Back to Journals » Neuropsychiatric Disease and Treatment » Volume 14
Anxiety and depression in paradoxical insomnia: a case–control study
Received 4 November 2017
Accepted for publication 5 December 2017
Published 8 January 2018 Volume 2018:14 Pages 231—238
DOI https://doi.org/10.2147/NDT.S156058
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Wai Kwong Tang
Jingping Liao,1,* Shichao Zhu,1,2,* Xiaolin Li1
1Department of Nursing, 2Department of Infection Control, West China Hospital, Sichuan University, Chengdu, People’s Republic of China
*These authors contributed equally to this work
Purpose: To compare anxiety and depression among patients with paradoxical insomnia (Para-I), patients with psychophysiological insomnia (Psy-I), and normal sleepers (NS).
Patients and methods: A cross-sectional case–control study was conducted in patients with Para-I (n=63), patients with Psy-I (n=63), and NS (n=63) from southwest China. The three groups were matched for age (mean age: 42.0±10.30 years), gender (per group: male =22; female =41), educational level, and nature of occupation. Anxiety and depression were evaluated using the Self-Rating Anxiety Scale and Self-Rating Depression Scale and compared among the groups.
Results: Concerning anxiety, patients with Para-I had a significantly higher mean Self-Rating Anxiety Scale score (P<0.05) and significantly higher incidence of anxiety compared to NS (P<0.0167). There were no significant differences between the two insomnia groups on anxiety measures. Concerning depression, patients with Para-I had a significantly higher mean Self-Rating Depression Scale score than patients with Psy-I and NS (both, P<0.05), as well as a significantly higher incidence of moderate to severe depression than in the Psy-I group and NS (both, P<0.0167).
Conclusion: Both Psy-I and Para-I patients had significantly higher anxiety and depression than NS. Compared to Psy-I, Para-I patients had slightly (but not significantly) lower anxiety and significantly higher depression.
Keywords: sleep disorders, psychological comorbidity, stress, paradoxical insomnia
Introduction
Insomnia is often defined as poor or unsatisfactory sleep. It is primarily viewed as a persistent condition that is characterized by severe distress as well as social, interpersonal, and occupational impairment.1 Prevalence of insomnia is high in the general population worldwide; conservative estimates of the prevalence of chronic insomnia range from 9% to 12% in adults, making insomnia the most commonly reported sleep complaint.2,3 Furthermore, insomnia has a high rate of comorbidity with a range of psychological disorders, particularly anxiety and depression.4–6
There are several different opinions concerning the relationship between anxiety/depression and insomnia. One viewpoint is that anxiety, depression, and insomnia simply co-occur, and can be explained by other precipitating or maintaining mechanisms.7,8 A second possibility is that insomnia is secondary to (ie, a symptom or epiphenomenon of) depression and anxiety, or conversely, that anxiety and depression are epiphenomenal to insomnia, which has been concluded to be unfounded.6 A third possibility is that anxiety and depression act as risk factors for insomnia,9–11 but also that insomnia may act as a risk factor for, and may even be causal in, the development of anxiety and depression.12 Finally, it is possible that the association between anxiety/depression and insomnia is not simply a cause–effect relationship, but instead a complex bidirectional one, whereby anxiety and depression influence insomnia, and vice versa.13 Thus, while insomnia evidently has a strong association with anxiety and depression, the nature of this relationship is as yet unclear.
According to the second edition of the International Classification of Sleep Disorders (ICSD-2), paradoxical insomnia (Para-I) is one subtype of chronic insomnia14 and is characterized by misperceptions of sleep quality and quantity. It is also known as subjective insomnia, pseudoinsomnia, subjective complaint of sleep initiation and maintenance difficulty without objective findings, insomnia without objective findings, sleep hypochondriasis, subjective sleep complaint, and, most widely, sleep state misperception. Para-I, which comprises about 5%–9% of all insomnia,14 is defined as complaints of severe insomnia that occur without obvious evidence of objective sleep disturbance and without a significant impairment of daytime functioning.15,16 Individuals suffering from Para-I complain about sleep difficulties while objective sleep measures (such as polysomnography, PSG) appear to be normal.17
Previous investigations of Para-I have mainly focused on its physiological characteristics,18,19 and psychological characteristics such as anxiety and depression remain unclear. This has hindered the understanding of and treatment for the emotional symptoms of Para-I; thus, a more detailed theoretical understanding of problems are required as is the development of intervention guidelines for treating anxiety and depression in patients with Para-I. To this end, a case–control study was performed to compare anxiety and depression in patients with Para-I, patients with psychophysiological insomnia (Psy-I), and normal sleepers (NS). We hypothesized that both Psy-I and Para-I groups would have higher anxiety and depression levels than NS. We also hypothesized that patients with Para-I would have different anxiety and depression levels to patients with Psy-I.
Methods
Subjects
A case–control study was conducted. Sixty-three consecutive patients with Para-I were recruited from the Sleep Medicine Center of West China Hospital of Sichuan University between March 2013 and December 2013. During the same period, gender-, age- (within 3 years), cultural level-, and nature of occupation-matched patients with Psy-I (n=63) and NS (n=63) were recruited as controls. The diagnostic criteria for Para-I and Psy-I were consistent with those of the ICSD-2.14 All subjects underwent a multistep screening evaluation, which consisted of a sleep history interview, an overnight PSG, a psychological assessment, and medical history taking with physical examination. These evaluations were conducted by a board-certified sleep specialist, a clinical psychologist, and a physician. Participants received no compensation for their participation in the study, nor did they pay for the screening evaluation.
Inclusion criteria for all participants were as follows: 1) over 18 years old; and 2) educated in primary school or above. Exclusion criteria for all participants were as follows: 1) presence of a significant current medical or neurological disorder that compromises sleep; 2) presence of a major psychopathology; 3) alcohol or drug abuse during the past year; 4) evidence of another sleep disorder such as sleep apnea or periodic limb movements during sleep; 5) use of a medication known to affect sleep; and 6) ongoing psychological treatment. Details for the specific groups are presented below.
Psy-I group
Individuals suffering from chronic Psy-I14,17 met the following inclusion criteria: 1) presence of a subjective complaint of insomnia, defined as difficulty initiating (ie, sleep-onset latency >30 min) and/or maintaining sleep (ie, time awake after sleep-onset >30 min) present at least three nights per week; 2) insomnia duration of at least 6 months; 3) insomnia or its perceived consequences causing marked distress or significant impairment of occupational or social functioning; and 4) presence of a subjective complaint of at least one negative daytime consequence attributed to insomnia.
Para-I group
In addition to the mentioned chronic insomnia criteria, participants in the Para-I group14,17 were required to meet the following criteria: 1) a total sleep time of more than 6 h 30 min and a sleep efficiency greater than 85% on nocturnal PSG; and 2) marked discrepancies between subjective and objective sleep measures (ie, a difference of 60 min or more for total sleep time, and/or a difference of at least 15% between subjective and objective measures of sleep efficiency).
NS
NS19 were community-dwelling adults recruited from the general population through advertisements. Inclusion criteria for NS were as follows: 1) a total sleep time of more than 7 h per night; 2) satisfaction with their sleep; 3) no subjective sleep complaints; and 4) an objective sleep efficiency of more than 85%.
Procedure
Evaluations were conducted by two graduate students through face-to-face interviews at the Sleep Medicine Center. Prior to the study, the investigators received training on the aim and procedure of the study, as well as the questionnaires and appropriate communication. First, the investigators explained the purpose and main content of the survey and evaluated whether participants met the eligibility criteria. After obtaining informed consent from eligible participants, the investigators explained how to complete the questionnaires. The participants were asked to complete the questionnaires independently and according to their true feelings. Finally, the investigators collected the questionnaires immediately after their completion to check the integrity of the data.
Ethical considerations
The study received approval from the Ethics Committee of West China Hospital of Sichuan University. The investigation complies with the principle of voluntariness, and written informed consent was obtained from all participants.
Measurements
PSG
Overnight PSG recording techniques and standard parameters were implemented according to the American Academy of Sleep Medicine Manual for the Scoring of Sleep and Associated Events.20 Sleep data were collected using an Alice 5 Diagnostic Sleep System (Philips Respironics, Bend, OR, USA). PSG recording included electroencephalography (F4-M1, C4-M1, O2-M1, F3-M2, C3-M2, and O1-M2), bilateral electro-oculography, electrocardiography, electromyography (submental, anterior tibialis), oral airflow, rib cage and abdominal movements, and an arterial oxygen saturation sensor on the left index finger. The observed sleep parameters included total time in bed, total sleep time, sleep-onset latency, sleep efficiency, and wake time after sleep-onset.
Pittsburgh Sleep Quality Index (PSQI)
The PSQI21 primarily assesses general sleep quality in the past month, and in the following seven domains: duration of sleep, sleep disturbance, sleep-onset latency, daytime dysfunction due to night sleep, sleep efficiency, need for medications to sleep, and overall sleep quality. A higher score indicates worse sleep quality and more severe insomnia, and a total score higher than five indicates insufficient sleep quality.22
Self-Rating Anxiety Scale (SAS)
The SAS23 is used to evaluate the level of anxiety-related symptoms in the past week. This self-administered test has 20 questions, with 15 items reflecting higher anxiety levels and five items reflecting lower anxiety levels. Each item is scored on a scale from 1 to 4 (rarely, sometimes, frequently, and always). The total score is computed as an original score, and then multiplied by 1.25 to get the standard score. Higher standard scores represent a higher intensity of symptoms. Scores less than 50 indicate no anxiety, scores between 50 and 59 indicate minimal to mild anxiety, scores between 60 and 69 indicate moderate to marked anxiety, and scores greater than 70 indicate severe anxiety.
Self-Rating Depression Scale (SDS)
The SDS24 is a 20-item questionnaire designed to assess depressive symptoms over the past week. Ten of the items express negative experience or symptoms (eg, “I have crying spells or feel like crying”) and ten express positive experiences and are reversed (eg, “My life is pretty full”). Each item is scored on a Likert scale ranging from 1 to 4. The raw total scores are converted to a 100-point scale by multiplying by 1.25. A higher score indicates more severe depression symptoms. Scores less than 50 indicate no depression, scores between 50 and 59 indicate minimal to mild depression, scores between 60 and 69 indicate moderate to marked depression, and scores greater than 70 indicate severe depression.
Statistics
The Statistical Package for SPSS 21.0 (SPSS, Chicago, IL, USA) was used for all data management and analyses. Data distribution was assessed using the Kolmogorov–Smirnov test. The data were expressed as means ± standard deviations for continuous variables and as frequencies (%) for categorical variables. One-way analysis of variance and χ2 tests were used to compare the three groups on sociodemographic variables, sleep parameters, and anxiety and depression. Two-way comparisons of continuous variables were performed using least significant difference procedure tests for equal variances or Tamhane’s T2 tests for unequal variances. Two-way comparisons of categorical variables were performed using Wilcoxon rank sum test for unequal variances. P-values presented as the result of Wilcoxon rank sum test were corrected (ie, P multiplied by the number of pairwise comparisons). Therefore, P-values under 0.0167 were considered significant. For all other results, P<0.05 was considered to be statistically significant.
Results
Sociodemographic variables
Sociodemographic variables for the three groups are shown in Table 1. The mean ages of Para-I, Psy-I, and NS groups were 42.0±10.2 (range 19–65), 42.8±9.6 (range 21–62), 41.4±11.1 (range 19–66) years, respectively. Each group consisted of 22 males (30.1%) and 41 females (69.9%). More than one-third of the participants were educated in college or above (39.1%) and the nature of their occupation was mainly mental (92.1%). No significant between-group differences were found in age, gender, education, or nature of occupation (all, P>0.05).
  | Table 1 Comparison of sociodemographic variables among the three groups |
Sleep parameters
Sleep parameters for the three groups are showed in Table 2. No significant between-group differences were found in total time in bed (P>0.05). There were significant between-group differences in total sleep time, sleep-onset latency, sleep efficiency, and wake time after sleep-onset (all, P<0.001); specifically, these sleep parameters were significantly higher in the Para-I and NS groups compared to the Psy-I group (all, P<0.001, Table 3). There were no significant differences in total sleep time, sleep-onset latency, sleep efficiency, and wake time after sleep-onset between Para-I and NS groups (all, P>0.05). However, there were significant between-group differences in PSQI scores (P<0.001); specifically, PSQI scores were significantly higher in the Para-I and Psy-I groups compared to the NS group (both P<0.001, Table 3). There was no significant difference in PSQI scores between Para-I and Psy-I groups (P>0.05). These results helped to categorize patients into groups.
SAS and SDS scores
The SAS and SDS scores are listed in Table 4. The mean SAS scores of the Para-I (51.1±10.5) and Psy-I (54.0±7.6) groups were indicative of minimal to mild levels of anxiety, and that of the NS group (27.1±1.7) was indicative of no anxiety. There were significant between-group differences in SAS scores (P<0.001); specifically, SAS scores were significantly higher in the Para-I and Psy-I groups compared to the NS group (both P<0.001, Table 5). There was no significant difference in SAS scores between Para-I and Psy-I groups (P>0.05).
The mean SDS scores of both the Para-I (55.2±13.1) and Psy-I (50.5±7.6) groups were indicative of minimal to mild levels of depression, and that of the NS group (29.0±2.0) was indicative of no depression. There were significant between-group differences in SDS scores (P<0.001); specifically, SDS scores in the Para-I and Psy-I groups were significantly higher than that of the NS group (P<0.001, Table 5). Moreover, the mean SDS score of the Para-I group was significantly higher than that of the Psy-I group (P<0.05).
Constituent ratio of anxiety and depression
The prevalence and severity of anxiety and depression according to the SAS and SDS are shown in Table 6. Overall, the prevalence of anxiety and depression in the Para-I group were 57.1% (36/63) and 63.5% (40/63), respectively, and 15.5% of cases (30/63) expressed symptoms of both anxiety and depression. Specifically, 42.9% of the patients with Para-I (27/63) were in the normal range for anxiety, 36.5% (23/63) were characterized by minimal to mild anxiety, 17.5% (11/63) by moderate to marked anxiety, and 3.2% (2/63) by severe anxiety. Furthermore, 36.5% of the patients with Para-I (23/63) showed no depression, 25.4% (16/63) minimal to mild depression, 25.4% (16/63) moderate to marked depression, and 12.7% (8/63) severe depression.
  | Table 6 Comparison of constituent ratio of anxiety and depression among the three groups |
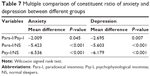
The χ2 test revealed a significant between-group difference in the constituent ratio of anxiety and depression (P<0.001). In addition, according to the results of multiple comparisons (Table 7), the constituent ratios of anxiety and depression of both the Para-I and Psy-I groups were significantly different to those of the NS group (P<0.0167). The incidence of anxiety in the Para-I group was slightly lower than that in patients with Psy-I, but this did not reach significance (P>0.0167). Moreover, even though the incidence of minimal to mild depression was lower in the Para-I group than that in the Psy-I group, the incidence of moderate to severe depression was significantly higher (P<0.0167).
Discussion
This study compared anxiety and depression between patients with Para-I, patients with Psy-I, and NS. As expected, both Psy-I and Para-I groups had higher anxiety and depression levels than NS. Interestingly, patients with Para-I had different anxiety and depression levels compared to those with Psy-I.
Our study showed that, compared to NS, both Psy-I and Para-I groups had significantly higher SAS scores (mean score: 54.0±7.6, 51.1±10.5 vs 27.1±1.7) and SDS scores (mean score: 50.5±7.6, 55.2±13.1 vs 29.0±2.0). Similarly, the incidences of anxiety and depression in the Psy-I (61.0%, 66.7%) and Para-I (57.1%, 63.5%) groups were significantly higher than that in NS (0%, 0%). These results indicate that patients with Psy-I and Para-I had significantly greater anxiety and depression than NS. This is consistent with a previous study,25 which also found that insomniacs had higher anxiety and depression than NS. Furthermore, our results indicated that patients with Para-I and with Psy-I experienced the same levels of anxiety and depression. These results go some way to support the idea that Para-I might be an extreme form of insomnia even though patients’ objective sleep measures are comparable to those of NS.26
Our study also showed that the Para-I group had a slightly (but not significantly) lower SAS than the Psy-I group (mean score: 51.1±10.5 vs 54.0±7.6). Similarly, the incidence of anxiety in the Para-I group (57.1%) was slightly (but not significantly) lower than that in Psy-I group (61.0%). In line with a previous study,27 these results support Salin-Pascual et al’s15 suggestion that Para-I may be a transitional pathophysiological state from healthy sleep to Psy-I, because the anxiety level of patients with Para-I is between those of NS and patients with Psy-I.15 Insomniacs often suffer from unpleasant intrusive thoughts and excessive and uncontrollable worry during the presleep period. One cognitive model of insomnia has suggested that individuals who suffer from insomnia tend to be overly worried about their sleep and about the daytime consequences of not getting enough sleep.28 This excessive negatively toned cognitive activity triggers both autonomic arousal and emotional distress.28 This anxious state triggers selective attention toward and monitoring of internal and external sleep-related threat cues.28 Together, the anxious state and the attentional processes may cause individuals to overestimate the extent of the perceived deficit in sleep and daytime performance.28 Anxious-ruminative traits, which may contribute to cognitive arousal, seem to mediate the underestimation of sleep duration.29
Another finding of our study was that the Para-I group had a significantly higher SDS score than the Psy-I group (mean score: 55.2±13.1 vs 50.5±7.6). Indeed, nearly a third of individuals with Para-I suffered from moderate to severe depression (38.1%), which was higher than Psy-I (7.9%). These results suggest that patients with Para-I experienced more severe depression than patients with Psy-I. These results are consistent with previous study.30 It is interesting to note that despite significantly higher levels of depression, individuals with Para-I had a slightly lower anxiety. As mentioned, it is widely accepted that anxious-ruminative traits play an important role in the underestimation of sleep duration. However, our results indicate that depression may have a stronger relationship with sleep underestimation than anxiety, and future studies could more fully investigate the mechanisms underlying this link. As we introduced in the background, there are many notions about the interrelationship between depression and insomnia. These notions may also be applied to the relationship between depression and Para-I.31
In light of several concerns, the ICSD-3 task force has elected to consolidate all insomnia diagnoses (ie, “primary” and “comorbid”) under a single, chronic insomnia disorder, and nullified the subtypes of chronic insomnia, including Psy-I and Para-I.32 However, this decision reflects the recognition that we are not yet able to reliably make such distinctions or translate them into customized therapeutic approaches rather than the belief that no pathophysiologic differences between chronic insomnia subtypes exist.33 The initiating mechanisms and characteristics are still important in understanding insomnia.34
Although the present study furthers our understanding of mood symptoms in patients with Para-I, its findings should be interpreted with caution in light of its potential limitations. First, this study was cross-sectional in nature; thus, identifying the complex causal relationships among anxiety, depression, and Para-I were not within the scope of this study. Second, the patients with Para-I and Psy-I in this study were voluntary sleep clinic outpatients, whereas NS participants were recruited from the community, which may lead to a selection bias. Third, all of the patients in the current study came from a single tertiary referral center, which may lead to another selection bias, and further multicenter longitudinal study may help to verify the current findings. Finally, we did not clinically diagnose depression and anxiety and relied only on self-rated questionnaires; thus, it might not reflect the true levels and incidences of anxiety and depression. Clinical diagnosis of depression and anxiety should be considered in future studies.
Conclusion
In conclusion, the present findings suggest that anxiety and depression should be routinely assessed in the diagnosis and treatment of sleep disorders. This may help clinicians to distinguish Para-I from NS (ie, higher anxiety and depression levels) as well as to distinguish Para-I from Psy-I (ie, slightly lower anxiety and significantly higher depression). This may also aid the development of tailored therapeutic interventions. However, future research on the role of anxiety and depression in sleep underestimation is warranted in the search for individualized treatment of Para-I.
Disclosure
The authors report no conflicts of interest in this work.
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Pub; 2013. | ||
Ancoli-Israel S, Roth T. Characteristics of insomnia in the United States: results of the 1991 National Sleep Foundation Survey. I. Sleep. 1999;22(Suppl 2):S347–S353. | ||
Hoch CC, Dew MA, Reynolds CF 3rd, et al. Longitudinal changes in diary- and laboratory-based sleep measures in healthy “old old” and “young old” subjects: a three-year follow-up. Sleep. 1997;20(3):192–202. | ||
Morin CM, Ware JC. Sleep and psychopathology. Appl Prevent Psychol. 1996;5(4):211–224. | ||
Ohayon MM, Caulet M, Lemoine P. Comorbidity of mental and insomnia disorders in the general population. Compr Psychiatry. 1998;39(4):185–197. | ||
Harvey AG. Insomnia: symptom or diagnosis? Clin Psychol Rev. 2001;21(7):1037–1059. | ||
Ford DE, Kamerow DB. Epidemiologic study of sleep disturbances and psychiatric disorders. An opportunity for prevention? JAMA. 1989;262(11):1479–1484. | ||
Mellinger GD, Balter MB, Uhlenhuth EH. Insomnia and its treatment. Prevalence and correlates. Arch Gen Psychiatry. 1985;42(3):225–232. | ||
Jansson M, Linton SJ. The role of anxiety and depression in the development of insomnia: cross-sectional and prospective analyses. Psychol Health. 2006;21(3):383–397. | ||
Johnson EO, Roth T, Breslau N. The association of insomnia with anxiety disorders and depression: exploration of the direction of risk. J Psychiatr Res. 2006;40(8):700–708. | ||
Ohayon MM, Roth T. Place of chronic insomnia in the course of depressive and anxiety disorders. J Psychiatr Res. 2003;37(1):9–15. | ||
Neckelmann D, Mykletun A, Dahl AA. Chronic insomnia as a risk factor for developing anxiety and depression. Sleep. 2007;30(7):873–880. | ||
Jansson-Frojmark M, Lindblom K. A bidirectional relationship between anxiety and depression, and insomnia? A prospective study in the general population. J Psychosom Res. 2008;64(4):443–449. | ||
American Academy of Sleep Medicine. International Classification of Sleep Disorders: Diagnostic and Coding Manual. 2nd ed. Westchester, IL: American Academy of Sleep Medicine; 2005:148–152. | ||
Salin-Pascual RJ, Roehrs TA, Merlotti LA, Zorick F, Roth T. Long-term study of the sleep of insomnia patients with sleep state misperception and other insomnia patients. Am J Psychiatry. 1992;149(7):904–908. | ||
Edinger JD, Fins AI. The distribution and clinical significance of sleep time misperceptions among insomniacs. Sleep. 1995;18(4):232–239. | ||
Edinger JD, Bonnet MH, Bootzin RR, et al. Derivation of research diagnostic criteria for insomnia: report of an American Academy of Sleep Medicine Work Group. Sleep. 2004;27(8):1567–1596. | ||
St-Jean G, Turcotte I, Perusse AD, Bastien CH. REM and NREM power spectral analysis on two consecutive nights in psychophysiological and paradoxical insomnia sufferers. Int J Psychophysiol. 2013;89(2):181–194. | ||
Turcotte I, St-Jean G, Bastien CH. Are individuals with paradoxical insomnia more hyperaroused than individuals with psychophysiological insomnia? Event-related potentials measures at the peri-onset of sleep. Int J Psychophysiol. 2011;81(3):177–190. | ||
Iber C, Ancoli-Israel S, Chesson A, Quan S. The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. Westchester, IL: American Academy of Sleep Medicine; 2007:48–49. | ||
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatr Res. 1989;28(2):193–213. | ||
Backhaus J, Junghanns K, Broocks A, Riemann D, Hohagen F. Test-retest reliability and validity of the Pittsburgh Sleep Quality Index in primary insomnia. J Psychosom Res. 2002;53(3):737–740. | ||
Zung WW. A rating instrument for anxiety disorders. Psychosomatics. 1971;12(6):371–379. | ||
Zung WW. A Self-rating depression scale. Arch Gen Psychiatry. 1965;12(12):63–70. | ||
Taylor DJ, Lichstein KL, Durrence HH, Reidel BW, Bush AJ. Epidemiology of insomnia, depression, and anxiety. Sleep. 2005;28(11):1457–1464. | ||
Attarian HP, Schuman C. Clinical Handbook of Insomnia. New York, NY: Springer; 2010. | ||
Dittoni S, Mazza M, Losurdo A, et al. Psychological functioning measures in patients with primary insomnia and sleep state misperception. Acta neurologica Scandinavica. 2013;128(1):54–60. | ||
Harvey AG. A cognitive model of insomnia. Behav Res Therapy. 2002;40(8):869–893. | ||
Fernandez-Mendoza J, Calhoun SL, Bixler EO, et al. Sleep misperception and chronic insomnia in the general population: role of objective sleep duration and psychological profiles. Psychosom Med. 2011;73(1):88–97. | ||
Vandeputte M, de Weerd A. Sleep disorders and depressive feelings: a global survey with the Beck depression scale. Sleep Med. 2003;4(4):343–345. | ||
Lovato N, Gradisar M. A meta-analysis and model of the relationship between sleep and depression in adolescents: recommendations for future research and clinical practice. Sleep Med Rev. 2014;18(6):521–529. | ||
American Academy of Sleep Medicine. International Classification of Sleep Disorders: Diagnostic and Coding Manual. 3rd ed. Darien, IL: American Academy of Sleep Medicine; 2014. | ||
Sateia MJ. International classification of sleep disorders-third edition: highlights and modifications. Chest. 2014;146(5):1387–1394. | ||
Thorpy M. International classification of sleep disorders. Sleep Disorders Medicine: Springer; 2017:475–484. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.