Back to Journals » Clinical Ophthalmology » Volume 16
Cost Burden of Endothelial Keratoplasty in Fuchs Endothelial Dystrophy: Real-World Analysis of a Commercially Insured US Population (2014–2019)
Authors Dhaliwal DK, Chirikov V, Schmier J, Rege S, Newton S
Received 18 January 2022
Accepted for publication 21 March 2022
Published 6 April 2022 Volume 2022:16 Pages 1055—1067
DOI https://doi.org/10.2147/OPTH.S358847
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Deepinder K Dhaliwal,1 Viktor Chirikov,2 Jordana Schmier,2 Sanika Rege,2 Schalon Newton3
1Department of Ophthalmology, University of Pittsburgh School of Medicine, Pittsburgh, PA, USA; 2OPEN Health Evidence & Access, Bethesda, MD, USA; 3Trefoil Therapeutics, Inc., San Diego, CA, USA
Correspondence: Schalon Newton, Trefoil Therapeutics, Inc., 6330 Nancy Ridge Dr Suite 103, San Diego, CA, 92121, USA, Tel +1 949 678 9572, Email [email protected]
Purpose: To assess the incremental burden of corneal transplant surgery for US commercially insured patients with Fuchs endothelial corneal dystrophy (FECD) treated with endothelial keratoplasty (EK) compared to controls.
Methods: The study design was retrospective cohort using IBM® MarketScan® claims (January 2014–September 2019) and included EK-treated (N=1562) and control patients (N=23,485) having ≥ 12 months’ enrollment before and after diagnosis, who were subsequently matched on select characteristics. The index date was the beginning of the pre-operative period (3 months before EK); synthetic EK index was assigned for controls. All-cause, eye-disease, and complication-related healthcare resource utilization (HCRU) and costs were compared up to 36 months post index. For a small subset of patients, patient data were linked to the Health and Productivity Management supplemental database, which integrates data on productivity loss and disability payments.
Results: Matched cohorts included 804 EK-treated and 1453 controls with average age 65.7 years, 1383 (61%) female. Over 12 months of follow-up, all-cause ($41,199 vs $20,222, p< 0.001) and eye-disease related costs ($22,951 vs $1389, p< 0.001) were higher among EK-treated patients than controls. The cost differential increased additionally by $1000–$2000 per annum by 36 months of follow-up. While balanced at baseline, over follow-up EK-treated patients had higher prevalence of glaucoma, elevated intraocular pressure, cataract, cataract surgery, diagnosis of cornea transplant rejection, retinal edema. By 36 month of follow-up, EK-treated patients had 9 more short-term disability days, resulting in $2992 additional burden of disability payments.
Conclusion: This study found a higher cost burden among FECD patients receiving EK treatment versus those who did not. With a shift in management of FECD, cost burden estimates generated in this study could serve as an important benchmark for future studies.
Keywords: cornea transplantation, cost analysis, glaucoma, retrospective study
Introduction
Fuchs endothelial corneal dystrophy (FECD) is a progressive degenerative disease characterized by the formation of guttae on the inner layer of the cornea, Descemet’s membrane thickening, and loss of endothelial cells.1–3 It is the most common corneal endothelial disorder, affecting approximately 4% of the US population over the age of 40 and results in diminished vision and, in severe cases, blindness.3,4 FECD is the leading cause of corneal transplantation in the US and worldwide as well as a contributor to poor outcomes in patients who subsequently undergo ocular surgery, including cataract surgery.4–6
Over the years, the surgical management of worsening FECD with corneal transplantation has evolved, with a shift from penetrating keratoplasties to lamellar procedures, primarily endothelial keratoplasties (EK), due to significant intraoperative and postoperative risks associated with penetrating keratoplasty.7,8 In 2018, EK procedures accounted for almost 62% of all corneal transplant surgeries performed in the US compared to 45% in 2010.9
As the elderly become a larger share of the US population, the demand for corneal tissue and corneal transplants is increasing.10 Previous reports suggest that the costs of transplants, pre- and post-operative care, and physician and facility services impose a significant financial burden to payers and patients’ caregivers8,11,12 but few studies estimate costs associated with EK.5,8 However, the costs of corneal transplant in prior literature8 may be underestimated as post-surgical care and ongoing steroid therapy typically continue for more than one year. Rejection risk varies by type of transplant with a mean time (± standard deviation) from keratoplasty to rejection of 19.8 ± 20.4 months, but rejection can occur up to 20 years post-transplant.13
Novel, alternative treatments based on regenerative medicine and cell-based therapies are being developed for FECD.14 For example, tissue-sparing surgical approaches, such as Descemet-stripping only (DSO), eliminate the need for transplanted endothelial replacement and provide an alternative strategy without the use of donor tissue.15 Given the gap since previous assessments and the availability of new treatment options, an up-to-date robust assessment of the cost of transplant surgery can provide benchmark information to evaluate the cost-effectiveness of new-generation FECD treatments. The objective of this study was to assess the incremental attributable burden of corneal transplant surgery for patients with FECD treated with EK, compared to matched controls who did not undergo EK, using real-world claims data of a commercially insured US population.
Methods
Data Source
This retrospective observational cohort study used IBM® MarketScan® Commercial Claims and Encounters and Medicare Supplemental and Coordination of Benefits database claims data from January 2014 to September 2019.16 The Health and Productivity Management supplemental database, which integrates data on productivity loss and disability payments, was also available and was linkable to <10% of patients identifiable in claims. Institutional review board approval was not sought for this research study, as it relied exclusively on secondary use of deidentified information. Due to the retrospective, de-identified nature of the MarketScan data, the study was considered exempt from IRB oversight.
Patient Population and Study Design
The study included patients with FECD diagnosis identified using International Classification of Diseases ICD-9-CM/ICD-10-CM codes of 371.57 and H18.51, respectively, and who were continuously enrolled for ≥12 months before and ≥24 months after diagnosis. Patients were excluded if <18 years, were treated with any keratoplasty 12 months before diagnosis, or were treated with penetrating keratoplasty following diagnosis.
The exposure of interest was the receipt of EK (identified by Current Procedural Terminology code 65756) after FECD diagnosis. Patients who were not treated with EK were considered untreated controls. The study index date was assigned as 3 months prior to the actual EK date (ie, start of pre-operative period). A synthetic EK date of treatment was assigned to controls (described in the section on matching below). The 12-month look-back period before the index was the baseline period (Figure 1) and patients were followed for ≥12 months after EK treatment.
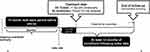 |
Figure 1 Retrospective study design. |
Outcomes
The following outcomes were compared between the two groups: i) all-cause, eye-related, and complications-related healthcare utilization resource utilization (HCRU) and cost burden; ii) prevalence of complications potentially related to EK treatment; iii) productivity loss. Costs were inflated to 2020 and categorized into medical (inpatient and outpatient) and prescription drug cost components. The medical component of the eye disease-related burden was flagged based on the presence of diagnosis codes covered under the ICD “Diseases of the eye and the adnexa” category. Complications prevalence and related costs were identified based on the presence of diagnosis codes covering glaucoma, cataract, cystoid macular edema, dry eye/ocular surface disease, endophthalmitis, globe rupture, infectious keratitis, infectious uveitis, persistent mydriasis, rejection of cornea transplant, retinal edema, suprachoroidal hemorrhage. The prescription drug cost definition was similar for the eye- and complications-related burden domains and included the cost of intraocular pressure (IOP) lowering medications, ophthalmic steroids, topical NSAIDs, steroids, antibiotics, and lubricants.
Matching of Demographics and Clinical Characteristics
Patient cohorts were characterized at baseline in terms of demographic and clinical characteristics, as well as HCRU and costs. Two rounds of matching were used to address imbalance in patient characteristics. In the first round, EK-treated and control patients were matched 1:10 based on characteristics identified in the 12 months prior to diagnosis (ie, glaucoma, cataract, age, diagnosis year), and the interval between diagnosis and treatment among the treated was then assigned to the matched controls to create a synthetic EK date. Subsequently, EK-treated and control patients were matched 1:2 based on characteristics identified in the 12-month pre-index period (ie, glaucoma, cataract, retinopathy, age, region, sex, use of IOP lowering meds, topical NSAIDS, ophthalmic steroids, cystoid macular edema, increased IOP diagnosis, prior cornea rejection diagnosis, number of visits to ophthalmologist, bullous keratopathy/corneal edema diagnosis, other dystrophies diagnosis). The choice of matching ratios was chosen to optimize retention of sample size with balancing baseline characteristics.
Statistical Analysis
Descriptive analyses assessed the baseline patient characteristics for the included patients and demographics tables were populated to compare baseline characteristics across the cohorts. Healthcare utilization, costs, prevalence of potential EK complications and productivity loss were compared between the matched cohorts at the following intervals: 3 months before EK treatment (ie, pre-op) as well as 12-, 24-, and 36-months post EK. In exploratory analysis, EK-treated patients were further stratified by whether they had only one eye treated with or without subsequent re-treatment (ie, use of EK on a different day but same eye) or both eyes were treated.
Results
We identified 94,259 patients with FECD in the period 1/1/2014–6/30/2019, of whom 1562 EK-treated and 23,485 without EK met continuous enrollment criteria. Time to EK treatment since diagnosis was on average 14 months. In the pre-diagnosis period, EK-treated patients had higher use of eye-related prescription drugs, which correlated with higher rates of corneal opacity, other disorders of the cornea, bullous keratopathy/corneal edema, as well as other dystrophies (available in the Supplemental Table 1).
After the first round of matching, patients who received EK were older (median of 65 vs 64 years of age), more of them were male, had a diagnosis of bullous keratopathy/corneal edema, cornea degeneration, other types of dystrophies, glaucoma, cataract, cataract surgery, used ophthalmic NSAIDS or steroids, and had higher average all-cause cost burden ($17,611 vs $16,091) than the untreated (Table 1). Differences in baseline cost burden were driven by higher eye disease-related medical ($2061 vs $1163) and prescription drug cost ($305 vs $142). After the second round of matching, both EK-treated and untreated cohorts were balanced with respect to the abovementioned characteristics. For any covariates where differences remained statistically significant, imbalances were small. Results from that analysis can be interpreted as adjusted compared to those from the first round of matching (ie, unadjusted), which was largely only used to assign a synthetic index date among controls.
 |
Table 1 Baseline Characteristics Captured 12 Months Before Index |
Cost Burden and Healthcare Resource Utilization
Healthcare resource utilization was higher for the EK-treated in every time period (Table 2). Over the first 12 months of follow-up, all-cause ($41,199 vs $20,222) and eye disease-related costs ($22,951 vs $1389) were higher among EK-treated patients than untreated. The difference in all-cause and eye disease-related costs was similar at 12 months since EK with $20,977 and $21,562, respectively. The difference in eye disease-related cost including the pre-op period and the day of EK treatment ($14,170) accounted for a large portion of the total cost difference. Additionally, most of the attributable cost difference was generated within 6 months since EK treatment and the cost burden differential increased by about $1000-$2000 per annum by 36 months, the practical end of follow-up for majority of patients. The majority of the attributable cost burden between EK-treated and untreated patients was due to differences in outpatient cost. The difference in prescription drug costs was <$1000 at 12 months post EK follow-up. The difference in complications-related costs was $10,214 at 12 months and remained stable by 36 months. The direct costs using claims with diagnosis code for FECD were $17,645 vs $222 at 12 months for treated and untreated patients, respectively. The cost differential burden correlated with a higher count of ophthalmologist visits, office visits for disease of the eye and the adnexa, as well as higher prescription drug use.
 |
Table 2 Cost and Health Care Utilization During Follow-Up Among Matched Cohorts |
When stratified by baseline diagnosis of cataract or glaucoma, the eye-disease related cost differential between EK treated and untreated at 12 months was $24,209 among those with cataract vs $20,639 without cataract and $22,897 among those with glaucoma vs $21,973 without glaucoma. Compared to control patients, EK-treated patients who underwent a triple procedure (n=411, 51%) on the day of EK procedure (ie, cataract surgery, intraocular lens implantation, and EK) had slightly higher eye disease-related cost differential at 12 months ($20,639 vs $18,414).
Prevalence of Potential EK Treatment Complications
While balanced at baseline, over the follow-up period EK-treated patients had higher prevalence of glaucoma, elevated IOP diagnosis, cataract, and cataract surgery, endophthalmitis, infectious keratitis, persistent mydriasis, diagnosis of cornea transplant rejection, retinal edema (Table 3).
 |
Table 3 Prevalence of Potential Complications at Baseline and Over the Follow-Up Among Matched Cohorts |
Productivity Loss
Among a subset of the matched sample that had information on disability and productivity, the EK-treated patients had approximately 7–9 more short-term disability days (resulting in $1500–2000 higher burden of short-term disability payments), whether based on the all-cause or eye-disease related definitions (Table 4).
 |
Table 4 Short Term Disability Payments and Days Missing from Work |
Outcomes by Number of Eyes Treated
When stratified by which eye may have received EK treatment in the study dataset, among n=584 EK-treated patients with available eye laterality information, about 15% had post-op retreatment with EK (n=86) and 38% (n=222) had both of their eyes treated with EK (Table 5). Due to difficulty of results interpretation and limited sample size, those with both eyes treated were not additionally broken down into whether they got further retreatment and for which eye. Over 36 months of follow-up since the first EK, rates of healthcare resource utilization, costs, as well as short term disability days and payment increased with the extent of EK treatment received.
 |
Table 5 Study Outcomes by 36 Months Since First EK, by Eye Treated |
Discussion
This retrospective cohort study assessed and compared the cost burden and healthcare resource utilization among FECD patients who were treated with EK versus those who were not. In addition, the study also assessed the prevalence of potential complications and indirect burden related to EK treatment among the two comparison groups. To our knowledge, this is the largest study of its type using recent billing data of commercially insured patients with FECD in the US. In our analysis, the cost burden of EK treatment was mostly driven by the cost of the surgical procedure and donor tissue on the day of treatment and post-operative care in the immediate few months after EK treatment. Our study found that glaucoma, use of IOP-lowering medications, cataract and cataract surgery, endophthalmitis, infectious keratitis, persistent mydriasis, diagnosis of cornea transplant rejection, retinal edema had a higher prevalence among EK-treated patients compared to untreated patients. One of the main strengths of this study is its large sample size for a relatively uncommon condition with a prevalence rate of 0.009.17 These results are consistent with previous literature that report complications with EK treatment.5,18–21 Generally, clinical characteristics at baseline such as glaucoma diagnosis, cataract diagnosis, and age had only moderate impact on the cost burden differential, that is, even in subgroups with glaucoma or cataract, the difference in costs between EK-treated and untreated patients was similar to the difference in the full study population.
As our study used a two-stage matching approach with a control group of untreated FECD patients to estimate the attributable burden of EK, it better addresses any measurement and confounding bias, compared to previous studies, which used a single cohort design. The Lewin Group estimated a per-patient average cost of corneal transplant of approximately $16,500 in 2013 USD ($20,138 if inflated to 2020 USD) for a year after the transplant, reflecting all related expenses, including corneal tissue, surgeon and anesthesia services, facility costs, pre-operative care, and related services.8 In our study, the comparable estimate to that of the Lewin Group is $22,951, representing the eye-related total cost at 12 months post-EK. A recent study by Bohm et al estimated direct cost during the episode of EK of around $14,000, including the cost of the cornea tissue and treatment costs, as well as 22 total office visits and long-term steroid use over a post-operative 15-year period.5 The itemized estimate reported by Bohm et al was based on a retrospective single-center study of 58 patients and covered treatment for only one eye with expected limited need for secondary EK (<3%), and the cost of post-operative complications was estimated to be low and approximately $500. The direct cost of EK treatment for one eye was therefore estimated by Bohm et al to be in the range of $13,886–$15,329.
Additionally, in terms of indirect burden, our study was only able to capture productivity loss over 3 years of follow-up. Therefore, future studies should fully capture the healthcare and societal perspectives over a longer period of time. The modelling study by Bohm et al covered EK societal burden in detail including informal healthcare costs (refraction and glasses, patient time-costs, unpaid caregiver time-costs, transportation costs) in addition to non-healthcare costs (productivity), although it did not include a control cohort without EK.5
Last but not least, benefit to society due to EK includes preventing further vision loss and possibly blindness, which develops over a long period of time in Fuchs patients. An estimate of the benefit of preventing vision loss would require longer follow-up than what was available in the study data source. A previous modelling study by the Lewin Group8 estimated an average of $77,000 direct medical benefit and $214,000 indirect benefit of avoiding blindness over the course of a person’s life, for a total of $118,000 in net lifetime benefit of corneal transplantation. A recent study by Rein et al estimated the annual economic burden of vision loss in the US to be $134.2 billion, which included $98.7 billion in direct costs (medical, nursing home, and supportive services) and $35.5 billion in indirect costs (absenteeism, lost household production, reduced labor force participation, and informal care).22 Patients with vision loss incurred a total of $16,838 per year in incremental burden with nursing home, other medical care services, and reduced labor force participation accounting for 66% of the total costs.22
In exploratory analysis we attempted to identify if the EK procedure could be assigned to a particular eye, based on an available CPT modifier code. The reason for this analysis to be considered exploratory is that claims data, used for billing purposes, may not be the best source of data to differentiate between which and whether both eyes are re/treated and as such results should be interpreted with caution. As evidenced by the findings from the exploratory analysis, more than half of FECD patients had either their eye re-treated with EK or had both of their eyes EK-treated and their cost burden close to doubled, compared to those that only had one eye treated with no post-operative re-treatment. Therefore, the average attributable eye disease-related cost burden of $21,562 at 12 months of follow-up and $23,698 at 36 months is more reflective of the real-world EK treatment, where a majority of patients receive re-treatment or have their second eye treated. In one study of FECD patients 99.1% had a bilateral diagnosis.2 Since FECD is a bilateral condition, most patients choose to have both eyes ultimately treated.4,23 FECD can be highly asymmetric, so the timing of second eye surgery varies; patients with complications post-treatment of their first eye may delay seeking treatment for the second eye.
There are limitations to our study, many of which are inherent to claims database analyses and the administrative nature of the data. Whenever possible, we tried to minimize the effect of these constraints. The retrospective, non-randomized claims data were analyzed using two-stage matching that addressed measurement bias resulting from missing treatment date among non-EK users, and confounding resulting from imbalance between patient characteristics across the EK and non-EK user groups. As progression of FECD results in increased swelling of the cornea leading to increasing burden over a longer period of time, our study was limited in evaluating long-term outcomes beyond 36 months. The claims data create a challenge to differentiate between number of eyes treated, specifically with the ICD-9 codes. Also, there may be nuances in coding that may limit perfect concordance. The cohort of patients who did not receive EK during the study period are likely to be treated in the future and patients delaying treatment are likely to have higher long-term costs and rates of complications. Additionally, claims data does not include information on preservation time and condition of corneal donor tissue, which would be a relevant variable to assess with corneal transplantation. Claims data is limited in terms of clinical information, including whether the presumed complications are related to EK, and whether steroids or IOP-lowering medications were prescribed for another problem or proactively. Also, there is a lack of clinical information about visual acuity, which may be related to other types of burdens and costs. Last but not least, data do not distinguish among the type of endothelial keratoplasty performed (DMEK or DSAEK) or capture the Descemet’s Stripping Only (DSO) procedure, the use of which was emerging towards the end of the study period. DSO could not be identified in the claims data. Outcomes of DMEK, DSAEK, or DSO administration are largely dependent on the skill of the surgeon as well as patient compliance during the recovery time, which was also not accounted for in our study.
Given that evidence from randomized clinical trials is missing, the clinical implication of our study is to provide the best possible estimate of the burden of EK, when the treatment landscape is expected to change in the coming years. While it is of high interest to compare the cost burden of DSO to that of EK, current data is not able to reliably identify DSO. Therefore, such analysis will need to be done by future studies when DSO has become an integral part of the treatment landscape and is reliably identifiable in billing claims data.
Conclusions
This study found a higher cost burden among FECD patients receiving EK treatment compared with those who did not. However, patients diagnosed with FECD will almost certainly be treated at some point in the future.4,23 A continued shift to DMEK may lead to lower complication costs, but the basic cost of the procedure will remain similar to the results of this study. New options in FECD management such as DSO, bioengineered corneal grafts, gene therapy, and cell injection are in development to assess their therapeutic potential.24 These new therapeutic options would all reduce the cost burden of the transplant tissue, which is a large cost component. Until new regimens become viable treatment options, EK may remain the standard of care for the management of FECD. The real-world cost burden estimates generated in this study, albeit reflective of the EK treatment paradigm, could serve as an important benchmark for future cost-effectiveness assessments of new approved health technologies for FECD management.
Acknowledgment(s)
Authors are grateful to David Eveleth for reviewer comments during manuscript preparation, Robert Bellizzi for patient burden contextualization, Jodi Luchs and Marian Macsai for assistance with study protocol development.
Disclosure
DKD is a consultant and clinical advisory board member for Trefoil Therapeutics and reports grants/personal fees from Glaukos, Kowa, Staar Surgical, advisory board for Tarsus, Horizon, Allergan, and Novartis; trainer for Johnson and Johnson, consultant for Haag Streit, and Ocular Therapeutix, outside the submitted work. VC, JS, SR are employees of OPEN Health, which received funding from Trefoil to conduct this study. SN is an employee of Trefoil Therapeutics, which funded the conduct of this study, and may own stock options. The authors report no other conflicts of interest in this work.
References
1. Soh YQ, Kocaba V, Pinto M, Mehta JS. Fuchs endothelial corneal dystrophy and corneal endothelial diseases: east meets west. Eye. 2020;34(3):427–441. doi:10.1038/s41433-019-0497-9
2. Goldberg RA, Raza S, Walford E, Feuer WJ, Goldberg JL. Fuchs endothelial corneal dystrophy: clinical characteristics of surgical and nonsurgical patients. Clin Ophthalmol. 2014;8:1761–1766. doi:10.2147/OPTH.S68217
3. Elhalis H, Azizi B, Jurkunas UV. Fuchs endothelial corneal dystrophy. Ocul Surf. 2010;8(4):173–184. doi:10.1016/S1542-0124(12)70232-X
4. Eghrari AO, Gottsch JD. Fuchs’ corneal dystrophy. Expert Rev Ophthalmol. 2010;5(2):147–159. doi:10.1586/eop.10.8
5. Böhm M, Leon P, Wylęgała A, Ong Tone S, Condron T, Jurkunas U. Cost-effectiveness analysis of preloaded versus non-preloaded Descemet membrane endothelial keratoplasty for the treatment of Fuchs endothelial corneal dystrophy in an academic centre. Br J Ophthalmol. 2021;
6. Bentley TS. U.S. Organ and Tissue Transplant Cost Estimates and Discussion. Milliman Research Report; Brookfield, WI: Milliman; 2014.
7. Blitzer AL, Colby KA. Update on the surgical management of fuchs endothelial corneal dystrophy. Ophthalmol Ther. 2020;9(4):757–765. doi:10.1007/s40123-020-00293-3
8. The Lewin Group, Inc. Cost-Benefit Analysis of Corneal Transplant. Eye Bank Association of America; 2013.
9. Zafar S, Wang P, Woreta FA, Aziz K, Makary M, Srikumaran D. Risk factors for repeat keratoplasty after endothelial keratoplasty in the medicare population. Am J Ophthalmol. 2021;221:287–298. doi:10.1016/j.ajo.2020.08.006
10. Gain P, Jullienne R, He Z, et al. Global survey of corneal transplantation and eye banking. JAMA Ophthalmol. 2016;134(2):167–173. doi:10.1001/jamaophthalmol.2015.4776
11. Bentley TS, Hanson SG. US organ and tissue transplant cost estimates and discussion. Milliman. Ref Type: Report. 2011.
12. Ortner NJ. US organ and tissue transplant cost estimates and discussion. Milliman. 7-28-2013. Ref Type: Report. 2005.
13. Guilbert E, Bullet J, Sandali O, Basli E, Laroche L, Borderie VM. Long-term rejection incidence and reversibility after penetrating and lamellar keratoplasty. Am J Ophthalmol. 2013;155(3):560–569.e2. doi:10.1016/j.ajo.2012.09.027
14. Ong HS, Ang M, Mehta J. Evolution of therapies for the corneal endothelium: past, present and future approaches. Br J Ophthalmol. 2021;105(4):454–467. doi:10.1136/bjophthalmol-2020-316149
15. Melles GRJ. Landmark study on Descemet stripping with endothelial keratoplasty: where has it led us? J Cataract Refract Surg. 2021;47(5):561–562. doi:10.1097/j.jcrs.0000000000000641
16. Kulaylat AS, Schaefer EW, Messaris E, Hollenbeak CS. Truven health analytics marketscan databases for clinical research in colon and rectal surgery. Clin Colon Rectal Surg. 2019;32(1):54–60. doi:10.1055/s-0038-1673354
17. Musch DC, Niziol LM, Stein JD, Kamyar RM, Sugar A. Prevalence of corneal dystrophies in the United States: estimates from claims data. Invest Ophthalmol Vis Sci. 2011;52(9):6959–6963. doi:10.1167/iovs.11-7771
18. Price MO, Feng MT, Price FW. Endothelial Keratoplasty update 2020. Cornea. 2021;40(5):541–547. doi:10.1097/ICO.0000000000002565
19. Gibbons A, Leung EH, Yoo SH. Cost-effectiveness analysis of Descemet’s membrane endothelial keratoplasty versus Descemet’s stripping endothelial keratoplasty in the United States. Ophthalmology. 2019;126(2):207–213. doi:10.1016/j.ophtha.2018.09.033
20. Thiel MA, Bochmann F, Schmittinger-Zirm A, Bänninger PB, Schmid MK, Kaufmann C. Komplikationen der Descemet Stripping Automatisierten Endothelkeratoplastik (DSAEK) [Complications of Descemet Stripping Automated Endothelial Keratoplasty (DSAEK)]. Ophthalmologe. 2015;112(12):969–973. doi:10.1007/s00347-015-0166-8
21. Sanchez PJ, Straiko MD, Terry MA. Complications of Descemet Stripping Automated Endothelial Keratoplasty (DSAEK) surgery. In: Cursiefen C, Jun A, editors. Current Treatment Options for Fuchs Endothelial Dystrophy. Springer, Cham;2017:189–204. doi:10.1007/978-3-319-43021-8_12
22. Rein DB, Wittenborn JS, Zhang P, et al. The economic burden of vision loss and blindness in the United States. Ophthalmology. 2021;120(9):1728–1735.
23. Matthaei M, Hribek A, Clahsen T, Bachmann B, Cursiefen C, Jun AS. Fuchs endothelial corneal dystrophy: clinical, genetic, pathophysiologic, and therapeutic aspects. Annu Rev Vis Sci. 2019;5:151–175. doi:10.1146/annurev-vision-091718-014852
24. Rocha-de-Lossada C, Rachwani-Anil R, Borroni D, et al. New horizons in the treatment of corneal endothelial dysfunction. J Ophthalmol. 2021;2021:6644114. doi:10.1155/2021/6644114
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
