abul A, Nair P, Behbehanei N, Sharma P. Hospital admissions and death rates from asthma in Kuwait during pre- and post-Gulf War periods. Ann Allergy, Asthma, Immunol. 2001;86(4):465–468. doi:10.1016/S1081-1206(10)62497-0
6. Center for Disease Control and Prevention. Most recent asthma state or territory data. Published 2020. Available from: https://www.cdc.gov/asthma/most_recent_data_states.htm.
7. Al-Qasem A, Smith F, Clifford S. Adherence to medication among chronic patients in Middle Eastern countries. East Mediterranean Heal J. 2011;17(4):356–363. doi:10.26719/2011.17.4.356
8. World Health Organization. Adherence to Long-Term Therapies: evidence for Action. 2003. Available from: https://www.who.int/chp/knowledge/publications/adherence_full_report.pdf.
9. Hassall D, Brealey N, Wright W, et al. Hair analysis to monitor adherence to prescribed chronic inhaler drug therapy in patients with asthma or COPD. Pulm Pharmacol Ther. 2018;51:59–64. doi:10.1016/j.pupt.2018.07.001
10. Foster J, Smith L, Bosnic-Anticevich S, et al. Identifying patient-specific beliefs and behaviours for conversations about adherence in asthma. Intern Med J. 2012;42(6):136–144. doi:10.1111/j.1445-5994.2011.02541.x
11. O’Dwyer S, Greene G, MacHale E, et al. Personalized biofeedback on inhaler adherence and technique by community pharmacists: a cluster randomized clinical trial. J Allergy Clin Immunol Pract. 2020;8(2):635–644. doi:10.1016/j.jaip.2019.09.008
12. Jentzsch N, Camargos P. Methods of assessing adherence to inhaled corticosteroid therapy in children and adolescents: adherence rates and their implications for clinical practice. J Bras Pneumol. 2008;34(8):614–621. doi:10.1590/S1806-37132008000800012
13. Van Boven J, Koponen M, Lalic S, et al. Trajectory analyses of adherence patterns in a real-life moderate to severe asthma population. J Allergy Clin Immunol Pract. 2019. doi:10.1016/j.jaip.2019.12.002
14. Rand C, Wise R. Measuring adherence to asthma medication regimens. Am J Respir Crit Care Med. 1994;149(2):69–78. doi:10.1164/ajrccm/149.2_Pt_2.S69
15. Apter A, Reisine S, Affleck G, Barrows E, ZuWallack R. Adherence with twice-daily dosing of inhaled steroids: socioeconomic and health-belief differences. Am J Respir Crit Care Med. 1998;157:1810–1817. doi:10.1164/ajrccm.157.6.9712007
16. Williams L, Peterson E, Wells K, et al. Quantifying the proportion of severe asthma exacerbations attributable to inhaled corticosteroid nonadherence. J Allergy Clin Immunol. 2011;128(6):1185–1191. doi:10.1016/j.jaci.2011.09.011
17. Williams L, Pladevall M, Xi H, et al. Relationship between adherence to inhaled corticosteroids and poor outcomes among adults with asthma. J Allergy Clin Immunol. 2004;114(6):1288–1293. doi:10.1016/j.jaci.2004.09.028
18. Williams L, Joseph C, Peterson E, Wells K, Wang M, Chowdhry V. Patients with asthma who do not fill their inhaled corticosteroids: a study of primary nonadherence. J Allergy Clin Immunol. 2007;120(5):1153–1159. doi:10.1016/j.jaci.2007.08.020
19. Bender B, Pedan A, Varasteh L. Adherence and persistence with fluticasone propionate/salmeterol combination therapy. J Allergy Clin Immunol. 2006;118(4):899–904. doi:10.1016/j.jaci.2006.07.002
20. Murphy A, Proeschal A, Brightling C, et al. The relationship between outcomes and medication adherence in difficult-to-control asthma. Thorax. 2012;67(8):751–753. doi:10.1136/thoraxjnl-2011-201096
21. Gamble J, Stevenson M, McClean E, Heaney L. The prevalence of nonadherence in difficult asthma. Am J Respir Crit Care Med. 2009;180(9):817–822. doi:10.1164/rccm.200902-0166OC
22. Ulrik C, Søes-Petersen U, Backer V, Lange P. Disease variability in asthma: how do the patients respond?–and why? J Asthma. 2008;45(6):507–511. doi:10.1080/02770900802017736
23. Al-Rawas O, Jayakrishnan B, Ben Abid F, George J, Baddar S, Al-Riyami B. Management and control of asthma in patients attending a specialist centre in Oman. Sultan Qaboos Univ Med J. 2009;9(2):132–139.
24. Rahimi A, Sulong S, Maimaiti N, Ghazi H. Asthma treatment adherence among asthmatic patients in Yazd. BMC Public Health. 2012;12:A39. doi:10.1186/1471-2458-12-S2-A39
25. Rifaat N, Abdel-Hady E, Hasan A. The golden factor in adherence to inhaled corticosteroid in asthma patients. Egypt J Chest Dis Tuberc. 2013;62(3):371–376. doi:10.1016/j.ejcdt.2013.07.010
26. World Health Organization. Statistics. Published 2018. Available from: http://www.who.int/countries/kwt/en/.
27. Horne R, Weinman J. Self regulation and self management in asthma: exploring the role of illness perceptions and treatment beliefs in explaining non-adherence to preventer medication. Psychol Heal. 2002;17(1):17–32. doi:10.1080/08870440290001502
28. Cohen J, Mann D, Wisnivesky J, et al. Assessing the validity of self-reported medication adherence among inner-city asthmatic adults: the medication adherence report scale for asthma. Ann Allergy, Asthma, Immunol. 2009;103(4):325–331. doi:10.1016/S1081-1206(10)60532-7
29. Alsous M, Alhalaiqa F, Abu Farha R, Abdel Jalil M. Reliability and validity of Arabic translation of medication adherence report scale (MARS) and beliefs about medication questionnaire (BMQ). PLoS One. 2017;12(2):e0171863. doi:10.1371/journal.pone.0171863
30. Hassan Mahboub B, Santhakumar S, Soriano J, Pawankar R. Asthma insights and reality in the United Arab Emirates. Ann Thorac Med. 2010;5(4):217–221. doi:10.4103/1817-1737.69109
31. BinSaeed A. Asthma control among adults in Saudi Arabia. Study of determinants. Saudi Med J. 2015;36(5):599–604. doi:10.15537/smj.2015.5.10929
32. Al-Jahdali HH, Al-Zahrani AI, Al-Otaibi ST, et al. Perception of the role of inhaled corticosteroids and factors affecting compliance among asthmatic adult patients. Saudi Med J. 2007;28(4):569–573.
33. Ponieman D, Wisnivesky J, Leventhal H, Musumeci-Szabó T, Halm E. Impact of positive and negative beliefs about inhaled corticosteroids on adherence in inner-city asthmatic patients. Ann Allergy, Asthma, Immunol. 2009;103(1):38–42. doi:10.1016/S1081-1206(10)60141-X
34. Roy A, Lurslurchachai L, Halm E, Li X, Leventhal H, Wisnivesky J. Use of herbal remedies and adherence to inhaled corticosteroids among inner-city asthmatic patients. Ann Allergy, Asthma, Immunol. 2010;104(2):132–138. doi:10.1016/j.anai.2009.11.024
35. Brandstetter S, Finger T, Fischer W, Brandl M. Differences in medication adherence are associated with beliefs about medicines in asthma and COPD. Clin Transl Allergy. 2017;7:39. doi:10.1186/s13601-017-0175-6
36. Sofianou A, Martynenko M, Wolf M, Wisnivesky J, Krauskopf K. Asthma beliefs are associated with medication adherence in older asthmatics. J Gen Intern Med. 2013;28(1):67–73. doi:10.1007/s11606-012-2160-z
37. Horne R, Weinman J. Patients’ beliefs about prescribed medicines and their role in adherence to treatment in chronic physical illness. J Psychosom Res. 1999;47(6):555–567. doi:10.1016/S0022-3999(99)00057-4
38. Halm E, Mora P, Leventhal H. No symptoms, no asthma: the acute episodic disease belief is associated with poor self-management among inner-city adults with persistent asthma. Chest. 2006;129(3):573–580. doi:10.1378/chest.129.3.573
39. Laforest L, El Hasnaoui A, Pribil C, et al. Asthma patients' self-reported behaviours toward inhaled corticosteroids. Respir Med. 2009;103(9):1366–1375.
40. Cooper V, Metcalf L, Versnel J, Upton J, Walker S, Horne R. Patient-reported side effects, concerns and adherence to corticosteroid treatment for asthma, and comparison with physician estimates of side-effect prevalence: a UK-wide, cross-sectional study. NPJ Prim Care Respir Med. 2015;25:15026. doi:10.1038/npjpcrm.2015.26
41. Menckeberg T, Bouvy M, Bracke M, et al. Beliefs about medicines predict refill adherence to inhaled corticosteroids. J Psychosom Res. 2008;64(1):47–54. doi:10.1016/j.jpsychores.2007.07.016
42. Brooks T, Leventhal H, Wolf M, O’Conor R, Morillo J. Strategies used by older adults with asthma for adherence to inhaled corticosteroids. J Gen Intern Med. 2014;29(11):1506–1512. doi:10.1007/s11606-014-2940-8
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
