Back to Journals » International Journal of Nanomedicine » Volume 15
Aptamers as Versatile Ligands for Biomedical and Pharmaceutical Applications
Received 6 November 2019
Accepted for publication 10 January 2020
Published 17 February 2020 Volume 2020:15 Pages 1059—1071
DOI https://doi.org/10.2147/IJN.S237544
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Baozhang Guan,1 Xingwang Zhang2
1Department of Nephrology, The First Affiliated Hospital of Jinan University, Guangzhou 510630, People’s Republic of China; 2Department of Pharmaceutics, College of Pharmacy, Jinan University, Guangzhou 510632, People’s Republic of China
Correspondence: Xingwang Zhang
Department of Pharmaceutics, College of Pharmacy, Jinan University, 601 West Huangpu Avenue, Guangzhou 510632, People’s Republic of China
Email [email protected]
Abstract: Aptamers are a class of targeting ligands that bind exclusively to biomarkers of interest. Aptamers have been identified as candidates for the construction of various smart systems for therapy, diagnosis, bioimaging, and drug delivery due to their high target affinity and specificity. Aptamers are accounted as chemical antibodies that can be readily linked to drugs, sensors, signal enhancers, or nanocarriers for functionalization. Use of aptamer-guided medications, especially nanomedicines, has resulted in encouraging outcomes compared to those use of aptamer-free counterparts. This article reviews recent advances in the use of aptamers as targeting ligands for various biomedical and pharmaceutical purposes. Special interests focus on aptamer-based theranostics, biosensing, bioimaging, drug potentiation, and targeted drug delivery.
Keywords: aptamer, theranostics, biosensor, biomedical imaging, drug delivery
Introduction
Aptamers are short RNA or single-stranded DNA comprise 20–80 nucleotides (Figure 1) that can fold into unique three-dimensional conformations to specifically bind to targets. Aptamers are intentionally designed oligonucleotide sequences that differ from naturally occurring ligands.1 They can specifically recognize and interact with diverse targets such as receptors, antigens, and biomarkers via van der Waals forces, hydrogen bonding, electrostatic interactions, stacking of flat moieties, and shape complementarity.2 Upon binding to targets, aptamers induce a series of biochemical effects, such as antagonism, agonism, suppression, and destruction.3,4 Aptamers can be synthesized through a systematic evolution of ligands by exponential enrichment (SELEX) technique, and have several advantages over commonly used small-molecule ligands, including excellent tissue permeability, unlimited availability of targets, good stability, and modifiability.5
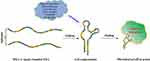 |
Figure 1 Aptamer structure, characteristics, and functional illustration for biomedical applications. |
Discovery and use of artificial antibodies have provided a viable platform for the development of precision medicines. In recent years, the potential for use of aptamers for biomedical purposes has been evaluated in a variety of fields. Aptamers have been evaluated for use in molecular therapy, diagnosis, biosensing, biomedical imaging, drug potentiation, and targeted drug delivery. Aptamers can be customized to target specific molecules using well-established techniques.6 The concept of a “magic bullet” has inspired practitioners to use aptamers to construct smart devices.7 For instance, a nucleolin-targeted intelligent DNA nanorobot that utilized an aptamer and DNA origami was constructed to obstruct vasculature feeding for cancer therapy.8 Hao et al developed aptamer-functionalized microfluidic devices based on dendrimers for sensitive and rapid detection of foodborne pathogenic bacteria.9 A study by Luo et al examined the delivery potential of CD30 aptamer-modified polymeric nanoparticles for targeted delivery of doxorubicin to anaplastic large cell lymphoma cells.10 These studies showed that aptamers have the versatility as specific functional ligands that can induce positive theranostic responses.
Aptamers are considered to be “chemical antibodies” based on their biomimetic nature and simple synthetic procedures. Aptamers are pleiotropic ligands that have potential for use in the development of smart and intelligent systems. Previous investigators have presented reviews on aptamer discovery and applications.11,12 However, these reports revolved around the synthesis of aptamers, aptamer-guided drug delivery, and targeted therapy. The biomedical and pharmaceutical applications of aptamers have not been fully reviewed. This article includes a full review of the application of aptamers in the biomedical and pharmaceutical fields. Aptamer-mediated theranostics, biosensing, bioimaging, drug potentiation, and targeted drug delivery are discussed in this work.
Use of Aptamers in Theranostics
Aptamers can specifically recognize molecular targets and modulate their biological activities, which allows aptamers to be used for therapeutic and diagnostic purposes. Aptamers can regulate biological pathways by interfering with pathological processes. Aptamers can also be used to target biomarkers for diagnostic purpose. Use of aptamers for therapeutic purposes began in 1990 when Tuerk and Gold performed the first SELEX experiment for the selection of an RNA aptamer against bacteriophage T4 DNA polymerase.13 Sullenger et al reported the inhibitory effect of trans-activation response (TAR) decoy, an RNA sequence, on HIV-1 viral infection in host cells.14 Presently, only one aptamer drug (Macugen) has been approved by the FDA, but several others are being evaluated in clinical trials.15 In addition, aptamers have been used to treat aging-related disorders,16 obesity and diabetes mellitus,17,18 cardiovascular diseases,19 infectious diseases,20,21 blood coagulation,22 bone diseases,23 immunological diseases,24 and cancers25 (Figure 2).
 |
Figure 2 Aptamers as theragnostic agents for various diseases by targeting to etiological loci and interfering with pathological processes. |
Aptamers used for diagnostic purposes are typically conjugated to suitable nanoparticles (NPs) to enable detection. Zhao and colleagues developed aptamer-modified fluorescent silica NPs (FSNPs) by conjugating amine-labeled Sgc8 aptamer to carboxyl-modified FSNPs via amide coupling to detect leukemia cells.26 They assessed the sensitivity and specificity of Sgc8-FSNPs using flow cytometry and fluorescence microscopy. The results showed that Sgc8-FSNPs could be used to accurately detect leukemia cells with high sensitivity, which indicated that Sgc8-FSNPs were a highly sensitive tool for specific detection of leukemia cells. Sakyi et al produced RNA aptamers that specifically bound to mycolactone for diagnosis of buruli ulcer (BU) using the SELEX technique.27 The results of this study indicated that diagnosis of BU using RNA aptamers was feasible. Recently, aptamers have received attention as potential agents for disease prevention and treatment. Fibroblast growth factor 2 (FGF2) plays a key role in bone remodeling and disease progression. Jin et al constructed a novel human FGF2-specific RNA aptamer, APT-F2,23 which blocked FGF2 binding to its four cellular receptors, inhibiting FGF2-induced downstream signaling. This resulted in decreased cell proliferation and restored osteoblast differentiation. Polyethylene glycol-modified APT-F2 (APT-F2P) effectively prevented arthritis and osteoporosis. These findings illustrated the dual therapeutic potential of APT-F2P for the treatment of bone disease and pain. In another case, AS1411 and modified AS1411 aptamers that bind to nucleolin were investigated for their potentials in treating hepatocellular carcinoma.28 Modified AS1411-aptamer significantly suppressed HCC cell growth in vitro and in vivo by up-regulating galectin-14 expressions. Introducing aptamers into the field of theranostics may provide more sensitive and precise alternatives to conventional approaches. Aptamer-based theranostics have advantages over traditional diagnosis and drug therapy. The underlying reason lies in the specificity and targeted regulation of an aptamer to a given disease.
Aptamer-Based Biosensors
Biosensors are comprised of a transducer and a biological element that recognizes a biological response. The transducer converts the signal to provide a diagnostic indicator. The biological element may be an enzyme, an antibody, or a nucleic acid (eg, aptamer) with or without a modified detectable molecule. Aptamers can bind to target molecules with high affinity and specificity, which is ideal for use as biosensor component. Aptamer-linked biosensors, also known as aptasensors, can be detected using fluorescence, chemiluminescence, electrochemistry, or immunoluminescence techniques.29 Figure 3 provides an illustration of components, detection techniques, and signal read-outs associated with aptasensors. Aptamer-based biosensors have been used for accurate and rapid detection of disease markers, pathogens, antibiotics, toxins, pollutes, and other chemicals or biomolecules.30–34 Biosensors utilize the specificity of aptamers to disease targets and the convertibility of transducers to translate the biological response into a detectable signal.
 |
Figure 3 Aptamer-mediated biosensors for analysis using fluorescence, chemiluminescence, electrochemistry, or immunoluminescence. |
Current diagnostic procedures for tumors typically involve biopsy. No mature techniques have been available for rapid and sensitive detection of tumors. Li et al developed aptamer-functionalized gold nanorods as a diagnostic biosensor for MCF-7 breast cancer cells.35 Gold nanorods with aptamers specific to mucin 1 protein (MUC-1) allowed for rapid diagnosis of human breast carcinoma MCF-7 cells with a detection limit of 100 cells/mL. The interactional signals between the biosensor and cancer cells were readily translated into unique localized surface plasmon resonance (LSPR) spectra. Ahmadzadeh-Raji et al developed an optically transparent aptasensor for diagnosis of colon cancer that consisted of gold NPs with electrodeposited indium tin oxide.36 The aptamer-containing electrodeposited gold NPs functioned as nanoprobes for cancer cell targeting and could be detected using a conventional optical microscope or an electrochemical method. Another study reported the development of aptamer-conjugated gold-coated magnetic NPs for intelligent detection of leukemia cancer cells.37 In addition to cancer detection, aptasensors have been used to trace microRNAs,38 C-reactive protein,39 and lysozyme.40
Aptamers for Biomedical Imaging
Non-invasive imaging of various disease markers using aptamer-modified probes has shown numerous advantages over traditional imaging techniques. Ligand-mediated non-invasive imaging allows for visual observation of diseases at the cellular and molecular levels.41 This technique provides high accuracy and predictability for early diagnosis and treatment, resulting in improved prognosis. Aptamer-aided biomedical imaging can be achieved through direct or activatable optical imaging by linking an aptamer to a fluorescent molecule or a fluorescent mediator (NPs or quantum dots), or through other tomographic techniques, such as MRI, ultrasound, and PET (Figure 4). Optical imaging can be used to detect fluorescence or bioluminescence of aptamer-linked signal transducers. Optical imaging is a simple, low-cost solution for visualization of diseases using aptamers. For example, Li et al synthesized a DNA aptamer, R13, and labeled it with FAM and Cy5 at the 5ʹ terminal of the DNA sequence for in vivo imaging of ovarian cancer.42 Aptamer R13 was highly specific for ovarian cancer, exhibited good stability, and was able to bind to extracellular membrane proteins. In addition, fluorescence-labeled R13 allowed for excellent ex vivo tissue and in vivo tumor imaging. Although aptamers possess good affinity to their targets, they are frequently off-target to specific tissues in vivo due to short half-life. To improve tissue targeting, aptamers are often conjugated with various nanovectors, including gold/silver NPs, silica NPs, and quantum dots (QDs). Chen et al constructed a versatile protein labeling platform with aptamer-mediated NPs for intracellular imaging and tracking of endocytosis dynamics.43 This platform exhibited good cellular tracing ability and provided valuable insights into NPs endocytosis, which may be promising for biomedical labeling of cell activities. Tan et al developed a novel theranostic system based on endoglin aptamer (YQ26)-modified fluorescent silica NPs (YQ26-FsiNPs) for tumor imaging, treatment, and monitoring.44 Use of YQ26-FSiNPs resulted in high targeting efficiency and excellent therapeutic effects via aptamer YQ26-mediated binding. In vitro and in vivo studies showed that YQ26-FSiNPs was a promising agent for clinical tumor diagnosis and treatment. Quantum dots are inorganic fluorescent semiconductor NPs used for imaging. Aptamers can be conjugated to QDs for bioimaging without the need for the addition of a fluorescent agent. Aptamer-conjugated QDs were used by Kaur to image cellular uptake in cancer cells.45 Fluorescence imaging showed that the conjugates containing QD-SL(2)-B aptamer, an aptamer that targets the heparin-binding domain of VEGF165 protein, specifically recognized Hep G2 cells and were internalized in the absence of an additional transfecting agent. This aptamer conjugate provided a smart strategy for liver cancer imaging and cellular labeling.
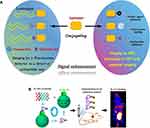 |
Figure 4 Design rationale and imaging methods for aptamer-facilitated biomedical imaging (A) with a demonstration of PET molecular imaging for HER2 in cancer (B). The illustration was used with permission from Zhu G, Zhang H, Jacobson O, et al. Combinatorial screening of DNA aptamers for molecular imaging of HER2 in cancer. Bioconjug Chem. 2017;28(4):1068–1075. doi:10.1021/acs.bioconjchem.6b00746, Copyright (2017) American Chemical Society.50 |
Activatable aptamer probes take advantage of the “on/off” rational of fluorescence resulting from biological interactions or during fluorescence resonance energy transfer (FRET) for imaging. Xu et al used the fluorescence quenching properties of graphene oxide (GO) to design an aptamer-based four-color fluorometric system for simultaneous detection and imaging of α-fetoprotein, VEGF-165, carcinoembryonic antigen, and human epidermal growth factor receptor (HER2) in living cells.46 Due to strong π interactions, the fluorescence of the labeled aptamers was quenched by GO. Four fluorophore-labeled aptamers targeting to tumor-associated proteins were adsorbed on GO to form four-color nanoprobes with quenched fluorescence. Upon being internalized into cells, the aptamer/GO nanoprobes interacted with the intracellular tumor-associated proteins, which resulted in dissociation of the aptamer from GO, and resulted in the activation of fluorescence. Aptamer-based probes allow for rapid discrimination of tumor cells from normal cells via imaging. Using hybridization chain reaction (HCR), Yuan et al constructed a multifluorescence FRET detection method for specific imaging of cell-surface glycosylation.47 Intramolecular multi-FRET occurred due to a short donor-acceptor distance when aptamer-tethered nanoassemblies bound to receptors through metabolic glycan labeling. Amplification effects and spatial flexibility of aptamer-linked HCR nanovectors enhanced the multi-FRET imaging resolution that could allow for real-time observation of cell-surface glycosylation.
Aptamer-mediated tomographic imaging has become an important arena of aptamer application. Aptamers can be conjugated to signal enhancers (not luminophores) used for targeted MRI, ultrasound, and PET imaging. Aptamer-based MRI contrast agents are generally designed by linking an aptamer with superparamagnetic iron oxide (SPIO) NPs. For example, a glypican-3 (GPC3)-specific aptamer (AP613-1) has been tethered to ultrafine SPIO NPs (USPIONPs) for targeted imaging of hepatocellular carcinoma.48 This study demonstrated specific uptake of Apt-USPIONPs into Huh-7 cells, and in vivo T2-weighted MRI showed significant negative enhancement of Huh-7 tumors through imaging of Apt-USPIONPs. The combination of aptamers and ultrasound contrast agents (UCA) can enhance ultrasonic signal detection. Commonly used UCAs include gas-containing micro/nanobubbles, which have intense ultrasonic scattering properties that can significantly enhance the ultrasonic echo signals following administration, resulting in improved imaging for tissues or organs. Conjugation of specific aptamers to UCAs can result in enhancive effects of UCAs due to targeting. Zhu et al developed carbonic anhydrase IX (CAIX) aptamer-immobilized targeted nanobubbles for ultrasound molecular imaging of various tumors.49 Targeted nanobubbles loaded with CAIX aptamer penetrated the tumor vasculature to allow for ultrasound molecular imaging (USMI) of tumor parenchymal cells. Nanobubbles containing CAIX aptamer exhibited higher peak intensity and larger area under the curve than non-targeted nanobubbles in 786-O and Hela xenograft tumor tissues. Aptamer-containing targeted nanobubbles exhibited superior imaging effects in tumor parenchymal cells, which resulted in enhanced diagnostic accuracy of malignant tumors in various organs. Radionuclide-based imaging techniques such as SPECT and PET have greater diagnostic capability than other molecular imaging techniques due to superb tissue penetration and quantifiability. Coupling of aptamers to radioisotopes (eg, 125I, 99Tc and 18F) can result in highly specific and sensitive radiopharmaceuticals. In one study, HER2-targeted DNA aptamers (heraptamers) were screened and labeled with 18F for PET imaging of ovarian cancer.50 Aptamers labeled with 18F exhibited high tumor uptake ratio and specific HER2 imaging in SKOV3 tumor-bearing mice. These studies demonstrated the potential of aptamers for targeted bioimaging. In addition, aptamers have been evaluated for use in other imaging applications, such as computed tomography (CT), transmission electron microscopy (TEM), surface plasmon resonance (SPR), and surface-enhanced Raman spectroscopy (SERS).
Drug Potentiation via Aptamer Modification
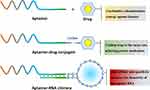
The potency of therapeutic agents can be compromised by limited selectivity, “off-target” effects, and lack of synergy. Targeted or combined therapy can enhance the selectivity and potency of medications. One approach is to utilize disease-specific aptamers with high affinity or bioactivity to potentiate the effects of therapeutic agents. Aptamers can achieve drug potentiation in three ways (Figure 5): (1) combinatorial administration using simple mixtures; (2) formation of aptamer-drug conjugates; and (3) development of aptamer-drug chimeras. Combinatorial administration utilizes the therapeutic properties of aptamers to synergistically enhance the therapeutic effects of the counterpart drugs. Aptamer-drug conjugates allow conjugated drugs to be specifically delivered to disease targets by aptamers. In the case of aptamer-drug chimeras, the “drug” is typically a therapeutic RNA (siRNA or miRNA). Aptamers are directly hybridized into therapeutic RNAs and serve as active targeting ligands to guide the therapeutic RNA to the target site. Cell- or receptor-specific chimeric RNAs have been shown to improve the in vivo pharmacokinetic distributions of prototype RNAs, and facilitate transport into cells by receptor-mediated endocytosis.51
Combined use of therapeutic aptamers and therapeutic drugs has been reported. Schilling et al investigated the radiosensitizing effect of a peptide aptamer, A17, which inhibits Hsp70, in combination with the Hsp90 inhibitor NVP-AUY922.52 Co-administration of A17 with NVP-AUY922 significantly enhanced the pro-apoptotic effects of NVP-AUY922, but it did not significantly affect the radiosensitivity of human lung or breast cancer cells. Inhibition of Hsp70 by A17 potentiated the radiosensitizing effect of NVP-AUY922. A combined strategy based on simultaneous inhibition of PD-1 and C5a/C5aR1 signaling using a RMP1-14 antibody and C5a L-aptamer to synergistically inhibit growth and metastasis of lung cancer was proposed by Ajona et al.53 This study showed that C5a and PD-1 inhibition by an RMP1-14 antibody and C5a L-aptamer significantly reduced tumor growth and metastasis, which resulted in increased survival.
Design of aptamer-drug conjugates is an innovative drug modification technique. In these conjugates, the aptamer does not exert pharmacological effects or modulate the activity of the parent compound, but targets the therapeutic molecule to the tissue of interest. Antibiotic resistance is a substantial clinical concern. Lijuan et al developed aptamer-ampicillin conjugates to penetrate bacterial biofilms, which helped to overcome antibiotic resistance.54 These conjugates exerted distinct synergistic antibacterial activity greater than that of either component alone. Coupling of an aptamer to a cytotoxic drug to treat local and metastatic tumor lesions is another important aptamer application. Powell et al synthesized tunable cytotoxic aptamer-drug conjugates for the treatment of prostate cancer.55 A prostate cancer-specific aptamer (E3) was produced using cell SELEX selection. The aptamer was internalized into prostate cancer cells, but not into normal prostate cells. Conjugation of E3 to MMAE and MMAF (monomethyl auristatin E and F) resulted in potent cytotoxic agents that efficiently killed prostate cancer cells, but not normal prostate epithelial cells, in vitro. In addition, the cytotoxicity of E3 aptamer-drug conjugates can be inhibited by an oligonucleotide against the E3 aptamer. This provides a means to intervene in the unexpected event of normal cell killing in vivo.
The therapeutic superiority of aptamer-RNA chimeras has been demonstrated in recent years.56 Wang et al constructed an aptamer-siRNA chimera targeted to epithelial cell adhesion molecule and survivin to deliver a high dose of siRNA and silence resistance to doxorubicin.57 Silencing of survivin (a key protein involved in drug resistance) resulted in a significant reversal of chemoresistance. This chimera, in combination with a low dose of doxorubicin, inhibited stemness, eliminated cancer stem cells via apoptosis, suppressed tumor growth, and prolonged survival of tumor-bearing mice. In brief, aptamers can be used to potentiate drugs or actives by synergistic interaction, inspiring specific drug delivery, and conferring affinity to the underperformed entities.
Aptamers for Targeted Drug Delivery
The in vivo distribution of medicines plays a key role in pharmacological action. Most of drugs exhibit diffuse biodistribution following administration, resulting in unsatisfactory therapeutic outcomes and pronounced side effects. Targeted drug delivery systems (TDDS) represent the most promising drug delivery strategy for drug accumulation in the tissues of interest in vivo. A targeted drug delivery system typically consists of a nanocarrier and a targeting moiety. The targeting component is referred to as “ligand.” Many ligand molecules are currently available for the development of TDDS,58,59 including vitamins, saccharides, bile salts, transferrin, lectins, oligopeptides, antibodies, and aptamers. Aptamers have been shown to be outstanding TDDS ligands due to facile synthesis, high specificity, and potential for simple modification. Aptamers are known as chemical antibodies with modifiable groups at the terminal end, which can be synthesized using the in vitro SELEX technique. In addition, aptamers can be flexibly anchored to a variety of nanocarriers via various conjugation approaches (Figure 6). Organic nanocarriers (liposomes, polymeric-lipid hybrid NPs, micelles, and dendrimers) and inorganic nanocarriers (Au/Ag NPs, silica NPs, carbon NPs, selenium NPs, and other inorganic NPs) can be modified with specific aptamers to achieve targeted drug delivery. Aptamer-mediated targeted drug delivery can be used to treat a broad spectrum of diseases, including tumors, immunological diseases, genetic diseases, infectious diseases, and cytopathic disorders.
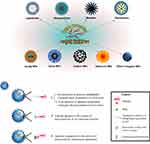 |
Figure 6 Various organic and inorganic nanocarriers functionalized using aptamers for targeted drug delivery, and commonly used strategies for aptamer conjugation with nanocarriers. |
Aptamer-mediated drug delivery has shown potential as a cancer treatment strategy. The primary considerations in the design of TDDS are to promote the accumulation of chemotherapeutic agents in the tumor tissue and overcome drug resistance. Ligands can guide nanocarriers to tumors and facilitate the transport of these nanocarriers into tumor cells, resulting in cell death. Liposomes are the most commonly used vehicles functionalized with aptamers for targeted drug delivery. To overcome drug resistance from erlotinib, Li et al developed lung cancer-specific liposomes using EGFR aptamer-chitosan conjugates to deliver erlotinib to the tumor.60 The nanocarrier showed good biostability and binding specificity for EGFR-mutated cancer cells, which facilitated liposomal erlotinib internalization into EGFR-mutated cancer cells. Aptamer-modified liposomes exhibited better delivery of erlotinib to EGFR-mutated cancer cells than standard delivery techniques, which resulted in greater cytotoxicity. In another study, tumoral delivery of cabazitaxel was improved using aptamer-modified liposomal delivery system (apt-liposomes).61 In this study, apt-liposomes increased the accumulation of cabazitaxel in the tumor tissue. These findings suggested that specific binding of aptamer (TLS1c) to the targeted cells (MEAR) facilitated the accumulation of liposomes in the target tissue. Coupling of aptamers to NPs, including polymeric and lipid-based NPs, is also a common delivery strategy to augment the anticancer efficacy of chemotherapeutic agents. Chen et al constructed docetaxel-loaded polymeric NPs (DTX-apt-NPs) targeting to prostate cancer using an aptamer-mediated delivery strategy.62 DTX-apt-NPs enhanced in vitro antitumor activity and cellular uptake compared with conventional NPs. Clathrin-dependent endocytosis was involved in intracellular trafficking of these NPs. Anti-PSMA aptamer-mediated delivery significantly improved the antitumor effect of docetaxel.
Micelles are likewise excellent drug delivery systems that can be aptamer-modified. Charbgoo et al engineered MUC1 aptamer-targeted DNA micelles for systemic delivery of doxorubicin and KLA peptide for dual tumor therapy.63 Anti-MUC1 aptamer specifically delivered micelles into MCF-7 cells by improving the cellular uptake. Co-delivery of doxorubicin and KLA using aptamer-modified micelles significantly enhanced anti-tumor efficacy through synergy and attenuation of drug combination. In addition, aptamers are often used to modify dendrimers for targeted drug delivery. Therapeutic RNA and small-molecule drugs can all be delivered to tumor targets using aptamer-modified dendrimers.64,65 In vitro and in vivo study results have shown that aptamer-modified dendrimers specifically bound to tumor cells and enhanced antitumor efficacy compared with aptamer-free dendrimers.
Aptamers have also been used to modify inorganic nanocarriers to optimize drug delivery. Inorganic nanocarriers that can be coated with functional aptamers include, but not limited to, gold/silver NPs, silicon NPs, carbon NPs, and selenium NPs. Chemical drugs or photosensitizers, or both, can be incorporated into aptamer-modified inorganic nanocarriers. For example, a nanoplatform based on aptamer-functionalized gold NPs was developed by Huang et al for co-delivery of a photosensitizer and doxorubicin.66 This tumor-targeted combinative therapy system resulted in significant necrocytosis of HeLa and Dox-resistant MCF-7R cell lines upon exposure to an exciting light. In addition, light-triggered release of doxorubicin resulted in enhanced cytotoxicity in target cells. To achieve controlled drug delivery and allow for radioimaging, MUC1 aptamer-capped mesoporous silica NPs (MSNs) were engineered by Pascual et al.67 Safranin O-loaded MSNs functionalized with aminopropyl groups and gated with the negatively charged MUC1 aptamer have been intended for specific delivery to target tissues and specific release of therapeutic agents in tumor cells. Confocal microscopy showed that MUC1 aptamer-capped NPs (MUC1-apt-NPs) were internalized into MDA-MB-231 breast cancer cells overexpressing the MUC1 receptor, which resulted in pore opening and cargo release. Radio-labeling of MUC1-apt-NPs with 99mTc resulted in tumor targeting in vivo, as evidenced using SPECT imaging in tumor-bearing Balb/c mice. Carbon NPs, which are inert, have also been evaluated in combination with functional aptamers for targeted drug delivery and bioimaging. Li et al reported use of aptamer-gated mesoporous carbon nanospheres for imaging of cellular mucin and on-demand targeted drug delivery.68 Aptamer-coupled carbon NPs can be used for real-time switchable imaging of cellular mucin through π-π stacking, and can also be used for targeted doxorubicin release in tumors. Selenium NPs (SeNPs) have recently received attention for use in drug delivery and biomedical applications.69 SeNPs can be functionalized with aptamers for the optimization of drug delivery. Recently, a targeted co-delivery system based on SeNPs was developed using 5TR1 aptamer as the targeting agent to enhance the in vitro and in vivo anti-tumor efficacy of epirubicin and NAS-24 aptamer (an apoptosis induction agent).70 Aptamer-modified SeNPs significantly reduced toxicity in non-target cells, but reduced cell viability of the target cancer cells. The in vivo anticancer efficacy of these SeNPs confirmed that this nanosystem significantly inhibited tumor growth of tumor-bearing mice to a greater extent than treatment with epirubicin alone. In addition, other aptamer-functionalized inorganic nanocarriers, such as calcium carbonate NPs,71 and iron oxide NPs,72 have also been evaluated for targeted drug delivery.
Aptamer-mediated active TDDS have been shown able to induce significant antitumor activities. In addition, aptamers can be used as targeting ligands for other drug delivery to treat non-tumor diseases. Danesh et al developed aptamer-modified gold NPs for targeted delivery and controllable release of daunorubicin to T-cell acute lymphoblastic leukemia.73 Targeted delivery of tacrolimus using pH-responsive aptamer-chitosan-PLGA nanocomplexes to suppress immunological rejection was also investigated.74 Besides, aptamer-functionalized lipid NPs were constructed for specific delivery of RNAi to osteoblasts to treat bone anabolic diseases.75 In two another studies, aptamer-modified nanocapsules and gold NPs were developed for targeted delivery of antimicrobial agents to bacteria via aptamer-receptor interactions to eradicate pathogens.76,77 Taken together, aptamers can be used to decorate a variety of drug delivery vehicles to confer targeting ability whereby to enhance the therapeutic efficacy of payloads against various diseases (Table 1). Nanocarriers solve the unmet properties of “problem” drugs by encapsulation, while aptamers push them to the endpoint of targeted therapy.
 |
Table 1 Representative Nanocarriers Functionalized with a Specific Aptamer Used for Targeted Drug Delivery |
Concluding Remarks
Aptamers are pleiotropic ligands that can be chemically synthesized in large quantities and exhibit high affinity for selected targets. Aptamers that can be considered as artificial antibodies have many advantages over naturally-occurring ligands such as scalability, high specificity, and ease of conjugation to other molecules. These properties make aptamers suitable for use in theranostics, biomedical imaging, drug potentiation, and targeted drug delivery. Aptamers can act as agonists or antagonists to modulate the functions of target effectors pertinent to a disease. Aptamers can also be coupled to signaling molecules for use in diagnostics. Aptamer-based biosensors, called aptasensors, can be used to detect a variety of biomarkers. Aptamers can be linked to luminophores or signal enhancers to allow for targeted imaging. Moreover, aptamers can be used as targeting ligands to functionalize non-targeted drugs, or as therapeutic molecules to potentiate the effects of other therapeutic molecules. Finally, aptamers can be used to construct intelligent drug delivery systems for the application of precision medicines. Aptamers have great potential for use in the biomedical/pharmaceutical field due to advances in the SELEX technique. Further studies may identify other potential uses of aptamers.
Although aptamers are extremely rich in resources, there are still some areas for improvement in the use of aptamers. The synthetic technology of oligonucleotides needs to be further developed so as to boost the output of aptamers. The aptamers screening technology needs to be further updated to facilitate the quick identification of target aptamers. It also requires to expand the modification strategies of aptamers to make them available for molecular coupling. Furthermore, the biological stability of aptamers in vivo should be taken into serious consideration, which is the prerequisite for the application of aptamers. We believe that these problems will be tackled properly with advances in aptamer technology.
Acknowledgments
This work was jointly supported by the National Natural Science Foundation of China (81673604), Scientific Research Foundation of the First Affiliated Hospital of Jinan University, China (2015210 and 2017305) and Medical Science and Technology Foundation of Guangdong Province (2018313 and A2016040).
Disclosure
The authors declare no conflicts of interest in this work.
References
1. Chushak YG, Martin JA, Chavez JL, Kelley-Loughnane N, Stone MO. Computational design of RNA libraries for in vitro selection of aptamers. Methods Mol Biol. 2014;1111:1–15.
2. Hayashi T, Oshima H, Mashima T, Nagata T, Katahira M, Kinoshita M. Binding of an RNA aptamer and a partial peptide of a prion protein: crucial importance of water entropy in molecular recognition. Nucleic Acids Res. 2014;42(11):6861–6875. doi:10.1093/nar/gku382
3. Li Y, Wu H, Niu Y, et al. Development of RNA aptamer-based therapeutic agents. Curr Med Chem. 2013;20(29):3655–3663. doi:10.2174/0929867311320290011
4. Wiraja C, Yeo D, Lio D, et al. Aptamer technology for tracking cells’ status & function. Mol Cell Ther. 2014;2:33. doi:10.1186/2052-8426-2-33
5. Sun H, Zu Y. A highlight of recent advances in aptamer technology and its application. Molecules. 2015;20(7):11959–11980. doi:10.3390/molecules200711959
6. Darmostuk M, Rimpelova S, Gbelcova H, Ruml T. Current approaches in SELEX: an update to aptamer selection technology. Biotechnol Adv. 2015;33(6 Pt 2):1141–1161. doi:10.1016/j.biotechadv.2015.02.008
7. Ding F, Gao Y, He X. Recent progresses in biomedical applications of aptamer-functionalized systems. Bioorg Med Chem Lett. 2017;27(18):4256–4269. doi:10.1016/j.bmcl.2017.03.032
8. Li H, Liu J, Gu H. Targeting nucleolin to obstruct vasculature feeding with an intelligent DNA nanorobot. J Cell Mol Med. 2019;23(3):2248–2250. doi:10.1111/jcmm.2019.23.issue-3
9. Hao X, Yeh P, Qin Y, et al. Aptamer surface functionalization of microfluidic devices using dendrimers as multi-handled templates and its application in sensitive detections of foodborne pathogenic bacteria. Anal Chim Acta. 2019;1056:96–107. doi:10.1016/j.aca.2019.01.035
10. Luo X, Yang Y, Kong F, Zhang L, Wei K. CD30 aptamer-functionalized PEG-PLGA nanoparticles for the superior delivery of doxorubicin to anaplastic large cell lymphoma cells. Int J Pharm. 2019;564:340–349. doi:10.1016/j.ijpharm.2019.04.013
11. Zhu G, Chen X. Aptamer-based targeted therapy. Adv Drug Del Rev. 2018;134:65–78. doi:10.1016/j.addr.2018.08.005
12. Alshaer W, Hillaireau H, Fattal E. Aptamer-guided nanomedicines for anticancer drug delivery. Adv Drug Del Rev. 2018;134:122–137. doi:10.1016/j.addr.2018.09.011
13. Tuerk C, Gold L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science. 1990;249(4968):505–510. doi:10.1126/science.2200121
14. Sullenger BA, Gallardo HF, Ungers GE, Gilboa E. Overexpression of TAR sequences renders cells resistant to human immunodeficiency virus replication. Cell. 1990;63(3):601–608. doi:10.1016/0092-8674(90)90455-N
15. Parashar A. Aptamers in therapeutics. J Clin Diagn Res. 2016;10(6):Be01–06. doi:10.7860/JCDR/2016/18712.7922
16. Yamagishi S, Taguchi K, Fukami K. DNA-aptamers raised against AGEs as a blocker of various aging-related disorders. Glycoconj J. 2016;33(4):683–690. doi:10.1007/s10719-016-9682-2
17. Helmling S, Maasch C, Eulberg D, et al. Inhibition of ghrelin action in vitro and in vivo by an RNA-Spiegelmer. Proc Natl Acad Sci U S A. 2004;101(36):13174–13179. doi:10.1073/pnas.0404175101
18. Vater A, Sell S, Kaczmarek P, et al. A mixed mirror-image DNA/RNA aptamer inhibits glucagon and acutely improves glucose tolerance in models of type 1 and type 2 diabetes. J Biol Chem. 2013;288(29):21136–21147. doi:10.1074/jbc.M112.444414
19. Wang P, Yang Y, Hong H, Zhang Y, Cai W, Fang D. Aptamers as therapeutics in cardiovascular diseases. Curr Med Chem. 2011;18(27):4169–4174. doi:10.2174/092986711797189673
20. Maier KE, Jangra RK, Shieh KR, et al. A new transferrin receptor aptamer inhibits new world hemorrhagic fever mammarenavirus entry. Mol Ther Nucleic Acids. 2016;5:e321. doi:10.1038/mtna.2016.32
21. Li W, Feng X, Yan X, Liu K, Deng L. A DNA aptamer against influenza a virus: an effective inhibitor to the hemagglutinin-glycan interactions. Nucleic Acid Ther. 2016;26(3):166–172. doi:10.1089/nat.2015.0564
22. Soule EE, Bompiani KM, Woodruff RS, Sullenger BA. Targeting two coagulation cascade proteases with a bivalent aptamer yields a potent and antidote-controllable anticoagulant. Nucleic Acid Ther. 2016;26(1):1–9. doi:10.1089/nat.2015.0565
23. Jin L, Nonaka Y, Miyakawa S, Fujiwara M, Nakamura Y. Dual therapeutic action of a neutralizing anti-FGF2 aptamer in bone disease and bone cancer pain. Mol Ther. 2016;24(11):1974–1986. doi:10.1038/mt.2016.158
24. Li W, Lan X. Aptamer oligonucleotides: novel potential therapeutic agents in autoimmune disease. Nucleic Acid Ther. 2015;25(4):173–179. doi:10.1089/nat.2014.0529
25. Zhu G, Ye M, Donovan MJ, Song E, Zhao Z, Tan W. Nucleic acid aptamers: an emerging frontier in cancer therapy. Chem Commun (Camb). 2012;48(85):10472–10480. doi:10.1039/c2cc35042d
26. Tan J, Yang N, Hu Z, et al. Aptamer-functionalized fluorescent silica nanoparticles for highly sensitive detection of leukemia cells. Nanoscale Res Lett. 2016;11(1):298. doi:10.1186/s11671-016-1512-8
27. Sakyi SA, Aboagye SY, Otchere ID, Liao AM, Caltagirone TG, Yeboah-Manu D. RNA aptamer that specifically binds to mycolactone and serves as a diagnostic tool for diagnosis of buruli ulcer. PLoS Negl Trop Dis. 2016;10(10):e0004950. doi:10.1371/journal.pntd.0004950
28. Cho Y, Lee YB, Lee JH, et al. Modified AS1411 aptamer suppresses hepatocellular carcinoma by up-regulating galectin-14. PLoS ONE. 2016;11(8):e0160822. doi:10.1371/journal.pone.0160822
29. Ravalli A, Voccia D, Palchetti I, Marrazza G. Electrochemical, electrochemiluminescence, and photoelectrochemical aptamer-based nanostructured sensors for biomarker analysis. Biosensors (Basel). 2016;6(3):e39. doi:10.3390/bios6030039
30. MacKay S, Wishart D, Xing JZ, Chen J. Developing trends in aptamer-based biosensor devices and their applications. IEEE Trans Biomed Circuits Syst. 2014;8(1):4–14. doi:10.1109/TBCAS.2014.2304718
31. Park KS. Nucleic acid aptamer-based methods for diagnosis of infections. Biosens Bioelectron. 2018;102:179–188. doi:10.1016/j.bios.2017.11.028
32. Mehlhorn A, Rahimi P, Joseph Y. Aptamer-based biosensors for antibiotic detection: a review. Biosensors (Basel). 2018;8(2):e54. doi:10.3390/bios8020054
33. Nguyen VT, Kwon YS, Gu MB. Aptamer-based environmental biosensors for small molecule contaminants. Curr Opin Biotechnol. 2017;45:15–23. doi:10.1016/j.copbio.2016.11.020
34. Zamay AS, Zamay GS, Kolovskaya OS, Zamay TN, Berezovski MV. Aptamer-based methods for detection of circulating tumor cells and their potential for personalized diagnostics. Adv Exp Med Biol. 2017;994:67–81.
35. Li Y, Zhang Y, Zhao M, et al. A simple aptamer-functionalized gold nanorods based biosensor for the sensitive detection of MCF-7 breast cancer cells. Chem Commun (Camb). 2016;52(20):3959–3961. doi:10.1039/C6CC01014H
36. Ahmadzadeh-Raji M, Ghafar-Zadeh E, Amoabediny G. An optically-transparent aptamer-based detection system for colon cancer applications using gold nanoparticles electrodeposited on indium tin oxide. Sensors (Basel). 2016;16(7):e1071. doi:10.3390/s16071071
37. Khoshfetrat SM, Mehrgardi MA. Amplified detection of leukemia cancer cells using an aptamer-conjugated gold-coated magnetic nanoparticles on a nitrogen-doped graphene modified electrode. Bioelectrochemistry. 2017;114:24–32. doi:10.1016/j.bioelechem.2016.12.001
38. Zhong W, Sczepanski JT. A mirror image fluorogenic aptamer sensor for live-cell imaging of microRNAs. ACS Sens. 2019;4(3):566–570. doi:10.1021/acssensors.9b00252
39. Wang J, Guo J, Zhang J, Zhang W, Zhang Y. RNA aptamer-based electrochemical aptasensor for C-reactive protein detection using functionalized silica microspheres as immunoprobes. Biosens Bioelectron. 2017;95:100–105. doi:10.1016/j.bios.2017.04.014
40. Khan NI, Maddaus AG, Song E. A low-cost inkjet-printed aptamer-based electrochemical biosensor for the selective detection of lysozyme. Biosensors (Basel). 2018;8(1):e7. doi:10.3390/bios8010007
41. Hassanzadeh L, Chen S, Veedu RN. Radiolabeling of nucleic acid aptamers for highly sensitive disease-specific molecular imaging. Pharmaceuticals (Basel). 2018;11(4):e106.
42. Li F, Wang Q, Zhang H, et al. Characterization of a DNA aptamer for ovarian cancer clinical tissue recognition and in vivo imaging. Cell Physiol Biochem. 2018;51(6):2564–2574. doi:10.1159/000495925
43. Chen LQ, Xiao SJ, Hu PP, et al. Aptamer-mediated nanoparticle-based protein labeling platform for intracellular imaging and tracking endocytosis dynamics. Anal Chem. 2012;84(7):3099–3110. doi:10.1021/ac202810b
44. Tan J, Yang N, Zhong L, et al. A new theranostic system based on endoglin aptamer conjugated fluorescent silica nanoparticles. Theranostics. 2017;7(19):4862–4876. doi:10.7150/thno.19101
45. Kaur H. Aptamer conjugated quantum dots for imaging cellular uptake in cancer cells. J Nanosci Nanotechnol. 2019;19(7):3798–3803. doi:10.1166/jnn.2019.16735
46. Xu J, Chen W, Shi M, et al. An aptamer-based four-color fluorometric method for simultaneous determination and imaging of alpha-fetoprotein, vascular endothelial growth factor-165, carcinoembryonic antigen and human epidermal growth factor receptor 2 in living cells. Mikrochim Acta. 2019;186(3):204. doi:10.1007/s00604-019-3312-1
47. Yuan B, Chen Y, Sun Y, et al. Enhanced imaging of specific cell-surface glycosylation based on multi-FRET. Anal Chem. 2018;90(10):6131–6137. doi:10.1021/acs.analchem.8b00424
48. Zhao M, Liu Z, Dong L, et al. A GPC3-specific aptamer-mediated magnetic resonance probe for hepatocellular carcinoma. Int J Nanomedicine. 2018;13:4433–4443. doi:10.2147/IJN.S168268
49. Zhu L, Wang L, Liu Y, Xu D, Fang K, Guo Y. CAIX aptamer-functionalized targeted nanobubbles for ultrasound molecular imaging of various tumors. Int J Nanomedicine. 2018;13:6481–6495. doi:10.2147/IJN
50. Zhu G, Zhang H, Jacobson O, et al. Combinatorial screening of DNA aptamers for molecular imaging of HER2 in cancer. Bioconjug Chem. 2017;28(4):1068–1075. doi:10.1021/acs.bioconjchem.6b00746
51. Yoon S, Rossi JJ. Aptamers: uptake mechanisms and intracellular applications. Adv Drug Deliv Rev. 2018;134:22–35. doi:10.1016/j.addr.2018.07.003
52. Schilling D, Garrido C, Combs SE, Multhoff G. The Hsp70 inhibiting peptide aptamer A17 potentiates radiosensitization of tumor cells by Hsp90 inhibition. Cancer Lett. 2017;390:146–152. doi:10.1016/j.canlet.2017.01.015
53. Ajona D, Ortiz-Espinosa S, Moreno H, et al. A combined PD-1/C5a blockade synergistically protects against lung cancer growth and metastasis. Cancer Discov. 2017;7(7):694–703. doi:10.1158/2159-8290.CD-16-1184
54. Lijuan C, Xing Y, Minxi W, Wenkai L, Le D. Development of an aptamer-ampicillin conjugate for treating biofilms. Biochem Biophys Res Commun. 2017;483(2):847–854. doi:10.1016/j.bbrc.2017.01.016
55. Powell Gray B, Kelly L, Ahrens DP, et al. Tunable cytotoxic aptamer-drug conjugates for the treatment of prostate cancer. Proc Natl Acad Sci U S A. 2018;115(18):4761–4766. doi:10.1073/pnas.1717705115
56. Kruspe S, Giangrande PH. Aptamer-siRNA chimeras: discovery, progress, and future prospects. Biomedicines. 2017;5(3):e45. doi:10.3390/biomedicines5030045
57. Wang T, Gantier MP, Xiang D, et al. EpCAM Aptamer-mediated survivin silencing sensitized cancer stem cells to doxorubicin in a breast cancer model. Theranostics. 2015;5(12):1456–1472. doi:10.7150/thno.11692
58. Zhang X, Wu W. Ligand-mediated active targeting for enhanced oral absorption. Drug Discov Today. 2014;19(7):898–904. doi:10.1016/j.drudis.2014.03.001
59. Srinivasarao M, Low PS. Ligand-targeted drug delivery. Chem Rev. 2017;117(19):12133–12164. doi:10.1021/acs.chemrev.7b00013
60. Li F, Mei H, Xie X, et al. Aptamer-conjugated chitosan-anchored liposomal complexes for targeted delivery of erlotinib to EGFR-mutated lung cancer cells. AAPS J. 2017;19(3):814–826. doi:10.1208/s12248-017-0057-9
61. Cheng Y, Ou Z, Li Q, et al. Cabazitaxel liposomes with aptamer modification enhance tumor-targeting efficacy in nude mice. Mol Med Rep. 2019;19(1):490–498.
62. Chen Z, Tai Z, Gu F, Hu C, Zhu Q, Gao S. Aptamer-mediated delivery of docetaxel to prostate cancer through polymeric nanoparticles for enhancement of antitumor efficacy. Eur J Pharm Biopharm. 2016;107:130–141. doi:10.1016/j.ejpb.2016.07.007
63. Charbgoo F, Alibolandi M, Taghdisi SM, Abnous K, Soltani F, Ramezani M. MUC1 aptamer-targeted DNA micelles for dual tumor therapy using doxorubicin and KLA peptide. Nanomedicine. 2018;14(3):685–697.
64. Ayatollahi S, Salmasi Z, Hashemi M, et al. Aptamer-targeted delivery of Bcl-xL shRNA using alkyl modified PAMAM dendrimers into lung cancer cells. Int J Biochem Cell Biol. 2017;92:210–217. doi:10.1016/j.biocel.2017.10.005
65. Barzegar Behrooz A, Nabavizadeh F, Adiban J, et al. Smart bomb AS1411 aptamer-functionalized/PAMAM dendrimer nanocarriers for targeted drug delivery in the treatment of gastric cancer. Clin Exp Pharmacol Physiol. 2017;44(1):41–51. doi:10.1111/1440-1681.12670
66. Shiao YS, Chiu HH, Wu PH, Huang YF. Aptamer-functionalized gold nanoparticles as photoresponsive nanoplatform for co-drug delivery. ACS Appl Mater Interfaces. 2014;6(24):21832–21841. doi:10.1021/am5026243
67. Pascual L, Cerqueira-Coutinho C, Garcia-Fernandez A, et al. MUC1 aptamer-capped mesoporous silica nanoparticles for controlled drug delivery and radio-imaging applications. Nanomedicine. 2017;13(8):2495–2505. doi:10.1016/j.nano.2017.08.006
68. Li C, Qian M, Wang S, et al. Aptavalve-gated mesoporous carbon nanospheres image cellular mucin and provide on-demand targeted drug delivery. Theranostics. 2017;7(13):3319–3325. doi:10.7150/thno.18692
69. Guan B, Yan R, Li R, Zhang X. Selenium as a pleiotropic agent for medical discovery and drug delivery. Int J Nanomedicine. 2018;13:7473–7490. doi:10.2147/IJN.S181343
70. Jalalian SH, Ramezani M, Abnous K, Taghdisi SM. Targeted co-delivery of epirubicin and NAS-24 aptamer to cancer cells using selenium nanoparticles for enhancing tumor response in vitro and in vivo. Cancer Lett. 2018;416:87–93. doi:10.1016/j.canlet.2017.12.023
71. Yazdian-Robati R, Arab A, Ramezani M, et al. Smart aptamer-modified calcium carbonate nanoparticles for controlled release and targeted delivery of epirubicin and melittin into cancer cells in vitro and in vivo. Drug Dev Ind Pharm. 2019;45(4):603–610.
72. Aghanejad A, Babamiri H, Adibkia K, Barar J, Omidi Y. Mucin-1 aptamer-armed superparamagnetic iron oxide nanoparticles for targeted delivery of doxorubicin to breast cancer cells. Bioimpacts. 2018;8(2):117–127.
73. Danesh NM, Lavaee P, Ramezani M, Abnous K, Taghdisi SM. Targeted and controlled release delivery of daunorubicin to T-cell acute lymphoblastic leukemia by aptamer-modified gold nanoparticles. Int J Pharm. 2015;489(1–2):311–317.
74. Mansouri A, Abnous K, Alibolandi M, Taghdisi SM, Ramezani M. Targeted delivery of tacrolimus to T cells by pH-responsive aptamer-chitosan- poly (lactic-co-glycolic acid) nanocomplex. J Cell Physiol. 2019;234(10):18262–18271.
75. Liang C, Guo B, Wu H, et al. Aptamer-functionalized lipid nanoparticles targeting osteoblasts as a novel RNA interference-based bone anabolic strategy. Nat Med. 2015;21(3):288–294. doi:10.1038/nm.3791
76. Kavruk M, Celikbicak O, Ozalp VC, et al. Antibiotic loaded nanocapsules functionalized with aptamer gates for targeted destruction of pathogens. Chem Commun (Camb). 2015;51(40):8492–8495.
77. Ocsoy I, Yusufbeyoglu S, Yilmaz V, McLamore ES, Ildiz N, Ulgen A. DNA aptamer functionalized gold nanostructures for molecular recognition and photothermal inactivation of methicillin-Resistant Staphylococcus aureus. Colloids Surf B Biointerfaces. 2017;159:16–22. doi:10.1016/j.colsurfb.2017.07.056
78. Moosavian SA, Abnous K, Akhtari J, Arabi L, Gholamzade Dewin A, Jafari M. 5TR1 aptamer-PEGylated liposomal doxorubicin enhances cellular uptake and suppresses tumour growth by targeting MUC1 on the surface of cancer cells. Artif Cells Nanomed Biotechnol. 2018;46(8):2054–2065.
79. Vandghanooni S, Eskandani M, Barar J, Omidi Y. AS1411 aptamer-decorated cisplatin-loaded poly (lactic-co-glycolic acid) nanoparticles for targeted therapy of miR-21-inhibited ovarian cancer cells. Nanomedicine (Lond). 2018;13(21):2729–2758. doi:10.2217/nnm-2018-0205
80. Yu Z, Chen F, Qi X, et al. Epidermal growth factor receptor aptamer-conjugated polymer-lipid hybrid nanoparticles enhance salinomycin delivery to osteosarcoma and cancer stem cells. Exp Ther Med. 2018;15(2):1247–1256. doi:10.3892/etm.2017.5578
81. Taghdisi SM, Danesh NM, Ramezani M, et al. Double targeting and aptamer-assisted controlled release delivery of epirubicin to cancer cells by aptamers-based dendrimer in vitro and in vivo. Eur J Pharm Biopharm. 2016;102:152–158. doi:10.1016/j.ejpb.2016.03.013
82. Baneshi M, Dadfarnia S, Shabani AMH, Sabbagh SK, Haghgoo S, Bardania H. A novel theranostic system of AS1411 aptamer-functionalized albumin nanoparticles loaded on iron oxide and gold nanoparticles for doxorubicin delivery. Int J Pharm. 2019;564:145–152. doi:10.1016/j.ijpharm.2019.04.025
83. Liang C, Li F, Wang L, et al. Tumor cell-targeted delivery of CRISPR/Cas9 by aptamer-functionalized lipopolymer for therapeutic genome editing of VEGFA in osteosarcoma. Biomaterials. 2017;147:68–85. doi:10.1016/j.biomaterials.2017.09.015
84. Wan Y, Wang L, Zhu C, et al. Aptamer-conjugated extracellular nanovesicles for targeted drug delivery. Cancer Res. 2018;78(3):798–808. doi:10.1158/0008-5472.CAN-17-2880
85. Yeom JH, Lee B, Kim D, et al. Gold nanoparticle-DNA aptamer conjugate-assisted delivery of antimicrobial peptide effectively eliminates intracellular Salmonella enterica serovar Typhimurium. Biomaterials. 2016;104:43–51. doi:10.1016/j.biomaterials.2016.07.009
86. Xie X, Li F, Zhang H, et al. EpCAM aptamer-functionalized mesoporous silica nanoparticles for efficient colon cancer cell-targeted drug delivery. Eur J Pharm Sci. 2016;83:28–35. doi:10.1016/j.ejps.2015.12.014
87. Taghavi S, Nia AH, Abnous K, Ramezani M. Polyethylenimine-functionalized carbon nanotubes tagged with AS1411 aptamer for combination gene and drug delivery into human gastric cancer cells. Int J Pharm. 2017;516(1–2):301–312. doi:10.1016/j.ijpharm.2016.11.027
88. Dong K, Wang Z, Zhang Y, Ren J, Qu X. Metal-organic framework-based nanoplatform for intracellular environment-responsive endo/lysosomal escape and enhanced cancer therapy. ACS Appl Mater Interfaces. 2018;10(38):31998–32005. doi:10.1021/acsami.8b11972
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.